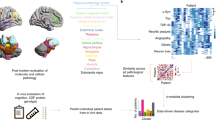
Key Points
-
Definite classification of dementia is based on the underlying neuropathology
-
Accumulation of abnormally folded proteins lies at the heart of dementia neuropathology
-
Alzheimer disease pathology can give rise to subtypes with focal onset in functional networks outside the memory system, such as language, visuospatial and behavioural executive domains
-
Frontotemporal lobar degeneration, associated with aggregates of tau, TDP-43 or FUS, can give rise to three core frontotemporal dementia syndromes and three associated syndromes
-
Clinical classification of dementia syndromes is based on diagnostic criteria that rely heavily on the specificity of affected domains and the evolution of deficits in these domains
-
In vivo biomarkers of disease include imaging findings of morphological, molecular and functional changes, both upstream and downstream of the disease processes
Abstract
The most definitive classification systems for dementia are based on the underlying pathology which, in turn, is categorized largely according to the observed accumulation of abnormal protein aggregates in neurons and glia. These aggregates perturb molecular processes, cellular functions and, ultimately, cell survival, with ensuing disruption of large-scale neural networks subserving cognitive, behavioural and sensorimotor functions. The functional domains affected and the evolution of deficits in these domains over time serve as footprints that the clinician can trace back with various levels of certainty to the underlying neuropathology. The process of phenotyping and syndromic classification has substantially improved over decades of careful clinicopathological correlation, and through the discovery of in vivo biomarkers of disease. Here, we present an overview of the salient features of the most common dementia subtypes — Alzheimer disease, vascular dementia, frontotemporal dementia and related syndromes, Lewy body dementias, and prion diseases — with an emphasis on neuropathology, relevant epidemiology, risk factors, and signature signs and symptoms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
28 July 2017
In the online biography for Fanny M. Elahi, 'American Academy of Neurology' was incorrectly written as 'American Association of Neurology'. This error has been corrected in the HTML version of the article.
References
Brun, A., Liu, X. & Erikson, C. Synapse loss and gliosis in the molecular layer of the cerebral cortex in Alzheimer's disease and in frontal lobe degeneration. Neurodegeneration 4, 171–177 (1995).
Miller, B. L. et al. Neuroanatomy of the self: evidence from patients with frontotemporal dementia. Neurology 57, 817–821 (2001).
Alzheimer's Disease International. World Alzheimer Report 2015: the Global Impact of Dementia. Alzheimer's Disease International https://www.alz.co.uk/research/world-report-2015 (2015).
Sloane, P. D. et al. The public health impact of Alzheimer's disease, 2000–2050: potential implication of treatment advances. Annu. Rev. Public Health 23, 213–231 (2002).
Katzman, R. Alzheimer's disease. N. Engl. J. Med. 314, 964–973 (1986).
McKhann, G. M. et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging–Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 7, 263–269 (2011).
Seeley, W. W., Crawford, R. K., Zhou, J., Miller, B. L. & Greicius, M. D. Neurodegenerative diseases target large-scale human brain networks. Neuron 62, 42–52 (2009).
McKinley, M. P., Masiarz, F. R. & Prusiner, S. B. Reversible chemical modification of the scrapie agent. Science 214, 1259–1261 (1981).
Prusiner, S. B. Novel proteinaceous infectious particles cause scrapie. Science 216, 136–144 (1982).
Jaunmuktane, Z. et al. Evidence for human transmission of amyloid-β pathology and cerebral amyloid angiopathy. Nature 525, 247–250 (2015).
Jucker, M. & Walker, L. C. Self-propagation of pathogenic protein aggregates in neurodegenerative diseases. Nature 501, 45–51 (2013).
Sanders, D. W. et al. Distinct tau prion strains propagate in cells and mice and define different tauopathies. Neuron 82, 1271–1288 (2014).
Li, J. Y. et al. Lewy bodies in grafted neurons in subjects with Parkinson's disease suggest host-to-graft disease propagation. Nat. Med. 14, 501–503 (2008).
Seeley, W. W. Selective functional, regional, and neuronal vulnerability in frontotemporal dementia. Curr. Opin. Neurol. 21, 701–707 (2008).
Jellinger, K. A. & Attems, J. Prevalence and impact of vascular and Alzheimer pathologies in Lewy body disease. Acta Neuropathol. 115, 427–436 (2008).
Matthews, F. E. et al. Epidemiological pathology of dementia: attributable-risks at death in the Medical Research Council Cognitive Function and Ageing Study. PLoS Med. 6, e1000180 (2009).
Lobo, A. et al. Prevalence of dementia and major subtypes in Europe: a collaborative study of population-based cohorts. Neurologic Diseases in the Elderly Research Group. Neurology 54, S4–S9 (2000).
Qiu, C., Kivipelto, M. & von Strauss, E. Epidemiology of Alzheimer's disease: occurrence, determinants, and strategies toward intervention. Dialogues Clin. Neurosci. 11, 111–128 (2009).
Panegyres, P. K. & Frencham, K. Course and causes of suspected dementia in young adults: a longitudinal study. Am. J. Alzheimers Dis. Other Demen. 22, 48–56 (2007).
Ratnavalli, E., Brayne, C., Dawson, K. & Hodges, J. R. The prevalence of frontotemporal dementia. Neurology 58, 1615–1621 (2002).
Harvey, R. J., Skelton-Robinson, M. & Rossor, M. N. The prevalence and causes of dementia in people under the age of 65 years. J. Neurol. Neurosurg. Psychiatry 74, 1206–1209 (2003).
Kelley, B. J., Boeve, B. F. & Josephs, K. A. Young-onset dementia: demographic and etiologic characteristics of 235 patients. Arch. Neurol. 65, 1502–1508 (2008).
Mercy, L., Hodges, J. R., Dawson, K., Barker, R. A. & Brayne, C. Incidence of early-onset dementias in Cambridgeshire, United Kingdom. Neurology 71, 1496–1499 (2008).
Schneider, J. A., Arvanitakis, Z., Bang, W. & Bennett, D. A. Mixed brain pathologies account for most dementia cases in community-dwelling older persons. Neurology 69, 2197–2204 (2007).
Nelson, P. T. et al. Modeling the association between 43 different clinical and pathological variables and the severity of cognitive impairment in a large autopsy cohort of elderly persons. Brain Pathol. 20, 66–79 (2010).
