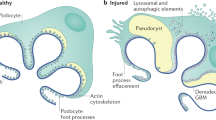
Abstract
Despite improvements in immunosuppressive therapy, long-term allograft survival after kidney transplantation remains as low as 50%. Chronic allograft nephropathy (CAN) is a major cause of late graft loss in renal transplant recipients. The histopathologic signs of CAN—interstitial fibrosis, tubular atrophy, glomerulopathy and vasculopathy—are nonspecific; therefore, the 2007 Banff classification dispensed with the term CAN in favor of 'interstitial fibrosis and tubular atrophy without evidence of any specific etiology'. In this Review, however, the term CAN is used to describe a clinical syndrome that is characterized by progressive decline in renal function from 3 months after transplantation, accompanied by the development of proteinuria and hypertension. The pathogenesis of CAN is complex and incompletely understood, and involves several immunological and non-immunological factors. We discuss the contributory roles of acute rejection, donor age, anti-human-leukocyte-antigen antibodies, calcineurin inhibitor nephrotoxic effects, viral infection, hypertension and hyperlipidemia. The prevention and treatment of CAN needs multidisciplinary strategies. Early detection by means of protocol biopsy and calculation of glomerular filtration rate is the first step, followed by management of modifiable risk factors.
Key Points
-
Chronic allograft nephropathy (CAN) is characterized by declining graft function, hypertension and proteinuria; the accompanying pathology is defined as 'interstitial fibrosis and tubular atrophy without evidence of any specific etiology'
-
Acute rejection, donor age, anti-human-leukocyte-antigen antibodies, viral infection, hypertension, hyperlipidemia and use of calcineurin inhibitors contribute to CAN
-
Prolonged endoplasmic reticulum stress caused by the calcineurin inhibitor ciclosporin induces apoptotic cell death in renal allografts by depleting molecular chaperones
-
Protocol biopsy and calculation of glomerular filtration rate are recommended to facilitate early diagnosis of CAN, and reduction of exposure to calcineurin inhibitors (for example, by substitution of non-nephrotoxic drugs) is advisable
-
Angiotensin-converting-enzyme inhibitors or angiotensin receptor blockers are recommended to treat hypertension and proteinuria, and ezetimibe, a novel inhibitor of intestinal cholesterol absorption, can be used in cases of statin-resistant hyperlipidemia
-
CAN in grafted kidneys mirrors chronic kidney disease in native kidneys; therefore, patients with CAN should be managed according to their stage of chronic kidney disease
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others

References
Meier-Kriesche, H. U., Schold, J. D., Srinivas, T. R. & Kaplan, B. Lack of improvement in renal allograft survival despite a marked decrease in acute rejection rates over the most recent era. Am. J. Transplant. 4, 378–383 (2004).
Harris, S. et al. TGF-β1 in chronic allograft nephropathy following renal transplantation. J. Nephrol. 20, 177–185 (2007).
Nankivell, B. J. et al. The natural history of chronic allograft nephropathy. N. Engl. J. Med. 349, 2326–2333 (2003).
Kasiske, B. L., Heim-Duthoy, K. L., Tortorice, K. L. & Rao, K. V. The variable nature of chronic declines in renal allograft function. Transplantation 51, 330–334 (1991).
Solez, K. et al. Banff 07 classification of renal allograft pathology: updates and future directions. Am. J. Transplant. 8, 753–760 (2008).
Massy, Z. A., Guijarro, C., Wiederkehr, M. R., Ma, J. Z. & Kasiske, B. L. Chronic renal allograft rejection: immunologic and nonimmunologic risk factors. Kidney Int. 49, 518–524 (1996).
Matas, A. J., Gillingham, K. J., Payne, W. D. & Najarian, J. S. The impact of an acute rejection episode on long-term renal allograft survival (t1/2). Transplantation 57, 857–859 (1994).
Cosio, F. G. et al. Impact of acute rejection and early allograft function on renal allograft survival. Transplantation 63, 1611–1615 (1997).
McDonald, S., Russ, G., Campbell, S. & Chadban, S. Kidney transplant rejection in Australia and New Zealand: relationships between rejection and graft outcome. Am. J. Transplant. 7, 1201–1208 (2007).
Nickerson, P. et al. Identification of clinical and histopathologic risk factors for diminished renal function 2 years post-transplant. J. Am. Soc. Nephrol. 9, 482–487 (1998).
