Abstract
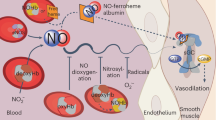
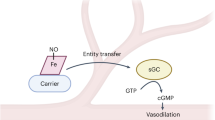
The nitrophorins comprise an unusual family of proteins that use ferric (Fe(III)) heme to transport highly reactive nitric oxide (NO) from the salivary gland of a blood sucking bug to the victim, resulting in vasodilation and reduced blood coagulation. We have determined structures of nitrophorin 4 in complexes with H2O, cyanide and nitric oxide. These structures reveal a remarkable feature: the nitrophorins have a broadly open distal pocket in the absence of NO, but upon NO binding, three or more water molecules are expelled and two loops fold into the distal pocket, resulting in the packing of hydrophobic groups around the NO molecule and increased distortion of the heme. In this way, the protein apparently forms a ‘hydrophobic trap’ for the NO molecule. The structures are very accurate, ranging between 1.6 and 1.4 Å resolutions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bredt, D.S. & Snyder, S.H. Annu. Rev. Biochem. 63, 175–195 (1994).
Law, J., Ribeiro, J.M.C. & Wells, M. Annu. Rev. Biochem. 61, 87–111 (1992).
Montfort, W.R., Weichsel, A. & Andersen, J.F. Biochim. Biophys. Acta, in the press (2000).
Kirchhoff, L.V. N. Engl. J. Med. 329, 639–644 (1993).
Sharma, V.S. & Magde, D. Methods 19, 494–505 (1999).
Ribeiro, J.M.C., Hazzard, J.M.H., Nussenzveig, R.H., Champagne, D.E. & Walker, F.A. Science 260, 539–541 (1993).
Andersen, J.F. et al. Biochemistry 36, 4423–4428 (1997).
Traylor, T.G. & Sharma, V.S. Biochemistry 31, 2847–2849 (1992).
Sharma, V.S., Isaacson, R.A., John, M.E., Waterman, M.R. & Chevion, M. Biochemsitry 22, 3897–3902 (1983).
Addison, A.W. & Stephanos, J.J. Biochemistry 25, 4104–4113 (1986).
Hoshino, M., Maeda, M., Konishi, R., Seki, H. & Ford, P.C. J. Am. Chem. Soc. 118, 5702–5707 (1996).
Andersen, J.F., Weichsel, A., Balfour, C.A., Champagne, D.E. & Montfort, W.R. Structure 6, 1315–1327 (1998).
Weichsel, A., Andersen, J.F., Champagne, D.E., Walker, F.A. & Montfort, W.R. Nature Struct. Biol. 5, 304–309 (1998).
Kaneko, Y., Yuda, M., Iio, T., Murase, T. & Chinzei, Y. Biochim. Biophys. Acta 1431, 492–499 (1999).
Sharma, V.S., Traylor, T.G., Gardiner, R. & Mizukami, H. Biochemistry 26, 3837–3843 (1987).
Ding, X.D. et al. J. Am. Chem. Soc. 121, 128–138 (1999).
Scheidt, W.R. & Ellison, M.K. Acc. Chem. Res. 32, 350–359 (1999).
Hodge, S.J. et al. Chem. Commun. 19, 2283–2284 (1996).
Edwards, S.L., Kraut, J. & Poulos, T.L. Biochemistry 27, 8074–8081 (1988).
Jewsbury, P., Yamamoto, S., Minato, T., Saito, M. & Kitagawa, T. J. Am. Chem. Soc. 116, 11586–11587 (1994).
Carducci, M.D., Pressprich, M.R. & Coppens, P. J. Am. Chem. Soc. 119, 2669–2678 (1997).
Shaw, A.W. & Vosper, A.J. J. Chem. Soc. Faraday Trans. 8, 1239–1244 (1977).
Kerwin, J.F., Jr., Lancaster, J.R., Jr. & Feldman, P.L. J. Med. Chem. 38, 4343–4362 (1995).
Walker, F.A., Emrick, D., Rivera, J.E., Hanquet, B.J. & Buttlaire, D.H. J. Am. Chem. Soc. 110, 6234–6240 (1988).
Shelnutt, J.A. et al. Chem. Soc. Rev. 27, 31–41 (1998).
Rohlfs, R.J. et al. J. Biol. Chem. 265, 3168–3176 (1990).
Messerschmidt, A. & Pflugrath, J.W. J. Appl. Crystallogr. 20, 306–315 (1987).
Kabsch, W. J. Appl. Crystallogr. 21, 916–934 (1988).
CCP4. Acta Crystallogr. D 50, 760–763 (1994).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjelgard, M. Acta Crystallogr. A 47, 110–119 (1991).
Brunger, A.T. X-PLOR: a system for X-ray crystallography and NMR (Yale University Press, New Haven, Connecticut; 1992).
Sheldrick, G.M. & Schneider, T.R. Methods Enzymol. 277, 319–343 (1997).
Andersen, J.F. et al. Biochemistry in the press (2000).
Acknowledgements
We thank C. Balfour for protein purification. Supported in part by grants from NIH, ACS, and ADCRC to W.R.M., and from the NIH to J.F.A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weichsel, A., Andersen, J., Roberts, S. et al. Nitric oxide binding to nitrophorin 4 induces complete distal pocket burial. Nat Struct Mol Biol 7, 551–554 (2000). https://doi.org/10.1038/76769
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/76769
This article is cited by
-
Architecture of the membrane-bound cytochrome c heme lyase CcmF
Nature Chemical Biology (2021)
-
NMR investigations of nitrophorin 2 belt side chain effects on heme orientation and seating of native N-terminus NP2 and NP2(D1A)
JBIC Journal of Biological Inorganic Chemistry (2014)
-
Assignment of the 1H NMR resonances of protein residues in close proximity to the heme of the nitrophorins: similarities and differences among the four proteins from the saliva of the adult blood-sucking insect Rhodnius prolixus
JBIC Journal of Biological Inorganic Chemistry (2012)
-
1H and 13C NMR spectroscopic studies of the ferriheme resonances of three low-spin complexes of wild-type nitrophorin 2 and nitrophorin 2(V24E) as a function of pH
JBIC Journal of Biological Inorganic Chemistry (2009)
-
Replacement of the axial histidine heme ligand with cysteine in nitrophorin 1: spectroscopic and crystallographic characterization
JBIC Journal of Biological Inorganic Chemistry (2009)