Ruitenberg, A., Ott, A., van Swieten, J. C., Hofman, A. & Breteler, M. M. Incidence of dementia: does gender make a difference? Neurobiol. Aging 22, 575–580 (2001).
Braak, H. & Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 82, 239–259 (1991).
Taylor, J. P., Hardy, J. & Fischbeck, K. H. Toxic proteins in neurodegenerative disease. Science 296, 1991–1995 (2002).
Braak, H., Alafuzoff, I., Arzberger, T., Kretzschmar, H. & Del Tredici, K. Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol. 112, 389–404 (2006).
Jack, C. R. Jr et al. Tracking pathophysiological processes in Alzheimer's disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 12, 207–216 (2013).
Jansen, W. J. et al. Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. JAMA 313, 1924–1938 (2015).
Thal, D. R., Rub, U., Orantes, M. & Braak, H. Phases of Aβ-deposition in the human brain and its relevance for the development of AD. Neurology 58, 1791–1800 (2002).
Braak, H. & Braak, E. Staging of Alzheimer's disease-related neurofibrillary changes. Neurobiol. Aging 16, 271–278 (1995).
Thal, D. R. et al. Sequence of Abeta-protein deposition in the human medial temporal lobe. J. Neuropathol. Exp. Neurol. 59, 733–748 (2000).
Braak, H., Thal, D. R., Ghebremedhin, E. & Del Tredici, K. Stages of the pathologic process in Alzheimer disease: age categories from 1 to 100 years. J. Neuropathol. Exp. Neurol. 70, 960–969 (2011).
Braak, H., Ghebremedhin, E., Rub, U., Bratzke, H. & Del Tredici, K. Stages in the development of Parkinson's disease-related pathology. Cell Tissue Res. 318, 121–134 (2004).
Grudzien, A. et al. Locus coeruleus neurofibrillary degeneration in aging, mild cognitive impairment and early Alzheimer's disease. Neurobiol. Aging 28, 327–335 (2007).
Theofilas, P., Dunlop, S., Heinsen, H. & Grinberg, L. T. Turning on the light within: subcortical nuclei of the isodentritic core and their role in Alzheimer's disease pathogenesis. J. Alzheimers Dis. 46, 17–34 (2015).
Simic, G. et al. Monoaminergic neuropathology in Alzheimer's disease. Prog. Neurobiol. 151, 101–138 (2017).
Petkova, A. T. et al. Self-propagating, molecular-level polymorphism in Alzheimer's β-amyloid fibrils. Science 307, 262–265 (2005).
Watts, J. C. et al. Serial propagation of distinct strains of Aβ prions from Alzheimer's disease patients. Proc. Natl Acad. Sci. USA 111, 10323–10328 (2014).
Cohen, M. L. et al. Rapidly progressive Alzheimer's disease features distinct structures of amyloid-β. Brain 138, 1009–1022 (2015).
Qiang, W., Yau, W. M., Lu, J. X., Collinge, J. & Tycko, R. Structural variation in amyloid-β fibrils from Alzheimer's disease clinical subtypes. Nature 541, 217–221 (2017).
Dubois, B. et al. Advancing research diagnostic criteria for Alzheimer's disease: the IWG-2 criteria. Lancet Neurol. 13, 614–629 (2014).
Petersen, R. C. et al. Mild cognitive impairment: clinical characterization and outcome. Arch. Neurol. 56, 303–308 (1999).
Albert, M. S. et al. The diagnosis of mild cognitive impairment due to Alzheimer's disease: recommendations from the National Institute on Aging–Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 7, 270–279 (2011).
Bertram, L. & Tanzi, R. E. The genetics of Alzheimer's disease. Prog. Mol. Biol. Transl Sci. 107, 79–100 (2012).
Bertram, L., Lill, C. M. & Tanzi, R. E. The genetics of Alzheimer disease: back to the future. Neuron 68, 270–281 (2010).
Bateman, R. J. et al. Clinical and biomarker changes in dominantly inherited Alzheimer's disease. N. Engl. J. Med. 367, 795–804 (2012).
Farrer, L. A. et al. Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease. A meta-analysis. APOE and Alzheimer Disease Meta Analysis Consortium. JAMA 278, 1349–1356 (1997).
Chouraki, V. & Seshadri, S. Genetics of Alzheimer's disease. Adv. Genet. 87, 245–294 (2014).
Desikan, R. S. et al. Genetic assessment of age-associated Alzheimer disease risk: development and validation of a polygenic hazard score. PLoS Med. 14, e1002258 (2017).
Baumgart, M. et al. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: a population-based perspective. Alzheimers Dement. 11, 718–726 (2015).
Osorio, R. S. et al. Sleep-disordered breathing advances cognitive decline in the elderly. Neurology 84, 1964–1971 (2015).
Iturria-Medina, Y. et al. Early role of vascular dysregulation on late-onset Alzheimer's disease based on multifactorial data-driven analysis. Nat. Commun. 7, 11934 (2016).
Haight, T. J. et al. Dissociable effects of Alzheimer disease and white matter hyperintensities on brain metabolism. JAMA Neurol. 70, 1039–1045 (2013).
Janelidze, S. et al. CSF Aβ42/Aβ40 and Aβ42/Aβ38 ratios: better diagnostic markers of Alzheimer disease. Ann. Clin. Transl. Neurol. 3, 154–165 (2016).
Ewers, M. et al. CSF biomarkers for the differential diagnosis of Alzheimer's disease: a large-scale international multicenter study. Alzheimers Dement. 11, 1306–1315 (2015).
Jack, C. R. Jr & Holtzman, D. M. Biomarker modeling of Alzheimer's disease. Neuron 80, 1347–1358 (2013).
Tosun, D. et al. Association between tau deposition and antecedent amyloid-beta accumulation rates in normal and early symptomatic individuals. Brain 140, 1499–1512 (2017).
Johnson, K. A. et al. Tau positron emission tomographic imaging in aging and early Alzheimer disease. Ann. Neurol. 79, 110–119 (2016).
Scholl, M. et al. PET imaging of tau deposition in the aging human brain. Neuron 89, 971–982 (2016).
Schwarz, A. J. et al. Regional profiles of the candidate tau PET ligand 18F-AV-1451 recapitulate key features of Braak histopathological stages. Brain 139, 1539–1550 (2016).
Cho, H. et al. In vivo cortical spreading pattern of tau and amyloid in the Alzheimer disease spectrum. Ann. Neurol. 80, 247–258 (2016).
