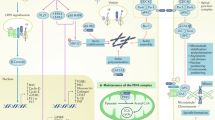
Key Points
-
Renin-expressing cells are evolutionarily conserved and have retained features to contribute to the defence against threats to survival such as bleeding, dehydration, hypotension and/or hypoxia
-
In the embryo, renin-expressing cells populate diverse tissues where they might have a role in tissue morphogenesis and growth; in the developing kidney, they are progenitors for arteriolar and mesangial cells and are crucial for arteriolar branching morphogenesis
-
In adults, cells derived from progenitors that previously expressed renin retain their developmental memory and can regenerate injured glomeruli or re-express renin to achieve blood pressure and fluid–electrolyte homeostasis
-
Renin-expressing cells are linked to other fundamental homeostatic systems, such as the haematopoietic and circulating leukocyte renin–angiotensin systems, through which they might contribute to bone marrow development and the local control of inflammation or infections
-
Renin-expressing cells intersect with erythropoietin-producing cells and are therefore at the crossroads of two key life-sustaining systems involved in the control of fluid volume, perfusion pressure and oxygen delivery to tissues
Abstract
An accumulating body of evidence suggests that renin-expressing cells have developed throughout evolution as a mechanism to preserve blood pressure and fluid volume homeostasis as well as to counteract a number of homeostatic and immunological threats. In the developing embryo, renin precursor cells emerge in multiple tissues, where they differentiate into a variety of cell types. The function of those precursors and their progeny is beginning to be unravelled. In the developing kidney, renin-expressing cells control the morphogenesis and branching of the renal arterial tree. The cells do not seem to fully differentiate but instead retain a degree of developmental plasticity or molecular memory, which enables them to regenerate injured glomeruli or to alter their phenotype to control blood pressure and fluid–electrolyte homeostasis. In haematopoietic tissues, renin-expressing cells might regulate bone marrow differentiation and participate in a circulating leukocyte renin–angiotensin system, which acts as a defence mechanism against infections or tissue injury. Furthermore, renin-expressing cells have an intricate lineage and functional relationship with erythropoietin-producing cells and are therefore central to two endocrine systems — the renin–angiotensin and erythropoietin systems — that sustain life by controlling fluid volume and composition, perfusion pressure and oxygen delivery to tissues. However, loss of the homeostatic control of these systems following dysregulation of renin-expressing cells can be detrimental, with serious pathological events.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Gomez, R. A. Fate of renin cells during development and disease. Hypertension 69, 387–395 (2017).
Gomez, R. A. & Sequeira-Lopez, M. L. Novel functions of renin precursors in homeostasis and disease. Physiology 31, 25–33 (2016).
Sequeira Lopez, M. L., Pentz, E. S., Nomasa, T., Smithies, O. & Gomez, R. A. Renin cells are precursors for multiple cell types that switch to the renin phenotype when homeostasis is threatened. Dev. Cell 6, 719–728 (2004). This paper presents the first genetic lineage tracing study showing that renin cells are precursors for renal and extrarenal cells.
Lee, G. et al. Homeostatic responses in the adrenal cortex to the absence of aldosterone in mice. Endocrinology 146, 2650–2656 (2005).
Makhanova, N., Sequeira-Lopez, M. L., Gomez, R. A., Kim, H. S. & Smithies, O. Disturbed homeostasis in sodium-restricted mice heterozygous and homozygous for aldosterone synthase gene disruption. Hypertension 48, 1151–1159 (2006).
Naruse, M., Naruse, K., Inagaki, T. & Inagami, T. Immunoreactive renin in mouse adrenal gland. Localization in the inner cortical region. Hypertension 6, 275–280 (1984).
Gomez, R. A. & Lopez, M. L. Plasticity of renin cells in the kidney vasculature. Curr. Hypertens. Rep. 19, 14 (2017).
Sequeira Lopez, M. L., Pentz, E. S., Robert, B., Abrahamson, D. R. & Gomez, R. A. Embryonic origin and lineage of juxtaglomerular cells. Am. J. Physiol. Renal Physiol. 281, F345–F356 (2001). This study shows that precursors for the kidney vasculature, including renin-expressing cells, are present in the embryonic kidney and assemble in situ to generate the kidney arterioles.
