Abstract
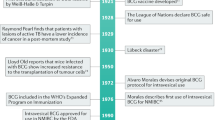
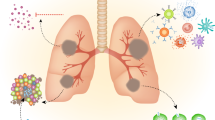
Genetic mutations have been progressively introduced to BCG by repeated serial passage over many decades of its culture and global dissemination. Thus, marked differences exist in the phenotype, antigenicity, reactogenicity, and clinical characteristics of the numerous substrains of BCG currently in use for bladder cancer immunotherapy. These differences influence proposed mycobacterial antitumour mechanisms and toxicity, potentially resulting in variations in clinical efficacy and adverse effects. However, although there is evidence of substrain-related differences in the clinical efficacy of BCG as a tuberculosis vaccine, evidence of an effect on bladder cancer immunotherapy remains elusive, owing to the lack of appropriately powered head-to-head comparative clinical trials, the nonstandardization of BCG manufacture, and variation in treatment protocols—possibly itself a response to underlying substrain differences. Advances in our understanding of mycobacterial genetics, structure and function, and host–pathogen interactions might explain differences in clinical practice and outcomes. These advances are guiding the identification of biomarkers for reactogenicity and efficacy, and the rational design of immunotherapeutic strategies to eliminate the use of live bacilli for bladder cancer therapy.
Key Points
-
Outcome of BCG immunotherapy for bladder cancer depends upon key host–pathogen interactions, which might be influenced by both human and bacterial genetics
-
BCG is not a defined strain but a family of regional substrains with marked genetic differences that have developed during manufacture and global dissemination
-
Differences in phenotype, antigenicity, reactogenicity, and clinical characteristics of BCG substrains have been observed during their use as a tuberculosis vaccine
-
Differences in bladder cancer outcomes between BCG substrains have been reported, but owing to the paucity of appropriately powered clinical trials the significance remains unconfirmed
-
Multiple dose levels, number of doses, and dosing schedules exist for bladder cancer BCG therapy, which complicates comparison of clinical trial outcomes with different strains
-
Appropriately powered head-to-head clinical trials using BCG substrains manufactured under current good manufacturing practice are required to determine if certain strains offer better efficacy or reduced reactogenicity for bladder cancer
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
19 May 2015
In the introduction of the originally published article, the names of the BCG substrains involved in recent shortages in BCG supply were transposed. The error has been corrected for the HTML and PDF versions of the article.
References
International Agency for Research on Cancer. Globocan 2008 [online], (2013).
Babjuk, M. et al. EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder, the 2011 update. Eur. Urol. 59, 997–1008 (2011).
Morales, A., Eidinger, D. & Bruce, A. W. Intracavitary Bacillus Calmette-Guerin in the treatment of superficial bladder tumors. J. Urol. 116, 180–183 (1976).
Gandhi, N. M., Morales, A. & Lamm, D. L. Bacillus Calmette-Guerin immunotherapy for genitourinary cancer. BJU Int. 112, 288–297 (2013).
Sylvester, R. J., van der Meijden, A. P. & Lamm, D. L. Intravesical bacillus Calmette-Guerin reduces the risk of progression in patients with superficial bladder cancer: a meta-analysis of the published results of randomized clinical trials. J. Urol. 168, 1964–1970 (2002).
Malmstrom, P. U. et al. An individual patient data meta-analysis of the long-term outcome of randomised studies comparing intravesical mitomycin C versus bacillus Calmette-Guerin for non-muscle-invasive bladder cancer. Eur. Urol. 56, 247–256 (2009).
Lamm, D. L. et al. Maintenance bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group Study. J. Urol. 163, 1124–1129 (2000).
van der Meijden, A. P. et al. Maintenance Bacillus Calmette-Guerin for Ta T1 bladder tumors is not associated with increased toxicity: results from a European Organisation for Research and Treatment of Cancer Genito-Urinary Group Phase III Trial. Eur. Urol. 44, 429–434 (2003).
Lukacs, S., Tschobotko, B., Szabo, N. A. & Symes, A. Systemic BCG-osis as a rare side effect of intravesical BCG treatment for superficial bladder cancer. Case Rep. Urol. 821526 (2013).
Martinez-Pineiro, J. A. et al. Has a 3-fold decreased dose of bacillus Calmette-Guerin the same efficacy against recurrences and progression of T1G3 and Tis bladder tumors than the standard dose? Results of a prospective randomized trial. J. Urol. 174, 1242–1247 (2005).