Wang, Y. et al. Development of a PET/SPECT agent for amyloid imaging in Alzheimer's disease. J. Mol. Neurosci. 24, 55–62 (2004).
Garibotto, V. et al. Clinical validity of brain fluorodeoxyglucose positron emission tomography as a biomarker for Alzheimer's disease in the context of a structured 5-phase development framework. Neurobiol. Aging 52, 183–195 (2017).
Ten Kate, M. et al. Clinical validity of medial temporal atrophy as a biomarker for Alzheimer's disease in the context of a structured 5-phase development framework. Neurobiol. Aging 52, 167–182.e1 (2017).
Chetelat, G. et al. Atrophy, hypometabolism and clinical trajectories in patients with amyloid-negative Alzheimer's disease. Brain 139, 2528–2539 (2016).
Reitz, C., Brayne, C. & Mayeux, R. Epidemiology of Alzheimer disease. Nat. Rev. Neurol. 7, 137–152 (2011).
Tang, M. et al. Neurological manifestations of autosomal dominant familial Alzheimer's disease: a comparison of the published literature with the Dominantly Inherited Alzheimer Network observational study (DIAN-OBS). Lancet Neurol. 15, 1317–1325 (2016).
Joshi, A., Ringman, J. M., Lee, A. S., Juarez, K. O. & Mendez, M. F. Comparison of clinical characteristics between familial and non-familial early onset Alzheimer's disease. J. Neurol. 259, 2182–2188 (2012).
Mendez, M. F., Lee, A. S., Joshi, A. & Shapira, J. S. Nonamnestic presentations of early-onset Alzheimer's disease. Am. J. Alzheimers Dis. Other Demen. 27, 413–420 (2012).
Mesulam, M. Primary progressive aphasia pathology. Ann. Neurol. 63, 124–125 (2008).
Mesulam, M. et al. Alzheimer and frontotemporal pathology in subsets of primary progressive aphasia. Ann. Neurol. 63, 709–719 (2008).
Gorno-Tempini, M. L. et al. Classification of primary progressive aphasia and its variants. Neurology 76, 1006–1014 (2011).
Spinelli, E. G. et al. Typical and atypical pathology in primary progressive aphasia variants. Ann. Neurol. 81, 430–443 (2017).
Benson, D. F., Davis, R. J. & Snyder, B. D. Posterior cortical atrophy. Arch. Neurol. 45, 789–793 (1988).
Crutch, S. J. et al. Shining a light on posterior cortical atrophy. Alzheimers Dement. 9, 463–465 (2013).
Whitwell, J. L. et al. Imaging correlates of posterior cortical atrophy. Neurobiol. Aging 28, 1051–1061 (2007).
Ossenkoppele, R. et al. The behavioural/dysexecutive variant of Alzheimer's disease: clinical, neuroimaging and pathological features. Brain 138, 2732–2749 (2015).
Binetti, G. et al. Disorders of visual and spatial perception in the early stage of Alzheimer's disease. Ann. NY Acad. Sci. 777, 221–225 (1996).
Back-Madruga, C. et al. Functional ability in executive variant Alzheimer's disease and typical Alzheimer's disease. Clin. Neuropsychol. 16, 331–340 (2002).
Snowden, J. S. et al. Cognitive phenotypes in Alzheimer's disease and genetic risk. Cortex 43, 835–845 (2007).
Dickerson, B. C., Wolk, D. A. & Alzheimer's Disease Neuroimaging Initiative. Dysexecutive versus amnesic phenotypes of very mild Alzheimer's disease are associated with distinct clinical, genetic and cortical thinning characteristics. J. Neurol. Neurosurg. Psychiatry 82, 45–51 (2011).
Mez, J. et al. Faster cognitive and functional decline in dysexecutive versus amnestic Alzheimer's subgroups: a longitudinal analysis of the National Alzheimer's Coordinating Center (NACC) database. PLoS ONE 8, e65246 (2013).
Varma, A. R. et al. Evaluation of the NINCDS–ADRDA criteria in the differentiation of Alzheimer's disease and frontotemporal dementia. J. Neurol. Neurosurg. Psychiatry 66, 184–188 (1999).
Rabinovici, G. D. et al. Amyloid versus FDG–PET in the differential diagnosis of AD and FTLD. Neurology 77, 2034–2042 (2011).
Ossenkoppele, R. et al. Impact of molecular imaging on the diagnostic process in a memory clinic. Alzheimers Dement. 9, 414–421 (2013).
Ryman, D. C. et al. Symptom onset in autosomal dominant Alzheimer disease: a systematic review and meta-analysis. Neurology 83, 253–260 (2014).
Grinberg, L. T. & Thal, D. R. Vascular pathology in the aged human brain. Acta Neuropathol. 119, 277–290 (2010).
Jellinger, K. A. The pathology of “vascular dementia”: a critical update. J. Alzheimers Dis. 14, 107–123 (2008).
Jellinger, K. A. Pathology and pathogenesis of vascular cognitive impairment — a critical update. Front. Aging Neurosci. 5, 17 (2013).
Gorelick, P. B. et al. Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 42, 2672–2713 (2011).
Kalaria, R. N. et al. Towards defining the neuropathological substrates of vascular dementia. J. Neurol. Sci. 226, 75–80 (2004).
Wardlaw, J. M. Blood–brain barrier and cerebral small vessel disease. J. Neurol. Sci. 299, 66–71 (2010).
Wardlaw, J. M. et al. Blood–brain barrier permeability and long-term clinical and imaging outcomes in cerebral small vessel disease. Stroke 44, 525–527 (2013).
Kalaria, R. N. Neuropathological diagnosis of vascular cognitive impairment and vascular dementia with implications for Alzheimer's disease. Acta Neuropathol. 131, 659–685 (2016).
McAleese, K. E. et al. Post-mortem assessment in vascular dementia: advances and aspirations. BMC Med. 14, 129 (2016).
Pendlebury, S. T. & Rothwell, P. M. Prevalence, incidence, and factors associated with pre-stroke and post-stroke dementia: a systematic review and meta-analysis. Lancet Neurol. 8, 1006–1018 (2009).
Zaccai, J., Ince, P. & Brayne, C. Population-based neuropathological studies of dementia: design, methods and areas of investigation — a systematic review. BMC Neurol. 6, 2 (2006).
Grinberg, L. T. et al. Prevalence of dementia subtypes in a developing country: a clinicopathological study. Clinics (Sao Paulo) 68, 1140–1145 (2013).
