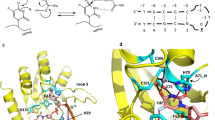
Abstract
APOBEC-catalyzed cytosine-to-uracil deamination of single-stranded DNA (ssDNA) has beneficial functions in immunity and detrimental effects in cancer. APOBEC enzymes have intrinsic dinucleotide specificities that impart hallmark mutation signatures. Although numerous structures have been solved, mechanisms for global ssDNA recognition and local target-sequence selection remain unclear. Here we report crystal structures of human APOBEC3A and a chimera of human APOBEC3B and APOBEC3A bound to ssDNA at 3.1-Å and 1.7-Å resolution, respectively. These structures reveal a U-shaped DNA conformation, with the specificity-conferring −1 thymine flipped out and the target cytosine inserted deep into the zinc-coordinating active site pocket. The −1 thymine base fits into a groove between flexible loops and makes direct hydrogen bonds with the protein, accounting for the strong 5′-TC preference. These findings explain both conserved and unique properties among APOBEC family members, and they provide a basis for the rational design of inhibitors to impede the evolvability of viruses and tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Conticello, S.G. The AID/APOBEC family of nucleic acid mutators. Genome Biol. 9, 229 (2008).
Harris, R.S. & Dudley, J.P. APOBECs and virus restriction. Virology 479–480, 131–145 (2015).
Di Noia, J.M. & Neuberger, M.S. Molecular mechanisms of antibody somatic hypermutation. Annu. Rev. Biochem. 76, 1–22 (2007).
Robbiani, D.F. & Nussenzweig, M.C. Chromosome translocation, B cell lymphoma, and activation-induced cytidine deaminase. Annu. Rev. Pathol. 8, 79–103 (2013).
Fossat, N. & Tam, P.P. Re-editing the paradigm of cytidine (C) to uridine (U) RNA editing. RNA Biol. 11, 1233–1237 (2014).
Koito, A. & Ikeda, T. Intrinsic immunity against retrotransposons by APOBEC cytidine deaminases. Front. Microbiol. 4, 28 (2013).
Malim, M.H. & Bieniasz, P.D. HIV restriction factors and mechanisms of evasion. Cold Spring Harb. Perspect. Med. 2, a006940 (2012).
Simon, V., Bloch, N. & Landau, N.R. Intrinsic host restrictions to HIV-1 and mechanisms of viral escape. Nat. Immunol. 16, 546–553 (2015).
Refsland, E.W. et al. Quantitative profiling of the full APOBEC3 mRNA repertoire in lymphocytes and tissues: implications for HIV-1 restriction. Nucleic Acids Res. 38, 4274–4284 (2010).
Koning, F.A. et al. Defining APOBEC3 expression patterns in human tissues and hematopoietic cell subsets. J. Virol. 83, 9474–9485 (2009).
Stenglein, M.D., Burns, M.B., Li, M., Lengyel, J. & Harris, R.S. APOBEC3 proteins mediate the clearance of foreign DNA from human cells. Nat. Struct. Mol. Biol. 17, 222–229 (2010).
Burns, M.B. et al. APOBEC3B is an enzymatic source of mutation in breast cancer. Nature 494, 366–370 (2013).
Helleday, T., Eshtad, S. & Nik-Zainal, S. Mechanisms underlying mutational signatures in human cancers. Nat. Rev. Genet. 15, 585–598 (2014).
Roberts, S.A. & Gordenin, D.A. Hypermutation in human cancer genomes: footprints and mechanisms. Nat. Rev. Cancer 14, 786–800 (2014).
Swanton, C., McGranahan, N., Starrett, G.J. & Harris, R.S. APOBEC enzymes: mutagenic fuel for cancer evolution and heterogeneity. Cancer Discov. 5, 704–712 (2015).
Carpenter, M.A., Rajagurubandara, E., Wijesinghe, P. & Bhagwat, A.S. Determinants of sequence-specificity within human AID and APOBEC3G. DNA Repair (Amst.) 9, 579–587 (2010).