Belyea, B. C. et al. Identification of renin progenitors in the mouse bone marrow that give rise to B-cell leukaemia. Nat. Commun. 5, 3273 (2014). This study shows that the bone marrow possesses B lymphocytes that express renin, which are the cell of origin of a highly penetrant form of leukaemia.
Sequeira Lopez, M. L. & Gomez, R. A. Development of the renal arterioles. J. Am. Soc. Nephrol. 22, 2156–2165 (2011).
Sequeira-Lopez, M. L. et al. The earliest metanephric arteriolar progenitors and their role in kidney vascular development. Am. J. Physiol. Regul. Integr. Comp. Physiol. 308, R138–R149 (2015). This study shows that the FOXD1+ stromal cell is a progenitor for renin-expressing cells in the kidney.
Hu, Y., Li, M., Gothert, J. R., Gomez, R. A. & Sequeira-Lopez, M. L. Hemovascular progenitors in the kidney require sphingosine-1-phosphate receptor 1 for vascular development. J. Am. Soc. Nephrol. 27, 1984–1995 (2016).
Robert, B., St John, P. L., Hyink, D. P. & Abrahamson, D. R. Evidence that embryonic kidney cells expressing flk-1 are intrinsic, vasculogenic angioblasts. Am. J. Physiol. 271, F744–F753 (1996).
Lin, E. E., Sequeira-Lopez, M. L. & Gomez, R. A. RBP-J in FOXD1+ renal stromal progenitors is crucial for the proper development and assembly of the kidney vasculature and glomerular mesangial cells. Am. J. Physiol. Renal Physiol. 306, F249–258 (2014).
Reddi, V., Zaglul, A., Pentz, E. S. & Gomez, R. A. Renin-expressing cells are associated with branching of the developing kidney vasculature. J. Am. Soc. Nephrol. 9, 63–71 (1998).
Gomez, R. A. et al. Distribution of renin mRNA and its protein in the developing kidney. Am. J. Physiol. 257, F850–F858 (1989).
Gomez, R. A. et al. Renin and angiotensinogen gene expression in maturing rat kidney. Am. J. Physiol. 254, F582–F587 (1988).
Gomez, R. A. Molecular biology of components of the renin-angiotensin system during development. Pediatr. Nephrol. 4, 421–423 (1990).
Gomez, R. A. et al. Renin and angiotensinogen gene expression and intrarenal renin distribution during ACE inhibition. Am. J. Physiol. 254, F900–F906 (1988).
Taugner, R. et al. in The Juxtaglomerular Apparatus: Structure and Function (eds Taugner, R. & Hackenthal, E.) 103–126 (Springer Verlag, 1989). This is an excellent monograph on the juxtaglomerular apparatus.
Celio, M. R., Groscurth, P. & Inagami, T. Ontogeny of renin immunoreactive cells in the human kidney. Anat. Embryol. 173, 149–155 (1985).
Celio, M. R. & Inagami, T. Renin in the human kidney. Immunohistochemical localization. Histochemistry 72, 1–10 (1981).
Egerer, G., Taugner, R. & Tiedemann, K. Renin immunohistochemistry in the mesonephros and metanephros of the pig embryo. Histochemistry 81, 385–390 (1984).
Nishimura, H. Renin-angiotensin system in vertebrates: phylogenetic view of structure and function. Anat. Sci. Int. 92, 215–247 (2017). This is an excellent review of the phylogeny of the RAS.
Nishimura, H., Ogawa, M. & Sawyer, W. H. Renin-angiotensin system in primitive bony fishes and a holocephalian. Am. J. Physiol. 224, 950–956 (1973).
Rider, S. A., Mullins, L. J., Verdon, R. F., MacRae, C. A. & Mullins, J. J. Renin expression in developing zebrafish is associated with angiogenesis and requires the Notch pathway and endothelium. Am. J. Physiol. Renal Physiol. 309, F531–F539 (2015). This study demonstrates conservation of the roles of renin-expressing cells in mammals and fish.
Rider, S. A. et al. Zebrafish mesonephric renin cells are functionally conserved and comprise two distinct morphological populations. Am. J. Physiol. Renal Physiol. 312, F778–F790 (2017).