Korczyn, A. D. The complex nosological concept of vascular dementia. J. Neurol. Sci. 203–204, 3–6 (2002).
James, B. D., Bennett, D. A., Boyle, P. A., Leurgans, S. & Schneider, J. A. Dementia from Alzheimer disease and mixed pathologies in the oldest old. JAMA 307, 1798–1800 (2012).
Hachinski, V. Preventable senility: a call for action against the vascular dementias. Lancet 340, 645–648 (1992).
Small, G. W. Revised Ischemic Score for diagnosing multi-infarct dementia. J. Clin. Psychiatry 46, 514–517 (1985).
Roman, G. C. et al. Vascular dementia: diagnostic criteria for research studies. Report of the NINDS–AIREN International Workshop. Neurology 43, 250–260 (1993).
Sachdev, P. et al. Diagnostic criteria for vascular cognitive disorders: a VASCOG statement. Alzheimer Dis. Assoc. Disord. 28, 206–218 (2014).
Garrett, K. D. et al. The neuropsychological profile of vascular cognitive impairment — no dementia: comparisons to patients at risk for cerebrovascular disease and vascular dementia. Arch. Clin. Neuropsychol. 19, 745–757 (2004).
Rosenberg, G. A. et al. Consensus statement for diagnosis of subcortical small vessel disease. J. Cereb. Blood Flow Metab. 36, 6–25 (2016).
Wardlaw, J. M. et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 12, 822–838 (2013).
Rosenberg, G. A., Bjerke, M. & Wallin, A. Multimodal markers of inflammation in the subcortical ischemic vascular disease type of vascular cognitive impairment. Stroke 45, 1531–1538 (2014).
Brun, A. Frontal lobe degeneration of non-Alzheimer type. I. Neuropathology. Arch. Gerontol. Geriatr. 6, 193–208 (1987).
Rascovsky, K. et al. Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain 134, 2456–2477 (2011).
Neumann, M. et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314, 130–133 (2006).
Baborie, A. et al. Pathological correlates of frontotemporal lobar degeneration in the elderly. Acta Neuropathol. 121, 365–371 (2011).
Mackenzie, I. R. et al. Nomenclature and nosology for neuropathologic subtypes of frontotemporal lobar degeneration: an update. Acta Neuropathol. 119, 1–4 (2010).
Mackenzie, I. R. et al. Nomenclature for neuropathologic subtypes of frontotemporal lobar degeneration: consensus recommendations. Acta Neuropathol. 117, 15–18 (2009).
Neumann, M. et al. A new subtype of frontotemporal lobar degeneration with FUS pathology. Brain 132, 2922–2931 (2009).
Lee, S. E. et al. Clinical characterization of bvFTD due to FUS neuropathology. Neurocase 18, 305–317 (2012).
Johnson, J. K. et al. Frontotemporal lobar degeneration: demographic characteristics of 353 patients. Arch. Neurol. 62, 925–930 (2005).
Yokoyama, J. S., Sirkis, D. W. & Miller, B. L. C9ORF72 hexanucleotide repeats in behavioral and motor neuron disease: clinical heterogeneity and pathological diversity. Am. J. Neurodegener. Dis. 3, 1–18 (2014).
Goldman, J. S. et al. Comparison of family histories in FTLD subtypes and related tauopathies. Neurology 65, 1817–1819 (2005).
Hodges, J. R. et al. Semantic dementia: demography, familial factors and survival in a consecutive series of 100 cases. Brain 133, 300–306 (2010).
Snowden, J. S. et al. Progranulin gene mutations associated with frontotemporal dementia and progressive non-fluent aphasia. Brain 129, 3091–3102 (2006).
Mesulam, M. et al. Progranulin mutations in primary progressive aphasia: the PPA1 and PPA3 families. Arch. Neurol. 64, 43–47 (2007).
Beck, J. et al. A distinct clinical, neuropsychological and radiological phenotype is associated with progranulin gene mutations in a large UK series. Brain 131, 706–720 (2008).
DeJesus-Hernandez, M. et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron 72, 245–256 (2011).
Freischmidt, A. et al. Haploinsufficiency of TBK1 causes familial ALS and fronto-temporal dementia. Nat. Neurosci. 18, 631–636 (2015).
Baker, M. et al. Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17. Nature 442, 916–919 (2006).
Chen-Plotkin, A. S. et al. Genetic and clinical features of progranulin-associated frontotemporal lobar degeneration. Arch. Neurol. 68, 488–497 (2011).
Snowden, J. S. et al. Distinct clinical and pathological phenotypes in frontotemporal dementia associated with MAPT, PGRN and C9orf72 mutations. Amyotroph. Lateral Scler. Frontotemporal Degener. 16, 497–505 (2015).
Watts, G. D. et al. Inclusion body myopathy associated with Paget disease of bone and frontotemporal dementia is caused by mutant valosin-containing protein. Nat. Genet. 36, 377–381 (2004).
Watts, G. D. et al. Novel VCP mutations in inclusion body myopathy associated with Paget disease of bone and frontotemporal dementia. Clin. Genet. 72, 420–426 (2007).
Rademakers, R. et al. High-density SNP haplotyping suggests altered regulation of tau gene expression in progressive supranuclear palsy. Hum. Mol. Genet. 14, 3281–3292 (2005).
Lanata, S. C. & Miller, B. L. The behavioural variant frontotemporal dementia (bvFTD) syndrome in psychiatry. J. Neurol. Neurosurg. Psychiatry 87, 501–511 (2016).
Mioshi, E., Bristow, M., Cook, R. & Hodges, J. R. Factors underlying caregiver stress in frontotemporal dementia and Alzheimer's disease. Dement. Geriatr. Cogn. Disord. 27, 76–81 (2009).
Wilson, R. S. et al. Life-span cognitive activity, neuropathologic burden, and cognitive aging. Neurology 81, 314–321 (2013).
Onyike, C. U. & Diehl-Schmid, J. The epidemiology of frontotemporal dementia. Int. Rev. Psychiatry 25, 130–137 (2013).
Coyle-Gilchrist, I. T. et al. Prevalence, characteristics, and survival of frontotemporal lobar degeneration syndromes. Neurology 86, 1736–1743 (2016).
Seeley, W. W. et al. Frontal paralimbic network atrophy in very mild behavioral variant frontotemporal dementia. Arch. Neurol. 65, 249–255 (2008).
