Abstract
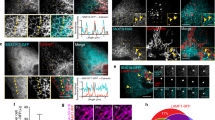
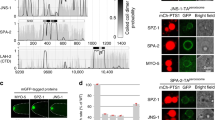
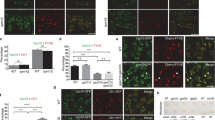
The endosomal sorting complex required for transport (ESCRT) machinery, including ESCRT-III, localizes to the midbody and participates in the membrane-abscission step of cytokinesis. The ESCRT-III protein charged multivesicular body protein 1B (CHMP1B) is required for recruitment of the MIT domain–containing protein spastin, a microtubule-severing enzyme, to the midbody. The 2.5-Å structure of the C-terminal tail of CHMP1B with the MIT domain of spastin reveals a specific, high-affinity complex involving a noncanonical binding site between the first and third helices of the MIT domain. The structural interface is twice as large as that of the MIT domain of the VPS4–CHMP complex, consistent with the high affinity of the interaction. A series of unique hydrogen-bonding interactions and close packing of small side chains discriminate against the other ten human ESCRT-III subunits. Point mutants in the CHMP1B binding site of spastin block recruitment of spastin to the midbody and impair cytokinesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Saksena, S., Sun, J., Chu, T. & Emr, S.D. ESCRTing proteins in the endocytic pathway. Trends Biochem. Sci. 32, 561–573 (2007).
Hurley, J.H. ESCRT complexes and the biogenesis of multivesicular bodies. Curr. Opin. Cell Biol. 20, 4–11 (2008).
Morita, E. & Sundquist, W.I. Retrovirus budding. Annu. Rev. Cell Dev. Biol. 20, 395–425 (2004).
Carlton, J.G. & Martin-Serrano, J. Parallels between cytokinesis and retroviral budding: a role for the ESCRT machinery. Science 316, 1908–1912 (2007).
Morita, E. et al. Human ESCRT and ALIX proteins interact with proteins of the midbody and function in cytokinesis. EMBO J. 26, 4215–4227 (2007).
Glotzer, M. The molecular requirements for cytokinesis. Science 307, 1735–1739 (2005).
Skop, A.R., Liu, H.B., Yates, J., Meyer, B.J. & Heald, R. Dissection of the mammalian midbody proteome reveals conserved cytokinesis mechanisms. Science 305, 61–66 (2004).
Gromley, A. et al. Centriolin anchoring of exocyst and SNARE complexes at the midbody is required for secretory-vesicle-mediated abscission. Cell 123, 75–87 (2005).
Pohl, C. & Jentsch, S. Final stages of cytokinesis and midbody ring formation are controlled by BRUCE. Cell 132, 832–845 (2008).
Hanson, P.I., Roth, R., Lin, Y. & Heuser, J.E. Plasma membrane deformation by circular arrays of ESCRT-III protein filaments. J. Cell Biol. 180, 389–402 (2008).
Lata, S. et al. Helical structures of ESCRT-III are disassembled by VPS4. Science 321, 1354–1357 (2008).
Dukes, J.D., Richardson, J.D., Simmons, R. & Whitley, P. A dominant-negative ESCRT-III protein perturbs cytokinesis and trafficking to lysosomes. Biochem. J. 411, 233–239 (2008).
Hurley, J.H. & Yang, D. MIT domainia. Dev. Cell 14, 6–8 (2008).
Stuchell-Brereton, M.D. et al. ESCRT-III recognition by VPS4 ATPases. Nature 449, 740–744 (2007).
Obita, T. et al. Structural basis for selective recognition of ESCRT-III by the AAA ATPase Vps4. Nature 449, 735–739 (2007).
Reid, E. et al. The hereditary spastic paraplegia protein spastin interacts with the ESCRT-III complex-associated endosomal protein CHMP1B. Hum. Mol. Genet. 14, 19–38 (2005).
Soderblom, C. & Blackstone, C. Traffic accidents: molecular genetic insights into the pathogenesis of the hereditary spastic paraplegias. Pharmacol. Ther. 109, 42–56 (2006).
Roll-Mecak, A. & Vale, R.D. Structural basis of microtubule severing by the hereditary spastic paraplegia protein spastin. Nature 451, 363–367 (2008).
Errico, A., Claudiani, P., D'Addio, M. & Rugarli, E.I. Spastin interacts with the centrosomal protein NA14, and is enriched in the spindle pole, the midbody and the distal axon. Hum. Mol. Genet. 13, 2121–2132 (2004).
Carlton, J.G., Agromayor, M. & Martin-Serrano, J. Differential requirements for Alix and ESCRT-III in cytokinesis and HIV-1 release. Proc. Natl. Acad. Sci. USA 105, 10541–10546 (2008).
Zamborlini, A. et al. Release of autoinhibition converts ESCRT-III components into potent inhibitors of HIV-1 budding. Proc. Natl. Acad. Sci. USA 103, 19140–19145 (2006).
