Key Points
-
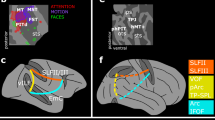
This review proposes that two networks of brain areas are involved in controlling attention. One network is primarily responsible for applying cognitive, top-down selection for stimuli and responses, whereas the other detects behaviourally relevant stimuli and might act as a 'circuit breaker' for the first system.
-
Humans use cognitive information to direct attention to relevant objects (targets) in a visual scene. Information such as the target's colour or location is represented as a 'perceptual set'. Similarly, advance information about the required response to a target is represented as a 'motor set'. These can be considered together as an 'attentional set', which aids the detection of and response to targets.
-
Such top-down control of attentional processes activates dorsal posterior parietal and frontal regions of the brain bilaterally in both monkeys and humans. This dorsal frontoparietal system is responsible for the generation of attentional sets.
-
Attention can also be driven by stimulus properties rather than cognitive processes. This 'bottom-up' control of attention explains why we find ourselves drawn to 'oddball' stimuli that are very different from the background, or to salient stimuli that share some sensory features, such as colour, with the target for which we are searching. The dorsal frontoparietal system seems to maintain a 'salience map' that combines bottom-up with top-down information during visual search.
-
Potentially important sensory stimuli, such as loud alarms or sudden movement, can attract our attention regardless of the ongoing task. This sensory orienting process seems to be mediated by the second attentional network, which is mainly lateralized to the right side of the brain and includes the temporoparietal junction and the ventral frontal cortex. This network seems to interrupt ongoing cognitive activity when a stimulus that might be behaviourally important is detected.
-
These two networks could interact in humans to control attention. It is possible that damage to these networks is responsible for the syndrome of neglect, in which patients that have suffered damage to the right side of the brain tend to ignore stimuli on the left side of space. The authors suggest that neglect results from damage to the ventral network that also 'functionally inactivates' the dorsal network.
Abstract
We review evidence for partially segregated networks of brain areas that carry out different attentional functions. One system, which includes parts of the intraparietal cortex and superior frontal cortex, is involved in preparing and applying goal-directed (top-down) selection for stimuli and responses. This system is also modulated by the detection of stimuli. The other system, which includes the temporoparietal cortex and inferior frontal cortex, and is largely lateralized to the right hemisphere, is not involved in top-down selection. Instead, this system is specialized for the detection of behaviourally relevant stimuli, particularly when they are salient or unexpected. This ventral frontoparietal network works as a 'circuit breaker' for the dorsal system, directing attention to salient events. Both attentional systems interact during normal vision, and both are disrupted in unilateral spatial neglect.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Eriksen, C. W. & Hoffman, J. E. The extent of processing of noise elements during selective encoding from visual displays. Percept. Psychophys. 14, 155–160 (1973).
Posner, M. I., Snyder, C. R. R. & Davidson, B. J. Attention and the detection of signals. J. Exp. Psychol. 109, 160–174 (1980).
Ball, K. & Sekuler, R. Models of stimulus uncertainty in motion perception. Psychol. Rev. 87, 435–469 (1980).
Egeth, H. E., Virzi, R. A. & Garbart, H. Searching for conjunctively defined targets. J. Exp. Psychol. Hum. Percept. Perform. 10, 32–39 (1984).
Dosher, B. A. & Lu, Z.-L. Mechanisms of perceptual attention in precuing of location. Vision Res. 40, 1269–1292 (2000).
Rosenbaum, D. A. Human movement initiation: specification of arm, direction and extent. J. Exp. Psychol. Gen. 109, 444–474 (1980).
Abrams, R. A. & Jonides, J. Programming saccadic eye movements. J. Exp. Psychol. Hum. Percept. Perform. 14, 428–443 (1988).References 1–7 provide evidence that preparatory attention to stimuli and responses facilitates perception and action.
Kanwisher, N. & Wojciulik, E. Visual attention: insights from brain imaging. Nature Rev. Neurosci. 1, 91–100 (2000).
