Abstract
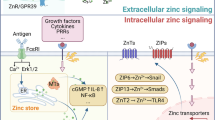
The healthy human prostate accumulates the highest level of zinc of any soft tissue in the body. This unique property is retained in BPH, but is lost in prostatic malignancy, which implicates changes in zinc and its transporters in carcinogenesis. Indeed, zinc concentrations diminish early in the course of prostate carcinogenesis, preceding histopathological changes, and continue to decline during progression toward castration-resistant disease. Numerous studies suggest that increased zinc intake might protect against progression of prostatic malignancy. In spite of increased dietary intake, zinc accumulation might be limited by the diminished expression of zinc uptake transporters, resulting in decreased intratumoural zinc levels. This finding can explain the conflicting results of various epidemiological studies evaluating the role of zinc supplementation on primary and secondary prostate cancer prevention. Overall, more research into the mechanisms of zinc homeostasis are needed to fully understand its impact on prostate carcinogenesis. Only then can the potential of zinc and zinc transport proteins be harnessed in the diagnosis and treatment of men with prostate cancer.
Key Points
-
In vitro and in vivo experimental studies have revealed a strong association between prostate cancer and the downregulation of intracellular zinc content
-
To escape the antitumour effects of intracellular zinc, malignant prostate cells can reduce expression of zinc uptake transporters through multiple mechanisms
-
Failure to accumulate zinc via the diminished expression of zinc uptake transporters can, in turn, result in decreased intratumoural zinc levels despite adequate dietary zinc intake
-
This finding can explain the conflicting results of various epidemiological studies evaluating the role of zinc supplementation on primary and secondary prostate cancer prevention
-
The importance of changes in zinc accumulation in prostate carcinogenesis emphasizes the potential roles of zinc and zinc transport proteins for the development of novel diagnostic and therapeutic modalities
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Chasapis, C. T., Loutsidou, A. C., Spiliopoulou, C. A. & Stefanidou, M. E. Zinc and human health: an update. Arch. Toxicol. 86, 521–534 (2012).
Rink, L. & Gabriel, P. Zinc and the immune system. Proc. Nutr. Soc. 59, 541–552 (2000).
Trumbo, P., Yates, A. A., Schlicker, S. & Poos, M. Dietary reference intakes: vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J. Am. Diet Assoc. 101, 294–301 (2001).
Costello, L. C., Feng, P., Milon, B., Tan, M. & Franklin, R. B. Role of zinc in the pathogenesis and treatment of prostate cancer: critical issues to resolve. Prostate Cancer Prostatic Dis. 7, 111–117 (2004).
Costello, L. C. & Franklin, R. B. The clinical relevance of the metabolism of prostate cancer; zinc and tumor suppression: connecting the dots. Mol. Cancer 5, 17 (2006).
Feng, P., Li, T. L., Guan, Z. X., Franklin, R. B. & Costello, L. C. Effect of zinc on prostatic tumorigenicity in nude mice. Ann. NY Acad. Sci. 1010, 316–320 (2003).
Franklin, R. B. & Costello, L. C. Zinc as an anti-tumor agent in prostate cancer and in other cancers. Arch. Biochem. Biophys. 463, 211–217 (2007).
Franklin, R. B., Milon, B., Feng, P. & Costello, L. C. Zinc and zinc transporters in normal prostate and the pathogenesis of prostate cancer. Front. Biosci. 10, 2230–2239 (2005).
Golovine, K. et al. Overexpression of the zinc uptake transporter hZIP1 inhibits nuclear factor-kappaB and reduces the malignant potential of prostate cancer cells in vitro and in vivo. Clin. Cancer Res. 14, 5376–5384 (2008).
Golovine, K. et al. Depletion of intracellular zinc increases expression of tumorigenic cytokines VEGF, IL-6 and IL-8 in prostate cancer cells via NF-kB-dependent pathway. Prostate 68, 1443–1449 (2008).
Uzzo, R. G. et al. Zinc Inhibits Nuclear Factor-kappaB Activation and Sensitizes Prostate Cancer Cells to Cytotoxic Agents. Clin. Cancer Res. 8, 3579–3583 (2002).
Chang, E. T., Hedelin, M., Adami, H. O., Grönberg, H. & Bälter, K. A. Re: Zinc supplement use and risk of prostate cancer. J. Natl Cancer Inst. 96, 1108–1109 (2004).
Gallus, S. et al. Dietary zinc and prostate cancer risk: a case-control study from Italy. Eur. Urol. 52, 1052–1056 (2007).
