Key Points
-
Amyotrophic lateral sclerosis (ALS) is a syndrome that can result from many possible underlying genetic variations; the rate of gene discovery in ALS is doubling every 4 years
-
ALS has a genetic archtecture in which a few rare variants contribute to risk in each patient, rather than a polygenic architecture whereby the cumulative effects of many common variants increase risk
-
Carrying a disease mutation does not inevitably lead to ALS, and many ALS-associated genes are also implicated in other conditions, including frontotemporal dementia and cerebellar disease
-
The distinction between familial and sporadic ALS is not clear-cut, which greatly complicates genetic counselling in ALS
-
Current data are consistent with a model in which multiple molecular steps are required to cause ALS; the causes of these steps could be genetic or environmental
Abstract
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease predominantly affecting upper and lower motor neurons. The disease leads to relentlessly progressive weakness of voluntary muscles, with death typically resulting from diaphragmatic failure within 2–5 years. Since the discovery of mutations in SOD1, which account for ∼2% of ALS cases, increasing efforts have been made to understand the genetic component of ALS risk, with the expectation that this insight will not only aid diagnosis and classification, but also guide personalized treatment and reveal the mechanisms that cause motor neuron death. In this Review, we outline previous and current efforts to characterize genes that are associated with ALS, describe current knowledge about the genetic architecture of ALS — including the relevance of family history — and the probable nature of future gene discoveries, and explore how our understanding of ALS genetics affects present and future clinical decisions. We observe that many gene variants associated with ALS have effect sizes between those of mutations that greatly increase risk and those of common variants that have a small effect on risk, and combine this observation with insights from next-generation sequencing to explore the implications for genetic counselling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
22 December 2016
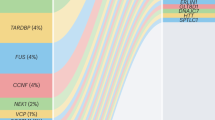
In the version of this article initially published online, Figure 3 erroneously depicted 6 items in the stalagmite plot, 2 of which did not correspond with the 4 items listed in the legend. Further, the placement of the headings in Figure 3 was erroneous. The errors have been corrected for the PDF, HTML and print versions of the article.
References
Phukan, J., Pender, N. P. & Hardiman, O. Cognitive impairment in amyotrophic lateral sclerosis. Lancet Neurol. 6, 994–1003 (2007).
Goldstein, L. H. & Abrahams, S. Changes in cognition and behaviour in amyotrophic lateral sclerosis: nature of impairment and implications for assessment. Lancet Neurol. 12, 368–380 (2013).
Johnston, C. A. et al. Amyotrophic lateral sclerosis in an urban setting: a population based study of inner city London. J. Neurol. 253, 1642–1643 (2006).
Marin, B. et al. Variation in worldwide incidence of amyotrophic lateral sclerosis: a meta-analysis. Int. J. Epidemiol. http://dx.doi.org/10.1093/ije/dyw061 (2016).
Al-Chalabi, A. & Hardiman, O. The epidemiology of ALS: a conspiracy of genes, environment and time. Nat. Rev. Neurol. 9, 617–628 (2013).
Manjaly, Z. R. et al. The sex ratio in amyotrophic lateral sclerosis: a population based study. Amyotroph. Lateral Scler. 11, 439–442 (2010).
Nakken, O., Lindstrom, J. C., Tysnes, O. B. & Holmoy, T. Mortality trends of amyotrophic lateral sclerosis in Norway 1951–2014: an age-period-cohort study. J. Neurol. http://dx.doi.org/10.1007/s00415-016-8273-2 (2016).
Chio, A. et al. Phenotypic heterogeneity of amyotrophic lateral sclerosis: a population based study. J. Neurol. Neurosurg. Psychiatry 82, 740–746 (2011).
Turner, M. R., Barnwell, J., Al-Chalabi, A. & Eisen, A. Young-onset amyotrophic lateral sclerosis: historical and other observations. Brain 135, 2883–2891 (2012).
Byrne, S. et al. Aggregation of neurologic and neuropsychiatric disease in amyotrophic lateral sclerosis kindreds: a population-based case–control cohort study of familial and sporadic amyotrophic lateral sclerosis. Ann. Neurol. 74, 699–708 (2013).
Al-Chalabi, A. et al. An estimate of amyotrophic lateral sclerosis heritability using twin data. J. Neurol. Neurosurg. Psychiatry 81, 1324–1326 (2010).
