Abstract
The Renal Tumour Study Group of the International Society of Paediatric Oncology (SIOP–RTSG) has developed a new protocol for the diagnosis and treatment of childhood renal tumours, the UMBRELLA SIOP–RTSG 2016 (the UMBRELLA protocol), to continue international collaboration in the treatment of childhood renal tumours. This protocol will support integrated biomarker and imaging research, focussing on assessing the independent prognostic value of genomic changes within the tumour and the volume of the blastemal component that survives preoperative chemotherapy. Treatment guidelines for Wilms tumours in the UMBRELLA protocol include recommendations for localized, metastatic, and bilateral disease, for all age groups, and for relapsed disease. These recommendations have been established by a multidisciplinary panel of leading experts on renal tumours within the SIOP–RTSG. The UMBRELLA protocol should promote international collaboration and research and serve as the SIOP–RTSG best available treatment standard.
Similar content being viewed by others
Main
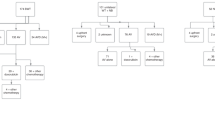
The Renal Tumour Study Group of the International Society of Paediatric Oncology (SIOP–RTSG) has developed a new protocol for diagnosis and treatment of childhood renal tumours, UMBRELLA SIOP-RTSG 2016 (referred to as the UMBRELLA protocol), to continue international collaboration in the treatment of childhood renal tumours1. The UMBRELLA protocol succeeds the SIOP−2001 protocol2. The name UMBRELLA signifies the ambitious aim to collect information concerning all paediatric primary renal tumours in a comprehensive multidimensional data registry, which includes embedded review of diagnostics, standardized biobanking, and treatment recommendations1 (Fig. 1). The UMBRELLA protocol will support integrated biomarker and imaging research, with a particular focus on assessing the independent prognostic value of genomic changes within the tumour (chromosomal gain of 1q and the extent of its intratumoral heterogeneity) and the volume of the blastemal component that survives preoperative chemotherapy3,4.
The UMBRELLA signifies the ambitious aim to collect information concerning all paediatric primary renal tumours in a comprehensive multidimensional data registry, which includes embedded review of diagnostics, standardized biobanking, and treatment recommendations. CCSK, clear cell sarcoma of the kidney, MRTK, malignant rhabdoid tumour of the kidney, CMN, congenital mesoblastic nephroma, RCC, renal cell carcinoma.
Childhood renal tumours are relatively uncommon, accounting for ∼5% of all paediatric malignancies. Of these tumours, around 80–90% are thought to be Wilms tumours, whereas other renal tumours (non-Wilms tumours), including clear cell sarcoma of the kidney, renal cell carcinoma, malignant rhabdoid tumour of the kidney, and congenital mesoblastic nephroma are even less common5. The exact incidence of non-Wilms tumours is unclear, owing to the probable under-registration of patients with these tumours in renal tumour protocols.
The UMBRELLA protocol addresses both Wilms tumours and non-Wilms tumours, and will be available on the SIOP–RTSG website (www.siop-rtsg.eu) after launch in 2017. All countries that are interested in joining the UMBRELLA protocol will be given full access to the treatment schedules, based on their commitment to register renal tumour patients. This Consensus Statement focuses on the rationale for treatment of Wilms tumours in the UMBRELLA protocol.
Treatment guidelines for Wilms tumours in the UMBRELLA protocol include recommendations for localized, metastatic (stage IV), and bilateral disease, for all age groups, and for relapsed disease. These recommendations were established by a multidisciplinary panel of leading experts on renal tumours within the SIOP–RTSG, including paediatric oncologists, radiologists, pathologists, surgeons, radiation oncologists, statisticians, and scientists involved in basic research. Thorough communications were undertaken with colleagues with similar expertise involved in the Children's Oncology Group (COG), to ensure all relevant evidence was applied when deciding how to implement the results of the SIOP−2001 randomized trial, which investigated the safety of omitting doxorubicin in treating stage II–III intermediate-risk Wilms tumours, and to refine recommendations for patients with Wilms tumour. Over the past 15 years, wide-ranging discussions on global strategies for children with renal tumours have evolved between SIOP–RTSG and COG during meetings and workshops. These conversations have resulted in sharing of data and knowledge, which has been used in the design of the current UMBRELLA guideline for diagnostics and treatment.
Treatment recommendations
In general, treatment of Wilms tumours is tailored to the patient based on tumour stage and histology, and involves a combination of chemotherapy, surgery, and, sometimes, radiotherapy. Since the first SIOP protocol started in 1971, treatment intensity has been successfully reduced for the majority of patients with Wilms tumours, and survival has risen to 90%2,6,7,8,9,10. Consequently, the identification of additional predictive and prognostic factors is increasingly important to improve the stratification of patients according to their individual risk. Approximately two-thirds of patients with Wilms tumour now receive chemotherapy consisting of only two drugs, actinomycin D and vincristine11. Other patients, including those with metastatic disease and high-risk histological subtypes, are believed to benefit from doxorubicin12,13,14,15,16. Moreover, as innovative techniques emerge, surgical and radiotherapeutic procedures are improving.