Gomez, R. A., Pentz, E. S., Jin, X., Cordaillat, M. & Sequeira Lopez, M. L. CBP and p300 are essential for renin cell identity and morphological integrity of the kidney. Am. J. Physiol. Heart Circ. Physiol. 296, H1255–H1262 (2009).
Neubauer, B. et al. Development of vascular renin expression in the kidney critically depends on the cyclic AMP pathway. Am. J. Physiol. Renal Physiol. 296, F1006–F1012 (2009). This study shows that the cAMP pathway is crucial for the development of the renin-expressing cell.
Pentz, E. S. et al. Histone acetyl transferases CBP and p300 are necessary for maintenance of renin cell identity and transformation of smooth muscle cells to the renin phenotype. Am. J. Physiol. Heart Circ. Physiol. 302, H2545–H2552 (2012).
Pentz, E. S., Lopez, M. L., Cordaillat, M. & Gomez, R. A. Identity of the renin cell is mediated by cAMP and chromatin remodeling: an in vitro model for studying cell recruitment and plasticity. Am. J. Physiol. Heart Circ. Physiol. 294, H699–H707 (2008). This study shows that the identity of the renin phenotype is mediated by cAMP and epigenetic changes.
Chen, L. et al. Regulation of renin in mice with Cre recombinase-mediated deletion of G protein Gsalpha in juxtaglomerular cells. Am. J. Physiol. Renal Physiol. 292, F27–37 (2007).
Castellanos-Rivera, R. M. et al. Recombination signal binding protein for Ig-kappaJ region regulates juxtaglomerular cell phenotype by activating the myo-endocrine program and suppressing ectopic gene expression. J. Am. Soc. Nephrol. 26, 67–80 (2015). This study demonstrates that the Notch transducer RBP-J is crucial for the acquisition and preservation of the endocrine-contractile phenotype of renin cells.
Everett, A. D., Carey, R. M., Chevalier, R. L., Peach, M. J. & Gomez, R. A. Renin release and gene expression in intact rat kidney microvessels and single cells. J. Clin. Invest. 86, 169–175 (1990). This study demonstrates that cAMP controls the number of renin-releasing cells.
Klar, J., Sandner, P., Muller, M. W. & Kurtz, A. Cyclic AMP stimulates renin gene transcription in juxtaglomerular cells. Pflugers Arch. 444, 335–344 (2002).
Borensztein, P. et al. cis-regulatory elements and trans-acting factors directing basal and cAMP-stimulated human renin gene expression in chorionic cells. Circ. Res. 74, 764–773 (1994).
Keeton, T. K. & Campbell, W. B. The pharmacologic alteration of renin release. Pharmacol Rev. 32, 81–227 (1980).
Lin, E. E., Pentz, E. S., Sequeira-Lopez, M. L. & Gomez, R. A. Aldo-keto reductase 1b7, a novel marker for renin cells, is regulated by cyclic AMP signaling. Am. J. Physiol. Regul. Integr. Comp. Physiol. 309, R576–R584 (2015).
Brunskill, E. W. et al. Genes that confer the identity of the renin cell. J. Am. Soc. Nephrol. 22, 2213–2225 (2011).
Chen, L. et al. Renal failure in mice with Gsα deletion in juxtaglomerular cells. Am. J. Nephrol. 32, 83–94 (2010).
Artavanis-Tsakonas, S., Matsuno, K. & Fortini, M. E. Notch signaling. Science 268, 225–232 (1995).
Artavanis-Tsakonas, S., Rand, M. D. & Lake, R. J. Notch signaling: cell fate control and signal integration in development. Science 284, 770–776 (1999).
Castellanos Rivera, R. M. et al. Transcriptional regulator RBP-J regulates the number and plasticity of renin cells. Physiol. Genom. 43, 1021–1028 (2011).
Yosypiv, I. V. Prorenin receptor in kidney development. Pediatr. Nephrol. 32, 383–392 (2017).
Peters, J. The (pro)renin receptor and its interaction partners. Pflugers Arch. 469, 1245–1256 (2017). This is an excellent review on the prorenin receptor and its interacting partners.
Sun, Y., Danser, A. H. J. & Lu, X. (Pro)renin receptor as a therapeutic target for the treatment of cardiovascular diseases? Pharmacol Res. 125, 48–56 (2017).