Wingo, T. S., Cutler, D. J., Yarab, N., Kelly, C. M. & Glass, J. D. The heritability of amyotrophic lateral sclerosis in a clinically ascertained United States research registry. PLoS ONE 6, e27985 (2011).
McLaughlin, R. L., Vajda, A. & Hardiman, O. Heritability of amyotrophic lateral sclerosis: insights from disparate numbers. JAMA Neurol. 72, 857–858 (2015).
Byrne, S., Elamin, M., Bede, P. & Hardiman, O. Absence of consensus in diagnostic criteria for familial neurodegenerative diseases. J. Neurol. Neurosurg. Psychiatry 83, 365–367 (2012). An important study showing that familial ALS has no formal or informal definition, with a wide range of opinions between physicians.
Al-Chalabi, A. & Lewis, C. M. Modelling the effects of penetrance and family size on rates of sporadic and familial disease. Hum. Hered. 71, 281–288 (2011).
Andersen, P. M. & Al-Chalabi, A. Clinical genetics of amyotrophic lateral sclerosis: what do we really know? Nat. Rev. Neurol. 7, 603–615 (2011).
Fecto, F. et al. SQSTM1 mutations in familial and sporadic amyotrophic lateral sclerosis. Arch. Neurol. 68, 1440–1446 (2011). This study, along with Hirano et al . (reference 105), Le Ber et al . (reference 106) and Rubino et al . (reference 107), showed that as well as TDP-43, sequestosome-1, another component of ubiquitin-immunoreactive inclusions in ALS, could be a primary cause of ALS, and also provided evidence that the proteasome system is important for motor neuron health.
Greenway, M. J. et al. ANG mutations segregate with familial and 'sporadic' amyotrophic lateral sclerosis. Nat. Genet. 38, 411–413 (2006).
Hubers, A. et al. De novo FUS mutations are the most frequent genetic cause in early-onset German ALS patients. Neurobiol. Aging 36, 3117.e1–3117.e6 (2015).
Jackson, M. et al. Copper/zinc superoxide dismutase 1 and sporadic amyotrophic lateral sclerosis: analysis of 155 cases and identification of a novel insertion mutation. Ann. Neurol. 42, 803–807 (1997).
Johnson, L. et al. Screening for OPTN mutations in a cohort of British amyotrophic lateral sclerosis patients. Neurobiol. Aging 33, 2948.e15–2948.e17 (2012).
Kabashi, E. et al. TARDBP mutations in individuals with sporadic and familial amyotrophic lateral sclerosis. Nat. Genet. 40, 572–574 (2008).
Miller, J. W. et al. Mutation analysis of VCP in British familial and sporadic amyotrophic lateral sclerosis patients. Neurobiol. Aging 33, 2721.e1–2721.e2 (2012).
DeJesus-Hernandez, M. et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron 72, 245–256 (2011). This key paper, along with Renton et al . (reference 46), provided the first identification of the most frequent cause of ALS, pathological expansion of a repeat in intron 1 of the C9orf72 gene. This finding was confirmed in linkage studies by Morita et al . (reference 39) and Vance et al . (reference 40), and in association studies by van Es et al . (reference 31) and Shatunov et al . (reference 32).
Diekstra, F. P. et al. C9orf72 and UNC13A are shared risk loci for amyotrophic lateral sclerosis and frontotemporal dementia: a genome-wide meta-analysis. Ann. Neurol. 76, 120–133 (2014).
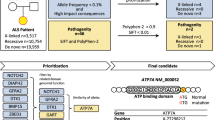
Kenna, K. P. et al. NEK1 variants confer susceptibility to amyotrophic lateral sclerosis. Nat. Genet. 48, 1037–1042 (2016). A study that used multiple methods, including machine learning and gene sequencing in isolated populations, to identify an ALS-associated gene.
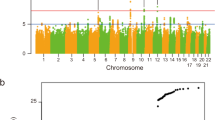
van Rheenen, W. et al. Genome-wide association analyses identify new risk variants and the genetic architecture of amyotrophic lateral sclerosis. Nat. Genet. 48, 1043–1048 (2016). A large genome-wide association study that found four new ALS loci, thee of which were replicated by the authors. The study also showed that rare genetic variants are disporportionately important in ALS.