Rathore, A. et al. The local dinucleotide preference of APOBEC3G can be altered from 5′-CC to 5′-TC by a single amino acid substitution. J. Mol. Biol. 425, 4442–4454 (2013).
Kohli, R.M. et al. Local sequence targeting in the AID/APOBEC family differentially impacts retroviral restriction and antibody diversification. J. Biol. Chem. 285, 40956–40964 (2010).
Wang, M., Rada, C. & Neuberger, M.S. Altering the spectrum of immunoglobulin V gene somatic hypermutation by modifying the active site of AID. J. Exp. Med. 207, 141–153 (2010).
Starrett, G.J. et al. The DNA cytosine deaminase APOBEC3H haplotype I likely contributes to breast and lung cancer mutagenesis. Nat. Commun. 7, 12918 (2016).
Caval, V., Suspène, R., Shapira, M., Vartanian, J.P. & Wain-Hobson, S. A prevalent cancer susceptibility APOBEC3A hybrid allele bearing APOBEC3B 3′ UTR enhances chromosomal DNA damage. Nat. Commun. 5, 5129 (2014).
Chan, K. et al. An APOBEC3A hypermutation signature is distinguishable from the signature of background mutagenesis by APOBEC3B in human cancers. Nat. Genet. 47, 1067–1072 (2015).
Nik-Zainal, S. et al. Association of a germline copy number polymorphism of APOBEC3A and APOBEC3B with burden of putative APOBEC-dependent mutations in breast cancer. Nat. Genet. 46, 487–491 (2014).
Burns, M.B., Temiz, N.A. & Harris, R.S. Evidence for APOBEC3B mutagenesis in multiple human cancers. Nat. Genet. 45, 977–983 (2013).
Sieuwerts, A.M. et al. Elevated APOBEC3B correlates with poor outcomes for estrogen-receptor-positive breast cancers. Horm. Cancer 5, 405–413 (2014).
Law, E.K. et al. The DNA cytosine deaminase APOBEC3B promotes tamoxifen resistance in ER-positive breast cancer. Sci. Adv. 2, e1601737 (2016).
Cescon, D.W., Haibe-Kains, B. & Mak, T.W. APOBEC3B expression in breast cancer reflects cellular proliferation, while a deletion polymorphism is associated with immune activation. Proc. Natl. Acad. Sci. USA 112, 2841–2846 (2015).
Yan, S. et al. Increased APOBEC3B predicts worse outcomes in lung cancer: a comprehensive retrospective study. J. Cancer 7, 618–625 (2016).
Walker, B.A. et al. Mutational spectrum, copy number changes, and outcome: results of a sequencing study of patients with newly diagnosed myeloma. J. Clin. Oncol. 33, 3911–3920 (2015).
Chen, K.M. et al. Structure of the DNA deaminase domain of the HIV-1 restriction factor APOBEC3G. Nature 452, 116–119 (2008).
Holden, L.G. et al. Crystal structure of the anti-viral APOBEC3G catalytic domain and functional implications. Nature 456, 121–124 (2008).
Kitamura, S. et al. The APOBEC3C crystal structure and the interface for HIV-1 Vif binding. Nat. Struct. Mol. Biol. 19, 1005–1010 (2012).
Li, M. et al. First-in-class small molecule inhibitors of the single-strand DNA cytosine deaminase APOBEC3G. ACS Chem. Biol. 7, 506–517 (2012).
Bohn, M.F. et al. Crystal structure of the DNA cytosine deaminase APOBEC3F: the catalytically active and HIV-1 Vif-binding domain. Structure 21, 1042–1050 (2013).
Byeon, I.J. et al. NMR structure of human restriction factor APOBEC3A reveals substrate binding and enzyme specificity. Nat. Commun. 4, 1890 (2013).
Bohn, M.F. et al. The ssDNA mutator APOBEC3A is regulated by cooperative dimerization. Structure 23, 903–911 (2015).