Halloran, P. F., Melk, A. & Barth, C. Rethinking chronic allograft nephropathy: the concept of accelerated senescence. J. Am. Soc. Nephrol. 10, 167–181 (1999).
Terasaki, P. I., Koyama, H., Cecka, J. M. & Gjertson, D. W. The hyperfiltration hypothesis in human renal transplantation. Transplantation 57, 1450–1454 (1994).
Humar, A. et al. Effect of initial slow graft function on renal allograft rejection and survival. Clin. Transplant. 11, 623–627 (1997).
Terasaki, P. I., Gjertson, D. W., Cecka, J. M., Takemoto, S. & Cho, Y. W. Significance of the donor age effect on kidney transplants. Clin. Transplant. 11, 366–372 (1997).
Kasiske, B. L., Snyder, J. J., Gilbertson, D. & Matas, A. J. Diabetes mellitus after kidney transplantation in the United States. Am. J. Transplant. 3, 178–185 (2003).
Heisel, O., Heisel, R., Balshaw, R. & Keown, P. New onset diabetes mellitus in patients receiving calcineurin inhibitors: a systematic review and meta-analysis. Am. J. Transplant. 4, 583–595 (2004).
Kahan, B. D. Efficacy of sirolimus compared with azathioprine for reduction of acute renal allograft rejection: a randomised multicentre study: The Rapamune US Study Group. Lancet 356, 194–202 (2000).
Johnston, O., Rose, C. L., Webster, A. C. & Gill, J. S. Sirolimus is associated with new-onset diabetes in kidney transplant recipients. J. Am. Soc. Nephrol. 19, 1411–1418 (2008).
Teutonico, A., Schena, P. F. & Di Paolo, S. Glucose metabolism in renal transplant recipients: effect of calcineurin inhibitor withdrawal and conversion to sirolimus. J. Am. Soc. Nephrol. 16, 3128–3135 (2005).
Guijarro, C., Massy, Z. A. & Kasiske, B. L. Clinical correlation between renal allograft failure and hyperlipidemia. Kidney Int. Suppl. 52, S56–S59 (1995).
Dimény, E. et al. The influence of pretransplant lipoprotein abnormalities on the early results of renal transplantation. Eur. J. Clin. Invest. 23, 572–579 (1993).
Isoniemi, H. et al. Risk factors predicting chronic rejection of renal allografts. Transplantation 57, 68–72 (1994).
Markell, M. S., Sumrani, N., DiBenedetto, A. & Friedman, E. A. Effect of early hyperlipidemia on graft and patient survival in cyclosporine-treated renal transplant patients. Am. J. Kidney Dis. 22, 233–239 (1993).
Opelz, G., Wujciak, T. & Ritz, E. Association of chronic kidney graft failure with recipient blood pressure. Collaborative Transplant Study. Kidney Int. 53, 217–222 (1998).
Mackenzie, H. S. et al. Candesartan cilexetil reduces chronic renal allograft injury in Fisher-->Lewis rats. J. Hypertens. Suppl. 15, S21–S25 (1997).
Amuchastegui, S. C. Chronic allograft nephropathy in the rat is improved by angiotensin II receptor blockade but not by calcium channel antagonism. J. Am. Soc. Nephrol. 9, 1948–1955 (1998).
Iñigo, P. et al. Effects of losartan and amlodipine on intrarenal hemodynamics and TGF-β(1) plasma levels in a crossover trial in renal transplant recipients. J. Am. Soc. Nephrol. 12, 822–827 (2001).
Lin, J. et al. Angiotensin converting enzyme inhibition in chronic allograft nephropathy. Transplantation 73, 783–788 (2002).
Gao, S. Z. et al. Early development of accelerated graft coronary artery disease: risk factors and course. J. Am. Coll. Cardiol. 28, 673–679 (1996).
Koskinen, P., Lemstrøm, K., Mattila, S., Häyry, P. & Nieminen, M. S. Cytomegalovirus infection associated accelerated heart allograft arteriosclerosis may impair the late function of the graft. Clin. Transplant. 10, 487–493 (1996).
O'Grady, J. G. et al. Cytomegalovirus infection and donor/recipient HLA antigens: interdependent co-factors in pathogenesis of vanishing bile-duct syndrome after liver transplantation. Lancet 2, 302–305 (1988).