Cirulli, E. T. et al. Exome sequencing in amyotrophic lateral sclerosis identifies risk genes and pathways. Science 347, 1436–1441 (2015).
Williams, K. L. et al. CCNF mutations in amyotrophic lateral sclerosis and frontotemporal dementia. Nat. Commun. 7, 11253 (2016).
Freischmidt, A. et al. Haploinsufficiency of TBK1 causes familial ALS and fronto-temporal dementia. Nat. Neurosci. 18, 631–636 (2015).
van Es, M. A. et al. Genome-wide association study identifies 19p13.3 (UNC13A) and 9p21.2 as susceptibility loci for sporadic amyotrophic lateral sclerosis. Nat. Genet. 41, 1083–1087 (2009).
Shatunov, A. et al. Chromosome 9p21 in sporadic amyotrophic lateral sclerosis in the UK and seven other countries: a genome-wide association study. Lancet Neurol. 9, 986–994 (2010).
Fogh, I. et al. A genome-wide association meta-analysis identifies a novel locus at 17q11.2 associated with sporadic amyotrophic lateral sclerosis. Hum. Mol. Genet. 23, 2220–2231 (2014).
Abel, O., Powell, J. F., Andersen, P. M. & Al-Chalabi, A. ALSoD: a user-friendly online bioinformatics tool for amyotrophic lateral sclerosis genetics. Hum. Mutat. 33, 1345–1351 (2012).
Wroe, R., Wai- Ling Butler, A., Andersen, P. M., Powell, J. F. & Al-Chalabi, A. ALSOD: the Amyotrophic Lateral Sclerosis Online Database. Amyotroph. Lateral Scler. 9, 249–250 (2008).
World Federation of Neurology. The ALS Online Genetics Database. ALSOD http://alsod.iop.kcl.ac.uk (2009).
Amberger, J. S., Bocchini, C. A., Schiettecatte, F., Scott, A. F. & Hamosh, A. OMIM.org: Online Mendelian Inheritance in Man (OMIM®), an online catalog of human genes and genetic disorders. Nucleic Acids Res. 43, D789–D798 (2015).
Visscher, P. M., Brown, M. A., McCarthy, M. I. & Yang, J. Five years of GWAS discovery. Am. J. Hum. Genet. 90, 7–24 (2012).
Morita, M. et al. A locus on chromosome 9p confers susceptibility to ALS and frontotemporal dementia. Neurology 66, 839–844 (2006).
Vance, C. et al. Familial amyotrophic lateral sclerosis with frontotemporal dementia is linked to a locus on chromosome 9p13.2-21.3. Brain 129, 868–876 (2006). References 39 and 40 showed, for the first time, that ALS and frontotemporal dementia should — at least in some cases — be considered as related diseases.
Hsiung, G. Y. et al. Clinical and pathological features of familial frontotemporal dementia caused by C9ORF72 mutation on chromosome 9p. Brain 135, 709–722 (2012).
Pearson, J. P. et al. Familial frontotemporal dementia with amyotrophic lateral sclerosis and a shared haplotype on chromosome 9p. J. Neurol. 258, 647–655 (2011).
Boxer, A. L. et al. Clinical, neuroimaging and neuropathological features of a new chromosome 9p-linked FTD–ALS family. J. Neurol. Neurosurg. Psychiatry 82, 196–203 (2011).
Gijselinck, I. et al. Identification of 2 loci at chromosomes 9 and 14 in a multiplex family with frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Arch. Neurol. 67, 606–616 (2010).
Valdmanis, P. N. et al. Three families with amyotrophic lateral sclerosis and frontotemporal dementia with evidence of linkage to chromosome 9p. Arch. Neurol. 64, 240–245 (2007).
Renton, A. E. et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS–FTD. Neuron 72, 257–268 (2011).
Greenway, M. J. et al. A novel candidate region for ALS on chromosome 14q11.2. Neurology 63, 1936–1938 (2004).
Elden, A. C. et al. Ataxin-2 intermediate-length polyglutamine expansions are associated with increased risk for ALS. Nature 466, 1069–1075 (2010). This study links the most frequent pathological finding in ALS, TDP-43 protein accumulation, with a genetic variation that cannot be detected with conventional genome-wide association methods — a trinucleotide repeat in a spinocerebellar ataxia-associated gene, which causes ALS when pathologically expanded to an intermediate size.
