Key Points
-
Multiple system atrophy (MSA) is a sporadic and rare neurodegenerative movement disorder with an estimated annual incidence of 0.6 per 100,000 (ORPHA102)
-
The clinical presentation is highly variable, with parkinsonism, cerebellar ataxia and autonomic failure being the most common — and often debilitating — symptoms
-
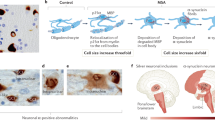
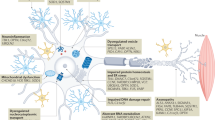
α-Synuclein-immunoreactive (oligodendro)glial cytoplasmic inclusions are the neuropathological hallmark of MSA
-
Permissive templating ('prion-like' propagation) of misfolded α-synuclein is currently thought to be the key event in the pathophysiological cascade
-
No disease-modifying therapies are currently available; symptomatic treatment options are limited, and the therapeutic benefits of these therapies is often only transient
-
International collaboration is gaining momentum with the recent formation of international study groups and novel funding opportunities dedicated exclusively to MSA research
Abstract
Multiple system atrophy (MSA) is a devastating and fatal neurodegenerative disorder. The clinical presentation of this disease is highly variable, with parkinsonism, cerebellar ataxia and autonomic failure being the most common — and often debilitating — symptoms. These symptoms progress rapidly, and patients die from MSA-related complications after 9 years of symptom duration on average. Unfortunately, the course of the disease cannot be improved by drug or surgical treatment. In addition, symptomatic treatment options are currently limited, and therapeutic benefits are often only transient. Thus, further interventional studies of candidate disease-modifying and symptomatic therapies are essential to improve patient care. In the past 15 years, the understanding of MSA-specific requirements in trial methodology has improved, resulting in a substantial increase in high-quality interventional studies. In this Review, we discuss MSA risk factors, clinical presentation and neuropathology, and we provide a hypothesis on key pathophysiological events, a summary of recent randomized controlled trials, and an overview of ongoing international collaborations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kollensperger, M. et al. Presentation, diagnosis, and management of multiple system atrophy in Europe: final analysis of the European multiple system atrophy registry. Mov. Disord. 25, 2604–2612 (2010).
Lin, D. J., Hermann, K. L. & Schmahmann, J. D. The diagnosis and natural history of multiple system atrophy, cerebellar type. Cerebellum 15, 663–679 (2016).
Wenning, G. K. et al. The natural history of multiple system atrophy: a prospective European cohort study. Lancet Neurol. 12, 264–274 (2013). A large prospective, multicentre cohort study from Europe that describes the natural history of MSA and studies predictors for aggressive disease course.
Low, P. A. et al. Natural history of multiple system atrophy in the USA: a prospective cohort study. Lancet Neurol. 14, 710–719 (2015). A large prospective, multicentre cohort study from the USA that explores the natural history and determining predictors of accelerated decline in MSA.
Trojanowski, J. Q., Revesz, T. & Neuropathology Working Group on MSA. Proposed neuropathological criteria for the post mortem diagnosis of multiple system atrophy. Neuropathol. Appl. Neurobiol. 33, 615–620 (2007). Current neuropathological criteria to diagnose definite MSA.
Stefanova, N. & Wenning, G. K. Multiple system atrophy: emerging targets for interventional therapies. Neuropathol. Appl. Neurobiol. 42, 20–32 (2016).
Lee, S. J. & Masliah, E. Neurodegeneration: aggregates feel the strain. Nature 522, 296–297 (2015).
McCann, H., Cartwright, H. & Halliday, G. M. Neuropathology of alpha-synuclein propagation and braak hypothesis. Mov. Disord. 31, 152–160 (2016).
Dehay, B., Vila, M., Bezard, E., Brundin, P. & Kordower, J. H. Alpha-synuclein propagation: new insights from animal models. Mov. Disord. 31, 161–168 (2016).
Fernagut, P. O. et al. Multiple system atrophy: a prototypical synucleinopathy for disease-modifying therapeutic strategies. Neurobiol. Dis. 67, 133–139 (2014).
Krismer, F. et al. Multiple system atrophy as emerging template for accelerated drug discovery in alpha-synucleinopathies. Parkinsonism Relat. Disord. 20, 793–799 (2014).