Key, T. J., Silcocks, P. B., Davey, G. K., Appleby, P. N. & Bishop, D. T. A case-control study of diet and prostate cancer. Br. J. Cancer 76, 678–687 (1997).
Kristal, A. R., Stanford, J. L., Cohen, J. H., Wicklund, K. & Patterson, R. E. Vitamin and mineral supplement use is associated with reduced risk of prostate cancer. Cancer Epidemiol. Biomarkers Prev. 8, 887–892 (1999).
Lee, M. M. et al. Case-control study of diet and prostate cancer in China. Cancer Causes Control 9, 545–552 (1998).
Leitzmann, M. F. et al. Zinc supplement use and risk of prostate cancer. J. Natl Cancer Inst. 95, 1004–1007 (2003).
Zhang, Y., Coogan, P., Palmer, J. R., Strom, B. L. & Rosenberg, L. Vitamin and mineral use and risk of prostate cancer: the case-control surveillance study. Cancer Causes Control 20, 691–698 (2009).
Zaichick, V. Ye., Sviridova, T. V. & Zaichick, S. V. Zinc in the human prostate gland: normal, hyperplastic and cancerous. Int. Urol. Nephrol. 29, 565–574 (1997).
Ogunlewe, J. O. & Osegbe, D. N. Zinc and cadmium concentrations in indigenous blacks with normal, hypertrophic, and malignant prostate. Cancer 63, 1388–1392 (1989).
Costello, L. C. & Franklin, R. B. Zinc is decreased in prostate cancer: an established relationship of prostate cancer! J. Biol. Inorg. Chem. 16, 3–8 (2011).
Christudoss, P., Selvakumar, R., Fleming, J. J. & Gopalakrishnan, G. Zinc status of patients with benign prostatic hyperplasia and prostate carcinoma. Indian J. Urol. 27, 14–18 (2011).
Costello, L. C. & Franklin, R. B. Novel role of zinc in the regulation of prostate citrate metabolism and its implications in prostate cancer. Prostate 35, 285–296 (1998).
Huang, L., Kirschke, C. P. & Zhang, Y. Decreased intracellular zinc in human tumorigenic prostate epithelial cells: a possible role in prostate cancer progression. Cancer Cell. Int. 6, 10 (2006).
Singh, K. K., Desouki, M. M., Franklin, R. B. & Costello, L. C. Mitochondrial aconitase and citrate metabolism in malignant and nonmalignant human prostate tissues. Mol. Cancer 5, 14 (2006).
Costello, L. C. & Franklin, R. B. The intermediary metabolism of the prostate: a key to understanding the pathogenesis and progression of prostate malignancy. Oncology 59, 269–282 (2000).
Liang, J. Y. et al. Inhibitory effect of zinc on human prostatic carcinoma cell growth. Prostate 40, 200–207 (1999).
Han, C. T., Schoene, N. W. & Lei, K. Y. Influence of zinc deficiency on Akt-Mdm2-p53 and Akt-p21 signaling axes in normal and malignant human prostate cells. Am. J. Physiol. Cell Physiol. 297, C1188–C1199 (2009).
Uzzo, R. G. et al. Diverse effects of zinc on NF-kappaB and AP-1 transcription factors: implications for prostate cancer progression. Carcinogenesis 27, 1980–1990 (2006).
Nardinocchi, L. et al. Zinc downregulates HIF-1α and inhibits its activity in tumor cells in vitro and in vivo. PLoS One 5, e15048 (2010).
Feng, P., Li, T. L., Guan, Z. X., Franklin, R. B. & Costello, L. C. Direct effect of zinc on mitochondrial apoptogenesis in prostate cells. Prostate 52, 311–318 (2002).
Feng, P. et al. Zinc induces mitochondria apoptogenesis in prostate cells. Mol. Urol. 4, 31–36 (2000).
Feng, P., Li, T., Guan, Z., Franklin, R. B. & Costello, L. C. The involvement of Bax in zinc-induced mitochondrial apoptogenesis in malignant prostate cells. Mol. Cancer 7, 25 (2008).
Ku, J. H., Seo, S. Y., Kwak, C. & Kim, H. H. The role of survivin and Bcl-2 in zinc-induced apoptosis in prostate cancer cells. Urol. Oncol. 30, 562–568 (2012).
Ishii, K. et al. Evidence that the prostate-specific antigen (PSA)/Zn2+ axis may play a role in human prostate cancer cell invasion. Cancer Lett. 207, 79–87 (2004).