Garcin, B. et al. Determinants of survival in behavioral variant frontotemporal dementia. Neurology 73, 1656–1661 (2009).
Diehl-Schmid, J., Perneczky, R., Koch, J., Nedopil, N. & Kurz, A. Guilty by suspicion? Criminal behavior in frontotemporal lobar degeneration. Cogn. Behav. Neurol. 26, 73–77 (2013).
Patel, A. N. & Sampson, J. B. Cognitive profile of C9orf72 in frontotemporal dementia and amyotrophic lateral sclerosis. Curr. Neurol. Neurosci. Rep. 15, 59 (2015).
Pletnikova, O. et al. Hippocampal sclerosis dementia with the C9ORF72 hexanucleotide repeat expansion. Neurobiol. Aging 35, 2419.e17–2419.e21 (2014).
Rankin, K. P. et al. Spontaneous social behaviors discriminate behavioral dementias from psychiatric disorders and other dementias. J. Clin. Psychiatry 69, 60–73 (2008).
Velakoulis, D., Walterfang, M., Mocellin, R., Pantelis, C. & McLean, C. Frontotemporal dementia presenting as schizophrenia-like psychosis in young people: clinicopathological series and review of cases. Br. J. Psychiatry 194, 298–305 (2009).
Kertesz, A. et al. Psychosis and hallucinations in frontotemporal dementia with the C9ORF72 mutation: a detailed clinical cohort. Cogn. Behav. Neurol. 26, 146–154 (2013).
Woolley, J. D. et al. Frontotemporal dementia and mania. Am. J. Psychiatry 164, 1811–1816 (2007).
Rosen, H. J. et al. Neuroanatomical correlates of behavioural disorders in dementia. Brain 128, 2612–2625 (2005).
Rankin, K. P. et al. Behavioral variant frontotemporal dementia with corticobasal degeneration pathology: phenotypic comparison to bvFTD with Pick's disease. J. Mol. Neurosci. 45, 594–608 (2011).
Lee, S. E. et al. Altered network connectivity in frontotemporal dementia with C9orf72 hexanucleotide repeat expansion. Brain 137, 3047–3060 (2014).
Khan, B. K. et al. Atypical, slowly progressive behavioural variant frontotemporal dementia associated with C9ORF72 hexanucleotide expansion. J. Neurol. Neurosurg. Psychiatry 83, 358–364 (2012).
Ranasinghe, K. G. et al. Distinct subtypes of behavioral variant frontotemporal dementia based on patterns of network degeneration. JAMA Neurol. 73, 1078–1088 (2016).
Galantucci, S. et al. White matter damage in primary progressive aphasias: a diffusion tensor tractography study. Brain 134, 3011–3029 (2011).
Rogalski, E. et al. Asymmetry of cortical decline in subtypes of primary progressive aphasia. Neurology 83, 1184–1191 (2014).
Mesulam, M. M. et al. Primary progressive aphasia and the evolving neurology of the language network. Nat. Rev. Neurol. 10, 554–569 (2014).
Gorno-Tempini, M. L. et al. Cognition and anatomy in three variants of primary progressive aphasia. Ann. Neurol. 55, 335–346 (2004).
Kramer, J. H. et al. Distinctive neuropsychological patterns in frontotemporal dementia, semantic dementia, and Alzheimer disease. Cogn. Behav. Neurol. 16, 211–218 (2003).
Edwards-Lee, T. et al. The temporal variant of frontotemporal dementia. Brain 120, 1027–1040 (1997).
Seeley, W. W. et al. The natural history of temporal variant frontotemporal dementia. Neurology 64, 1384–1390 (2005).
Chan, D. et al. The clinical profile of right temporal lobe atrophy. Brain 132, 1287–1298 (2009).
Geschwind, N. & Galaburda, A. M. Cerebral lateralization. Biological mechanisms, associations, and pathology: III. A hypothesis and a program for research. Arch. Neurol. 42, 634–654 (1985).
Gorno-Tempini, M. L. et al. Cognitive and behavioral profile in a case of right anterior temporal lobe neurodegeneration. Cortex 40, 631–644 (2004).
Snowden, J. S. et al. The clinical diagnosis of early-onset dementias: diagnostic accuracy and clinicopathological relationships. Brain 134, 2478–2492 (2011).
Mesulam, M. M., Wieneke, C., Thompson, C., Rogalski, E. & Weintraub, S. Quantitative classification of primary progressive aphasia at early and mild impairment stages. Brain 135, 1537–1553 (2012).
Mesulam, M. M. et al. Asymmetry and heterogeneity of Alzheimer's and frontotemporal pathology in primary progressive aphasia. Brain 137, 1176–1192 (2014).
Gorno-Tempini, M. L., Murray, R. C., Rankin, K. P., Weiner, M. W. & Miller, B. L. Clinical, cognitive and anatomical evolution from nonfluent progressive aphasia to corticobasal syndrome: a case report. Neurocase 10, 426–436 (2004).
Lomen-Hoerth, C., Anderson, T. & Miller, B. The overlap of amyotrophic lateral sclerosis and frontotemporal dementia. Neurology 59, 1077–1079 (2002).
Burrell, J. R., Kiernan, M. C., Vucic, S. & Hodges, J. R. Motor neuron dysfunction in frontotemporal dementia. Brain 134, 2582–2594 (2011).
Strong, M. J. et al. Consensus criteria for the diagnosis of frontotemporal cognitive and behavioural syndromes in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 10, 131–146 (2009).
Devenney, E., Vucic, S., Hodges, J. R. & Kiernan, M. C. Motor neuron disease–frontotemporal dementia: aclinical continuum. Expert Rev. Neurother. 15, 509–522 (2015).
Höglinger, G. U. et al. Clinical diagnosis of progressive supranuclear palsy: the Movement Disorder Society criteria. Mov. Disord. 32, 853–864 (2017).
Williams, D. R. et al. Pathological tau burden and distribution distinguishes progressive supranuclear palsy-parkinsonism from Richardson's syndrome. Brain 130, 1566–1576 (2007).
Williams, D. R. et al. Characteristics of two distinct clinical phenotypes in pathologically proven progressive supranuclear palsy: Richardson's syndrome and PSP-parkinsonism. Brain 128, 1247–1258 (2005).
Boxer, A. L. et al. Saccade abnormalities in autopsy-confirmed frontotemporal lobar degeneration and Alzheimer disease. Arch. Neurol. 69, 509–517 (2012).