Shim, S., Kimpler, L.A. & Hanson, P.I. Structure/function analysis of four core ESCRT-III proteins reveals common regulatory role for extreme C-terminal domain. Traffic 8, 1068–1079 (2007).
Lata, S. et al. Structural basis for autoinhibition of ESCRT-III CHMP3. J. Mol. Biol. 378, 818–825 (2008).
Xiao, J. et al. Structural basis of Vta1 function in the multi-vesicular body sorting pathway. Dev. Cell 14, 37–49 (2008).
Azmi, I.F. et al. ESCRT-III family members stimulate Vps4 ATPase activity directly or via Vta1. Dev. Cell 14, 50–61 (2008).
Shim, S., Merrill, S.A. & Hanson, P.I. Novel interactions of ESCRT-III with LIP5 and VPS4 and their implications for ESCRT-III disassembly. Mol. Biol. Cell 19, 2661–2672 (2008).
Tsang, H.T.H. et al. A systematic analysis of human CHMP protein interactions: additional MIT domain-containing proteins bind to multiple components of the human ESCRT III complex. Genomics 88, 333–346 (2006).
Agromayor, M. & Martin-Serrano, J. Interaction of AMSH with ESCRT-III and deubiquitination of endosomal cargo. J. Biol. Chem. 281, 23083–23091 (2006).
Ma, Y.M. et al. Targeting of AMSH to endosomes is required for epidermal growth factor degradation. J. Biol. Chem. 282, 9805–9812 (2007).
Row, P.E. et al. The MIT domain of UBPY constitutes a CHMP binding and endosomal localization signal required for efficient EGF receptor degradation. J. Biol. Chem. 282, 30929–30937 (2007).
Kieffer, C. et al. Two distinct modes of ESCRT-III recognition are required for VPS4 functions in lysosomal protein targeting and HIV-1 budding. Dev. Cell 15, 62–73 (2008).
Blackstone, C., Roberts, R.G., Seeburg, D.P. & Sheng, M. Interaction of the deafness-dystonia protein DDP/TIMM8a with the signal transduction adaptor molecule STAM1. Biochem. Biophys. Res. Commun. 305, 345–352 (2003).
Yu, W. et al. The microtubule-severing proteins spastin and katanin participate differently in the formation of axonal branches. Mol. Biol. Cell 19, 1485–1498 (2008).
Studier, F.W. Protein production by auto-induction in high-density shaking cultures. Protein Expr. Purif. 41, 207–234 (2005).
Terwilliger, T.C. & Berendzen, J. Automated MAD and MIR structure solution. Acta Crystallogr. D Biol. Crystallogr. 55, 849–861 (1999).
Terwilliger, T.C. Maximum-likelihood density modification. Acta Crystallogr. D Biol. Crystallogr. 56, 965–972 (2000).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Brunger, A.T. et al. Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum- likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Acknowledgements
We thank N. Elia for discussions, the staff of SER-CAT for user support at the Advance Photon Source (APS), C.-R. Chang for technical assistance, E. Tyler for generating Figure 6 and D. Davies for comments on the manuscript. Use of the APS was supported by the US Department of Energy, Basic Energy Sciences, Office of Science, under Contract No.W-31-109-Eng-38. This project was funded by the Intramural Research Programs of US National Institute of Diabetes and Digestive and Kidney Diseases, US National Institute of Neurological Disorders and Stroke and the US National Institute of Child Health and Human Development, and the Bench-to-Bedside program of the US National Institutes of Health (NIH).
Author information
Authors and Affiliations
Contributions
D.Y. carried out binding experiments, crystallization and structure determination; N.R. and B.R. carried out knockdown and cell-imaging experiments; J.L.-S. interpreted data; C.B. carried out yeast two-hybrid experiments; C.B. and J.H.H. designed research, interpreted data and wrote the paper.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Discussion. (PDF 12460 kb)
Rights and permissions
About this article
Cite this article
Yang, D., Rismanchi, N., Renvoisé, B. et al. Structural basis for midbody targeting of spastin by the ESCRT-III protein CHMP1B. Nat Struct Mol Biol 15, 1278–1286 (2008). https://doi.org/10.1038/nsmb.1512
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1512
This article is cited by
-
Cytokinetic abscission requires actin-dependent microtubule severing
Nature Communications (2024)
-
Evolutionary conserved relocation of chromatin remodeling complexes to the mitotic apparatus
BMC Biology (2022)
-
The enhanced association between mutant CHMP2B and spastin is a novel pathological link between frontotemporal dementia and hereditary spastic paraplegias
Acta Neuropathologica Communications (2022)
-
The ATPase SRCAP is associated with the mitotic apparatus, uncovering novel molecular aspects of Floating-Harbor syndrome
BMC Biology (2021)
-
The many functions of ESCRTs
Nature Reviews Molecular Cell Biology (2020)