Ishii, K. et al. Aminopeptidase N regulated by zinc in human prostate participates in tumor cell invasion. Int. J. Cancer 92, 49–54 (2001).
Wickstrom, M., Larsson, R., Nygren, P. & Gullbo, J. Aminopeptidase N (CD13) as a target for cancer chemotherapy. Cancer Sci. 102, 501–508 (2011).
Goel, T. & Sankhwar, S. N. Comparative study of zinc levels in benign and malignant lesions of the prostate. Scand. J. Urol. Nephrol. 40, 108–112 (2006).
McMahon, R. J. & Cousins, R. J. Regulation of the zinc transporter ZnT-1 by dietary zinc. Proc. Natl Acad. Sci. USA 95, 4841–4846 (1998).
Cousins, R. J., Liuzzi, J. P. & Lichten, L. A. Mammalian zinc transport, trafficking, and signals. J. Biol. Chem. 281, 24085–24089 (2006).
Kambe, T., Yamaguchi-Iwai, Y., Sasaki, R. & Nagao, M. Overview of mammalian zinc transporters. Cell. Mol. Life Sci. 61, 49–68 (2004).
Liuzzi, J. P. & Cousins, R. J. Mammalian zinc transporters. Annu. Rev. Nutr. 24, 151–172 (2004).
Foster, M., Hancock, D., Petocz, P. & Samman, S. Zinc transporter genes are coordinately expressed in men and women independently of dietary or plasma zinc. J. Nutr. 141, 1195–1201 (2011).
Hasumi, M. et al. Regulation of metallothionein and zinc transporter expression in human prostate cancer cells and tissues. Cancer Lett. 200, 187–195 (2003).
Majumder, S. et al. Chromium(VI) down-regulates heavy metal-induced metallothionein gene transcription by modifying transactivation potential of the key transcription factor, metal-responsive transcription factor 1. J. Biol. Chem. 278, 26216–26226 (2003).
Iguchi, K. et al. Zinc and metallothionein levels and expression of zinc transporters in androgen-independent subline of LNCaP cells. J. Androl. 25, 154–161 (2004).
Tepaamorndech, S., Huang, L. & Kirschke, C. P. A null-mutation in the Znt7 gene accelerates prostate tumor formation in a transgenic adenocarcinoma mouse prostate model. Cancer Lett. 308, 33–42 (2011).
Gaither, L. A. & Eide, D. J. The human ZIP1 transporter mediates zinc uptake in human K562 erythroleukemia cells. J. Biol. Chem. 276, 22258–22264 (2001).
Gaither, L. A. & Eide, D. J. Functional expression of the human hZIP2 zinc transporter. J. Biol. Chem. 275, 5560–5564 (2000).
Costello, L. C., Liu, Y., Zou, J. & Franklin, R. B. Evidence for a zinc uptake transporter in human prostate cancer cells which is regulated by prolactin and testosterone. J. Biol. Chem. 274, 17499–17504 (1999).
Eide, D. J. The SLC39 family of metal ion transporters. Pflugers Arch. 447, 796–800 (2004).
Dufner-Beattie, J., Huang, Z. L., Geiser, J., Xu, W. & Andrews, G. K. Mouse ZIP1 and ZIP3 genes together are essential for adaptation to dietary zinc deficiency during pregnancy. Genesis 44, 239–251 (2006).
Kim, B. E. et al. Zn2+-stimulated endocytosis of the mZIP4 zinc transporter regulates its location at the plasma membrane. J. Biol. Chem. 279, 4523–4530 (2004).
Desouki, M. M., Geradts, J., Milon, B., Franklin, R. B. & Costello, L. C. hZip2 and hZip3 zinc transporters are down regulated in human prostate adenocarcinomatous glands. Mol. Cancer 6, 37 (2007).
Chen, Q. G. et al. The role of zinc transporter ZIP4 in prostate carcinoma. Urol. Oncol. 30, 906–911 (2012).
Rishi, I. et al. Prostate cancer in African American men is associated with downregulation of zinc transporters. Appl. Immunohistochem. Mol. Morphol. 11, 253–260 (2003).
Franklin, R. B. et al. hZIP1 zinc uptake transporter down regulation and zinc depletion in prostate cancer. Mol. Cancer 4, 32 (2005).
Franklin, R. B. et al. Human ZIP1 is a major zinc uptake transporter for the accumulation of zinc in prostate cells. J. Inorg. Biochem. 96, 435–442 (2003).
Makhov, P. B. et al. Reversal of epigenetic silencing of AP-2α results in increased zinc uptake in DU-145 and LNCaP prostate cancer cells. Carcinogenesis 32, 1773–1781 (2011).