Massey, L. A. et al. The midbrain to pons ratio: a simple and specific MRI sign of progressive supranuclear palsy. Neurology 80, 1856–1861 (2013).
Lee, S. E. et al. Clinicopathological correlations in corticobasal degeneration. Ann. Neurol. 70, 327–340 (2011).
Armstrong, M. J. et al. Criteria for the diagnosis of corticobasal degeneration. Neurology 80, 496–503 (2013).
Sha, S. J. et al. Predicting amyloid status in corticobasal syndrome using modified clinical criteria, magnetic resonance imaging and fluorodeoxyglucose positron emission tomography. Alzheimers Res. Ther. 7, 8 (2015).
George, S., Rey, N. L., Reichenbach, N., Steiner, J. A. & Brundin, P. α-synuclein: the long distance runner. Brain Pathol. 23, 350–357 (2013).
Braak, H. et al. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol. Aging 24, 197–211 (2003).
Mulak, A. & Bonaz, B. Brain–gut–microbiota axis in Parkinson's disease. World J. Gastroenterol. 21, 10609–10620 (2015).
Svensson, E. et al. Vagotomy and subsequent risk of Parkinson's disease. Ann. Neurol. 78, 522–529 (2015).
Jellinger, K. A. & Attems, J. Prevalence and pathology of dementia with Lewy bodies in the oldest old: a comparison with other dementing disorders. Dement. Geriatr. Cogn. Disord. 31, 309–316 (2011).
Walker, L. et al. Neuropathologically mixed Alzheimer's and Lewy body disease: burden of pathological protein aggregates differs between clinical phenotypes. Acta Neuropathol. 129, 729–748 (2015).
Barnes, L. L. et al. Mixed pathology is more likely in black than white decedents with Alzheimer dementia. Neurology 85, 528–534 (2015).
Hall, H. et al. Hippocampal Lewy pathology and cholinergic dysfunction are associated with dementia in Parkinson's disease. Brain 137, 2493–2508 (2014).
Horvath, J., Herrmann, F. R., Burkhard, P. R., Bouras, C. & Kovari, E. Neuropathology of dementia in a large cohort of patients with Parkinson's disease. Parkinsonism Relat. Disord. 19, 864–868 (2013).
Irwin, D. J., Lee, V. M. & Trojanowski, J. Q. Parkinson's disease dementia: convergence of α-synuclein, tau and amyloid-β pathologies. Nat. Rev. Neurosci. 14, 626–636 (2013).
Meeus, B. et al. DLB and PDD: a role for mutations in dementia and Parkinson disease genes? Neurobiol. Aging 33, 629.e5–629.e18 (2012).
Tsuang, D. et al. APOE ε4 increases risk for dementia in pure synucleinopathies. JAMA Neurol. 70, 223–228 (2013).
Berge, G., Sando, S. B., Rongve, A., Aarsland, D. & White, L. R. Apolipoprotein E ε2 genotype delays onset of dementia with Lewy bodies in a Norwegian cohort. J. Neurol. Neurosurg. Psychiatry 85, 1227–1231 (2014).
Hyun, C. H., Yoon, C. Y., Lee, H. J. & Lee, S. J. LRRK2 as a potential genetic modifier of synucleinopathies: interlacing the two major genetic factors of Parkinson's disease. Exp. Neurobiol. 22, 249–257 (2013).
Bras, J. et al. Genetic analysis implicates APOE. SNCA and suggests lysosomal dysfunction in the etiology of dementia with Lewy bodies. Hum. Mol. Genet. 23, 6139–6146 (2014).
Nalls, M. A. et al. A multicenter study of glucocerebrosidase mutations in dementia with Lewy bodies. JAMA Neurol. 70, 727–735 (2013).
McKeith, I. Dementia with Lewy bodies and Parkinson's disease with dementia: where two worlds collide. Pract. Neurol. 7, 374–382 (2007).
Parkinson, J. An essay on the shaking palsy. 1817. J. Neuropsychiatry Clin. Neurosci. 14, 223–236 (2002).
Albanese, A. Diagnostic criteria for Parkinson's disease. Neurol. Sci. 24 (Suppl. 1), S23–S26 (2003).
Sauerbier, A., Jenner, P., Todorova, A. & Chaudhuri, K. R. Non motor subtypes and Parkinson's disease. Parkinsonism Relat. Disord. 22 (Suppl. 1), S41–S46 (2016).
Aarsland, D. et al. Cognitive impairment in incident, untreated Parkinson disease: the Norwegian ParkWest study. Neurology 72, 1121–1126 (2009).
Gaig, C. et al. Rapidly progressive diffuse Lewy body disease. Mov. Disord. 26, 1316–1323 (2011).
Schneider, J. A. et al. Cognitive impairment, decline and fluctuations in older community-dwelling subjects with Lewy bodies. Brain 135, 3005–3014 (2012).
Tiraboschi, P. et al. What best differentiates Lewy body from Alzheimer's disease in early-stage dementia? Brain 129, 729–735 (2006).
Cagnin, A. et al. High specificity of MMSE pentagon scoring for diagnosis of prodromal dementia with Lewy bodies. Parkinsonism Relat. Disord. 21, 303–305 (2015).
Hamilton, J. M. et al. Early visuospatial deficits predict the occurrence of visual hallucinations in autopsy-confirmed dementia with Lewy bodies. Am. J. Geriatr. Psychiatry 20, 773–781 (2012).
Boot, B. P. et al. Risk factors for dementia with Lewy bodies: a case–control study. Neurology 81, 833–840 (2013).
Postuma, R. B., Gagnon, J. F., Pelletier, A. & Montplaisir, J. Prodromal autonomic symptoms and signs in Parkinson's disease and dementia with Lewy bodies. Mov. Disord. 28, 597–604 (2013).
Auning, E. et al. Early and presenting symptoms of dementia with lewy bodies. Dement. Geriatr. Cogn. Disord. 32, 202–208 (2011).
Chiba, Y. et al. Retrospective survey of prodromal symptoms in dementia with Lewy bodies: comparison with Alzheimer's disease. Dement. Geriatr. Cogn. Disord. 33, 273–281 (2012).
Ferman, T. J. et al. Inclusion of RBD improves the diagnostic classification of dementia with Lewy bodies. Neurology 77, 875–882 (2011).