Kastner, S. & Ungerleider, L. G. Mechanisms of visual attention in the human cortex. Annu. Rev. Neurosci. 23, 315–341 (2000).References 8 and 9 review the effects of attention on neural mechanisms in the human visual cortex, and discuss the interaction between the frontoparietal network and the visual cortex.
Corbetta, M., Kincade, J. M., Ollinger, J. M., McAvoy, M. P. & Shulman, G. L. Voluntary orienting is dissociated from target detection in human posterior parietal cortex. Nature Neurosci. 3, 292–297 (2000).Dissociates preparatory goal-directed activity in the IPs for stimulus location from stimulus-driven activity in the TPJ during reorienting to unattended targets.
Kastner, S., Pinsk, M. A., De Weerd, P., Desimone, R. & Ungerleider, L. G. Increased activity in human visual cortex during directed attention in the absence of visual stimulation. Neuron 22, 751–761 (1999).
Hopfinger, J. B., Buonocore, M. H. & Mangun, G. R. The neural mechanisms of top-down attentional control. Nature Neurosci. 3, 284–291 (2000).
Corbetta, M., Kincade, J. M. & Shulman, G. L. Neural systems for visual orienting and their relationship with working memory. J. Cogn. Neurosci. 14 (in the press).
Shulman, G. L. et al. Areas involved in encoding and applying directional expectations to moving objects. J. Neurosci. 19, 9480–9496 (1999).References 11–14 provide neuroimaging evidence that the human dorsal frontoparietal network is involved in preparing and maintaining expectations for stimulus location and motion.
Corbetta, M., Miezin, F. M., Shulman, G. L. & Petersen, S. E. A PET study of visuospatial attention. J. Neurosci. 13, 1202–1226 (1993).
Nobre, A. C. et al. Functional localization of the system for visuospatial attention using positron emission tomography. Brain 120, 515–533 (1997).
Vandenberghe, R. et al. Attention to one or two features in left and right visual field: a positron emission tomography study. J. Neurosci. 17, 3739–3750 (1997).
Culham, J. C. et al. Cortical fMRI activation produced by attentive tracking of moving targets. J. Neurophysiol. 80, 2657–2670 (1998).
Wojciulik, E. & Kanwisher, N. The generality of parietal involvement in visual attention. Neuron 23, 747–764 (1999).
Gitelman, D. R. et al. A large-scale distributed network for covert spatial attention: further anatomical delineation based on stringent behavioural and cognitive controls. Brain 122, 1093–1106 (1999).
Perry, R. J. & Zeki, S. The neurology of saccades and covert shifts in spatial attention: an event-related fMRI study. Brain 123, 2273–2288 (2000).
Corbetta, M. Frontoparietal cortical networks for directing attention and the eye to visual locations: identical, independent, or overlapping neural systems. Proc. Natl Acad. Sci. USA 95, 831–838 (1998).
Van Essen, D. C. et al. Mapping visual cortex in monkeys and humans using surface-based atlases. Vision Res. 41, 1359–1378 (2001).
Paus, T. Location and function of the human frontal eye-field: a selective review. Neuropsychologia 34, 475–483 (1996).
Bushnell, M. C., Goldberg, M. E. & Robinson, D. L. Behavioral enhancement of visual responses in monkey cerebral cortex. I. Modulation in posterior parietal cortex related to selective attention. J. Neurophysiol. 46, 755–772 (1981).
Colby, C. L., Duhamel, J. R. & Goldberg, M. E. Visual, presaccadic, and cognitive activation of single neurons in monkey lateral intraparietal area. J. Neurophysiol. 76, 2841–2852 (1996).Shows how preparatory signals for visual attention and eye movements are combined in LIP neurons.
Nakamura, K. & Colby, C. L. Visual, saccade-related, and cognitive activation of single neurons in monkey exstrastriate area V3A. J. Neurophysiol. 84, 677–692 (2000).
Pashler, H. E. The Psychology of Attention (MIT Press, Cambridge, Massachusetts, 1998).