Hughes, A. J., Daniel, S. E., Ben-Shlomo, Y. & Lees, A. J. The accuracy of diagnosis of parkinsonian syndromes in a specialist movement disorder service. Brain 125, 861–870 (2002).
Lee, P. H. et al. A randomized trial of mesenchymal stem cells in multiple system atrophy. Ann. Neurol. 72, 32–40 (2012).
Bower, J. H., Maraganore, D. M., McDonnell, S. K. & Rocca, W. A. Incidence of progressive supranuclear palsy and multiple system atrophy in Olmsted County, Minnesota, 1976 to 1990. Neurology 49, 1284–1288 (1997).
Bjornsdottir, A., Gudmundsson, G., Blondal, H. & Olafsson, E. Incidence and prevalence of multiple system atrophy: a nationwide study in Iceland. J. Neurol. Neurosurg. Psychiatry 84, 136–140 (2013).
Linder, J., Stenlund, H. & Forsgren, L. Incidence of Parkinson's disease and parkinsonism in northern Sweden: a population-based study. Mov. Disord. 25, 341–348 (2010).
Winter, Y. et al. Incidence of Parkinson's disease and atypical parkinsonism: Russian population-based study. Mov. Disord. 25, 349–356 (2010).
Chio, A., Magnani, C. & Schiffer, D. Prevalence of Parkinson's disease in Northwestern Italy: comparison of tracer methodology and clinical ascertainment of cases. Mov. Disord. 13, 400–405 (1998).
Schrag, A., Ben-Shlomo, Y. & Quinn, N. P. Prevalence of progressive supranuclear palsy and multiple system atrophy: a cross-sectional study. Lancet 354, 1771–1775 (1999).
Tison, F., Yekhlef, F., Chrysostome, V. & Sourgen, C. Prevalence of multiple system atrophy. Lancet 355, 495–496 (2000).
Wermuth, L., Joensen, P., Bunger, N. & Jeune, B. High prevalence of Parkinson's disease in the Faroe Islands. Neurology 49, 426–432 (1997).
The Multiple-System Atrophy Research Collaboration. Mutations in COQ2 in familial and sporadic multiple-system atrophy. N. Engl. J. Med. 369, 233–244 (2013). The first study to identify relevant functional mutations in the COQ2 gene that are associated with an increased risk of developing MSA.
Wullner, U. et al. Probable multiple system atrophy in a German family. J. Neurol. Neurosurg. Psychiatry 75, 924–925 (2004).
Hara, K. et al. Multiplex families with multiple system atrophy. Arch. Neurol. 64, 545–551 (2007).
Jeon, B. S., Farrer, M. J., Bortnick, S. F. & Korean Canadian Alliance on Parkinson's Disease and Related Disorders. Mutant COQ2 in multiple-system atrophy. N. Engl. J. Med. 371, 80 (2014).
Chen, Y. P. et al. Mutation scanning of the COQ2 gene in ethnic Chinese patients with multiple-system atrophy. Neurobiol. Aging 36, 1222.e7–1222.e11 (2015).
Zhao, Q. et al. Association of the COQ2 V393A variant with risk of multiple system atrophy in East Asians: a case–control study and meta-analysis of the literature. Neurol. Sci. 37, 423–430 (2016).
Lin, C. H., Tan, E. K., Yang, C. C., Yi, Z. & Wu, R. M. COQ2 gene variants associate with cerebellar subtype of multiple system atrophy in Chinese. Mov. Disord. 30, 436–437 (2015).
Schottlaender, L. V., Houlden, H. & Multiple-System Atrophy Brain Bank Collaboration. Mutant COQ2 in multiple-system atrophy. N. Engl. J. Med. 371, 81 (2014).
Sharma, M., Wenning, G., Kruger, R. & European Multiple-System Atrophy Study Group. Mutant COQ2 in multiple-system atrophy. N. Engl. J. Med. 371, 80–81 (2014).
Federoff, M. et al. Genome-wide estimate of the heritability of multiple system atrophy. Parkinsonism Relat. Disord. 22, 35–41 (2016).
Scholz, S. W. et al. SNCA variants are associated with increased risk for multiple system atrophy. Ann. Neurol. 65, 610–614 (2009).
Al-Chalabi, A. et al. Genetic variants of the alpha-synuclein gene SNCA are associated with multiple system atrophy. PLoS ONE 4, e7114 (2009).