Simpson, C. L. et al. Variants of the elongator protein 3 (ELP3) gene are associated with motor neuron degeneration. Hum. Mol. Genet. 18, 472–481 (2009).
Blauw, H. M. et al. A large genome scan for rare CNVs in amyotrophic lateral sclerosis. Hum. Mol. Genet. 19, 4091–4099 (2010).
Al-Chalabi, A. et al. Deletions of the heavy neurofilament subunit tail in amyotrophic lateral sclerosis. Hum. Mol. Genet. 8, 157–164 (1999).
Consortium Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature 511, 421–427 (2014).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 536, 285–291 (2016). A superb genetic and statistical analysis of the genome, providing a framework for the interpretation of exome sequence changes.
Appel, S. H. & Rowland, L. P. Amyotrophic lateral sclerosis, frontotemporal lobar dementia, and p62: a functional convergence? Neurology 79, 1526–1527 (2012).
Rosen, D. R. et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362, 59–62 (1993).
Deng, H. X. et al. Amyotrophic lateral sclerosis and structural defects in Cu,Zn superoxide dismutase. Science 261, 1047–1051 (1993).
Andersen, P. M. et al. Amyotrophic lateral sclerosis associated with homozygosity for an Asp90Ala mutation in CuZn-superoxide dismutase. Nat. Genet. 10, 61–66 (1995).
Tsuda, T. et al. Analysis of the functional effects of a mutation in SOD1 associated with familial amyotrophic lateral sclerosis. Neuron 13, 727–736 (1994).
Shaw, C. E. et al. Mutations in all five exons of SOD-1 may cause ALS. Ann. Neurol. 43, 390–394 (1998).
Exome Aggregation Consortium. ExAC Browser. ExAC http://exac.broadinstitute.org (2016).
Ionita-Laza, I., McCallum, K., Xu, B. & Buxbaum, J. D. A spectral approach integrating functional genomic annotations for coding and noncoding variants. Nat. Genet. 48, 214–220 (2016).
Rao, S. S. et al. A 3D map of the human genome at kilobase resolution reveals principles of chromatin looping. Cell 159, 1665–1680 (2014).
Carvunis, A. R. et al. Evidence for a common evolutionary rate in metazoan transcriptional networks. eLife 4, e11615 (2015).
Campbell, I. M., Shaw, C. A., Stankiewicz, P. & Lupski, J. R. Somatic mosaicism: implications for disease and transmission genetics. Trends Genet. 31, 382–392 (2015).
Glass, C. K., Saijo, K., Winner, B., Marchetto, M. C. & Gage, F. H. Mechanisms underlying inflammation in neurodegeneration. Cell 140, 918–934 (2010).
Fang, X. et al. The NEK1 interactor, C21ORF2, is required for efficient DNA damage repair. Acta Biochim. Biophys. Sin. (Shanghai) 47, 834–841 (2015).
Lai, C. K. et al. Functional characterization of putative cilia genes by high-content analysis. Mol. Biol. Cell 22, 1104–1119 (2011).
Ma, X., Peterson, R. & Turnbull, J. Adenylyl cyclase type 3, a marker of primary cilia, is reduced in primary cell culture and in lumbar spinal cord in situ in G93A SOD1 mice. BMC Neurosci. 12, 71 (2011).
Smith, B. N. et al. Exome-wide rare variant analysis identifies TUBA4A mutations associated with familial ALS. Neuron 84, 324–331 (2014).
Puls, I. et al. Mutant dynactin in motor neuron disease. Nat. Genet. 33, 455–456 (2003).
Kwiatkowski, T. J. Jr et al. Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science 323, 1205–1208 (2009).
Sreedharan, J. et al. TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis. Science 319, 1668–1672 (2008). This study, along with Kabashi et al . (reference 22), showed that TDP-43, a protein found as a pathological accumulation in motor neurons in 98% of cases of ALS, could be a primary cause of the disease, and was not just a downstream manifestation of the disease process. The study also highlighted, for the first time, that RNA-processing defects could result in motor neuron degeneration.
Vance, C. et al. Mutations in FUS, an RNA processing protein, cause familial amyotrophic lateral sclerosis type 6. Science 323, 1208–1211 (2009).