Shulman, G. L., d'Avossa, G., Tansy, A. P. & Corbetta, M. Two attentional processes in the parietal lobe. Soc. Neurosci. Abstr. 27, 722.20 (2001).
Serences, J. T., Schwarzbach, J. & Yantis, S. Control mechanisms of object-based visual attention in human cortex. Soc. Neurosci. Abstr. 27, 348.9 (2001).
Le, T. H., Pardo, J. V. & Hu, X. 4T-fMRI study of nonspatial shifting of selective attention: cerebellar and parietal contributions. J. Neurophysiol. 79, 1535–1548 (1998).
Assad, J. A. & Maunsell, J. H. R. Neuronal correlates of inferred motion in primate posterior parietal cortex. Nature 373, 518–521 (1995).
Toth, L. J. & Assad, J. A. Dynamic coding of behaviourally relevant stimuli in parietal cortex. Nature 415, 165–168 (2002).
Blake, R., Cepeda, N. J. & Hiris, E. Memory for visual motion. J. Exp. Psychol. Hum. Percept. Perform. 23, 353–369 (1997).
Magnussen, S., Greenlee, M. W., Asplund, R. & Dyrnes, S. Stimulus-specific mechanisms of visual short-term memory. Vision Res. 31, 1213–1219 (1991).
Awh, E. & Jonides, J. Overlapping mechanisms of attention and spatial working memory. Trends Cogn. Sci. 5, 119–126 (2001).Reviews the relationship between spatial working memory and attention.
Courtney, S. M., Ungerleider, L. G., Keil, K. & Haxby, J. V. Transient and sustained activity in a distributed neural system for human working memory. Nature 386, 608–611 (1997).
Gnadt, J. W. & Andersen, R. A. Memory related motor planning activity in posterior parietal cortex of macaque. Exp. Brain Res. 70, 216–220 (1988).
Funahashi, S., Bruce, C. J. & Goldman-Rakic, P. S. Neuronal activity related to saccadic eye movements in the monkey's dorsolateral prefrontal cortex. J. Neurophysiol. 65, 1464–1483 (1991).
Desimone, R. & Duncan, J. Neural mechanisms of selective visual attention. Annu. Rev. Neurosci. 18, 193–222 (1995).
Miller, E. K. & Cohen, J. D. An integrative theory of prefrontal cortex function. Annu. Rev. Neurosci. 24, 167–202 (2001).References 40 and 41 review the role of the prefrontal cortex in attention and executive control.
Savage-Rumbaugh, S., Shanker, S. G. & Talbot, J. T. Apes, Language, and the Human Mind (Oxford Univ. Press, New York, 1998).
Snyder, L. H., Batista, A. P. & Andersen, R. A. Coding of intention in the posterior parietal cortex. Nature 386, 167–170 (1997).Presents evidence of different preparatory response mechanisms for eye and arm in the macaque posterior parietal cortex.
Sakata, H., Taira, M., Kusunoki, M., Murata, A. & Tanaka, Y. The parietal association cortex in depth perception and visual control of hand action. Trends Neurosci. 20, 350–357 (1997).
Bruce, C. J. & Goldberg, M. E. Primate frontal eye fields. I. Single neurons discharging before saccades. J. Neurophysiol. 53, 603–635 (1985).
Wise, S. P., Weinrich, M. & Mauritz, K. H. Motor aspects of cue-related neuronal activity in premotor cortex of the rhesus monkey. Brain Res. 260, 301–305 (1983).
Kawashima, R., Roland, P. E. & O'Sullivan, B. Functional anatomy of reaching and visuomotor learning: a positron emission tomography study. Cereb. Cortex 5, 111–122 (1995).
Petit, L., Clark, V. P., Ingeholm, J. & Haxby, J. V. Dissociation of saccade-related and pursuit-related activation in human frontal eye fields as revealed by fMRI. J. Neurophysiol. 77, 3386–3390 (1997).