Sailer, A. et al. A genome-wide association study in multiple system atrophy. Neurology 87, 1591–1598 (2016). The first MSA genome-wide association study. Despite the fact that none of the single nucleotide polymorphisms reached genome-wide significance, single nucleotide polymorphisms in four genes ( FBXO47, ELOVL7, EDN1 and MAPT ) emerged as potentially interesting for follow-up.
Reuter, J. A., Spacek, D. V. & Snyder, M. P. High-throughput sequencing technologies. Mol. Cell 58, 586–597 (2015).
Mills, J. D., Ward, M., Kim, W. S., Halliday, G. M. & Janitz, M. Strand-specific RNA-sequencing analysis of multiple system atrophy brain transcriptome. Neuroscience 322, 234–250 (2016).
Mills, J. D., Kim, W. S., Halliday, G. M. & Janitz, M. Transcriptome analysis of grey and white matter cortical tissue in multiple system atrophy. Neurogenetics 16, 107–122 (2015).
Nee, L. E. et al. Environmental-occupational risk factors and familial associations in multiple system atrophy: a preliminary investigation. Clin. Auton. Res. 1, 9–13 (1991).
Vanacore, N. et al. Case-control study of multiple system atrophy. Mov. Disord. 20, 158–163 (2005).
Cho, J. W. et al. Association between parkinsonism and participation in agriculture in Korea. J. Clin. Neurol. 4, 23–28 (2008).
Seo, J. H. et al. A case-control study of multiple system atrophy in Korean patients. Mov. Disord. 25, 1953–1959 (2010).
Vidal, J. S. et al. Risk factors of multiple system atrophy: a case-control study in French patients. Mov. Disord. 23, 797–803 (2008).
Vanacore, N. et al. Smoking habits in multiple system atrophy and progressive supranuclear palsy. European Study Group on Atypical Parkinsonisms. Neurology 54, 114–119 (2000).
Jellinger, K. A. & Lantos, P. L. Papp-Lantos inclusions and the pathogenesis of multiple system atrophy: an update. Acta Neuropathol. 119, 657–667 (2010).
Papp, M. I., Kahn, J. E. & Lantos, P. L. Glial cytoplasmic inclusions in the CNS of patients with multiple system atrophy (striatonigral degeneration, olivopontocerebellar atrophy and Shy-Drager syndrome). J. Neurol. Sci. 94, 79–100 (1989).
Yoshida, M. Multiple system atrophy: alpha-synuclein and neuronal degeneration. Neuropathology 27, 484–493 (2007).
Daniel, S. in Autonomic Failure: A Textbook of Clinical Disorders of the Autonomic Nervous System (eds Mathias, C. J. & Bannister, R.) 321–328 (Oxford Univ. Press, 1999).
Wenning, G. K., Ben-Shlomo, Y., Magalhaes, M., Daniel, S. E. & Quinn, N. P. Clinicopathological study of 35 cases of multiple system atrophy. J. Neurol. Neurosurg. Psychiatry 58, 160–166 (1995).
Ozawa, T. et al. The spectrum of pathological involvement of the striatonigral and olivopontocerebellar systems in multiple system atrophy: clinicopathological correlations. Brain 127, 2657–2671 (2004).
Benarroch, E. E., Schmeichel, A. M., Sandroni, P., Low, P. A. & Parisi, J. E. Involvement of vagal autonomic nuclei in multiple system atrophy and Lewy body disease. Neurology 66, 378–383 (2006).
Ozawa, T. Morphological substrate of autonomic failure and neurohormonal dysfunction in multiple system atrophy: impact on determining phenotype spectrum. Acta Neuropathol. 114, 201–211 (2007).
Hlavanda, E. et al. Brain-specific p25 protein binds to tubulin and microtubules and induces aberrant microtubule assemblies at substoichiometric concentrations. Biochemistry 41, 8657–8664 (2002).
Lindersson, E. et al. p25alpha stimulates alpha-synuclein aggregation and is co-localized with aggregated alpha-synuclein in alpha-synucleinopathies. J. Biol. Chem. 280, 5703–5715 (2005).
Song, Y. J. et al. p25alpha relocalizes in oligodendroglia from myelin to cytoplasmic inclusions in multiple system atrophy. Am. J. Pathol. 171, 1291–1303 (2007).