Localized disease. Similar to the SIOP−2001 protocol, the UMBRELLA protocol continues to recommend preoperative actinomycin D and vincristine for patients newly diagnosed with Wilms tumour aged ≥6 months, based on results of previous SIOP trials that showed tumour downstaging using this regimen2,6,8,9,14,17. This benefit was also independently observed in the randomized, controlled UKW3 trial conducted by the UK Children's Cancer and Leukaemia Group (UKCCLG, previously known as the UK Children's Cancer Study Group)18. In patients receiving preoperative chemotherapy, the use of radiotherapy or doxorubicin could be reduced by 20% compared with those treated with direct nephrectomy, with no significant difference in survival18. The SIOP–RTSG accounts for the risk of misdiagnosis of Wilms tumour by recommending direct surgery instead of preoperative chemotherapy for children <6 months old, and the consideration of fine-needle biopsy for patients who have unusual clinical presentations or unusual findings on imaging. To avoid treatment delay, routine histological assessment before treatment is not advocated. This approach has been shown to be safe and identifies the vast majority of patients with non-Wilms tumours who are at risk of being unnecessarily treated with preoperative chemotherapy.
Furthermore, preoperative chemotherapy enables personalized assessment of tumour chemosensitivity, including identification of the high-risk, blastemal-type Wilms tumours. The centralized review process of histology, undertaken in the SIOP–RTSG, has shown that identification of the blastemal subtype is feasible and clinically relevant. Yet, the definition of blastemal-type histology might be improved by considering the absolute residual volume of blastema rather than the relative percentage, as will be investigated in the UMBRELLA protocol19.
Patients registered in the UMBRELLA protocol will continue to be stratified for postoperative treatment according to tumour stage and histological risk group, as was the protocol in SIOP−2001 (Table 1). Prospective data from patients who are stratified and treated based on standardized recommendations will be collected and analysed. This data collection, in combination with the results of planned integrated biomarker and imaging studies (which will assess the relative contribution of gain of 1q and assessment of residual blastemal volume), might be used to guide stratification in future protocols.
The therapeutic regimen of the experimental arm of the SIOP−2001 trial has been adopted as the new standard management regimen for most patients in the UMBRELLA protocol with stage II−III intermediate-risk Wilms tumours. This regimen consists of 27 weeks of vincristine and actinomycin D without doxorubicin. This schedule resulted in a nonsignificant small decrease in event-free survival (EFS) and had no effect on overall survival compared with 27 weeks of vincristine and actinomycin D plus five doses of doxorubicin at 50 mg/m2 (the standard arm) in the SIOP-2001 trial2.
Post hoc analysis of data from SIOP−2001 was carried out to examine the association between omitting doxorubicin and the outcomes of patients with large-volume tumours (defined as tumours with a volume >500 ml after preoperative chemotherapy). Stromal and epithelial tumour types, which have excellent prognosis20, were excluded from this analysis, leaving only stage II–III regressive, mixed, and focal anaplasia-type tumours (n = 429). In Kaplan Meier analysis, patients with large-volume tumours had an estimated 5-year EFS of 80% versus 90% for patients with small-volume tumours (log rank P = 0.01) (Fig. 2). Most importantly, EFS was significantly improved (93% versus 67%, log rank P = 0.0005) when doxorubicin was added to the treatment regimen for large-volume (≥500 ml) tumours (Fig. 3). Thus, the inclusion of doxorubicin in postoperative treatment of patients with large-volume (≥500 ml) stage II–III nonstromal, nonepithelial tumours is recommended in the UMBRELLA protocol.
Association of tumour volume with a | event-free survival (EFS) and b | overall survival (OS). Kaplan Meier curves.
Association of tumour volume and treatment with a | event-free survival (EFS) and b | overall survival (OS). Kaplan Meier curves. AV-2, actinomycin D, vincristine (27 weeks); AVD, actinomycin D, vincristine and doxorubicin.
Furthermore, the UMBRELLA protocol will continue treatment for blastemal-type tumours according to the regimen used in SIOP−2001. A comparison of the results of the SIOP−2001 and SIOP−93–01 trials showed that in SIOP−2001, in which treatment was intensified by changing to the high-risk tumour treatment schedule for patients with blastemal-type Wilms tumour, EFS increased from 67% to 80% (log rank P = 0.006) avoiding intensive treatment for relapse in a considerable number of patients12.
Metastatic disease (stage IV). Overall, ∼17% of patients with Wilms tumours present with stage IV disease at diagnosis, which is defined as haematogenous metastases to the lungs, liver, or other sites, or extra-abdominal lymph node metastases. Pulmonary metastases are by far the most frequently observed21,22,23. The increasing use of chest CT as routine imaging for staging has resulted in the detection of small pulmonary nodules not visible on chest radiography (CT-only nodules). Similar to the COG protocol, CT-only nodules are included in the definition of lung nodules and treated as metastases in the UMBRELLA protocol if they have a transverse diameter of at least 3 mm (Refs 22,24,25). Presence of these CT-only nodules was associated with increased relapse risk and reduced survival in a SIOP–RTSG analysis comparing the outcomes of patients with CT-only lung lesions with those with true localized disease24. Results from the COG National Wilms Tumor Study Group (NWTS)-4 and NWTS-5 trials showed that patients with CT-only nodules who were treated with vincristine and actinomycin D plus doxorubicin had superior EFS to those who received vincristine and actinomycin D only, but overall survival was similar in both groups25. Including CT-only nodules in the definition of metastatic disease will benefit patients with intermediate-risk or low-risk histology who achieve a rapid complete response of their CT-only nodules. These patients do not need pulmonary radiotherapy and have, therefore, a reduced risk of severe long-term sequelae such as lung disease, cardiac complications or secondary malignancies.