Krop, M., Lu, X., Danser, A. H. & Meima, M. E. The (pro)renin receptor. A decade of research: what have we learned? Pflugers Arch. 465, 87–97 (2013).
Sealey, J. E. et al. Specific prorenin/renin binding (ProBP). Identification and characterization of a novel membrane site. Am. J. Hypertens. 9, 491–502 (1996).
Glorioso, N., Atlas, S. A., Laragh, J. H., Jewelewicz, R. & Sealey, J. E. Prorenin in high concentrations in human ovarian follicular fluid. Science 233, 1422–1424 (1986).
Nguyen, G. et al. Pivotal role of the renin/prorenin receptor in angiotensin II production and cellular responses to renin. J. Clin. Invest. 109, 1417–1427 (2002).
Oshima, Y. et al. Prorenin receptor is essential for normal podocyte structure and function. J. Am. Soc. Nephrol. 22, 2203–2212 (2011).
Song, R. et al. Prorenin receptor is critical for nephron progenitors. Dev. Biol. 409, 382–391 (2016).
Hunt, M. K. et al. Colocalization and release of angiotensin and renin in renal cortical cells. Am. J. Physiol. 263, F363–373 (1992).
Geary, K. M., Hunt, M. K., Peach, M. J., Gomez, R. A. & Carey, R. M. Effects of angiotensin converting enzyme inhibition, sodium depletion, calcium, isoproterenol, and angiotensin II on renin secretion by individual renocortical cells. Endocrinology 131, 1588–1594 (1992).
Matsusaka, T. et al. Chimeric mice carrying 'regional' targeted deletion of the angiotensin type 1A receptor gene. Evidence against the role for local angiotensin in the in vivo feedback regulation of renin synthesis in juxtaglomerular cells. J. Clin. Invest. 98, 1867–1877 (1996).
Cantin, M., Araujo-Nascimento, M. D., Benchimol, S. & Desormeaux, Y. Metaplasia of smooth muscle cells into juxtaglomerular cells in the juxtaglomerular apparatus, arteries, and arterioles of the ischemic (endocrine) kidney. An ultrastructural-cytochemical and autoradiographic study. Am. J. Pathol. 87, 581–602 (1977). This paper presents the first description of the ability of renal arteriolar cells to switch phenotype into renin-expressing cells.
Tufro-McReddie, A., Chevalier, R. L., Everett, A. D. & Gomez, R. A. Decreased perfusion pressure modulates renin and ANG II type 1 receptor gene expression in the rat kidney. Am. J. Physiol. 264, R696–R702 (1993).
Tufro-McReddie, A. et al. Effect of CsA on the expression of renin and angiotensin type 1 receptor genes in the rat kidney. Kidney Int. 43, 615–622 (1993).
el-Dahr, S. S. et al. Expression of renin and its mRNA in the adult rat kidney with chronic ureteral obstruction. Am. J. Kidney Dis. 15, 575–582 (1990).
Kim, H. S. et al. Homeostasis in mice with genetically decreased angiotensinogen is primarily by an increased number of renin-producing cells. J. Biol. Chem. 274, 14210–14217 (1999). This study demonstrates that the number of renin-producing cells determines blood pressure and fluid–electrolyte homeostasis.
Norwood, V. F. et al. Neonatal ureteral obstruction stimulates recruitment of renin-secreting renal cortical cells. Kidney Int. 45, 1333–1339 (1994).
Berg, A. C., Chernavvsky-Sequeira, C., Lindsey, J., Gomez, R. A. & Sequeira-Lopez, M. L. Pericytes synthesize renin. World J. Nephrol. 2, 11–16 (2013).
Gomez, R. A. et al. Recruitment of renin gene-expressing cells in adult rat kidneys. Am. J. Physiol. 259, F660–F665 (1990). This study shows that the increase in renin cells along the kidney vasculature is due to increased renin gene expression by smooth muscle cells and not to absorption of the circulating protein.
Ray, P. E. et al. Renal vascular induction of TGF-beta 2 and renin by potassium depletion. Kidney Int. 44, 1006–1013 (1993).
Chen, M. et al. Renin and renin mRNA in proximal tubules of the rat kidney. J. Clin. Invest. 94, 237–243 (1994).
Rohrwasser, A. et al. Renin and kallikrein in connecting tubule of mouse. Kidney Int. 64, 2155–2162 (2003).