Kovacs, G. G. et al. The brain-specific protein TPPP/p25 in pathological protein deposits of neurodegenerative diseases. Acta Neuropathol. 113, 153–161 (2007).
Braak, H. et al. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol. Aging 24, 197–211 (2003).
Kordower, J. H., Chu, Y., Hauser, R. A., Freeman, T. B. & Olanow, C. W. Lewy body-like pathology in long-term embryonic nigral transplants in Parkinson's disease. Nat. Med. 14, 504–506 (2008).
Kordower, J. H., Chu, Y., Hauser, R. A., Olanow, C. W. & Freeman, T. B. Transplanted dopaminergic neurons develop PD pathologic changes: a second case report. Mov. Disord. 23, 2303–2306 (2008).
Desplats, P. et al. Inclusion formation and neuronal cell death through neuron-to-neuron transmission of alpha-synuclein. Proc. Natl Acad. Sci. USA 106, 13010–13015 (2009).
Kordower, J. H. et al. Transfer of host-derived alpha synuclein to grafted dopaminergic neurons in rat. Neurobiol. Dis. 43, 552–557 (2011).
Luk, K. C. et al. Pathological alpha-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science 338, 949–953 (2012).
Luk, K. C. et al. Intracerebral inoculation of pathological alpha-synuclein initiates a rapidly progressive neurodegenerative alpha-synucleinopathy in mice. J. Exp. Med. 209, 975–986 (2012).
Watts, J. C. et al. Transmission of multiple system atrophy prions to transgenic mice. Proc. Natl Acad. Sci. USA 110, 19555–19560 (2013).
Prusiner, S. B. et al. Evidence for alpha-synuclein prions causing multiple system atrophy in humans with parkinsonism. Proc. Natl Acad. Sci. USA 112, E5308–E5317 (2015). Preclinical evidence that α -synuclein assemblies derived from MSA brains induce an aggregation pathology that is consistent with MSA.
Bernis, M. E. et al. Prion-like propagation of human brain-derived alpha-synuclein in transgenic mice expressing human wild-type alpha-synuclein. Acta Neuropathol. Commun. 3, 75 (2015).
Danzer, K. M. et al. Different species of alpha-synuclein oligomers induce calcium influx and seeding. J. Neurosci. 27, 9220–9232 (2007).
Danzer, K. M., Krebs, S. K., Wolff, M., Birk, G. & Hengerer, B. Seeding induced by alpha-synuclein oligomers provides evidence for spreading of alpha-synuclein pathology. J. Neurochem. 111, 192–203 (2009).
Luk, K. C. et al. Exogenous alpha-synuclein fibrils seed the formation of Lewy body-like intracellular inclusions in cultured cells. Proc. Natl Acad. Sci. USA 106, 20051–20056 (2009).
Peelaerts, W. et al. Alpha-synuclein strains cause distinct synucleinopathies after local and systemic administration. Nature 522, 340–344 (2015). The first study to describe how different α -synuclein strains can cause different pathologies.
Melki, R. Role of different alpha-synuclein strains in synucleinopathies, similarities with other neurodegenerative diseases. J. Parkinsons Dis. 5, 217–227 (2015).
Binolfi, A. et al. Intracellular repair of oxidation-damaged alpha-synuclein fails to target C-terminal modification sites. Nat. Commun. 7, 10251 (2016).
Theillet, F. X. et al. Structural disorder of monomeric alpha-synuclein persists in mammalian cells. Nature 530, 45–50 (2016).
Gilman, S. et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 71, 670–676 (2008). Report on the current consensus diagnostic criteria for MSA.
Kim, H. J., Jeon, B. S., Lee, J. Y. & Yun, J. Y. Survival of Korean patients with multiple system atrophy. Mov. Disord. 26, 909–912 (2011).
Saito, Y., Matsuoka, Y., Takahashi, A. & Ohno, Y. Survival of patients with multiple system atrophy. Intern. Med. 33, 321–325 (1994).
Watanabe, H. et al. Progression and prognosis in multiple system atrophy: an analysis of 230 Japanese patients. Brain 125, 1070–1083 (2002).
Wenning, G. K., Ben Shlomo, Y., Magalhaes, M., Daniel, S. E. & Quinn, N. P. Clinical features and natural history of multiple system atrophy. An analysis of 100 cases. Brain 117, 835–845 (1994).