Ojea, A. et al. A multicentre, randomised prospective trial comparing three intravesical adjuvant therapies for intermediate-risk superficial bladder cancer: low-dose bacillus Calmette-Guerin (27 mg) versus very low-dose bacillus Calmette-Guerin (13.5 mg) versus mitomycin C. Eur. Urol. 52, 1398–1406 (2007).
Hall, M. C. et al. Guideline for the management of nonmuscle invasive bladder cancer (stages Ta, T1, and Tis): 2007 update. J. Urol. 178, 2314–2330 (2007).
Kassouf, W. et al. Canadian guidelines for treatment of non-muscle invasive bladder cancer: a focus on intravesical therapy. Can. Urol. Assoc. J. 4, 168–173 (2010).
Committee for Establishment of the Clinical Practice Guidelines for the Management of Bladder Cancer and the Japanese Urological Association. Evidence-based clinical practice guidelines for bladder cancer (summary—JUA 2009 Edition). Int. J. Urol. 17, 102–124 (2010).
British Association of Urological Surgeons (BAUS) Section of Oncology. MDT (Multi-disciplinary team) guidance for managing bladder cancer [online], (2013).
Shortage of BCG vaccine. East & South East England Specialist Pharmacy Services [online], (2013).
Shaaf, H. S. & Zumla, A. Tuberculosis A Comprehensive Clinical Reference (Saunders Elsevier, 2009).
Guerin, C. in BCG Vaccine: Tuberculosis-Cancer (ed. Rosenthal, S.) 35 (PSG Publishing, 1980).
Venkataswamy, M. M. et al. In vitro culture medium influences the vaccine efficacy of Mycobacterium bovis BCG. Vaccine 30, 1038–1049 (2012).
Milstien, J. B. & Gibson, J. J. Quality control of BCG vaccine by WHO: a review of factors that may influence vaccine effectiveness and safety. Bull. World Health Organ. 68, 93–108 (1990).
Ritz, N., Hanekom, W. A., Robins-Browne, R., Britton, W. J. & Curtis, N. Influence of BCG vaccine strain on the immune response and protection against tuberculosis. FEMS Microbiol. Rev. 32, 821–841 (2008).
Lotte, A. et al. BCG complications. Estimates of the risks among vaccinated subjects and statistical analysis of their main characteristics. Adv. Tuberc. Res. 21, 107–193 (1984).
The BCG World Atlas [online], (2013).
Ritz, N. & Curtis, N. Mapping the global use of different BCG vaccine strains. Tuberculosis (Edinb.) 89, 248–251 (2009).
Zwerling, A. et al. The BCG World Atlas: a database of global BCG vaccination policies and practices. PLoS Med. 8, e1001012 (2011).
Assis, A. Novas perspectivas de Calmettização. Hospital (RioJ) 37, 337–353 (1950).
Quirino, R., Ornellas, A. A. & Wisnescky, A. Treatment of patients with recurrent superficial bladder cancer with lyophilized Moureau-Rio de Janeiro BCG strain. Brazillian National Cancer Institute experience [French]. Prog. Urol. 15, 667–673 (2005).
Fonseca, F. P. et al. Treatment of patients with superficial bladder cancer stratified by risk groups treated with lyophilized Moreau-Rio de Janeiro BCG strain. Int. Braz. J. Urol. 28, 426–435 (2002).
Fine, P. E. The BCG story: lessons from the past and implications for the future. Rev. Infect. Dis. 11 (Suppl. 2), S353–S359 (1989).
Brosch, R. et al. Genome plasticity of BCG and impact on vaccine efficacy. Proc. Natl Acad. Sci. USA 104, 5596–5601 (2007).
Leung, A. S. et al. Novel genome polymorphisms in BCG vaccine strains and impact on efficacy. BMC Genomics 9, 413 (2008).
Schwarzer, K., Foerster, M., Steiner, T., Hermann, I. M. & Straube, E. BCG strain S4-Jena: An early BCG strain is capable to reduce the proliferation of bladder cancer cells by induction of apoptosis. Cancer Cell. Int. 10, 21 (2010).
Torrado, E., Robinson, R. T. & Cooper, A. M. Cellular response to mycobacteria: balancing protection and pathology. Trends Immunol. 32, 66–72 (2011).