Prieto-Carrasquero, M. C. et al. Collecting duct renin is upregulated in both kidneys of 2-kidney, 1-clip goldblatt hypertensive rats. Hypertension 51, 1590–1596 (2008).
Kang, J. J. et al. The collecting duct is the major source of prorenin in diabetes. Hypertension 51, 1597–1604 (2008).
Gomez, R. A., Belyea, B., Medrano, S., Pentz, E. S. & Sequeira-Lopez, M. L. Fate and plasticity of renin precursors in development and disease. Pediatr. Nephrol. 29, 721–726 (2014).
Kurt, B., Karger, C., Wagner, C. & Kurtz, A. Control of renin secretion from kidneys with renin cell hyperplasia. Am. J. Physiol. Renal Physiol. 306, F327–F332 (2014).
Cantin, M. et al. Metaplastic and mitotic activity of the ischemic (endocrine) kidney in experimental renal hypertension. Am. J. Pathol. 96, 545–566 (1979).
Wagner, C. et al. Selective deletion of Connexin 40 in renin-producing cells impairs renal baroreceptor function and is associated with arterial hypertension. Kidney Int. 78, 762–768 (2010). This study demonstrates the mislocation of renin cells and malignant hypertension in CX40 deficient mice.
Sequeira Lopez, M. L. & Gomez, R. A. Novel mechanisms for the control of renin synthesis and release. Curr. Hypertens. Rep. 12, 26–32 (2010).
Sequeira-Lopez, M. L. et al. The microRNA-processing enzyme dicer maintains juxtaglomerular cells. J. Am. Soc. Nephrol. 21, 460–467 (2010). This study demonstrates that microRNAs are required for the acquisition and maintenance of renin-expressing cells.
Medrano, S., Monteagudo, M. C., Sequeira-Lopez, M. L., Pentz, E. S. & Gomez, R. A. Two microRNAs, miR-330 and miR-125b-5p, mark the juxtaglomerular cell and balance its smooth muscle phenotype. Am. J. Physiol. Renal Physiol. 302, F29–F37 (2012).
Hickmann, L. et al. Persistent and inducible neogenesis repopulates progenitor renin lineage cells in the kidney. Kidney Int. 92, 1419–1432 (2017). This paper presents the first description of neogenesis, a mechanism to repopulate renin cell progenitors during development, adult life and in response to homeostatic challenge.
Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Wang, H. et al. Adult renal mesenchymal stem cell-like cells contribute to juxtaglomerular cell recruitment. J. Am. Soc. Nephrol. 24, 1263–1273 (2013).
Yang, Y. et al. Salt restriction leads to activation of adult renal mesenchymal stromal cell-like cells via prostaglandin E2 and E-prostanoid receptor 4. Hypertension 65, 1047–1054 (2015).
Peti-Peterdi, J. Newly stemming functions of macula densa-derived prostanoids. Hypertension 65, 987–988 (2015).
Qian, H., Le Blanc, K. & Sigvardsson, M. Primary mesenchymal stem and progenitor cells from bone marrow lack expression of CD44 protein. J. Biol. Chem. 287, 25795–25807 (2012).
Lattion, A. L., Michel, J. B., Arnauld, E., Corvol, P. & Soubrier, F. Myocardial recruitment during ANF mRNA increase with volume overload in the rat. Am. J. Physiol. 251, H890–H896 (1986).
Gerber, H. et al. Transformation of normal thyroids into colloid goiters in rats and mice by diphenylthiohydantoin. Endocrinology 135, 2688–2699 (1994).
Gerber, H., Peter, H. J., Bachmeier, C., Kaempf, J. & Studer, H. Progressive recruitment of follicular cells with graded secretory responsiveness during stimulation of the thyroid gland by thyrotropin. Endocrinology 120, 91–96 (1987).
Schuit, F. C., In' t Veld, P. A. & Pipeleers, D. G. Glucose stimulates proinsulin biosynthesis by a dose-dependent recruitment of pancreatic beta cells. Proc. Natl Acad. Sci. USA 85, 3865–3869 (1988).
Eckardt, K. U. et al. Distribution of erythropoietin producing cells in rat kidneys during hypoxic hypoxia. Kidney Int. 43, 815–823 (1993).