Connolly, J. D., Goodale, M. A., Desouza, J. F., Menon, R. S. & Vilis, T. A comparison of frontoparietal fMRI activation during anti-saccades and anti-pointing. J. Neurophysiol. 84, 1645–1655 (2000).
Rowe, J. B., Toni, I., Josephs, O., Frackowiak, R. S. & Passingham, R. E. The prefrontal cortex: response selection or maintenance within working memory? Science 288, 1656–1660 (2000).
Rizzolatti, G., Riggio, L., Dascola, I. & Umiltá, C. Reorienting attention across the horizontal and vertical meridians: evidence in favor of a premotor theory of attention. Neuropsychologia 25, 31–40 (1987).
Corbetta, M. et al. A common network of functional areas for attention and eye movements. Neuron 21, 761–773 (1998).
Nobre, A. C., Gitelman, D. R., Dias, E. C. & Mesulam, M. M. Covert visual spatial orienting and saccades: overlapping neural systems. Neuroimage 11, 210–216 (2000).
Rushworth, M. F., Paus, T. & Sipila, P. K. Attention systems and the organization of the human parietal cortex. J. Neurosci. 21, 5262–5271 (2001).
Allport, A., Styles, E. A. & Hsieh, S. in Attention and Performance XV (eds Umilta, C. & Moscovitch, M.) 421–452 (Erlbaum, Hillsdale, New Jersey, 1994).
Rogers, R. D. & Monsell, S. Costs of a predictable switch between simple cognitive tasks. J. Exp. Psychol. 124, 207–231 (1995).
Meiran, N., Chorev, Z. & Sapir, A. Component processes in task switching. Cognit Psychol 41, 211–253 (2000).References 55–57 describe psychological processes involved in task switching.
Fuster, J. M. in Cerebral Cortex (eds Jones, E. G. & Peters, A.) 151–177 (Plenum, New York, 1985).
Goldman-Rakic, P. S. in Handbook of Physiology, Section 1. Higher Functions of the Brain (eds Plum, F. & Mountcastle, V.) 373–417 (American Physiological Society, Bethesda, Maryland, 1987).
Kimberg, D. Y., Aguirre, G. K. & D'Esposito, M. Modulation of task-related neural activity in task-switching: an fMRI study. Brain Res. Cogn. Brain Res. 10, 189–196 (2000).
Sohn, M.-H., Ursu, S., Anderson, J. R., Stenger, V. A. & Carter, C. S. The role of prefrontal cortex and posterior parietal cortex in task switching. Proc. Natl Acad. Sci. USA 97, 13448–13453 (2000).
James, W. Principles of Psychology Vol. 1 (Henry-Holt & Co., New York, 1890).
Duncan, J. & Humphreys, G. W. Visual search and stimulus similarity. Psychol. Rev. 96, 433–458 (1989).
Posner, M. I. & Cohen, Y. in Attention and Performance X (eds Bouman, H. & Bowhuis, D.) 55–66 (Erlbaum, Hillsdale, New Jersey, 1984).
Muller, H. J. & Rabbitt, M. A. Reflexive and voluntary orienting of visual attention: time course of activation and resistance to interruption. J. Exp. Psychol. 15, 315–330 (1989).
Klein, R. M. Inhibition of return. Trends Cogn. Sci. 4, 138–147 (2000).
Jonides, J. in Attention and Performance XI (eds Posner, M. I. & Marin, O.) 187–205 (Erlbaum, Hillsdale, New Jersey, 1981).
Yantis, S. & Jonides, J. Abrupt visual onsets and selective attention: voluntary versus automatic allocation. J. Exp. Psychol. Hum. Percept. Perform. 16, 121–134 (1990).
Folk, C. L., Remington, R. W. & Johnston, J. C. Involuntary covert orienting is contingent on attentional control settings. J. Exp. Psychol. Hum. Percept. Perform. 18, 1030–1044 (1992).References 68 and 69 discuss cognitive influences on stimulus-driven orienting.
Wolfe, J. M. Guided search 2.0: a revised model of visual search. Psychon. Bull. Rev. 1, 202–238 (1994).