Shi, K., Carpenter, M.A., Kurahashi, K., Harris, R.S. & Aihara, H. Crystal structure of the DNA deaminase APOBEC3B catalytic domain. J. Biol. Chem. 290, 28120–28130 (2015).
Nakashima, M. et al. Structural insights into HIV-1 Vif-APOBEC3F interaction. J. Virol. 90, 1034–1047 (2015).
Shaban, N.M., Shi, K., Li, M., Aihara, H. & Harris, R.S. 1.92 angstrom zinc-free APOBEC3F catalytic domain crystal structure. J. Mol. Biol. 428, 2307–2316 (2016).
Byeon, I.J. et al. Nuclear magnetic resonance structure of the APOBEC3B catalytic domain: structural basis for substrate binding and DNA deaminase activity. Biochemistry 55, 2944–2959 (2016).
Xiao, X., Li, S.X., Yang, H. & Chen, X.S. Crystal structures of APOBEC3G N-domain alone and its complex with DNA. Nat. Commun. 7, 12193 (2016).
Carpenter, M.A. et al. Methylcytosine and normal cytosine deamination by the foreign DNA restriction enzyme APOBEC3A. J. Biol. Chem. 287, 34801–34808 (2012).
Caval, V. et al. Molecular basis of the attenuated phenotype of human APOBEC3B DNA mutator enzyme. Nucleic Acids Res. 43, 9340–9349 (2015).
Chen, H. et al. APOBEC3A is a potent inhibitor of adeno-associated virus and retrotransposons. Curr. Biol. 16, 480–485 (2006).
Logue, E.C. et al. A DNA sequence recognition loop on APOBEC3A controls substrate specificity. PLoS One 9, e97062 (2014).
Harjes, S. et al. Impact of H216 on the DNA binding and catalytic activities of the HIV restriction factor APOBEC3G. J. Virol. 87, 7008–7014 (2013).
Pham, P., Landolph, A., Mendez, C., Li, N. & Goodman, M.F. A biochemical analysis linking APOBEC3A to disparate HIV-1 restriction and skin cancer. J. Biol. Chem. 288, 29294–29304 (2013).
Rausch, J.W., Chelico, L., Goodman, M.F. & Le Grice, S.F. Dissecting APOBEC3G substrate specificity by nucleoside analog interference. J. Biol. Chem. 284, 7047–7058 (2009).
Harris, R.S. et al. DNA deamination mediates innate immunity to retroviral infection. Cell 113, 803–809 (2003).
Yu, Q. et al. Single-strand specificity of APOBEC3G accounts for minus-strand deamination of the HIV genome. Nat. Struct. Mol. Biol. 11, 435–442 (2004).
Kim, E.Y. et al. Human APOBEC3 induced mutation of human immunodeficiency virus type-1 contributes to adaptation and evolution in natural infection. PLoS Pathog. 10, e1004281 (2014).
Roberts, S.A. et al. An APOBEC cytidine deaminase mutagenesis pattern is widespread in human cancers. Nat. Genet. 45, 970–976 (2013).
Alexandrov, L.B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421 (2013).
Mitra, M. et al. Structural determinants of human APOBEC3A enzymatic and nucleic acid binding properties. Nucleic Acids Res. 42, 1095–1110 (2014).
Fu, Y. et al. DNA cytosine and methylcytosine deamination by APOBEC3B: enhancing methylcytosine deamination by engineering APOBEC3B. Biochem. J. 471, 25–35 (2015).
Demorest, Z.L., Li, M. & Harris, R.S. Phosphorylation directly regulates the intrinsic DNA cytidine deaminase activity of activation-induced deaminase and APOBEC3G protein. J. Biol. Chem. 286, 26568–26575 (2011).
Holtz, C.M., Sadler, H.A. & Mansky, L.M. APOBEC3G cytosine deamination hotspots are defined by both sequence context and single-stranded DNA secondary structure. Nucleic Acids Res. 41, 6139–6148 (2013).