Brincks, E. L., Risk, M. C. & Griffith, T. S. PMN and anti-tumor immunity—the case of bladder cancer immunotherapy. Semin. Cancer Biol. 23, 183–189 (2013).
Dorhoi, A., Reece, S. T. & Kaufmann, S. H. For better or for worse: the immune response against Mycobacterium tuberculosis balances pathology and protection. Immunol. Rev. 240, 235–251 (2011).
Cosgrove, C. A. et al. Boosting of cellular immunity against Mycobacterium tuberculosis and modulation of skin cytokine responses in healthy human volunteers by Mycobacterium bovis BCG substrain Moreau Rio de Janeiro oral vaccine. Infect. Immun. 74, 2449–2452 (2006).
Miura, K., Nagai, S., Kinomoto, M., Haga, S. & Tokunaga, T. Comparative studies with various substrains of Mycobacterium bovis BCG on the production of an antigenic protein, MPB70. Infect. Immun. 39, 540–545 (1983).
Charlet, D. et al. Reduced expression of antigenic proteins MPB70 and MPB83 in Mycobacterium bovis BCG strains due to a start codon mutation in sigK. Mol. Microbiol. 56, 1302–1313 (2005).
Said-Salim, B., Mostowy, S., Kristof, A. S. & Behr, M. A. Mutations in Mycobacterium tuberculosis Rv0444c, the gene encoding anti-SigK, explain high level expression of MPB70 and MPB83 in Mycobacterium bovis. Mol. Microbiol. 62, 1251–1263 (2006).
Ritz, N. et al. Susceptibility of Mycobacterium bovis BCG vaccine strains to antituberculous antibiotics. Antimicrob. Agents Chemother. 53, 316–318 (2009).
Malhotra, P. & Farber, B. F. Isoniazid resistance among Bacillus Calmette Guerin strains: implications on bladder cancer immunotherapy related infections. Can. J. Urol. 18, 5671–5675 (2011).
Dinadayala, P. et al. Revisiting the structure of the anti-neoplastic glucans of Mycobacterium bovis Bacille Calmette-Guerin. Structural analysis of the extracellular and boiling water extract-derived glucans of the vaccine substrains. J. Biol. Chem. 279, 12369–12378 (2004).
Bedwell, J., Kairo, S. K., Behr, M. A. & Bygraves, J. A. Identification of substrains of BCG vaccine using multiplex PCR. Vaccine 19, 2146–2151 (2001).
Honda, I. et al. Identification of two subpopulations of Bacillus Calmette-Guerin (BCG) Tokyo172 substrain with different RD16 regions. Vaccine 24, 4969–4974 (2006).
Naka, T. et al. Lipid phenotype of two distinct subpopulations of Mycobacterium bovis Bacillus Calmette-Guerin Tokyo 172 substrain. J. Biol. Chem. 286, 44153–44161 (2011).
Luo, Y. Blocking IL-10 enhances bacillus Calmette-Guerin induced T helper Type 1 immune responses and anti-bladder cancer immunity. Oncoimmunology 1, 1183–1185 (2012).
Ritz, N. et al. The influence of bacille Calmette-Guerin vaccine strain on the immune response against tuberculosis: a randomized trial. Am. J. Respir. Crit. Care Med. 185, 213–222 (2012).
Aronson, N. E. et al. Long-term efficacy of BCG vaccine in American Indians and Alaska Natives: A 60-year follow-up study. JAMA 291, 2086–2091 (2004).
Chan, S., Allen, G. & Pio, A. Comparison of the efficacy of two strains of BCG vaccine for prevention of tuberculosis among newborn children in Hong Kong. Bull. Int. Union Tuberc. 61, 36–37 (1966).
Ratliff, T. L., Gillen, D. & Catalona, W. J. Requirement of a thymus dependent immune response for BCG-mediated antitumor activity. J. Urol. 137, 155–158 (1987).
Lima, L., Dinis-Ribeiro, M., Longatto-Filho, A. & Santos, L. Predictive biomarkers of bacillus calmette-guerin immunotherapy response in bladder cancer: where are we now? Adv. Urol. 232609 (2012).
Biot, C. et al. Preexisting BCG-specific T cells improve intravesical immunotherapy for bladder cancer. Sci. Transl. Med. 4, 137ra72 (2012).
Brandau, S. & Bohle, A. Activation of natural killer cells by Bacillus Calmette-Guerin. Eur. Urol. 39, 518–524 (2001).