Starke, C. et al. Renin lineage cells repopulate the glomerular mesangium after injury. J. Am. Soc. Nephrol. 26, 48–54 (2015). This study demonstrates mesangial cell regeneration by renin-expressing cells.
Kaverina, N. V. et al. Tracking the stochastic fate of cells of the renin lineage after podocyte depletion using multicolor reporters and intravital imaging. PLoS ONE 12, e0173891 (2017).
Pippin, J. W. et al. Cells of renin lineage are progenitors of podocytes and parietal epithelial cells in experimental glomerular disease. Am. J. Pathol. 183, 542–557 (2013). This study demonstrates that juxtaglomerular cells repopulate podocytes in experimental glomerulonephritis.
Lahiri, S., Cherniack, N. S. & Fitzgerald, R. S. Response and Adaptation to Hypoxia: Organ to Organelle. (Oxford Univ. Press, 1991).
Ritthaler, T., Schricker, K., Kees, F., Kramer, B. & Kurtz, A. Acute hypoxia stimulates renin secretion and renin gene expression in vivo but not in vitro. Am. J. Physiol. 272, R1105–R1111 (1997).
Kurtz, A. Endocrine functions of the renal interstitium. Pflugers Arch. 469, 869–876 (2017).
Haase, V. H. Regulation of erythropoiesis by hypoxia-inducible factors. Blood Rev. 27, 41–53 (2013).
Zeisberg, M. & Kalluri, R. Physiology of the renal interstitium. Clin. J. Am. Soc. Nephrol. 10, 1831–1840 (2015).
Stefanska, A. et al. Cells of renin lineage express hypoxia inducible factor 2alpha following experimental ureteral obstruction. BMC Nephrol. 17, 5 (2016).
Stefanska, A. et al. Human kidney pericytes produce renin. Kidney Int. 90, 1251–1261 (2016).
Kurt, B. et al. Deletion of von Hippel-Lindau protein converts renin-producing cells into erythropoietin-producing cells. J. Am. Soc. Nephrol. 24, 433–444 (2013). This paper presents an extreme example of plasticity — the transformation of one endocrine cell into another.
Kurt, B., Gerl, K., Karger, C., Schwarzensteiner, I. & Kurtz, A. Chronic hypoxia-inducible transcription factor-2 activation stably transforms juxtaglomerular renin cells into fibroblast-like cells in vivo. J. Am. Soc. Nephrol. 26, 587–596 (2015).
Lundby, C. & Olsen, N. V. Effects of recombinant human erythropoietin in normal humans. J. Physiol. 589, 1265–1271 (2011).
Olsen, N. V. et al. Erythropoietin down-regulates proximal renal tubular reabsorption and causes a fall in glomerular filtration rate in humans. J. Physiol. 589, 1273–1281 (2011).
Gomez, R. A. et al. Leukocytes synthesize angiotensinogen. Hypertension 21, 470–475 (1993).
Haznedaroglu, I. C. & Beyazit, Y. Local bone marrow renin-angiotensin system in primitive, definitive and neoplastic haematopoiesis. Clin. Sci. 124, 307–323 (2013).
Bernstein, K. E. et al. Overexpression of angiotensin-converting enzyme in myelomonocytic cells enhances the immune response. F1000Res 5, 393 (2016).
Harrison, D. G. The immune system in hypertension. Trans. Am. Clin. Climatol Assoc. 125, 130–140 (2014).
Harrison, D. G. et al. Inflammation, immunity, and hypertension. Hypertension 57, 132–140 (2011).
Harrison, D. G., Marvar, P. J. & Titze, J. M. Vascular inflammatory cells in hypertension. Front. Physiol. 3, 128 (2012).
Okwan-Duodu, D. et al. Angiotensin-converting enzyme overexpression in mouse myelomonocytic cells augments resistance to Listeria and methicillin-resistant Staphylococcus aureus. J. Biol. Chem. 285, 39051–39060 (2010). This study demonstrates that ACE overexpression in monocytes confers resistance to major infectious agents.
Klickstein, L. B., Kaempfer, C. E. & Wintroub, B. U. The granulocyte-angiotensin system. Angiotensin I-converting activity of cathepsin G. J. Biol. Chem. 257, 15042–15046 (1982).