O'Sullivan, S. S. et al. Clinical outcomes of progressive supranuclear palsy and multiple system atrophy. Brain 131, 1362–1372 (2008).
Kollensperger, M. et al. Red flags for multiple system atrophy. Mov. Disord. 23, 1093–1099 (2008).
Beck, R. O., Betts, C. D. & Fowler, C. J. Genitourinary dysfunction in multiple system atrophy: clinical features and treatment in 62 cases. J. Urol. 151, 1336–1341 (1994).
Freeman, R. et al. Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin. Auton. Res. 21, 69–72 (2011).
Benarroch, E. E. et al. Involvement of medullary regions controlling sympathetic output in Lewy body disease. Brain 128, 338–344 (2005).
Benarroch, E. E., Schmeichel, A. M. & Parisi, J. E. Involvement of the ventrolateral medulla in parkinsonism with autonomic failure. Neurology 54, 963–968 (2000).
Forno, L. S. Neuropathology of Parkinson's disease. J. Neuropathol. Exp. Neurol. 55, 259–272 (1996).
Mathias, C. J. & Kimber, J. R. Postural hypotension: causes, clinical features, investigation, and management. Annu. Rev. Med. 50, 317–336 (1999).
Low, P. A. & Singer, W. Management of neurogenic orthostatic hypotension: an update. Lancet Neurol. 7, 451–458 (2008).
Donadio, V. et al. Anhidrosis in multiple system atrophy: a preganglionic sudomotor dysfunction? Mov. Disord. 23, 885–888 (2008).
Lipp, A. et al. Prospective differentiation of multiple system atrophy from Parkinson disease, with and without autonomic failure. Arch. Neurol. 66, 742–750 (2009).
Wenning, G. K., Colosimo, C., Geser, F. & Poewe, W. Multiple system atrophy. Lancet Neurol. 3, 93–103 (2004).
Higo, R., Nito, T. & Tayama, N. Swallowing function in patients with multiple-system atrophy with a clinical predominance of cerebellar symptoms (MSA-C). Eur. Arch. Otorhinolaryngol. 262, 646–650 (2005).
Wenning, G. K. et al. What clinical features are most useful to distinguish definite multiple system atrophy from Parkinson's disease? J. Neurol. Neurosurg. Psychiatry 68, 434–440 (2000).
Boesch, S. M., Wenning, G. K., Ransmayr, G. & Poewe, W. Dystonia in multiple system atrophy. J. Neurol. Neurosurg. Psychiatry 72, 300–303 (2002).
Quinn, N. Disproportionate antecollis in multiple system atrophy. Lancet 1, 844 (1989).
Ghorayeb, I. et al. Toward a primate model of l-dopa-unresponsive parkinsonism mimicking striatonigral degeneration. Mov. Disord. 15, 531–536 (2000).
Kaindlstorfer, C., Granata, R. & Wenning, G. K. Tremor in multiple system atrophy — a review. Tremor Other Hyperkinet. Mov. http://dx.doi.org/10.7916/D8NV9GZ9 (2013).
Silber, M. H. & Levine, S. Stridor and death in multiple system atrophy. Mov. Disord. 15, 699–704 (2000).
Iranzo, A. et al. Neurodegenerative disease status and post-mortem pathology in idiopathic rapid-eye-movement sleep behaviour disorder: an observational cohort study. Lancet Neurol. 12, 443–453 (2013).
Iranzo, A. et al. Characteristics of idiopathic REM sleep behavior disorder and that associated with MSA and PD. Neurology 65, 247–252 (2005).
Rascol, O. & Schelosky, L. 123I-metaiodobenzylguanidine scintigraphy in Parkinson's disease and related disorders. Mov. Disord. 24, S732–S741 (2009).
Paviour, D. C., Williams, D., Fowler, C. J., Quinn, N. P. & Lees, A. J. Is sphincter electromyography a helpful investigation in the diagnosis of multiple system atrophy? A retrospective study with pathological diagnosis. Mov. Disord. 20, 1425–1430 (2005).
Krismer, F. et al. Sniffing the diagnosis: olfactory testing in neurodegenerative parkinsonism. Parkinsonism Relat. Disord. 35, 36–41 (2017).