Similar to SIOP−2001, preoperative treatment for metastatic (stage IV) disease in the UMBRELLA protocol includes a combined vincristine, actinomycin D, and doxorubicin regimen for 6 weeks, followed by reassessment imaging of local tumour (using MRI) and metastatic sites (using CT and/or MRI) before surgery. With this preoperative regimen, 61–67% of patients have complete metastatic response before surgery21,23. Detailed guidelines are provided for the stratification of postoperative chemotherapy, in which the cumulative dose of doxorubicin has been lowered in order to reduce cardiac toxicity. The cumulative doxorubicin dose for patients with metastatic disease was 300 mg/m2 in SIOP−2001, preliminary data from the COG AREN0533 trial suggest that using a cumulative doxorubicin dose of 150 mg/m2 for patients with favourable histology does not considerably affect survival25,26. For this reason, the UMBRELLA protocol recommends stratifying patients to either vincristine and actinomycin D plus doxorubicin with a cumulative doxorubicin dose of 150 mg/m2, vincristine, and actinomycin D plus doxorubicin with cumulative doxorubicin of 250 mg/m2, or a four-drug regimen including etoposide (150 mg/m2/day), carboplatin (200 mg/m2/day), cyclophosphamide (450 mg/m2/day), and doxorubicin (cumulative dose 300 mg/m2). Stratification is based on local stage of the primary tumour, histology of the primary tumour and the metastatic tumour (if resected), the size of metastatic lesions, and their response to preoperative treatment and surgery (Table 2).
Notably, patients with metastatic disease and high-risk characteristics on histological examination are a rare subgroup, with recognized unfavourable prognosis. Only a few patients per year will be stratified into this category. Thus, UMBRELLA protocol advises that local centres discuss the best current treatment approach with the principle investigator for stage IV disease. Currently, the SIOP–RTSG board suggests a regimen based on unpublished but presented data from the COG27, including combinations of vincristine, irinotecan, cyclophosphamide, carboplatin, etoposide, and doxorubicin, followed by high-dose chemotherapy and autologous stem cell transplantation at the discretion of the treating physician. The role of upfront high-dose chemotherapy for this subgroup is under debate, but a trend towards favourable outcomes has been reported by several groups in the primary and relapsed settings28,29,30. Details of this suggested regimen were added as an appendix to the UMBRELLA protocol. Data on the use of this regimen and outcomes will be prospectively captured in the SIOP database and can, therefore, be evaluated in a descriptive study.
Bilateral disease (stage V). Synchronous bilateral Wilms tumour (stage V) accounts for ∼5–8% of instances of Wilms tumour and long-term overall survival is currently ∼80%31,32,33,34,35. End-stage renal disease (ESRD) is the most clinically significant morbidity for patients with bilateral Wilms tumours and can be caused by underlying germline genetic aberrations as well as treatment-related loss of functional renal tissue. Aronson et al.35 observed that functional renal outcome was considerably better after bilateral nephron sparing surgery (NSS) than when other types of surgery were used35. Independently of the type of treatment, children with Wilms tumour, aniridia, genitourinary anomalies, and retardation (WAGR), Denys-Drash or other syndromes associated with WT1 mutations, are at increased risk of ESRD36. Thus, avoiding total nephrectomy at initial surgery is advised for bilateral tumours in the UMBRELLA protocol35. However, other important causes of ESRD exist, including tumour recurrence requiring bilateral nephrectomy or renal irradiation. Long-term monitoring of renal function is required after treatment of bilateral disease. In the SIOP−2001 study, patients with bilateral disease received preoperative chemotherapy including vincristine and actinomycin D until NSS was deemed feasible, with response evaluations performed every 4 weeks. However, several studies have shown that prolonged preoperative chemotherapy is often ineffective (especially as many bilateral tumours are the chemotherapy-insensitive stromal subtype) and could even result in an increased risk of the presence of anaplasia, disease progression, and development of metastases31,32,34. Thus, the UMBRELLA protocol limits preoperative chemotherapy to a maximum of 12 weeks, with time intervals for evaluation fixed to 6 weeks, to be comparable with the COG approach for future studies. The occurrence of misdiagnosis, in which synchronous bilateral renal tumours other than Wilms tumours are present, is, from experience, extremely rare. In instances of tumour nonresponsiveness or inoperability switching to treatment with etoposide and carboplatin is recommended, to avoid use of anthracyclines, and biopsy can be considered to determine histology.
Relapsed Wilms tumour. The UMBRELLA protocol provides structured guidelines for the treatment of patients with relapsed Wilms tumours. In retrospective studies, the best prognostic factors were initial histology and the first-line treatment used37,38,39. Thus, patients with relapsed tumours will be prospectively classified into three groups in the UMBRELLA protocol, group AA, group BB, and group CC, based on these factors.