Wintroub, B. U., Klickstein, L. B., Dzau, V. J. & Watt, K. W. Granulocyte-angiotensin system. Identification of angiotensinogen as the plasma protein substrate of leukocyte cathepsin G. Biochemistry 23, 227–232 (1984).
Gomez, D. & Owens, G. K. Smooth muscle cell phenotypic switching in atherosclerosis. Cardiovasc. Res. 95, 156–164 (2012).
Khan, Z. et al. Angiotensin-converting enzyme enhances the oxidative response and bactericidal activity of neutrophils. Blood 130, 328–339 (2017). This study demonstrates that ACE in neutrophils confers enhanced resistance to infection independently of angiotensin generation.
Fournier, D., Luft, F. C., Bader, M., Ganten, D. & Andrade-Navarro, M. A. Emergence and evolution of the renin-angiotensin-aldosterone system. J. Mol. Med. 90, 495–508 (2012). This paper is an excellent review on the evolution of the RAS.
Sokabe, H. & Ogawa, M. Comparative studies of the juxtaglomerular apparatus. Int. Rev. Cytol. 37, 271–327 (1974).
Hilgers, K. F., Reddi, V., Krege, J. H., Smithies, O. & Gomez, R. A. Aberrant renal vascular morphology and renin expression in mutant mice lacking angiotensin-converting enzyme. Hypertension 29, 216–221 (1997).
Oka, M., Medrano, S., Sequeira-Lomicronpez, M. L. S. & Gomez, R. A. Chronic stimulation of renin cells leads to vascular pathology. Hypertension 70, 119–128 (2017). This study demonstrates that cells of the renin lineage contribute to concentric renal arteriolar hypertrophy when overly and chronically stimulated.
Okubo, S. et al. Angiotensinogen gene null-mutant mice lack homeostatic regulation of glomerular filtration and tubular reabsorption. Kidney Int. 53, 617–625 (1998).
Takahashi, N. et al. Ren1c homozygous null mice are hypotensive and polyuric, but heterozygotes are indistinguishable from wild-type. J. Am. Soc. Nephrol. 16, 125–132 (2005).
Tanimoto, K. et al. Angiotensinogen-deficient mice with hypotension. J. Biol. Chem. 269, 31334–31337 (1994).
Tsuchida, S. et al. Murine double nullizygotes of the angiotensin type 1A and 1B receptor genes duplicate severe abnormal phenotypes of angiotensinogen nullizygotes. J. Clin. Invest. 101, 755–760 (1998).
Tufro-McReddie, A., Romano, L. M., Harris, J. M., Ferder, L. & Gomez, R. A. Angiotensin II regulates nephrogenesis and renal vascular development. Am. J. Physiol. 269, F110–F115 (1995). This paper presents the first experimental demonstration that administration of angiotensin-receptor inhibitors in early life results in serious vascular abnormalities both in rodents and frogs.
Gribouval, O. et al. Spectrum of mutations in the renin-angiotensin system genes in autosomal recessive renal tubular dysgenesis. Hum. Mutat. 33, 316–326 (2012).
Esther, C. R. et al. Mice lacking angiotensin-converting enzyme have low blood pressure, renal pathology, and reduced male fertility. Lab Invest. 74, 953–965 (1996).
Krege, J. H. et al. Male-female differences in fertility and blood pressure in ACE-deficient mice. Nature 375, 146–148 (1995).
Yanai, K. et al. Regulated expression of human angiotensinogen gene by hepatocyte nuclear factor 4 and chicken ovalbumin upstream promoter-transcription factor. J. Biol. Chem. 274, 34605–34612 (1999).
Nagata, M. et al. Nephrogenesis and renovascular development in angiotensinogen-deficient mice. Lab. Invest. 75, 745–753 (1996).
Moniwa, N. et al. Hemodynamic and hormonal changes to dual renin-angiotensin system inhibition in experimental hypertension. Hypertension 61, 417–424 (2013).
Sequeira-Lopez, M. L., Nagalakshmi, V. K., Li, M., Sigmund, C. D. & Gomez, R. A. Vascular versus tubular renin: role in kidney development. Am. J. Physiol. Regul. Integr. Comp. Physiol. 309, R650–R657 (2015).