Nadasdy, T. et al. Absence of association between cytomegalovirus infection and obliterative transplant arteriopathy in renal allograft rejection. Mod. Pathol. 7, 289–294 (1994).
Nickeleit, V. et al. Testing for polyomavirus type BK DNA in plasma to identify renal-allograft recipients with viral nephropathy. N. Engl. J. Med. 342, 1309–1315 (2000).
Colvin, R. B. & Mauiyyedi, S. Differential diagnosis between infection and rejection in renal allografts. Transplant. Proc. 33, 1778–1779 (2001).
Purighalla, R., Shapiro, R., McCauley, J. & Randhawa, P. BK virus infection in a kidney allograft diagnosed by needle biopsy. Am. J. Kidney Dis. 26, 671–673 (1995).
Binet, I. et al. Polyomavirus disease under new immunosuppressive drugs: a cause of renal graft dysfunction and graft loss. Transplantation 67, 918–922 (1999).
Hogan, T. F., Borden, E. C., McBain, J. A., Padgett, B. L. & Walker, D. L. Human polyomavirus infections with JC virus and BK virus in renal transplant patients. Ann. Intern. Med. 92, 373–378 (1980).
Gardner, S. D., MacKenzie, E. F., Smith, C. & Porter, A. A. Prospective study of the human polyomaviruses BK and JC and cytomegalovirus in renal transplant recipients. J. Clin. Pathol. 37, 578–586 (1984).
Dunn, J. et al. Causes of graft loss beyond two years in the cyclosporine era. Transplantation 49, 349–353 (1990).
Waltzer, W. C., Miller, F., Arnold, A., Anaise, D. & Rapaport, F. T. Immunohistologic analysis of human renal allograft dysfunction. Transplantation 43, 100–105 (1987).
de Mattos, A. M., Olyaei, A. J. & Bennett, W. M. Nephrotoxicity of immunosuppressive drugs: long-term consequences and challenges for the future. Am. J. Kidney Dis. 35, 333–346 (2000).
Olyaei, A. J., de Mattos, A. M. & Bennett, W. M. Nephrotoxicity of immunosuppressive drugs: new insight and preventive strategies. Curr. Opin. Crit. Care 7, 384–389 (2001).
Yang, C. W. et al. Influence of the renin-angiotensin system on epidermal growth factor expression in normal and cyclosporine-treated rat kidney. Kidney Int. 60, 847–857 (2001).
Myers, B. D. et al. The long-term course of cyclosporine-associated chronic nephropathy. Kidney Int. 33, 590–600 (1988).
Liptak, P. & Ivanyi, B. Primer: Histopathology of calcineurin-inhibitor toxicity in renal allografts. Nat. Clin. Pract. Nephrol. 2, 398–404 (2006).
Shimizu, T. et al. Clinical and histological analysis of chronic tacrolimus nephrotoxicity in renal allografts. Transplant. Proc. 40, 2370–2372 (2008).
Nankivell, B. J. et al. Natural history, risk factors, and impact of subclinical rejection in kidney transplantation. Transplantation 78, 242–249 (2004).
Li, C. et al. Pravastatin treatment attenuates interstitial inflammation and fibrosis in a rat model of chronic cyclosporine-induced nephropathy. Am. J. Physiol. Renal Physiol. 286, F46–F57 (2004).
Li, C. et al. Reversibility of chronic cyclosporine nephropathy in rats after withdrawal of cyclosporine. Am. J. Physiol. Renal Physiol. 284, F389–F398 (2003).
Li, C. et al. Inhibitory effect of pravastatin on transforming growth factor β1-inducible gene h3 expression in a rat model of chronic cyclosporine nephropathy. Am. J. Nephrol. 25, 611–620 (2005).
Sun, B. K. et al. Expression of transforming growth factor-β-inducible gene-h3 in normal and cyclosporine-treated rat kidney. J. Lab. Clin. Med. 143, 175–183 (2004).
Li, C. et al. Combined effects of losartan and pravastatin on interstitial inflammation and fibrosis in chronic cyclosporine-induced nephropathy. Transplantation 79, 1522–1529 (2005).
Yang, C. W. et al. Synergistic effects of mycophenolate mofetil and losartan in a model of chronic cyclosporine nephropathy. Transplantation 75, 309–315 (2003).