Treatment of group AA relapsed Wilms tumours, defined as patients with initial stage I−II low-risk or intermediate-risk tumours, who received only vincristine and/or actinomycin D (no radiotherapy) in their first-line treatment, will include four drugs (combinations of doxorubicin and/or cyclophosphamide and carboplatin and/or etoposide). The combination of these drugs has already been tested in two comprehensive studies, the UKW-R protocol and the NWTS-5 relapse protocol, but drug combinations and doses varied40,41.
Patients without initial diffuse anaplasia or blastemal-type histology, who have already received doxorubicin in their initial treatment, will be classified as group BB and receive an intensive reinduction drug regimen (including the combination of etoposide and carboplatin with either ifosfamide or cyclophosphamide), followed by either high-dose melphalan and autologous stem cell rescue (ASCR) or two further reinduction courses, at the discretion of the local physician28,37. Acceptable response rates have been observed with both cyclophosphamide and etoposide and carboplatin and etoposide combinations, but ifosfamide showed an increased response rate in early-phase trials42. In an effort to reduce the risk of ifosfamide-related nephrotoxicity, cyclophosphamide will be alternated with ifosfamide in the group BB protocol recommendations. The heterogeneous settings in which the role of high-dose chemotherapy and ASCR has been explored and the inconclusive results reported led us to propose a flexible approach to the consolidation phase, and high-dose chemotherapy is at the discretion of the treating physician, aiming to describe the results in a prospective observational fashion28.
Relapsed group CC includes patients with initial diffuse anaplasia or blastemal-type tumours. For patients in this category, and for the other relapsing patients showing no response to salvage treatment, the UMBRELLA protocol advises trying camptothecins (irinotecan or topotecan) or novel compounds, as these patients will have already received most conventional active agents in their first-line therapy and are likely to develop chemotherapy-resistant disease43. In the UMBRELLA protocol, the SIOP–RTSG commits to endorse initiatives dedicated to new drug development in children, such as those launched by the Innovative Therapies for Children with Cancer consortium.
Importantly, approaches to local treatment (including radiotherapy and surgical excision of relapsing tumour masses) have not been systematically explored, so the UMBRELLA protocol provides structured guidelines for administering radiotherapy and surgery at relapse. These guidelines include considering resection after proven reduction of relapsed disease after chemotherapy, independently of histological subtype or risk group, when radical surgery seems possible or when it is useful to evaluate histological tumour response. Applying radiotherapy to initially nonirradiated sites is uniformly accepted, but developing standard recommendations for the approach to previously irradiated sites is difficult, because of the many different situations encountered. For these instances, the UMBRELLA protocol recommends contacting radiotherapists on the SIOP−RTSG panel.
Infant Wilms tumours. Infants, defined as patients younger than 6 months (182 days), should be considered for primary surgery according to the UMBRELLA protocol, unless tumours are judged not amenable to immediate nephrectomy in a multidisciplinary team consensus. The reason for upfront nephrectomy is that, compared with older children, a higher proportion of renal tumours in infants are congenital mesoblastic nephroma or malignant rhabdoid tumours that either need surgery alone (congenital mesoblastic nephroma) or alternative chemotherapy at the outset (more intensive chemotherapy than actinomycin D and vincristine)44,45. Percutaneous cutting needle biopsy is recommended in instances of stage IV disease or when immediate surgery is deemed difficult. Postoperative chemotherapy for Wilms tumour is similar in infants to that in older children who underwent direct nephrectomy, with adjustment of drug doses according to age and body weight based on the experience from previous SIOP studies44.
Adult Wilms tumours. The UMBRELLA protocol registers and provides comprehensive guidelines for the management of adults with Wilms tumours, recognizing the long treatment delays and associated poor outcomes experienced by adult patients in the past46,47,48,49. The diagnosis of Wilms tumour in adults is exceptional and treatment recommendations are based on literature review and broad international and multidisciplinary consensus, as published in 2011 (Ref. 46). Adult Wilms tumour is often diagnosed unexpectedly after nephrectomy for a suspected renal cell carcinoma. In rare instances in which the diagnosis of Wilms tumour is histologically proven before surgery, preoperative chemotherapy is recommended, similar to treatment strategies for Wilms tumour diagnosed in childhood. In contrast to the histological classification of childhood Wilms tumours, both focal and diffuse anaplasia are considered high-risk subtypes in adults, as no evidence exists that patients with focal anaplasia have better outcomes50.
Adult patients often experience considerable delays before starting postoperative chemotherapy, owing to the time needed to verify histology46,47,48,49. For this reason, postoperative treatment recommendations for children cannot simply be applied to adult patients. For instance, the treatment regimen for paediatric stage I disease of actinomycin D and vincristine is only advised for a selected group of adult stage I patients without anaplasia. All other adult patients will receive more intensive treatment, either consisting of vincristine and actinomycin D plus doxorubicin for patients with nonanaplastic subtypes, or four drugs (carboplatin, cyclophosphamide, etoposide, and doxorubicin) for anaplastic tumours of any stage. Exceptions can be made in individual instances, in which diagnosis of a stage II Wilms tumour with favourable histology is timely and postoperative chemotherapy can start within 14 days after surgery. Notably, vincristine dose intensity is decreased in guidelines for treatment of adult Wilms tumours compared with standard guidelines for children, as adults more frequently develop severe neurological toxicities48.