Schenck, C. H., Boeve, B. F. & Mahowald, M. W. Delayed emergence of a parkinsonian disorder or dementia in 81% of older men initially diagnosed with idiopathic rapid eye movement sleep behavior disorder: a 16-year update on a previously reported series. Sleep Med. 14, 744–748 (2013).
Litvan, I. et al. Diagnostic criteria for mild cognitive impairment in Parkinson's disease: Movement Disorder Society Task Force guidelines. Mov. Disord. 27, 349–356 (2012).
Svenningsson, P., Westman, E., Ballard, C. & Aarsland, D. Cognitive impairment in patients with Parkinson's disease: diagnosis, biomarkers, and treatment. Lancet Neurol. 11, 697–707 (2012).
Aarsland, D., Zaccai, J. & Brayne, C. A systematic review of prevalence studies of dementia in Parkinson's disease. Mov. Disord. 20, 1255–1263 (2005).
Janvin, C. C., Larsen, J. P., Aarsland, D. & Hugdahl, K. Subtypes of mild cognitive impairment in Parkinson's disease: progression to dementia. Mov. Disord. 21, 1343–1349 (2006).
Emre, M. et al. Clinical diagnostic criteria for dementia associated with Parkinson's disease. Mov. Disord. 22, 1689–1707 (2007).
Olde Dubbelink, K. T. et al. Predicting dementia in Parkinson disease by combining neurophysiologic and cognitive markers. Neurology 82, 263–270 (2014).
Mok, W., Chow, T. W., Zheng, L., Mack, W. J. & Miller, C. Clinicopathological concordance of dementia diagnoses by community versus tertiary care clinicians. Am. J. Alzheimers Dis. Other Demen. 19, 161–165 (2004).
Toledo, J. B. et al. Clinical and multimodal biomarker correlates of ADNI neuropathological findings. Acta Neuropathol. Commun. 1, 65 (2013).
Kehagia, A. A., Barker, R. A. & Robbins, T. W. Neuropsychological and clinical heterogeneity of cognitive impairment and dementia in patients with Parkinson's disease. Lancet Neurol. 9, 1200–1213 (2010).
Williams-Gray, C. H. et al. The CamPaIGN study of Parkinson's disease: 10-year outlook in an incident population-based cohort. J. Neurol. Neurosurg. Psychiatry 84, 1258–1264 (2013).
Teune, L. K. et al. Typical cerebral metabolic patterns in neurodegenerative brain diseases. Mov. Disord. 25, 2395–2404 (2010).
Jokinen, P. et al. [11C]PIB-, [18F]FDG–PET and MRI imaging in patients with Parkinson's disease with and without dementia. Parkinsonism Relat. Disord. 16, 666–670 (2010).
Lim, S. M. et al. The 18F-FDG PET cingulate island sign and comparison to 123I-β-CIT SPECT for diagnosis of dementia with Lewy bodies. J. Nucl. Med. 50, 1638–1645 (2009).
Lim, X., Yeo, J. M., Green, A. & Pal, S. The diagnostic utility of cerebrospinal fluid alpha-synuclein analysis in dementia with Lewy bodies — a systematic review and meta-analysis. Parkinsonism Relat. Disord. 19, 851–858 (2013).
Kantarci, K. et al. Antemortem amyloid imaging and β-amyloid pathology in a case with dementia with Lewy bodies. Neurobiol. Aging 33, 878–885 (2012).
Brunnstrom, H., Hansson, O., Zetterberg, H., Londos, E. & Englund, E. Correlations of CSF tau and amyloid levels with Alzheimer pathology in neuropathologically verified dementia with Lewy bodies. Int. J. Geriatr. Psychiatry 28, 738–744 (2013).
Thomas, A. J. et al. Autopsy validation of 123I-FP-CIT dopaminergic neuroimaging for the diagnosis of DLB. Neurology 88, 276–283 (2017).
Prusiner, S. B. Prions. Proc. Natl Acad. Sci. USA 95, 13363–13383 (1998).
Safar, J., Roller, P. P., Gajdusek, D. C. & Gibbs, C. J. Jr. Conformational transitions, dissociation, and unfolding of scrapie amyloid (prion) protein. J. Biol. Chem. 268, 20276–20284 (1993).
Puoti, G. et al. Sporadic human prion diseases: molecular insights and diagnosis. Lancet Neurol. 11, 618–628 (2012).
Brown, K. & Mastrianni, J. A. The prion diseases. J. Geriatr. Psychiatry Neurol. 23, 277–298 (2010).
Heath, C. A. et al. Diagnosing variant Creutzfeldt–Jakob disease: a retrospective analysis of the first 150 cases in the UK. J. Neurol. Neurosurg. Psychiatry 82, 646–651 (2011).
Brown, P., Brandel, J. P., Preece, M. & Sato, T. Iatrogenic Creutzfeldt–Jakob disease: the waning of an era. Neurology 67, 389–393 (2006).
Will, R. G. Acquired prion disease: iatrogenic CJD, variant CJD, kuru. Br. Med. Bull. 66, 255–265 (2003).
Yamada, M. et al. An inherited prion disease with a PrP P105L mutation: clinicopathologic and PrP heterogeneity. Neurology 53, 181–188 (1999).
Giaccone, G. et al. Neurofibrillary tangles of the Indiana kindred of Gerstmann–Straussler–Scheinker disease share antigenic determinants with those of Alzheimer disease. Brain Res. 530, 325–329 (1990).
Gambetti, P., Kong, Q., Zou, W., Parchi, P. & Chen, S. G. Sporadic and familial CJD: classification and characterisation. Br. Med. Bull. 66, 213–239 (2003).
Tsuji, S. & Kuroiwa, Y. Creutzfeldt–Jakob disease in Japan. Neurology 33, 1503–1506 (1983).
Will, R. G. & Matthews, W. B. A retrospective study of Creutzfeldt-Jakob disease in England and Wales 1970–1979. I: clinical features. J. Neurol. Neurosurg. Psychiatry 47, 134–140 (1984).
Brown, P., Cathala, F., Castaigne, P. & Gajdusek, D. C. Creutzfeldt–Jakob disease: clinical analysis of a consecutive series of 230 neuropathologically verified cases. Ann. Neurol. 20, 597–602 (1986).
Rabinovici, G. D. et al. First symptom in sporadic Creutzfeldt–Jakob disease. Neurology 66, 286–287 (2006).