Sun, B. K. et al. Blockade of angiotensin II with losartan attenuates transforming growth factor-β1 inducible gene-h3 (betaig-h3) expression in a model of chronic cyclosporine nephrotoxicity. Nephron Exp. Nephrol. 99, e9–e16 (2005).
Yang, C. W. et al. Expression of apoptosis-related genes in chronic cyclosporine nephrotoxicity in mice. Am. J. Transplant. 2, 391–399 (2002).
Yang, C. W. et al. Cyclosporine withdrawal and mycophenolate mofetil treatment effects on the progression of chronic cyclosporine nephrotoxicity. Kidney Int. 62, 20–30 (2002).
Han, S. W. et al. Prolonged endoplasmic reticulum stress induces apoptotic cell death in an experimental model of chronic cyclosporine nephropathy. Am. J. Nephrol. 28, 707–714 (2008).
Li, C., Lim, S. W., Sun, B. K. & Yang, C. W. Chronic cyclosporine nephrotoxicity: new insights and preventive strategies. Yonsei Med. J. 45, 1004–1016 (2004).
Li, C. et al. Expression of apoptosis-related factors in chronic cyclosporine nephrotoxicity after cyclosporine withdrawal. Acta Pharmacol. Sin. 25, 401–411 (2004).
Li, C. et al. Colchicine decreases apoptotic cell death in chronic cyclosporine nephrotoxicity. J. Lab. Clin. Med. 139, 364–371 (2002).
Thomas, S. E. et al. Accelerated apoptosis characterizes cyclosporine-associated interstitial fibrosis. Kidney Int. 53, 897–908 (1998).
Yang, C. W. et al. Oral supplementation of L-arginine prevents chronic cyclosporine nephrotoxicity in rats. Exp. Nephrol. 6, 50–56 (1998).
Lim, S. W. et al. Cyclosporine-induced renal injury induces toll-like receptor and maturation of dendritic cells. Transplantation 80, 691–699 (2005).
Ahn, K. O. et al. Influence of angiotensin II on expression of toll-like receptor 2 and maturation of dendritic cells in chronic cyclosporine nephropathy. Transplantation 83, 938–947 (2007).
Kim, J. Y. et al. Effect of FTY720 on chronic cyclosporine nephropathy in rats. Transplantation 80, 1323–1330 (2005).
Ahn, K. O. et al. Infiltration of nestin-expressing cells in interstitial fibrosis in chronic cyclosporine nephropathy. Transplantation 86, 571–517 (2008).
Isoniemi, H. M. et al. Histopathological findings in well-functioning, long-term renal allografts. Kidney Int. 41, 155–160 (1992).
Isoniemi, H. M. et al. Long-term consequences of different immunosuppressive regimens for renal allografts. Transplantation 55, 494–499 (1993).
Pallet, N. et al. Cyclosporine-induced endoplasmic reticulum stress triggers tubular phenotypic changes and death. Am. J. Transplant. 8, 2283–2296 (2008).
Mao, Q. et al. Extremely high association between appearance of HLA antibodies and failure of kidney grafts in a five-year longitudinal study. Am. J. Transplant. 7, 864–871 (2007).
Gerbase-DeLima, M. et al. Anti-HLA class II antibodies and chronic allograft nephropathy. Clin. Transpl. 201–205 (2006).
Haas, M. et al. Subclinical acute antibody-mediated rejection in positive crossmatch renal allografts. Am. J. Transplant. 7, 576–585 (2007).
Chapman, J. R., O'Connell, P. J. & Nankivell, B. J. Chronic renal allograft dysfunction. J. Am. Soc. Nephrol. 16, 3015–3026 (2005).
Zarkhin, V. et al. Characterization of intra-graft B cells during renal allograft rejection. Kidney Int. 74, 664–673 (2008).
Choi, B. S. et al. Clinical significance of an early protocol biopsy in living-donor renal transplantation: ten-year experience at a single center. Am. J. Transplant. 5, 1354–1360 (2005).
Vincenti, F. et al. Costimulation blockade with belatacept in renal transplantation. N. Engl. J. Med. 353, 770–781 (2005).
Oberbauer, R. et al. Early cyclosporine withdrawal from a sirolimus-based regimen results in better renal allograft survival and renal function at 48 months after transplantation. Transpl. Int. 18, 22–28 (2005).