Ticozzi, N. et al. Mutational analysis reveals the FUS homolog TAF15 as a candidate gene for familial amyotrophic lateral sclerosis. Am. J. Med. Genet. B Neuropsychiatr. Genet. 156B, 285–290 (2011).
Hand, C. K. et al. Compound heterozygous D90A and D96N SOD1 mutations in a recessive amyotrophic lateral sclerosis family. Ann. Neurol. 49, 267–271 (2001).
van Blitterswijk, M. et al. Evidence for an oligogenic basis of amyotrophic lateral sclerosis. Hum. Mol. Genet. 21, 3776–3784 (2012). The finding of an oligogenic component in a significant percentage of ALS cases has implications for genetic counselling, and provides evidence of an additive effect of gene variations.
van Blitterswijk, M. et al. VAPB and C9orf72 mutations in 1 familial amyotrophic lateral sclerosis patient. Neurobiol. Aging 33, 2950.e1–2950.e4 (2012).
Bury, J. J. et al. Oligogenic inheritance of optineurin (OPTN) and C9ORF72 mutations in ALS highlights localisation of OPTN in the TDP-43-negative inclusions of C9ORF72-ALS. Neuropathology 36, 125–134 (2016).
Kramer, N. J. et al. Spt4 selectively regulates the expression of C9orf72 sense and antisense mutant transcripts. Science 353, 708–712 (2016).
Chio, A. et al. Genetic counselling in ALS: facts, uncertainties and clinical suggestions. J. Neurol. Neurosurg. Psychiatry 85, 478–485 (2014).
Smith, B. N. et al. The C9ORF72 expansion mutation is a common cause of ALS+/−FTD in Europe and has a single founder. Eur. J. Hum. Genet. 21, 102–108 (2013).
Al-Chalabi, A. et al. Analysis of amyotrophic lateral sclerosis as a multistep process: a population-based modelling study. Lancet Neurol. 13, 1108–1113 (2014). A large European study showing that ALS is a multistep process like cancer, and requires, on average, six molecular steps, at least one of which is likely to be genetic in most people.
Agosta, F. et al. The El Escorial criteria: strengths and weaknesses. Amyotroph. Lateral Scler. Frontotemporal Degener. 16, 1–7 (2015).
Escott-Price, V. et al. Common polygenic variation enhances risk prediction for Alzheimer's disease. Brain 138, 3673–3684 (2015).
Zou, Z. Y., Liu, M. S., Li, X. G. & Cui, L. Y. H46R SOD1 mutation is consistently associated with a relatively benign form of amyotrophic lateral sclerosis with slow progression. Amyotroph. Lateral Scler. Frontotemporal Degener. http://dx.doi.org/10.1080/21678421.2016.1199698 (2016).
Juneja, T., Pericak-Vance, M. A., Laing, N. G., Dave, S. & Siddique, T. Prognosis in familial amyotrophic lateral sclerosis: progression and survival in patients with glu100gly and ala4val mutations in Cu,Zn superoxide dismutase. Neurology 48, 55–57 (1997).
Murray, M. E. et al. Clinical and neuropathologic heterogeneity of c9FTD/ALS associated with hexanucleotide repeat expansion in C9ORF72. Acta Neuropathol. 122, 673–690 (2011).
Fogh, I. et al. Association of a locus in the CAMTA1 gene with survival in patients with sporadic amyotrophic lateral sclerosis. JAMA Neurol. 73, 812–820 (2016).
Hoglinger, G. U. et al. Identification of common variants influencing risk of the tauopathy progressive supranuclear palsy. Nat. Genet. 43, 699–705 (2011).
Miller, T. M. et al. An antisense oligonucleotide against SOD1 delivered intrathecally for patients with SOD1 familial amyotrophic lateral sclerosis: a phase 1, randomised, first-in-man study. Lancet Neurol. 12, 435–442 (2013). An important step in the development of personalized, gene-based therapies for ALS.
Atanasio, A. et al. C9orf72 ablation causes immune dysregulation characterized by leukocyte expansion, autoantibody production, and glomerulonephropathy in mice. Sci. Rep. 6, 23204 (2016).
Lopez, E. S. et al. Full ablation of C9orf72 in mice causes immune system-related pathology and neoplastic events but no motor neuron defects. Acta Neuropathol. 132, 145–147 (2016).