Nik-Zainal, S. et al. Landscape of somatic mutations in 560 breast cancer whole-genome sequences. Nature 534, 47–54 (2016).
Matthews, M.M. et al. Structures of human ADAR2 bound to dsRNA reveal base-flipping mechanism and basis for site selectivity. Nat. Struct. Mol. Biol. 23, 426–433 (2016).
Shirakawa, K. et al. Phosphorylation of APOBEC3G by protein kinase A regulates its interaction with HIV-1 Vif. Nat. Struct. Mol. Biol. 15, 1184–1191 (2008).
Losey, H.C., Ruthenburg, A.J. & Verdine, G.L. Crystal structure of Staphylococcus aureus tRNA adenosine deaminase TadA in complex with RNA. Nat. Struct. Mol. Biol. 13, 153–159 (2006).
Conticello, S.G., Langlois, M.A. & Neuberger, M.S. Insights into DNA deaminases. Nat. Struct. Mol. Biol. 14, 7–9 (2007).
Almog, R., Maley, F., Maley, G.F., Maccoll, R. & Van Roey, P. Three-dimensional structure of the R115E mutant of T4-bacteriophage 2′-deoxycytidylate deaminase. Biochemistry 43, 13715–13723 (2004).
Teh, A.H. et al. The 1.48 Å resolution crystal structure of the homotetrameric cytidine deaminase from mouse. Biochemistry 45, 7825–7833 (2006).
Ireton, G.C., Black, M.E. & Stoddard, B.L. The 1.14 Å crystal structure of yeast cytosine deaminase: evolution of nucleotide salvage enzymes and implications for genetic chemotherapy. Structure 11, 961–972 (2003).
Ko, T.P. et al. Crystal structure of yeast cytosine deaminase. Insights into enzyme mechanism and evolution. J. Biol. Chem. 278, 19111–19117 (2003).
Kabsch, W. Xds. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Sheldrick, G.M. Experimental phasing with SHELXC/D/E: combining chain tracing with density modification. Acta Crystallogr. D Biol. Crystallogr. 66, 479–485 (2010).
Vagin, A. & Teplyakov, A. Molecular replacement with MOLREP. Acta Crystallogr. D Biol. Crystallogr. 66, 22–25 (2010).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Crooks, G.E., Hon, G., Chandonia, J.M. & Brenner, S.E. WebLogo: a sequence logo generator. Genome Res. 14, 1188–1190 (2004).
Gaborek, T.J., Chipot, C. & Madura, J.D. Conformational free-energy landscapes for a peptide in saline environments. Biophys. J. 103, 2513–2520 (2012).
Narvaiza, I. et al. Deaminase-independent inhibition of parvoviruses by the APOBEC3A cytidine deaminase. PLoS Pathog. 5, e1000439 (2009).
Bulliard, Y. et al. Structure-function analyses point to a polynucleotide-accommodating groove essential for APOBEC3A restriction activities. J. Virol. 85, 1765–1776 (2011).
Acknowledgements
We thank D. Largaespada and D. Yee for insightful comments, R. Moorthy for oligonucleotide sample preparations, and J. Stivers (Pharmacology and Molecular Sciences Department, Johns Hopkins University, Baltimore, Maryland, USA) for providing the human UNG2 expression construct and purification protocol. This work was supported by grants from the US National Institutes of Health (NIGMS R01-GM118000 to R.S.H. and H.A., NIGMS R35-GM118047 to H.A., NIGMS R01-GM110129 to D.A.H., NCI R21-CA206309 to R.S.H., and DP2-OD007237 and NIGMS P41-GM103426 to R.E.A.), the NSF (CHE060073N to R.E.A.), the Prospect Creek Foundation (R.S.H. and D.A.H.), and the University of Minnesota Masonic Cancer Center (Spore-Program-Project-Planning Seed Grant to R.S.H.). This work is based upon research conducted at the Northeastern Collaborative Access Team beamlines, which are funded by the US National Institutes of Health (NIGMS P41-GM103403). The Pilatus 6M detector on the 24-ID-C beamline is funded by an NIH-ORIP HEI grant (S10 RR029205). This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. R.S.H. is supported as the Margaret Harvey Schering Land Grant Chair for Cancer Research and as an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
R.S.H. and H.A. conceived and designed the studies. K.S., N.M.S., K.K., J.V.D., and H.A. purified proteins and established crystallization conditions. S.B. collected X-ray diffraction data. K.S. solved the crystal structures. M.A.C., D.J.S., J.L.M., and G.J.S. performed the deep-deamination studies. M.A.C. performed biochemical experiments. D.A.H. designed modified DNA substrates. O.D. and R.E.A. provided computational and structural insights. K.S., M.A.C., N.M.S., R.S.H., and H.A. drafted the manuscript, and all authors contributed to revisions and figure preparation.