Thompson, K. G., Bichot, N. P. & Schall, J. D. Dissociation of visual discrimination from saccade programming in macaque frontal eye field. J. Neurophysiol. 77, 1046–1050 (1997).
Bichot, N. P. & Schall, J. D. Effects of similarity and history on neural mechanisms of visual selection. Nature Neurosci. 2, 549–554 (1999).
Gottlieb, J. P., Kusunoki, M. & Goldberg, M. E. The representation of visual salience in monkey parietal cortex. Nature 391, 481–484 (1998).References 71–73 show that FEF and LIP neurons are modulated by stimulus salience and task relevance.
Corbetta, M., Shulman, G. L., Miezin, F. M. & Petersen, S. E. Superior parietal cortex activation during spatial attention shifts and visual feature conjunction. Science 270, 802–805 (1995).
Leonards, U., Sunaert, S., Van Hecke, P. & Orban, G. A. Attention mechanisms in visual search — an fMRI study. J. Cogn. Neurosci. 12, 61–75 (2000).
Shulman, G. L., Ollinger, J. M., Linenweber, M., Petersen, S. E. & Corbetta, M. Multiple neural correlates of detection in the human brain. Proc. Natl Acad. Sci. USA 98, 313–318 (2001).
Huettel, S. A., Guzeldere, G. & McCarthy, G. Dissociating the neural mechanisms of visual attention in change detection using functional MRI. J. Cogn. Neurosci. 13, 1006–1018 (2001).
Beck, D. M., Rees, G., Frith, C. D. & Lavie, N. Neural correlates of change detection and change blindness. Nature Neurosci. 4, 645–650 (2001).References 76–78 show that attentional search and target detection modulate the dorsal frontoparietal network and the visual cortex.
Arrington, C. M., Carr, T. H., Mayer, A. R. & Rao, S. M. Neural mechanisms of visual attention: object-based selection of a region in space. J. Cogn. Neurosci. 12, 106–117 (2000).
Kirino, E., Belger, A., Goldman-Rakic, P. & McCarthy, G. Prefrontal activation evoked by infrequent target and novel stimuli in a visual target detection task: an event-related functional magnetic resonance imaging study. J. Neurosci. 20, 6612–6618 (2000).
Marois, R., Leung, H. C. & Gore, J. C. A stimulus-driven approach to object identity and location processing in the human brain. Neuron 25, 717–728 (2000).
Downar, J., Crawley, A. P., Mikulis, D. J. & Davis, K. D. A multimodal cortical network for the detection of changes in the sensory environment. Nature Neurosci. 3, 277–283 (2000).Presents evidence that the ventral frontoparietal network responds to stimulus changes in different sensory modalities.
Braver, T. S., Barch, D. M., Gray, J. R., Molfese, D. L. & Snyder, A. Anterior cingulate cortex and response conflict: effects of frequency, inhibition and errors. Cereb. Cortex 11, 825–836 (2001).
Knight, R. T. & Scabini, D. Anatomic bases of event-related potentials and their relationship to novelty detection in humans. J. Clin. Neurophysiol. 15, 3–13 (1998).
Daffner, K. R. et al. The central role of the prefrontal cortex in directing attention to novel events. Brain 123, 927–939 (2000).
Wilkins, A. J., Shallice, T. & McCarthy, R. Frontal lesions and sustained attention. Neuropsychologia 25, 359–365 (1987).
Downar, J., Crawley, A. P., Mikulis, D. J. & Davis, K. D. The effect of task relevance on the cortical response to changes in visual and auditory stimuli: an event-related fMRI study. Neuroimage 14, 1256–1267 (2001).
Serences, J., Shomstein, S., Leber, A., Yantis, S. & Egeth, H. E. Neural mechanisms of goal-directed and stimulus-driven attentional control. Psychon. Soc. Abstr. 42, 135 (2001).
Clark, V. P., Fannon, S., Lai, S., Benson, R. & Bauer, L. Responses to rare visual target and distractor stimuli using event-related fMRI. J. Neurophysiol. 83, 3133–3139 (2000).