Doudna, J. A. & Charpentier, E. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014).
Heidenreich, M. & Zhang, F. Applications of CRISPR–Cas systems in neuroscience. Nat. Rev. Neurosci. 17, 36–44 (2016).
Do, R., Kathiresan, S. & Abecasis, G. R. Exome sequencing and complex disease: practical aspects of rare variant association studies. Hum. Mol. Genet. 21, R1–R9 (2012).
Acknowledgements
The authors would like to thank The EU Joint Programme — Neurodegenerative Disease Research (JPND) projects: STRENGTH, SOPHIA and ALS-CarE. The authors' work is supported by the following funding organizations under the aegis of JPND: the Medical Research Council and the Economic and Social Research Council in the UK, and ZonMW in the Netherlands. A.A.-C. receives salary support from the National Institute for Health Research Dementia Biomedical Research Unit at South London and Maudsley NHS Foundation Trust and King's College London, UK. The work leading up to this publication was funded by the European Community's Health Seventh Framework Programme (FP7/2007–2013; grant agreement number 259867) and Horizon 2020 Programme (H2020-PHC-2014-two-stage; grant agreement number 633413). The authors would also like to thank Dr William Sproviero who kindly provided the data used in Fig. 2. The authors' work is supported by ZonMW under the frame of E-Rare-2, the ERA Net for Research on Rare Diseases (PYRAMID).
Author information
Authors and Affiliations
Contributions
A.A,-C., L.H.B and J.V. contributed equally to researching the data for the article, discussions of the content, writing the article, and review and/or editing of the manuscript before submission
Corresponding author
Ethics declarations
Competing interests
A.A.-C. declares associations with OrionPharma, Cytokinetics, Mitsubishi-Tanabe Pharma, OneWorld Publications and Cold Spring Harbor Laboratory Press. L.H.B. received a grant from the Netherlands Organization for Health Research and Development (Vici scheme); serves on scientific advisory boards for Prinses Beatrix Spierfonds, Thierry Latran Foundation, Baxalta, Cytokinetics and Biogen. J.V. declares no competing interests.
Related links
FURTHER INFORMATION
Supplementary information
Supplementary information S1 (table)
ALS-associated genes as listed in OMIM, supplemented with recent discoveries (PDF 211 kb)
Glossary
- Linkage
-
A statistical technique whereby the transmission and sharing of genetic variations within a family is used to home in on the disease gene. Linkage analysis is conducted by comparing the observed frequency of transmissions with the pattern that would be expected if there was no relationship with disease status.
- Somatic mosaicism
-
Mutations that arise during mitotic cell division are inherited in all the daughter cells of that line. Although individual mutations are rare, human cells undergo so many divisions that we are all mosaics of cells that are nearly, but not quite, genetically identical.
- Penetrance
-
The conditional probability of a phenotype, such as amyotrophic lateral sclerosis, being associated with a given genotype.
- El Escorial criteria
-
Research criteria used to define the certainty that the phenotype will ultimately be amyotrophic lateral sclerosis rather than some other variant of motor neuron disease. The criteria are based on the distribution and pattern of upper and lower motor neuron signs, and have recently been revised to incorporate neurophysiological findings.
Rights and permissions
About this article
Cite this article
Al-Chalabi, A., van den Berg, L. & Veldink, J. Gene discovery in amyotrophic lateral sclerosis: implications for clinical management. Nat Rev Neurol 13, 96–104 (2017). https://doi.org/10.1038/nrneurol.2016.182
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2016.182
This article is cited by
-
PCDHA9 as a candidate gene for amyotrophic lateral sclerosis
Nature Communications (2024)
-
Abnormal Brain Protein Abundance and Cross-tissue mRNA Expression in Amyotrophic Lateral Sclerosis
Molecular Neurobiology (2024)
-
Genetics screening in an Italian cohort of patients with Amyotrophic Lateral Sclerosis: the importance of early testing and its implication
Journal of Neurology (2024)
-
Genetics of amyotrophic lateral sclerosis: seeking therapeutic targets in the era of gene therapy
Journal of Human Genetics (2023)
-
BL-918, a small-molecule activator of ULK1, induces cytoprotective autophagy for amyotrophic lateral sclerosis therapy
Acta Pharmacologica Sinica (2023)