Abele, M., Riet, A., Hummel, T., Klockgether, T. & Wullner, U. Olfactory dysfunction in cerebellar ataxia and multiple system atrophy. J. Neurol. 250, 1453–1455 (2003).
Garland, E. M., Raj, S. R., Peltier, A. C., Robertson, D. & Biaggioni, I. A cross-sectional study contrasting olfactory function in autonomic disorders. Neurology 76, 456–460 (2011).
Wenning, G. K. et al. Olfactory function in atypical parkinsonian syndromes. Acta Neurol. Scand. 91, 247–250 (1995).
Brooks, D. J. & Seppi, K. Proposed neuroimaging criteria for the diagnosis of multiple system atrophy. Mov. Disord. 24, 949–964 (2009).
Tang, C. C. et al. Differential diagnosis of parkinsonism: a metabolic imaging study using pattern analysis. Lancet Neurol. 9, 149–158 (2010).
Poston, K. L. et al. Network correlates of disease severity in multiple system atrophy. Neurology 78, 1237–1244 (2012).
Poston, K. L. & Eidelberg, D. Functional brain networks and abnormal connectivity in the movement disorders. Neuroimage 62, 2261–2270 (2012).
Osaki, Y., Ben-Shlomo, Y., Lees, A. J., Wenning, G. K. & Quinn, N. P. A validation exercise on the new consensus criteria for multiple system atrophy. Mov. Disord. 24, 2272–2276 (2009).
Koga, S. et al. When DLB, PD, and PSP masquerade as MSA: an autopsy study of 134 patients. Neurology 85, 404–412 (2015). Clinicopathological study showing that misdiagnosis of MSA is common, particularly when diagnostic criteria are not applied.
Stankovic, I. et al. Cognitive impairment in multiple system atrophy: a position statement by the Neuropsychology Task Force of the MDS Multiple System Atrophy (MODIMSA) study group. Mov. Disord. 29, 857–867 (2014).
Asi, Y. T. et al. Neuropathological features of multiple system atrophy with cognitive impairment. Mov. Disord. 29, 884–888 (2014).
Abele, M., Minnerop, M., Urbach, H., Specht, K. & Klockgether, T. Sporadic adult onset ataxia of unknown etiology: a clinical, electrophysiological and imaging study. J. Neurol. 254, 1384–1389 (2007).
Wenning, G. K. & Working Group on Atypical Parkinsonism of the Austrian Parkinson's Society. Placebo-controlled trial of amantadine in multiple-system atrophy. Clin. Neuropharmacol. 28, 225–227 (2005).
Colosimo, C., Merello, M. & Pontieri, F. E. Amantadine in parkinsonian patients unresponsive to levodopa: a pilot study. J. Neurol. 243, 422–425 (1996).
Jain, S., Dawson, J., Quinn, N. P. & Playford, E. D. Occupational therapy in multiple system atrophy: a pilot randomized controlled trial. Mov. Disord. 19, 1360–1364 (2004).
Muller, J., Wenning, G. K., Wissel, J., Seppi, K. & Poewe, W. Botulinum toxin treatment in atypical parkinsonian disorders associated with disabling focal dystonia. J. Neurol. 249, 300–304 (2002).
Thobois, S., Broussolle, E., Toureille, L. & Vial, C. Severe dysphagia after botulinum toxin injection for cervical dystonia in multiple system atrophy. Mov. Disord. 16, 764–765 (2001).
Halaska, M. et al. Controlled, double-blind, multicentre clinical trial to investigate long-term tolerability and efficacy of trospium chloride in patients with detrusor instability. World J. Urol. 20, 392–399 (2003).
Fowler, C. J. & O'Malley, K. J. Investigation and management of neurogenic bladder dysfunction. J. Neurol. Neurosurg. Psychiatry 74 (Suppl. 4), iv27–iv31 (2003).
Shannon, J. R. et al. Water drinking as a treatment for orthostatic syndromes. Am. J. Med. 112, 355–360 (2002).
Young, T. M. & Mathias, C. J. The effects of water ingestion on orthostatic hypotension in two groups of chronic autonomic failure: multiple system atrophy and pure autonomic failure. J. Neurol. Neurosurg. Psychiatry 75, 1737–1741 (2004).