Corresponding authors
Ethics declarations
Competing interests
R.S.H. and D.A.H. are cofounders, shareholders, and consultants of ApoGen Biotechnologies Inc. H.A. and R.E.A. are consultants for ApoGen Biotechnologies Inc. R.E.A. is a cofounder of Actavalon Inc. The other authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Protein sequence alignment of human A3A and the C-terminal catalytic domain of human A3B.
Clustal Omega amino acid alignment of human A3A 13-199 and A3B 193-382 (catalytic domain) showing secondary structures (α-helices in red, β-strands in yellow) and loop regions. Blue boxes indicate amino acids that were altered in order to obtain soluble proteins for structural studies and, in the case of the catalytic glutamate, to prevent genotoxicity to E. coli during protein expression and substrate turnover during crystallization.
Supplementary Figure 2 Additional representations of A3A–ssDNA complexes.
(a) Overlay of the 4 distinct A3A-ssDNA complexes in the asymmetric unit of the crystal.
(b) Composite omit 2Fo-Fc map contoured at 1.0σ for the 4 complexes in the asymmetric unit (protein electron density is shown in green, and ssDNA in blue).
(c) Enlarged view of the composite omit 2Fo-Fc map contoured at 1.0σ (blue mesh) for the ssDNA oligonucleotides bound in the 4 distinct A3A molecules (differently colored) captured in the asymmetric unit of the crystal. The orange mesh (composite omit 2Fo-Fc map contoured at 8.0σ) represents the position of the single zinc atom (gray sphere shown at 0.6x scale of the van der Waals radius).
Supplementary Figure 3 Additional representations of A3Bctd*–ssDNA complexes.
(a) Composite omit 2Fo-Fc map for the single A3Bctd*-ssDNA complex contoured at 1.0σ in the asymmetric unit (electron density for protein and ssDNA is shown in green and blue, respectively).
(b) Enlarged view of the composite omit 2Fo-Fc map contoured at 1.0σ (blue mesh) for the ssDNA oligonucleotide (yellow sticks) bound to the active site of A3B (magenta). The orange mesh (composite omit 2Fo-Fc map contoured at 8.0σ) represents the position of the single zinc atom (gray sphere, shown at 0.6x scale of the van der Waals radius).
Supplementary Figure 4 Human A3A deaminates ssDNA containing –1 Super-T.
(a) Letter format of the single-stranded DNA sequence modeled in panels b and c with 5-hydroxybutynl-2'-deoxyuridine (Super T) at the -1 position relative to the target cytidine.
(b) Chemical structures of normal deoxy-thymidine and Super T differing only at the 5 position of the cytosine ring.
(c) Predicted conformation of ssDNA containing -1 Super T bound to human A3A. A semi-transparent molecular surface is shown for the protein.
(d) Raw dose response data for human A3A and ssDNA substrates with a normal T or Super T at the -1 position relative to the target cytosine (0.1 - 100 nM A3A with 100 nM A3A-E72A and no enzyme reactions shown as negative controls). The wild-type A3A data are identical to those in Supplementary Fig. 5 to facilitate cross-comparisons.