Pascual, J. Concentration-controlled everolimus (Certican): combination with reduced dose calcineurin inhibitors. Transplantation 79, S76–S79 (2005).
Kirk, A. D. et al. Results from a human renal allograft tolerance trial evaluating T-cell depletion with alemtuzumab combined with deoxyspergualin. Transplantation 80, 1051–1059 (2005).
Ekberg, H. et al. Reduced exposure to calcineurin inhibitors in renal transplantation. N. Engl. J. Med. 357, 2562–2575 (2007).
Heinze, G. et al. Angiotensin-converting enzyme inhibitor or angiotensin II type 1 receptor antagonist therapy is associated with prolonged patient and graft survival after renal transplantation. J. Am. Soc. Nephrol. 17, 889–899 (2006).
Opelz, G., Zeier, M., Laux, G., Morath, C. & Döhler, B. No improvement of patient or graft survival in transplant recipients treated with angiotensin-converting enzyme inhibitors or angiotensin II type 1 receptor blockers: a collaborative transplant study report. J. Am. Soc. Nephrol. 17, 3257–3262 (2006).
Midtvedt, K. et al. Sustained improvement of renal graft function for two years in hypertensive renal transplant recipients treated with nifedipine as compared to lisinopril. Transplantation 72, 1787–1792 (2001).
Rahn, K. H. et al. Effect of nitrendipine on renal function in renal-transplant patients treated with cyclosporin: a randomised trial. Lancet 354, 1415–1420 (1999).
van Riemsdijk, I. C. et al. Addition of isradipine (Lomir) results in a better renal function after kidney transplantation: a double-blind, randomized, placebo-controlled, multi-center study. Transplantation 70, 122–126 (2000).
Madsen, J. K., Sørensen, S. S., Hansen, H. E. & Pedersen, E. B. The effect of felodipine on renal function and blood pressure in cyclosporin-treated renal transplant recipients during the first three months after transplantation. Nephrol. Dial. Transplant. 13, 2327–2334 (1998).
Holdaas, H. et al. Effect of fluvastatin on cardiac outcomes in renal transplant recipients: a multicentre, randomised, placebo-controlled trial. Lancet 361, 2024–2031 (2003).
Palinski, W. Immunomodulation: a new role for statins? Nat. Med. 6, 1311–1312 (2006).
Kwak, B., Mulhaupt, F., Myit, S. & Mach, F. Statins as a newly recognized type of immunomodulator. Nat. Med. 6, 1399–1402 (2000).
Jones, T. The effect of HMG-CoA reductase inhibitors on chronic allograft rejection. Expert Opin. Emerg. Drugs 6, 95–109 (2001).
Chuang, P. & Langone, A. J. Ezetimibe reduces low-density lipoprotein cholesterol (LDL-C) in renal transplant patients resistant to HMG-CoA reductase inhibitors. Am. J. Ther. 14, 438–441 (2007).
Hur, K. Y. et al. Risk factors associated with the onset and progression of post-transplantation diabetes in renal allograft recipients. Diabetes Care 30, 609–615 (2007).
Pascual, M., Theruvath, T., Kawai, T., Tolkoff-Rubin, N. & Cosimi, A. B. Strategies to improve long-term outcomes after renal transplantation. N. Engl. J. Med. 346, 580–590 (2002).
Terasaki, P. I. Humoral theory of transplantation. Am. J. Transplant. 3, 665–673 (2003).
Nankivell, B. J. et al. Calcineurin inhibitor nephrotoxicity: longitudinal assessment by protocol histology. Transplantation 78, 557–565 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Li, C., Yang, C. The pathogenesis and treatment of chronic allograft nephropathy. Nat Rev Nephrol 5, 513–519 (2009). https://doi.org/10.1038/nrneph.2009.113
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2009.113
This article is cited by
-
Calcineurin and mTOR inhibitors in kidney transplantation: integrative metamodeling on transplant survival and kidney function
International Urology and Nephrology (2023)
-
Expression of brain-derived neurotrophic factor in kidneys from normal and cyclosporine-treated rats
BMC Nephrology (2018)
-
Early treatment with xenon protects against the cold ischemia associated with chronic allograft nephropathy in rats
Kidney International (2014)
-
A reproducible mouse model of chronic allograft nephropathy with vasculopathy
Kidney International (2012)
-
Targets of new immunosuppressants in renal transplantation
Kidney International Supplements (2011)