Mihelich, B. L. et al. miR-183-96-182 cluster is overexpressed in prostate tissue and regulates zinc homeostasis in prostate cells. J. Biol. Chem. 286, 44503–44511 (2011).
Milon, B. C., Agyapong, A., Bautista, R., Costello, L. C. & Franklin, R. B. Ras responsive element binding protein-1 (RREB-1) down-regulates hZIP1 expression in prostate cancer cells. Prostate 70, 288–296 (2010).
Zou, J., Milon, B. C., Desouki, M. M., Costello, L. C. & Franklin, R. B. hZIP1 zinc transporter down-regulation in prostate cancer involves the overexpression of ras responsive element binding protein-1 (RREB-1). Prostate doi:10.1002/pros.21368.
Costello, L. C. & Franklin, R. B. Re: Silvano Gallus, Roberto Foschi, Eva Negri et al. Dietary zinc and prostate cancer risk: a case-control study from Italy. Eur. Urol. 2007;52:1052–7. Eur. Urol. 52, 1262–1263 (2007).
Costello, L. C., Franklin, R. B., Feng, P. & Tan, M. Re: zinc supplement use and risk of prostate cancer. J. Natl Cancer Inst. 96, 239–240 (2004).
Costello, L. C., Franklin, R. B., Feng, P., Tan, M. & Bagasra, O. Zinc and prostate cancer: a critical scientific, medical, and public interest issue (United States). Cancer Causes Control 16, 901–915 (2005).
Franklin, R. B. & Costello, L. C. Re: Vitamin and mineral use and risk of prostate cancer: the case-control surveillance: Zhang et al. Cancer Causes Control 20, 1529–1531 (2009).
Prasad, A. S. et al. Dietary zinc and prostate cancer in the TRAMP mouse model. J. Med. Food 13, 70–76 (2010).
Banudevi, S. et al. Chemopreventive effects of zinc on prostate carcinogenesis induced by N-methyl-N-nitrosourea and testosterone in adult male Sprague-Dawley rats. J. Cancer Res. Clin. Oncol. 137, 677–686 (2011).
Banudevi, S. et al. Protective effect of zinc on N-methyl-N-nitrosourea and testosterone-induced prostatic intraepithelial neoplasia in the dorsolateral prostate of Sprague Dawley rats. Exp. Biol. Med. (Maywood) 236, 1012–1021 (2011).
Platz, E. A. et al. Prediagnostic toenail cadmium and zinc and subsequent prostate cancer risk. Prostate 52, 288–296 (2002).
Epstein, M. M. et al. Dietary zinc and prostate cancer survival in a Swedish cohort. Am. J. Clin. Nutr. 93, 586–593 (2011).
Gonzalez, A., Peters, U., Lampe, J. W. & White, E. Zinc intake from supplements and diet and prostate cancer. Nutr. Cancer 61, 206–215 (2009).
Mayo Clinic. Zinc [online], (2012).
Chandra, R. K. Excessive intake of zinc impairs immune responses. JAMA 252, 1443–1446 (1984).
Samman, S. & Roberts, D. C. The effect of zinc supplements on lipoproteins and copper status. Atherosclerosis 70, 247–252 (1988).
King, J. C., Shames, D. M. & Woodhouse, L. R. Zinc homeostasis in humans. J. Nutr. 130 (5S Suppl.), 1360S–1366S (2000).
Park, S. Y., Wilkens, L. R., Morris, J. S., Henderson, B. E. & Kolonel, L. N. Serum zinc and prostate cancer risk in a nested case-control study: the multiethnic cohort. Prostate 73, 261–266 (2013).
Lawson, K. A. et al. Multivitamin use and risk of prostate cancer in the National Institutes of Health-AARP Diet and Health Study. J. Natl Cancer Inst. 99, 754–764 (2007).
Uzzo, R. G. et al. Prevalence and patterns of self-initiated nutritional supplementation in men at high risk of prostate cancer. BJU Int. 93, 955–960 (2004).
Krone, C. A. & Harms, L. C. Re: Zinc supplement use and risk of prostate cancer. J. Natl Cancer Inst. 95, 1556–1557 (2003).
Krone, C. A., Wyse, E. J. & Ely, J. T. Cadmium in zinc-containing mineral supplements. Int. J. Food Sci. Nutr. 52, 379–382 (2001).
Achanzar, W. E. et al. Cadmium-induced malignant transformation of human prostate epithelial cells. Cancer Res. 61, 455–458 (2001).