Kiehl, K. A., Laurens, K. R., Duty, T. L., Forster, B. B. & Liddle, P. F. Neural sources involved in auditory target detection and novelty processing: an event-related fMRI study. Psychophysiology 38, 133–142 (2001).
Stuss, D. T. & Benson, D. F. The Frontal Lobes (Raven, New York, 1986).
Yokoyama, K., Jennings, R., Ackles, P., Hood, P. & Boller, F. Lack of heart rate changes during an attention-demanding task after right hemisphere lesions. Neurology 37, 624–630 (1987).
Steinmetz, M. A. & Constantinidis, C. Neurophysiological evidence for a role of posterior parietal cortex in redirecting visual attention. Cereb. Cortex 5, 448–456 (1995).
Robinson, D. L., Bowman, E. M. & Kertzman, C. Covert orienting of attention in macaques. II. Contributions of parietal cortex. J. Neurophysiol. 74, 698–721 (1995).
Constantinidis, C. & Steinmetz, M. A. Neuronal responses in area 7a to multiple stimulus displays. II. Responses are suppressed at the cued location. Cereb. Cortex 11, 592–597 (2001).
Shulman, G. L. et al. Reactivation of networks involved in preparatory states. Cereb. Cortex (in the press).
Posner, M. I. & Petersen, S. E. The attention system of the human brain. Annu. Rev. Neurosci. 13, 25–42 (1990).
Mesulam, M. M. Spatial attention and neglect: parietal, frontal and cingulate contributions to the mental representation and attentional targeting of salient extrapersonal events. Philos Trans R Soc Lond B Biol Sci 354, 1325–1346 (1999).Reviews the pathophysiology and anatomy of unilateral neglect.
Morrison, J. H. & Foote, S. L. Noradrenergic and serotoninergic innervation of cortical, thalamic and tectal visual structures in old and new world monkeys. J. Comp. Neurol. 243, 117–128 (1986).
Oke, A., Keller, R., Mefford, I. & Adams, R. N. Lateralization of norepinephrine in human thalamus. Science 200, 1411–1413 (1978).
Pardo, J. V., Fox, P. T. & Raichle, M. E. Localization of a human system for sustained attention by positron emission tomography. Nature 349, 61–64 (1991).
Aston-Jones, G., Foote, S. L. & Bloom, F. E. in Frontiers of Clinical Neuroscience (ed. Ziegler, M. G.) (Williams & Wilkins, Baltimore, Maryland, 1984).
Dalley, J. W. et al. Distinct changes in cortical acetylcholine and noradrenaline efflux during contingent and noncontingent performance of a visual attentional task. J. Neurosci. 21, 4908–4914 (2001).Provides a possible link between activity in ventral frontoparietal networks and noradrenaline projection systems.
Heilman, K. M., Watson, R. T. & Valenstein, E. in Clinical Neuropsychology (eds Heilman, K. M. & Valenstein, E.) 243–293 (Oxford Univ. Press, New York, 1985).
Robertson, I. H., Mattingley, J. B., Rorden, C. & Driver, J. Phasic alerting of neglect patients overcomes their spatial deficit in visual awareness. Nature 395, 169–172 (1998).
Corbetta, M., Kincade, M. J. & Shulman, G. L. in The Cognitive and Neural Basis of Spatial Neglect (eds Karnath, H. O., Milner, D. & Vallar, G.) (Oxford Univ. Press, Oxford, in the press).
Vallar, G. & Perani, D. in Neurophysiological and Neuropsychological Aspects of Spatial Neglect (ed. Jeannerod, M.) 235–258 (Elsevier, North-Holland, Amsterdam, 1987).
Karnath, H. O., Ferber, S. & Himmelbach, M. Spatial awareness is a function of the temporal not the posterior parietal lobe. Nature 411, 950–953 (2001).
Husain, M. & Kennard, C. Visual neglect associated with frontal lobe infarction. J. Neurol. 243, 652–657 (1996).