Surgical recommendations
After preoperative chemotherapy, radical tumour nephrectomy is the standard of care for children with Wilms tumour. The UMBRELLA protocol specifies surgical guidelines and emphasizes the importance of lymph node sampling, stating that the aim should be to sample seven locoregional lymph nodes, for the purpose of accurate staging51,52,53.
NSS is now acceptable for nonsyndromic unilateral Wilms tumours under certain conditions, specified in the UMBRELLA protocol, that include small tumour volume (<300 ml) and the expectation of a substantial remnant kidney function in patients with tumours <300 ml who never had lymph node involvement52. A new classification system for NSS, developed by a group of surgeons and pathologists from SIOP–RTSG, was adopted in the UMBRELLA protocol to optimize comparison of patient outcomes54 (Table 3). For bilateral Wilms tumours, discussion with the SIOP−RTSG surgical panel is strongly recommended, in order to assess the feasibility of NSS and minimize the risk of upstaging by incomplete resection of the tumour. Where possible, surgery should be performed in identified reference centres. Issues related to minimally invasive or laparoscopic surgery are addressed in the UMBRELLA protocol, and although not advocated, owing to lack of evidence supporting its safety, these techniques will be acceptable in selected circumstances, including small, central tumours with a rim of nonmalignant renal tissue, which still enable lymph node sampling. Minimally invasive or laparoscopic surgery should not be done in patients in whom NSS can be safely performed.
Radiotherapy recommendations
In SIOP−2001, around 25% of children with Wilms tumours underwent radiotherapy to the flank and/or metastatic sites. For the UMBRELLA protocol, the radiotherapy guidelines used in SIOP−2001 were refined based on the experience from a 2017 SIOP−2001 analysis and prior COG–NWTS trials2,8,14,55 (Table 4,5). The boost dose to the area of lymph node involvement for stage III intermediate-risk tumours is omitted in the UMBRELLA protocol, based on a 2017 analysis of SIOP−2001 data in which no locoregional control or survival benefit was observed (Davila Fajardo et al. unpublished data, manuscript submitted). Moreover, the dosage of whole-lung irradiation was decreased from 15 Gy to 12 Gy in the UMBRELLA protocol, to be in line with previous NWTS experience demonstrating high relapse-free and overall survival (72% and 78% respectively) for favourable-histology tumours after treatment with doxorubicin, actinomycin-D, vincristine, and 12 Gy to the lungs13.
Whole-abdominal radiotherapy is indicated for intermediate-risk or high-risk histology tumours with major (visible on imaging or during surgery) preoperative or intraoperative tumour rupture, or macroscopic peritoneal deposits.
Pulmonary radiotherapy is administered for lung metastases lacking complete response by postoperative week 10. Evidence suggests that the majority of patients achieving a complete response after induction chemotherapy with or without surgery do not need radiotherapy to the lungs, as they have excellent survival even without radiotherapy (5-year EFS 84%, 5-year OS 92%)21. Patients with viable metastases at surgery or high-risk histology, both of which are associated with poor survival of <40%, are the exception and receive radiotherapy to the lungs23. Given the inferior outcome with second-line treatment for patients with disease recurrence in the lung, whole-lung irradiation is recommended for patients who did not receive lung irradiation during first-line treatment, irrespective of histology56.
Radiotherapy recommendations are similar for adults and children with Wilms tumours, with the exception of stage II disease. In adult protocols, unlike paediatric protocols, radiotherapy is indicated for all stage II Wilms tumours, as lymph node sampling is often not performed46. Only for adult patients that are enrolled in UMBRELLA in time to confirm negative lymph nodes and intermediate-risk histology can the avoidance of radiotherapy be discussed.
The UMBRELLA protocol also provides a detailed description of the radiotherapy target volumes so that advanced radiotherapy techniques can be applied if they are available. The potential role of proton therapy for flank irradiation in treating Wilms tumours has only been suggested in a dosimetric study, and needs further investigation before implementation in the UMBRELLA protocol57.
International collaboration
The UMBRELLA protocol will guide treatment of Wilms tumour treatment in over 50 countries in Europe and beyond, making it the largest collaborative SIOP renal tumour protocol published to date, enabling international research to be conducted. In Europe alone, about 1,000 instances of paediatric renal tumours are diagnosed each year. In general, survival is excellent, but the SIOP–RTSG aims to address the current geographic inequalities in childhood cancer survival by providing a standardized approach to diagnosis, risk stratification, and treatment. Furthermore, estimates suggest that ∼300 instances of complex renal tumours in Europe would benefit from multidisciplinary discussion of treatment with clinicians at centres of expertise5. Examples include most instances of bilateral Wilms tumours, patients with extensive intravascular tumour thrombus or complicated metastatic sites, and advanced diffuse anaplastic Wilms tumours. Patients with these diseases could benefit from international collaboration to access specialized surgical techniques, cardiothoracic expertise, innovative radiotherapy options, and guidance for phase I/II trials. European initiatives like the European Expert Paediatric Oncology Reference Network for Diagnostics and Treatment (ExPO-r-Net) pilot (http://www.expornet.eu/) aim to enhance such collaboration. The EXPO-R-Net is an online consultation platform for which national reference centres have been identified (Fig. 4), and this platform should contribute to the establishment of international tumour boards, funding for coordinators, IT platforms and logistics, and future outreach to low-income countries.