Johnson, L. A., Kanak, M. A., Kajdacsy-Balla, A., Pestaner, J. P. & Bagasra, O. Differential zinc accumulation and expression of human zinc transporter 1 (hZIP1) in prostate glands. Methods 52, 316–321 (2010).
Ghosh, S. K. et al. A novel imaging approach for early detection of prostate cancer based on endogenous zinc sensing. Cancer Res. 70, 6119–6127 (2010).
Cortesi, M. et al. Clinical assessment of the cancer diagnostic value of prostatic zinc: a comprehensive needle-biopsy study. Prostate 68, 994–1006 (2008).
Liguori, P. F. et al. Non-classical anticancer agents: synthesis and biological evaluation of zinc(II) heteroleptic complexes. Dalton Trans. 39, 4205–4212 (2010).
Ding, W. Q. & Lind, S. E. Metal ionophores—an emerging class of anticancer drugs. IUBMB Life 61, 1013–1018 (2009).
Costello, L. C. & Franklin, R. B. Cytotoxic/tumor suppressor role of zinc for the treatment of cancer: an enigma and an opportunity. Expert Rev. Anticancer Ther. 12, 121–128 (2012).
Killilea, A. N., Downing, K. H. & Killilea, D. W. Zinc deficiency reduces paclitaxel efficacy in LNCaP prostate cancer cells. Cancer Lett. 258, 70–79 (2007).
Magda, D. et al. Synthesis and anticancer properties of water-soluble zinc ionophores. Cancer Res. 68, 5318–5325 (2008).
Shah, M. R. et al. Direct intra-tumoral injection of zinc-acetate halts tumor growth in a xenograft model of prostate cancer. J. Exp. Clin. Cancer Res. 28, 84 (2009).
Makhov, P. et al. Docetaxel-mediated apoptosis in myeloid progenitor TF-1 cells is mitigated by zinc: potential implication for prostate cancer therapy. Prostate 71, 1413–1419 (2011).
Makhov, P. et al. Zinc chelation induces rapid depletion of the X-linked inhibitor of apoptosis and sensitizes prostate cancer cells to TRAIL-mediated apoptosis. Cell Death Differ. 15, 1745–1751 (2008).
Schimmer, A. D., Dalili, S., Batey, R. A. & Riedl, S. J. Targeting XIAP for the treatment of malignancy. Cell Death Differ. 13, 179–188 (2006).
Schimmer, A. D. et al. Small-molecule antagonists of apoptosis suppressor XIAP exhibit broad antitumor activity. Cancer Cell 5, 25–35 (2004).
Zuo, J. et al. Novel polypyridyl chelators deplete cellular zinc and destabilize the X-linked inhibitor of apoptosis protein (XIAP) prior to induction of apoptosis in human prostate and breast cancer cells. J. Cell. Biochem. 113, 2567–2575 (2012).
Acknowledgements
This work was supported in part by the National Institutes of Health Grants (RO1 CA134463, RO3 CA167671, CCSG, P30 CA006927) to V. Kolenko, the American Institute for Cancer Research Grant (09A023) to R. Uzzo and the Department of Defense Physician Research Training Award (W81XWH-10-1-0187) and Bucks County Board of Associates Award to A. Kutikov.
Author information
Authors and Affiliations
Contributions
V. Kolenko and A. Kutikov researched the data for the article. A. Kutikov and R. Uzzo discussed the article's content. V. Kolenko and E. Teper wrote the manuscript, after which V. Kolenko and R. Uzzo edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Kolenko, V., Teper, E., Kutikov, A. et al. Zinc and zinc transporters in prostate carcinogenesis. Nat Rev Urol 10, 219–226 (2013). https://doi.org/10.1038/nrurol.2013.43
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2013.43
This article is cited by
-
Novel Mononuclear Zinc(II) Complex with Niacin: Crystal Structure, DNA/Protein Interaction, and Cytotoxicity Studies
Journal of Applied Spectroscopy (2022)
-
Associations of trace element levels in paired serum, whole blood, and tissue: an example of esophageal squamous cell carcinoma
Environmental Science and Pollution Research (2022)
-
ZnT7 RNAi favors RafGOFscrib−/−-induced tumor growth and invasion in Drosophila through JNK signaling pathway
Oncogene (2021)
-
The Roles of ZnT1 and ZnT4 in Glucose-Stimulated Zinc Secretion in Prostate Epithelial Cells
Molecular Imaging and Biology (2021)
-
Tumour metabolism and its unique properties in prostate adenocarcinoma
Nature Reviews Urology (2020)