Stone, S. P., Patel, P., Greenwood, R. J. & Halligan, P. W. Measuring visual neglect in acute stroke and predicting its recovery: the visual neglect recovery index. J. Neurol. Neurosurg. Psychiatry 55, 431–436 (1992).
Weinberg, J. et al. Visual scanning training effects on reading-related tasks in acquired brain-damage. Arch. Phys. Med. Rehabil. 58, 479–486 (1977).
Posner, M. I., Walker, J. A., Friedrich, F. J. & Rafal, R. D. Effects of parietal injury on covert orienting of attention. J. Neurosci. 4, 1863–1874 (1984).
Smania, N. et al. The spatial distribution of visual attention in hemineglect. Brain 121, 1759–1770 (1998).
Mattingley, J. B., Husain, M., Rorden, C., Kennard, C. & Driver, J. Motor role of human inferior parietal lobe revealed in unilateral neglect patients. Nature 392, 179–182 (1998).
Kinsbourne, M. in Unilateral Neglect: Clinical and Experimental Studies (eds Robertson, I. H. & Marshall, J.) 63–86 (Erlbaum, Hillsdale, New Jersey, 1993).
Witte, E. A., Villareal, M. & Marrocco, R. T. Visual orienting and alerting in rhesus monkeys: comparison with humans. Behav Brain Res 82, 103–112 (1996).
Drury, H. A. et al. Computerized mappings of the cerebral cortex: a multiresolution flattening method and a surface-based coordinate system. J. Cogn. Neurosci. 8, 1–28 (1996).
Thompson, K. G., Bichot, N. P. & Schall, J. D. in Visual Attention and Cortical Circuits (eds Braun, J., Koch, C. & Davis, J. L.) (MIT Press, Cambridge, Massachusetts, 2001).
Acknowledgements
This work was supported by the National Institutes of Health and The J. S. McDonnell Foundation. We thank M. Kincade, A. Tansy, M. Linenweber, S. Astafiev and G. d'Avossa for scientific collaboration; M. Cowan for figure preparation; C. Stanley for scanning; and L. Snyder for contributing data to figure 4a.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- TOP-DOWN PROCESSING
-
The flow of information from 'higher' to 'lower' centres, conveying knowledge derived from previous experience rather than sensory stimulation.
- BOTTOM-UP PROCESSING
-
Information processing that proceeds in a single direction from sensory input, through perceptual analysis, towards motor output, without involving feedback information flowing backwards from 'higher' centres to 'lower' centres.
- FRONTAL EYE FIELD
-
An area in the frontal lobe that receives visual inputs and produces movements of the eye.
- SACCADIC EYE MOVEMENT
-
A rapid eye movement that brings the point of maximal visual acuity — the fovea — to the image of interest.
- NEGLECT
-
A neurological syndrome (often involving damage to the right parietal cortex) in which patients show a marked deficit in the ability to detect or respond to information in the contralesional field.
- LOCUS COERULEUS
-
A nucleus of the brainstem. The main supplier of noradrenaline to the brain.
Rights and permissions
About this article
Cite this article
Corbetta, M., Shulman, G. Control of goal-directed and stimulus-driven attention in the brain. Nat Rev Neurosci 3, 201–215 (2002). https://doi.org/10.1038/nrn755
Issue Date:
DOI: https://doi.org/10.1038/nrn755
This article is cited by
-
High-intensity sweet taste as a predictor of subjective alcohol responses to the ascending limb of an intravenous alcohol prime: an fMRI study
Neuropsychopharmacology (2024)
-
The role of attitudes towards contradiction in psychological resilience: the cortical mechanism of conflicting resolution networks
Scientific Reports (2024)
-
Orthogonal neural encoding of targets and distractors supports multivariate cognitive control
Nature Human Behaviour (2024)
-
Neural evidence for attentional capture by salient distractors
Nature Human Behaviour (2024)
-
Mapping causal links between prefrontal cortical regions and intra-individual behavioral variability
Nature Communications (2024)