Conclusions
As well as providing a useful guideline for routine clinical practice, the UMBRELLA protocol should stimulate international collaboration and research. By standardizing the treatment for all Wilms tumour types, prospectively collected data from a large, homogenous cohort of patients will be available for future validation of biomarkers, treatment stratification, and therapeutic targets. Moreover, the UMBRELLA protocol can serve as the SIOP–RTSG best-available treatment standard. It will be the backbone for new treatment approaches in future phase I/II and randomized trials, in which collaboration with the COG will continue to be sought. Global collaboration is still necessary for finding effective treatments for the most unfavourable Wilms tumours, such as refractory metastatic, bilateral, and relapsed high-risk disease, and the UMBRELLA protocol will contribute to this aim.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
SIOP Renal Tumour Study Group. Paediatric renal tumours: perspectives from the SIOP-RTSG. Nat. Rev. Urol. 14, 3–4 (2017).
Pritchard-Jones, K. et al. Omission of doxorubicin from the treatment of stage II-III, intermediate-risk Wilms' tumour (SIOP WT 2001): an open-label, non-inferiority, randomised controlled trial. Lancet 386, 1156–1164 (2015).
Chagtai, T. et al. Gain of 1q as a prognostic biomarker in Wilms tumors (WTs) treated with preoperative chemotherapy in the International Society of Paediatric Oncology (SIOP) WT 2001 trial: a SIOP Renal Tumours Biology Consortium study. J. Clin. Oncol. 34, 3195–3203 (2016).
Cresswell, G. D. et al. Intra-tumor genetic heterogeneity in Wilms tumor: clonal evolution and clinical implications. EBioMedicine 9, 120–129 (2016).
Pastore, G. et al. Malignant renal tumours incidence and survival in European children (1978–1997): report from the Automated Childhood Cancer Information System project. Eur. J. Cancer 42, 2103–2114 (2006).
Weirich, A. et al. Survival in nephroblastoma treated according to the trial and study SIOP-9/GPOH with respect to relapse and morbidity. Ann. Oncol. 15, 808–820 (2004).
Pritchard-Jones, K. et al. Treatment and outcome of Wilms' tumour patients: an analysis of all cases registered in the UKW3 trial. Ann. Oncol. 23, 2457–2463 (2012).
de Kraker, J. et al. Reduction of postoperative chemotherapy in children with stage I intermediate-risk and anaplastic Wilms' tumour (SIOP 93–01 trial): a randomised controlled trial. Lancet 364, 1229–1235 (2004).
Graf, N. et al. Characteristics and outcome of stage II and III non-anaplastic Wilms' tumour treated according to the SIOP trial and study 93–01. Eur. J. Cancer 48, 3240–3248 (2012).
Green, D. M. et al. Outcome of patients with Stage II/favorable histology Wilms tumor with and without local tumor spill: a report from the National Wilms Tumor Study Group. Pediatr. Blood Cancer 61, 134–139 (2014).
Spreafico, F. & Bellani, F. F. Wilms' tumor: past, present and (possibly) future. Expert Rev. Anticancer Ther. 6, 249–258 (2006).
van den Heuvel-Eibrink, M. M. et al. Outcome of localised blastemal-type Wilms tumour patients treated according to intensified treatment in the SIOP WT 2001 protocol, a report of the SIOP Renal Tumour Study Group (SIOP-RTSG). Eur. J. Cancer 51, 498–506 (2015).
D'Angio, G. J. et al. Treatment of Wilms' tumor. Results of the Third National Wilms' Tumor Study. Cancer 64, 349–360 (1989).
Tournade, M. F. et al. Optimal duration of preoperative therapy in unilateral and nonmetastatic Wilms' tumor in children older than 6 months: results of the Ninth International Society of Pediatric Oncology Wilms' Tumor Trial and Study. J. Clin. Oncol. 19, 488–500 (2001).
Breslow, N. E. et al. Doxorubicin for favorable histology, stage II-III Wilms tumor: results from the National Wilms Tumor Studies. Cancer 101, 1072–1080 (2004).
D'Angio, G. J. et al. The treatment of Wilms' tumor: results of the Second National Wilms' Tumor Study. Cancer 47, 2302–2311 (1981).
Tournade, M. F. et al. Results of the Sixth International Society of Pediatric Oncology Wilms' Tumor Trial and Study: a risk-adapted therapeutic approach in Wilms' tumor. J. Clin. Oncol. 11, 1014–1023 (1993).
Mitchell, C. et al. Immediate nephrectomy versus preoperative chemotherapy in the management of non-metastatic Wilms' tumour: results of a randomised trial (UKW3) by the UK Children's Cancer Study Group. Eur. J. Cancer 42, 2554–2562 (2006).
Graf, N. et al. Is the absolute blastema volume after preoperative chemotherapy in nephroblastoma relevant for prognosis? [abstract]. Pediatr. Blood Cancer 57, 741–742 (2011).