Deguchi, K. et al. Effects of daily water drinking on orthostatic and postprandial hypotension in patients with multiple system atrophy. J. Neurol. 254, 735–740 (2007).
Freeman, R. Clinical practice. Neurogenic orthostatic hypotension. N. Engl. J. Med. 358, 615–624 (2008).
Fanciulli, A. et al. Elastic abdominal binders attenuate orthostatic hypotension in Parkinson's disease. Mov. Disord. Clin. Pract. 3, 156–160 (2016).
Jankovic, J. et al. Neurogenic orthostatic hypotension: a double-blind, placebo-controlled study with midodrine. Am. J. Med. 95, 38–48 (1993).
Low, P. A., Gilden, J. L., Freeman, R., Sheng, K. N. & McElligott, M. A. Efficacy of midodrine versus placebo in neurogenic orthostatic hypotension. A randomized, double-blind multicenter study. Midodrine Study Group. JAMA 277, 1046–1051 (1997).
Biaggioni, I. et al. Randomized withdrawal study of patients with symptomatic neurogenic orthostatic hypotension responsive to droxidopa. Hypertension 65, 101–107 (2015).
Kaufmann, H. et al. Droxidopa for neurogenic orthostatic hypotension: a randomized, placebo-controlled, phase 3 trial. Neurology 83, 328–335 (2014).
Singer, W. et al. Pyridostigmine treatment trial in neurogenic orthostatic hypotension. Arch. Neurol. 63, 513–518 (2006).
Mathias, C. J., Fosbraey, P., da Costa, D. F., Thornley, A. & Bannister, R. The effect of desmopressin on nocturnal polyuria, overnight weight loss, and morning postural hypotension in patients with autonomic failure. Br. Med. J. (Clin. Res. Ed.) 293, 353–354 (1986).
Fanciulli, A. et al. Supine hypertension in Parkinson's disease and multiple system atrophy. Clin. Auton. Res. 26, 97–105 (2016).
Fanciulli, A. & Wenning, G. K. Multiple-system atrophy. N. Engl. J. Med. 372, 249–263 (2015).
Sakakibara, R. et al. Calcium polycarbophil improves constipation in primary autonomic failure and multiple system atrophy subjects. Mov. Disord. 22, 1672–1673 (2007).
Liu, Z. et al. Mosapride citrate, a novel 5-HT4 agonist and partial 5-HT3 antagonist, ameliorates constipation in parkinsonian patients. Mov. Disord. 20, 680–686 (2005).
Eichhorn, T. E. & Oertel, W. H. Macrogol 3350/electrolyte improves constipation in Parkinson's disease and multiple system atrophy. Mov. Disord. 16, 1176–1177 (2001).
Hussain, I. F., Brady, C. M., Swinn, M. J., Mathias, C. J. & Fowler, C. J. Treatment of erectile dysfunction with sildenafil citrate (Viagra) in parkinsonism due to Parkinson's disease or multiple system atrophy with observations on orthostatic hypotension. J. Neurol. Neurosurg. Psychiatry 71, 371–374 (2001).
Poewe, W., Mahlknecht, P. & Krismer, F. Therapeutic advances in multiple system atrophy and progressive supranuclear palsy. Mov. Disord. 30, 1528–1538 (2015).
Holmberg, B. et al. Safety and tolerability of growth hormone therapy in multiple system atrophy: a double-blind, placebo-controlled study. Mov. Disord. 22, 1138–1144 (2007).
Bensimon, G. et al. Riluzole treatment, survival and diagnostic criteria in Parkinson plus disorders: the NNIPPS study. Brain 132, 156–171 (2009). The largest clinical trial in atypical parkinsonian disorders to date.
Dodel, R. et al. Minocycline 1-year therapy in multiple-system-atrophy: effect on clinical symptoms and [11C] (R)-PK11195 PET (MEMSA-trial). Mov. Disord. 25, 97–107 (2010).
Sacca, F. et al. A randomized clinical trial of lithium in multiple system atrophy. J. Neurol. 260, 458–461 (2013).
Low, P. A. et al. Efficacy and safety of rifampicin for multiple system atrophy: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 13, 268–275 (2014). A large randomized controlled trial confirming that rifampicin does not mediate neuroprotective effects in patients with MSA.