Ikeda, N., Toida, I., Iwasaki, A., Kawai, K. & Akaza, H. Surface antigen expression on bladder tumor cells induced by bacillus Calmette-Guerin (BCG): A role of BCG internalization into tumor cells. Int. J. Urol. 9, 29–35 (2002).
Poggi, M. M., Johnstone, P. A. & Conner, R. J. Glycosaminoglycan content of human bladders. a method of analysis using cold-cup biopsies. Urol. Oncol. 5, 234–237 (2000).
Hudson, M. A., Ritchey, J. K., Catalona, W. J., Brown, E. J. & Ratliff, T. L. Comparison of the fibronectin-binding ability and antitumor efficacy of various mycobacteria. Cancer Res. 50, 3843–3847 (1990).
Gan, H. et al. Mycobacterium tuberculosis blocks crosslinking of annexin-1 and apoptotic envelope formation on infected macrophages to maintain virulence. Nat. Immunol. 9, 1189–1197 (2008).
Secanella-Fandos, S., Luquin, M. & Julian, E. Connaught and Russian strains showed the highest direct antitumor effects of different Bacillus Calmette-Guerin substrains. J. Urol. 189, 711–718 (2013).
de Boer, E. C. et al. Role of interleukin-8 in onset of the immune response in intravesical BCG therapy for superficial bladder cancer. Urol. Res. 25, 31–34 (1997).
Shintani, Y. et al. Intravesical instillation therapy with bacillus Calmette-Guerin for superficial bladder cancer: study of the mechanism of bacillus Calmette-Guerin immunotherapy. Int. J. Urol. 14, 140–146 (2007).
Chen, J. M., Islam, S. T., Ren, H. & Liu, J. Differential productions of lipid virulence factors among BCG vaccine strains and implications on BCG safety. Vaccine 25, 8114–8122 (2007).
Thalmann, G. N. et al. Urinary Interleukin-8 and 18 predict the response of superficial bladder cancer to intravesical therapy with bacillus Calmette-Guerin. J. Urol. 164, 2129–2133 (2000).
Gonzalez, O. Y. et al. Spectrum of bacille Calmette-Guerin (BCG) infection after intravesical BCG immunotherapy. Clin. Infect. Dis. 36, 140–148 (2003).
Ajili, F., Kourda, N., Darouiche, A., Chebil, M. & Boubaker, S. Prognostic value of tumor-associated macrophages count in human non-muscle-invasive bladder cancer treated by BCG immunotherapy. Ultrastruct. Pathol. 37, 56–61 (2013).
De Boer, E. C. et al. Induction of urinary interleukin-1 (IL-1), IL-2, IL-6, and tumour necrosis factor during intravesical immunotherapy with bacillus Calmette-Guerin in superficial bladder cancer. Cancer Immunol. Immunother. 34, 306–312 (1992).
Birkhaeuser, F., Rentsch, C., Studer, U., Albert, M. & Thalmann, G. A randomized phase III study comparing Immucyst® versus Oncotice®: the BCG strain used makes a difference. J. Urol. 187, e676 (2012).
Sengiku, A. et al. A prospective comparative study of intravesical Bacillus Calmette-Guerin therapy with the Tokyo or Connaught strains for non-muscle invasive bladder cancer. J. Urol. 190, 50–54 (2013).
Witjes, W. P., Witjes, J. A., Oosterhof, G. O. & Debruyne, M. J. Update on the Dutch Cooperative Trial: mitomycin versus bacillus Calmette-Guerin-Tice versus bacillus Calmette-Guerin RIVM in the treatment of patients with pTA–pT1 papillary carcinoma and carcinoma in situ of the urinary bladder. Dutch South East Cooperative Urological Group. Semin. Urol. Oncol. 14, 10–16 (1996).
Fellows, G. J. et al. Marker tumour response to Evans and Pasteur bacille Calmette-Guerin in multiple recurrent pTa/pT1 bladder tumours: report from the Medical Research Council Subgroup on Superficial Bladder Cancer (Urological Cancer Working Party). Br. J. Urol. 73, 639–644 (1994).
Mukherjee, A., Persad, R. & Smith, P. J. Intravesical BCG treatment for superficial bladder cancer: long-term results using two different strains of BCG. Br. J. Urol. 69, 147–150 (1992).