Verschuur, A. et al. Stromal and epithelial predominant Wilms tumours have an excellent outcome: the SIOP 93 01 experience. Pediatr. Blood Cancer 55, 233–238 (2010).
Verschuur, A. et al. Treatment of pulmonary metastases in children with stage IV nephroblastoma with risk-based use of pulmonary radiotherapy. J. Clin. Oncol. 30, 3533–3539 (2012).
Grundy, P. E. et al. Clinical significance of pulmonary nodules detected by CT and Not CXR in patients treated for favorable histology Wilms tumor on national Wilms tumor studies-4 and -5: a report from the Children's Oncology Group. Pediatr. Blood Cancer 59, 631–635 (2012).
Warmann, S. W. et al. Tumor biology influences the prognosis of nephroblastoma patients with primary pulmonary metastases: results from SIOP 93-01/GPOH and SIOP 2001/GPOH. Ann. Surg. 254, 155–162 (2011).
Smets, A. M. et al. The contribution of chest CT-scan at diagnosis in children with unilateral Wilms' tumour. Results of the SIOP 2001 study. Eur. J. Cancer 48, 1060–1065 (2012).
Dome, J. S. et al. Advances in Wilms tumor treatment and biology: progress through international collaboration. J. Clin. Oncol. 33, 2999–3007 (2015).
Dix, D. B. et al. Treatment of stage IV favorable histology Wilms tumor with incomplete lung metastasis response after chemotherapy: a report from Children's Oncology Group study AREN0533 [abstract]. J. Clin. Oncol. 32, 10001 (2014).
Daw, N. C. et al. A phase 2 study of vincristine and irinotecan in metastatic diffuse anaplastic Wilms tumor: results from the Children's Oncology Group AREN0321 study [abstract]. J Clin. Oncol. 32, 10032 (2014).
Ha, T. C. et al. An international strategy to determine the role of high dose therapy in recurrent Wilms' tumour. Eur. J. Cancer 49, 194–210 (2013).
Kremens, B. et al. High-dose chemotherapy with autologous stem cell rescue in children with nephroblastoma. Bone Marrow Transplant. 30, 893–898 (2002).
Furtwangler, R. et al. Update on relapses in unilateral nephroblastoma registered in 3 consecutive SIOP/GPOH studies — a report from the GPOH-nephroblastoma study group. Klin. Padiatr. 223, 113–119 (2011).
Hamilton, T. E. et al. The management of synchronous bilateral Wilms tumor: a report from the National Wilms Tumor Study Group. Ann. Surg. 253, 1004–1010 (2011).
Sudour, H. et al. Bilateral Wilms tumors (WT) treated with the SIOP 93 protocol in France: epidemiological survey and patient outcome. Pediatr. Blood Cancer 59, 57–61 (2012).
Indolfi, P. et al. Synchronous bilateral Wilms tumor: a report from the Associazione Italiana Ematologia Oncologia Pediatrica (AIEOP). Cancer 119, 1586–1592 (2013).
Furtwängler, R. et al. Pretreatment for bilateral nephroblastomatosis is an independent risk factor for progressive disease in patients with stage V nephroblastoma. Klin. Padiatr. 226, 175–181 (2014).
Aronson, D. C., Slaar, A., Heinen, R. C., de Kraker, J. & Heij, H. A. Long-term outcome of bilateral Wilms tumors (BWT). Pediatr. Blood Cancer 56, 1110–1113 (2011).
Breslow, N. E. et al. End stage renal disease in patients with Wilms tumor: results from the National Wilms Tumor Study Group and the United States Renal Data System. J. Urol. 174, 1972–1975 (2005).
Spreafico, F. et al. Treatment of relapsed Wilms tumors: lessons learned. Expert Rev. Anticancer Ther. 9, 1807–1815 (2009).
Grundy, P. et al. Prognostic factors for children with recurrent Wilms' tumor: results from the Second and Third National Wilms' Tumor Study. J. Clin. Oncol. 7, 638–647 (1989).
Dome, J. S. et al. Improved survival for patients with recurrent Wilms tumor: the experience at St. Jude Children's Research Hospital. J. Pediatr. Hematol. Oncol. 24, 192–198 (2002).
Hale, J., Hobson, R., Moroz, V. & Sartori, P. Results of UK Children's Cancer and Leukemia Group (CCLG) protocol for relapsed Wilms tumor (UKWR): unified relapse strategy improves outcome. Proceeding of the 40th meeting of International Society of Paediatric Oncology 62, (2008).
Green, D. M. et al. Treatment of Wilms tumor relapsing after initial treatment with vincristine and actinomycin D: a report from the National Wilms Tumor Study Group. Pediatr. Blood Cancer 48, 493–499 (2007).
Tournade, M. F. et al. Ifosfamide is an active drug in Wilms' tumor: a phase II study conducted by the French Society of Pediatric Oncology. J. Clin. Oncol. 6, 793–796 (1988).