Krasnianski, A. et al. Clinical findings and diagnostic tests in the MV2 subtype of sporadic CJD. Brain 129, 2288–2296 (2006).
Goldfarb, L. G. et al. Fatal familial insomnia and familial Creutzfeldt–Jakob disease: disease phenotype determined by a DNA polymorphism. Science 258, 806–808 (1992).
Parchi, P. et al. Classification of sporadic Creutzfeldt–Jakob disease based on molecular and phenotypic analysis of 300 subjects. Ann. Neurol. 46, 224–233 (1999).
Hill, A. F. et al. Investigation of variant Creutzfeldt–Jakob disease and other human prion diseases with tonsil biopsy samples. Lancet 353, 183–189 (1999).
Steinhoff, B. J. et al. Diagnostic value of periodic complexes in Creutzfeldt–Jakob disease. Ann. Neurol. 56, 702–708 (2004).
Forner, S. A. et al. Comparing CSF biomarkers and brain MRI in the diagnosis of sporadic Creutzfeldt–Jakob disease. Neurol. Clin. Pract. 5, 116–125 (2015).
Martindale, J. et al. Sporadic Creutzfeldt–Jakob disease mimicking variant Creutzfeldt–Jakob disease. Arch. Neurol. 60, 767–770 (2003).
Hamlin, C. et al. A comparison of tau and 14-3-3 protein in the diagnosis of Creutzfeldt–Jakob disease. Neurology 79, 547–552 (2012).
Kim, M. O. & Geschwind, M. D. Clinical update of Jakob–Creutzfeldt disease. Curr. Opin. Neurol. 28, 302–310 (2015).
Stoeck, K. et al. Cerebrospinal fluid biomarker supported diagnosis of Creutzfeldt–Jakob disease and rapid dementias: a longitudinal multicentre study over 10 years. Brain 135, 3051–3061 (2012).
Foutz, A. et al. Diagnostic and prognostic value of human prion detection in cerebrospinal fluid. Ann. Neurol. 81, 79–92 (2017).
McGuire, L. I. et al. Cerebrospinal fluid real-time quaking-induced conversion is a robust and reliable test for sporadic Creutzfeldt–Jakob disease: an international study. Ann. Neurol. 80, 160–165 (2016).
Lehmann, M. et al. Intrinsic connectivity networks in healthy subjects explain clinical variability in Alzheimer's disease. Proc. Natl Acad. Sci. USA 110, 11606–11611 (2013).
Smith, B. N. et al. The C9ORF72 expansion mutation is a common cause of ALS+/−FTD in Europe and has a single founder. Eur. J. Hum. Genet. 21, 102–108 (2013).
Snowden, J. S. et al. Distinct clinical and pathological characteristics of frontotemporal dementia associated with C9ORF72 mutations. Brain 135, 693–708 (2012).
Author information
Authors and Affiliations
Contributions
F.M.E. researched data for the article and wrote the manuscript. Both authors made substantial contributions to discussions of the content, and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (box)
Diagnostic criteria for selected dementia syndromes. (DOC 72 kb)
Glossary
- Acalculia
-
Inability to perform calculations.
- Phonemic paraphasias
-
Errors in speech resulting in substitution of parts of the intended word by other phonemes, leading to generation of a — sometimes non-existent — word sounding similar to the target word (for example, pipe for pile, loan for moan, or papple for apple).
- Visual agnosia
-
Inability to recognize or interpret visual stimuli despite intact vision.
- Alexia
-
Inability to read, which comprises inability to read out loud and/or comprehend.
- Ideomotor apraxia
-
Deficit in the ability to voluntarily plan or complete a motor task, with preservation of involuntary (automatic) motor planning when the subject is cued. This preserved ability to perform automated motoric responses to cuing contrasts with 'ideational apraxia', in which the ability to select the appropriate motor programme or sequencial steps, even in the presence of cuing, is lost.
- Prosopagnosia
-
Inability to recognize faces, also known as 'face blindness'.
- Pseudobulbar affect
-
Also referred to as marked emotional lability or emotional incontinence. This symptom is characterized by uncontrollable episodes of crying or laughter, proportionately in excess of the valence of an emotional stimulus.
- Virchow–Robin spaces
-
Perivascular spaces surrounding the penetrating vessels that arise from the subarachnoid space and perforate the brain parenchyma. Prominence — in terms of visibility and numbers — of enlarged Virchow–Robin spaces has been associated with cognitive ageing, small vessel disease, and neurodegeneration.
- CADASIL
-
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is an autosomal dominantly inherited small vessel disease, with notable dysregulation of inflammatory markers and pathognomonic T2/FLAIR white matter hyperintensities in anterior temporal lobes.
- Anticipation
-
Genetic phenomenon relating to the gradual expansion of a mutation with each generation, usually resulting in earlier age of onset and more-severe symptoms when passed on to the next generation.
- Surface dyslexia and dysgraphia
-
Impairment in the ability to read and write words that are considered 'irregular' with regard to their spelling-to-sound correspondence (for example, friend, island or yacht), as opposed to 'regular' words (for example, fire, lemon or computer). This impairment can result in regularization errors, that is, words are erroneously spelled according to the regular phonetic rules.
- Associative agnosia
-
Impaired recognition of visually presented objects despite intact visual perception of these objects. Also known as 'visual object agnosia'.
- Hypergraphia
-
Compulsive and overwhelming urge to write, with potential intraindividual variability in style and content during the disease course.
- Astereognosis
-
Inability to recognize an object by active touch alone, in the absence of primary sensory deficit.
Rights and permissions
About this article
Cite this article
Elahi, F., Miller, B. A clinicopathological approach to the diagnosis of dementia. Nat Rev Neurol 13, 457–476 (2017). https://doi.org/10.1038/nrneurol.2017.96
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2017.96
This article is cited by
-
Social support and cognitive activity and their associations with incident cognitive impairment in cognitively normal older adults
BMC Geriatrics (2024)
-
Factors promoting and impeding efforts to deprescribe antidepressants among nursing home residents with dementia– a process evaluation guided by normalization process theory
BMC Nursing (2024)
-
Identification of clinical disease trajectories in neurodegenerative disorders with natural language processing
Nature Medicine (2024)
-
Age-specific prevalence of the different clinical presentations of AD and FTD in young-onset dementia
Journal of Neurology (2024)
-
The Association Between Neurocognitive Disorders and Gustatory Dysfunction: A Systematic Review and Meta-Analysis
Neuropsychology Review (2024)