Pentz, E. S., Moyano, M. A., Thornhill, B. A., Sequeira Lopez, M. L. & Gomez, R. A. Ablation of renin-expressing juxtaglomerular cells results in a distinct kidney phenotype. Am. J. Physiol. Regul. Integr. Comp. Physiol. 286, R474–R483 (2004).
Gomez, R. A. & Sequeira Lopez, M. L. Who and where is the renal baroreceptor?: the connexin hypothesis. Kidney Int. 75, 460–462 (2009).
Acknowledgements
The authors thank H. Watanabe, B. Belyea and E. Brown (University of Virginia, Charlottesville, USA) for their comments on the manuscript and figures. Studies were funded by the US National Institutes of Health, Grants DK-096373 and HL-096735 to R.A.G. and DK-091330 and DK-096373 to M.L.S.S.-L.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to researching data for the article, discussion of the content, writing the article and revising and/or editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Juxtaglomerular cells
-
A specialized group of renin-producing, myo-epithelioid, granulated cells located at the tip of the afferent arterioles at the entrance to the glomeruli.
- Juxtaglomerular apparatus
-
The functional sensory and responding unit of the renin–angiotensin system; it is composed of the afferent and efferent arterioles, the macula densa and the extraglomerular mesangium.
- Plasticity
-
The ability of cells to switch phenotypes.
- Metanephros
-
The definitive kidney of adult mammals.
- Fetal zone
-
A large, eosinophilic steroidogenic group of cells of the fetal adrenal gland, which is believed to involute after birth.
- Mural cells
-
Cells located within the wall(s) of the renal arterioles, including smooth muscle cells and renin-expressing cells such as juxtaglomerular cells. In larger arteries, adventitial cells such as fibroblasts and perivascular pericytes are sometimes considered mural cells. Although part of the arterial wall, endothelial cells are not usually considered mural cells.
- Stromal compartment
-
One of the three main compartments of the developing kidney; it contains FOXD1+ progenitors of vascular smooth muscle cells, renin-expressing cells, mesangial cells and pericytes as well as SCL+ progenitors of endothelial cells of arteries and arterioles, glomeruli and interstitial capillaries.
- Vasculogenesis
-
De novo, local differentiation and assembly of blood vessels from resident progenitor cells that are usually derived from multiple clones.
- Angiogenesis
-
The formation of new blood vessels, usually by sprouting of new branches from pre-existing vessels. This process usually involves the proliferation and/or elongation of cells from single clones. In the case of arterioles, it is followed by the recruitment of perivascular smooth muscle cells.
- Fractal
-
A shape or structure made of smaller parts or units that repeat themselves and resemble the whole.
- Developmental memory
-
The ability of cells to expressly re-acquire a phenotype previously expressed during development.
- Metaplastic transformation
-
Pathological transformation of one cell type into another not related by lineage.
- Hyperplasia
-
Increase in cell number, usually due to increased cell proliferation.
- Neogenesis
-
De novo expression of a gene (for example, renin) in a cell that has not previously expressed the gene.
- Direct transdifferentiation
-
Transformation of one cell into another without reprogramming into induced pluripotent stem cells.
- Erythrocytosis
-
Increase in red cell mass.
- Leukaemic blast cells
-
Immature forms of white blood cells that fail to differentiate normally and proliferate in an uncontrolled manner, leading to tissue infiltration and, if untreated, death.
Rights and permissions
About this article
Cite this article
Gomez, R., Sequeira-Lopez, M. Renin cells in homeostasis, regeneration and immune defence mechanisms. Nat Rev Nephrol 14, 231–245 (2018). https://doi.org/10.1038/nrneph.2017.186
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2017.186
This article is cited by
-
Piezo2 expression and its alteration by mechanical forces in mouse mesangial cells and renin-producing cells
Scientific Reports (2022)
-
Flexible and multifaceted: the plasticity of renin-expressing cells
Pflügers Archiv - European Journal of Physiology (2022)
-
Anticipated pharmacological role of Aviptadil on COVID-19
Environmental Science and Pollution Research (2022)
-
Endocrine disorders in patients with Fabry disease: insights from a reference centre prospective study
Endocrine (2022)
-
A cross-species analysis of systemic mediators of repair and complex tissue regeneration
npj Regenerative Medicine (2021)