(e) A plot quantifying product accumulation for the experiment shown in panel d. These data indicate that the methyl group at the 5-position of the thymine ring is solvent exposed and unlikely to be involved in an interaction with the enzyme.
Supplementary Figure 5 Hydrogen-bonding potential of ssDNA nucleobases +1 to +3.
(a) A schematic showing a ssDNA substrate containing an optimal A3A target site (5'-ATCGGG) and a derivative substrate with 5-nitroindole bases substituted at the +1 to +3 positions.
(b) Representative endpoint data for human A3A showing catalytic activity with normal or 5-nitroindole substituted ssDNA substrates (S, substrate; P, product).
(c) Raw dose response data for human A3A and ssDNA substrates with normal GGG or XXX at the +1 to +3 positions relative to the target cytosine (0.1 - 100 nM A3A with 100 nM A3A-E72A and no enzyme reactions shown as a negative controls). The wild-type A3A data are identical to those in Supplementary Fig. 4 to facilitate cross-comparisons.
(d) A plot quantifying product accumulation for the experiment shown in panel c. A3A shows a modest 2-fold preference for normal ssDNA substrate in comparison to the 5-nitroindole substituted ssDNA substrate. The data in panels b-d combine to suggest that base stacking of the +1 to +3 nucleotides may be more relevant for the ssDNA deamination mechanism than nucleobase hydrogen-bonding with enzyme.
Supplementary Figure 6 Comparison of A3A–ssDNA and A3Gntd–poly dT structures.
(a) Ribbon schematics of A3A-ssDNA (this study) and A3Gntd-poly dT (pdb 5K83) with active site regions positioned at similar angles to facilitate comparisons.
(b) Superposition of A3A-ssDNA and A3Gntd-poly dT structures showing a lack of congruency in the binding conformations. A3A-bound ssDNA is shown in yellow.
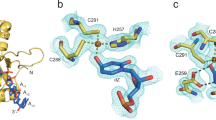
Supplementary Figure 7 APOBEC3 subfamily superposition.
(a) Superposition of ribbon schematics of crystal structures for A3A in cyan (pdb 4XXO), A3Bctd in magenta (pdb 5CQH), A3C in yellow (pdb 3VOW), A3Fctd in gray and green (pdb 5HX5 and pdb 3WUS), and A3Gctd in salmon (pdb 3V4K).
(b) A superposition of key active site amino acid residues. The zinc-coordinating residues and those that line the active site, including the Trp-Ser-Pro-Cys-X2-4-Cys motif and Thr31 (Thr214) that directly interact with the target cytosine, show high structural conservation. In contrast, Tyr130 (Tyr313) from loop 7 and Asn57 (Asn240) preceding loop 3, which both make critical ssDNA backbone contacts, can adopt more variable conformations. A3A residue numbers are indicated and those for A3B are shown in parentheses.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 1331 kb)
Supplementary Data Set 1
Raw gel images for data in Figures 3e and 5a,b (PDF 8507 kb)
Source data
Rights and permissions
About this article
Cite this article
Shi, K., Carpenter, M., Banerjee, S. et al. Structural basis for targeted DNA cytosine deamination and mutagenesis by APOBEC3A and APOBEC3B. Nat Struct Mol Biol 24, 131–139 (2017). https://doi.org/10.1038/nsmb.3344
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3344
This article is cited by
-
Engineering APOBEC3A deaminase for highly accurate and efficient base editing
Nature Chemical Biology (2024)
-
Mesoscale DNA features impact APOBEC3A and APOBEC3B deaminase activity and shape tumor mutational landscapes
Nature Communications (2024)
-
APOBEC3-mediated mutagenesis in cancer: causes, clinical significance and therapeutic potential
Journal of Hematology & Oncology (2023)
-
Re-engineering the adenine deaminase TadA-8e for efficient and specific CRISPR-based cytosine base editing
Nature Biotechnology (2023)
-
Direct enzymatic sequencing of 5-methylcytosine at single-base resolution
Nature Chemical Biology (2023)