Kaisary, A. V. Intravesical BCG therapy in the management of multiple superficial bladder carcinoma. Comparison between Glaxo and Pasteur strains. Br. J. Urol. 59, 554–558 (1987).
Ikeda, N., Honda, I., Yano, I., Koyama, A. & Toida, I. Bacillus calmette-guerin Tokyo172 substrain for superficial bladder cancer: characterization and antitumor effect. J. Urol. 173, 1507–1512 (2005).
Chouchkova, M. & Engibarov, A. Studies on the biological activity of two strains of BCG: influence of production conditions. Dev. Biol. Stand. 58 (Pt A), 105–118 (1986).
Hesseling, A. C. et al. Danish bacille Calmette-Guerin vaccine-induced disease in human immunodeficiency virus-infected children. Clin. Infect. Dis. 37, 1226–1233 (2003).
Bonifachich, E. et al. Protective effect of Bacillus Calmette-Guerin (BCG) vaccination in children with extra-pulmonary tuberculosis, but not the pulmonary disease. A case-control study in Rosario, Argentina. Vaccine 24, 2894–2899 (2006).
Liu, J., Tran, V., Leung, A. S., Alexander, D. C. & Zhu, B. BCG vaccines: their mechanisms of attenuation and impact on safety and protective efficacy. Hum. Vaccin. 5, 70–78 (2009).
Lamm, D. L. et al. Incidence and treatment of complications of bacillus Calmette-Guerin intravesical therapy in superficial bladder cancer. J. Urol. 147, 596–600 (1992).
Lotte, A. et al. A bibliography of the complications of BCG vaccination. A comprehensive list of the world literature since the introduction of BCG up to July 1982, supplemented by over 100 personal communications. Adv. Tuberc. Res. 21, 194–245 (1984).
Kaufmann, S. H. Tuberculosis vaccines: Time to think about the next generation. Semin. Immunol. http://dx.doi.org/10.1016/j.smim.2013.04.006.
Sampson, S. L. et al. Extended safety and efficacy studies of a live attenuated double leucine and pantothenate auxotroph of Mycobacterium tuberculosis as a vaccine candidate. Vaccine 29, 4839–4847 (2011).
Kawai, K., Miyazaki, J., Joraku, A., Nishiyama, H. & Akaza, H. Bacillus Calmette-Guerin (BCG) immunotherapy for bladder cancer: current understanding and perspectives on engineered BCG vaccine. Cancer Sci. 104, 22–27 (2013).
Begnini, K. R. et al. Auxotrophic recombinant Mycobacterium bovis BCG overexpressing Ag85B enhances cytotoxicity on superficial bladder cancer cells in vitro. Appl. Microbiol. Biotechnol. 97, 1543–1552 (2013).
Kato, T. et al. Bacillus Calmette-Guerin and BCG cell wall skeleton suppressed viability of bladder cancer cells in vitro. Anticancer Res. 30, 4089–4096 (2010).
Hayashi, A. et al. Immunotherapy of ovarian cancer with cell wall skeleton of Mycobacterium bovis Bacillus Calmette-Guerin: effect of lymphadenectomy. Cancer Sci. 100, 1991–1995 (2009).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article and contributed to the discussion of content. C. Gan and D. J. M. Lewis wrote the article. All authors reviewed the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Gan, C., Mostafid, H., Khan, M. et al. BCG immunotherapy for bladder cancer—the effects of substrain differences. Nat Rev Urol 10, 580–588 (2013). https://doi.org/10.1038/nrurol.2013.194
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2013.194
This article is cited by
-
100 years of Bacillus Calmette–Guérin immunotherapy: from cattle to COVID-19
Nature Reviews Urology (2021)
-
Current status and future perspectives of immunotherapy in bladder cancer treatment
Science China Life Sciences (2021)
-
Efficacy of three BCG strains (Connaught, TICE and RIVM) with or without secondary resection (re-TUR) for intermediate/high-risk non-muscle-invasive bladder cancers: results from a retrospective single-institution cohort analysis
Journal of Cancer Research and Clinical Oncology (2021)
-
Trained immunity as a molecular mechanism for BCG immunotherapy in bladder cancer
Nature Reviews Urology (2020)
-
Angiotensin II type 2 receptor promotes apoptosis and inhibits angiogenesis in bladder cancer
Journal of Experimental & Clinical Cancer Research (2017)