Metzger, M. L. et al. Topotecan is active against Wilms' tumor: results of a multi-institutional phase II study. J. Clin. Oncol. 25, 3130–3136 (2007).
van den Heuvel-Eibrink, M. M. et al. Characteristics and survival of 750 children diagnosed with a renal tumor in the first seven months of life: a collaborative study by the SIOP/GPOH/SFOP, NWTSG, and UKCCSG Wilms tumor study groups. Pediatr. Blood Cancer 50, 1130–1134 (2008).
Brok, J., Treger, T. D., Gooskens, S. L., van den Heuvel-Eibrink, M. M. & Pritchard-Jones, K. Biology and treatment of renal tumours in childhood. Eur. J. Cancer 68, 179–195 (2016).
Segers, H. et al. Management of adults with Wilms' tumor: recommendations based on international consensus. Expert Rev. Anticancer Ther. 11, 1105–1113 (2011).
Terenziani, M. et al. Adult Wilms' tumor: a monoinstitutional experience and a review of the literature. Cancer 101, 289–293 (2004).
Reinhard, H. et al. Wilms' tumor in adults: results of the Society of Pediatric Oncology (SIOP) 93-01/Society for Pediatric Oncology and Hematology (GPOH) study. J. Clin. Oncol. 22, 4500–4506 (2004).
Kalapurakal, J. A. et al. Treatment outcomes in adults with favorable histologic type Wilms tumor — an update from the National Wilms Tumor Study Group. Int. J. Radiat. Oncol. Biol. Phys. 60, 1379–1384 (2004).
Faria, P. et al. Focal versus diffuse anaplasia in Wilms tumor — new definitions with prognostic significance: a report from the National Wilms Tumor Study Group. Am. J. Surg. Pathol. 20, 909–920 (1996).
Shamberger, R. C. et al. Surgery-related factors and local recurrence of Wilms tumor in National Wilms Tumor Study 4. Ann. Surg. 229, 292–297 (1999).
Godzinski, J. et al. Nephroblastoma: does the decrease in tumor volume under preoperative chemotherapy predict the lymph nodes status at surgery? Pediatr. Blood Cancer 57, 1266–1269 (2011).
Kieran, K. et al. Lymph node involvement in Wilms tumor: results from National Wilms Tumor Studies 4 and 5. J. Pediatr. Surg. 47, 700–706 (2012).
Godzinski, J., Graf, N. & Audry, G. Current concepts in surgery for Wilms tumor — the risk and function-adapted strategy. Eur. J. Pediatr. Surg. 24, 457–460 (2014).
Burgers, J. M. et al. Abdominal recurrences in Wilms' tumours: a report from the SIOP Wilms' tumour trials and studies. Radiother. Oncol. 5, 175–182 (1986).
Malogolowkin, M. et al. Treatment of Wilms tumor relapsing after initial treatment with vincristine, actinomycin D, and doxorubicin. A report From Natl Wilms Tumor Study Group. Pediatr. Blood Cancer 50, 236–241 (2008).
Vogel, J. et al. Pencil beam scanning proton therapy for treatment of the retroperitoneum after nephrectomy for Wilms tumor: a dosimetric comparison study. Pediatr. Blood Cancer 64, 39–45 (2017).
Acknowledgements
The SIOP–RTSG wishes to acknowledge the outstanding contribution of Prof. Dr Ivo Leuschner, a SIOP Pathology Panel member and cochair who was dedicated to improving the outcomes of children with cancer. In this respect, he served as a reference pathologist for many countries. His sudden death is a great loss for us and our society. We will miss him as a friend and colleague and we will keep him in our lasting memories. We further acknowledge all members of the SIOP-RTSG for their dedicated contribution to development of the UMBRELLA protocol.
Author information
Authors and Affiliations
Consortia
Contributions
J.A.H. and H.v.T. researched data for the article. M.M.v.d.H.-E. and J.A.H. wrote the manuscript. M.M.v.d.H.-E., J.A.H., K.P.-J., H.v.T., R.F., A.C.V., G.M.V., I.L. J.B., C.R., A.M.S., G.O.J., J.G., B.d.C., H.S., P.C., M.G., C.B., F.S. and N.G. made substantial contributions to discussions of content. M.M.v.d.H.-E., K.P.-J., R.F., A.C.V., G.M.V., I.L. J.B., C.R., A.M.S., G.O.J., J.G., G.L.R.-V., B.d.C., H.S., P.C., M.G., C.B., F.S. and N.G. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van den Heuvel-Eibrink, M., Hol, J., Pritchard-Jones, K. et al. Rationale for the treatment of Wilms tumour in the UMBRELLA SIOP–RTSG 2016 protocol. Nat Rev Urol 14, 743–752 (2017). https://doi.org/10.1038/nrurol.2017.163
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2017.163
This article is cited by
-
Automated segmentation and volume prediction in pediatric Wilms’ tumor CT using nnu-net
BMC Pediatrics (2024)
-
A comprehensive overview of liquid biopsy applications in pediatric solid tumors
npj Precision Oncology (2024)
-
Nierentumoren bei Kindern und Jugendlichen
Die Radiologie (2024)
-
Direct correlation of MR-DWI and histopathology of Wilms’ tumours through a patient-specific 3D-printed cutting guide
European Radiology (2024)
-
The rationale for nephron-sparing surgery in unilateral non-syndromic Wilms tumour
Pediatric Nephrology (2024)