Poewe, W. et al. Efficacy of rasagiline in patients with the parkinsonian variant of multiple system atrophy: a randomised, placebo-controlled trial. Lancet Neurol. 14, 145–152 (2015). A large RCT assessing the disease-modifying efficacy of rasagiline in patients with MSA.
Dehay, B. et al. Targeting alpha-synuclein: therapeutic options. Mov. Disord. 31, 882–888 (2016).
Groft, S. C. & Rubinstein, Y. R. New and evolving rare diseases research programs at the National Institutes of Health. Public Health Genomics 16, 259–267 (2013).
Norcliffe-Kaufmann, L., Palma, J. A. & Krismer, F. Multiple system atrophy: the case for an international collaborative effort. Clin. Auton. Res. 25, 81–83 (2015).
Wenning, G. K. et al. Development and validation of the Unified Multiple System Atrophy Rating Scale (UMSARS). Mov. Disord. 19, 1391–1402 (2004). Report on the development and validation of the unified Multiple System Atrophy Rating Scale, a disease-specific and commonly used rating scale to determine the severity of motor and nonmotor symptoms in MSA.
Asi, Y. T. et al. Alpha-synuclein mRNA expression in oligodendrocytes in MSA. Glia 62, 964–970 (2014).
Reyes, J. F. et al. Alpha-synuclein transfers from neurons to oligodendrocytes. Glia 62, 387–398 (2014).
Rockenstein, E. et al. Neuronal to oligodendroglial alpha-synuclein redistribution in a double transgenic model of multiple system atrophy. Neuroreport 23, 259–264 (2012).
Miller, D. W. et al. Absence of alpha-synuclein mRNA expression in normal and multiple system atrophy oligodendroglia. J. Neural Transm. (Vienna) 112, 1613–1624 (2005).
Author information
Authors and Affiliations
Contributions
G.K.W and F.K. contributed equally to writing the article. G.K.W researched the data for the article, and provided a substantial contribution to discussions of the content and to review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Permissive templating
-
Also known as seeding, permissive templating is a phenomenon whereby some protein assemblies can induce folding of the same (or similar) protein into a disease-causing confirmation.
- Orphan disease
-
A disease that affects fewer than 5 in 10,000 people (European Union) or fewer than 7.5 in 10,000 people (USA).
- Argyrophilic
-
A histological term used to describe an affinity to silver, making the protein amenable to silver-staining techniques.
- Dysarthria
-
Slurred or slow speech that can be difficult to understand.
- Nystagmus
-
Involuntary, rapid and repetitive movement of the eyes.
- Hypometric saccades
-
Saccades refer to conjugate and voluntary eye movements that shift the eyes from one target to another. A hypometric saccade is a undershoot movement of the eyes.
- Neurogenic orthostatic hypotension
-
A sign of cardiovascular autonomic dysfunction with substantial blood pressure drops on postural challenge.
- Anhidrosis
-
Inability to sweat.
- Camptocormia
-
Abnormal forward flexion of the trunk that disappears when the patient is lying down.
- Pisa syndrome
-
Lateral flexion of the trunk when sitting or standing.
- Antecollis
-
Also known as dropped head sign: a marked forward flexion of the head and neck.
- Polyminimyoclonus
-
Small-amplitude myoclonic movements of the hands and/or fingers.
- Supranuclear gaze palsy
-
Inability to look in a particular direction, although reflex eye movements remain intact.
- Focal dystonia
-
Involuntary muscular contractions affecting a muscle or a group of muscles in a circumscribed part of the body.
Rights and permissions
About this article
Cite this article
Krismer, F., Wenning, G. Multiple system atrophy: insights into a rare and debilitating movement disorder. Nat Rev Neurol 13, 232–243 (2017). https://doi.org/10.1038/nrneurol.2017.26
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2017.26
This article is cited by
-
The comorbidity profiles and medication issues of patients with multiple system atrophy: a systematic cross-sectional analysis
Journal of Neurology (2024)
-
Multidimensional biomarkers for multiple system atrophy: an update and future directions
Translational Neurodegeneration (2023)
-
Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction
Cell Death & Disease (2023)
-
Oral appliance therapy for obstructive sleep apnea in multiple system atrophy with floppy epiglottis: a case series of three patients
Sleep and Breathing (2023)
-
The ‘α-synucleinopathy syndicate’: multiple system atrophy and Parkinson’s disease
Journal of Neural Transmission (2023)