Key Points
-
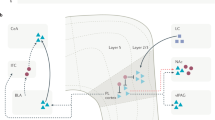
The spinal dorsal horn is innervated by primary afferent fibres and contains a large number of excitatory (glutamatergic) and inhibitory (GABA (γ-aminobutyric acid)-ergic or glycinergic) interneurons, as well as projection neurons that convey sensory information to several brain areas. The interneurons regulate transmission of this information to projection cells and to local reflex pathways. There are also descending modulatory inputs from the brainstem.
-
The dorsal horn can be divided into six parallel laminae, each of which has a distinctive pattern of primary afferent input — for example, nociceptive primary afferents terminate mainly in lamina I and lamina II. Projection neurons are concentrated in lamina I and scattered through laminae III–VI. In all laminae, interneurons make up the great majority of the neuronal population.
-
Despite its importance in pain mechanisms, we still know little about the neuronal organisation and synaptic circuitry of the dorsal horn. This is largely because of the diversity of the neurons, which has made it difficult to recognize functional populations.
-
Recent studies have begun to reveal discrete classes of inhibitory and excitatory interneurons, as well as certain distinctive types of projection neuron. There have also been important advances in our understanding of the structure and function of primary afferents.
-
Based on these studies, we can now begin to map some of the neuronal circuits. For example, many projection neurons in lamina I and some of those in lamina III receive a powerful direct input from nociceptive primary afferents. There is also evidence that projection neurons are selectively innervated by particular types of interneuron.
-
Several changes that could contribute to chronic pain have been identified in the dorsal horn following inflammation or nerve injury. Proposed mechanisms include changes affecting inhibitory interneurons or their synapses, development of long-term potentiation and alterations in the excitability of neurons.
-
Future studies will need to investigate the synaptic organization of the dorsal horn, and the expression of receptors and ion channels on different neuronal populations. These should lead to the identification of new molecular targets for pain treatment, as well as allowing us to identify (and ideally prevent) changes in the dorsal horn that underlie chronic pain.
Abstract
Neurons in the spinal dorsal horn process sensory information, which is then transmitted to several brain regions, including those responsible for pain perception. The dorsal horn provides numerous potential targets for the development of novel analgesics and is thought to undergo changes that contribute to the exaggerated pain felt after nerve injury and inflammation. Despite its obvious importance, we still know little about the neuronal circuits that process sensory information, mainly because of the heterogeneity of the various neuronal components that make up these circuits. Recent studies have begun to shed light on the neuronal organization and circuitry of this complex region.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sivilotti, L. & Woolf, C. J. The contribution of GABAA and glycine receptors to central sensitization: disinhibition and touch-evoked allodynia in the spinal cord. J. Neurophysiol. 72, 169–179 (1994).
Yaksh, T. L. Behavioral and autonomic correlates of the tactile evoked allodynia produced by spinal glycine inhibition: effects of modulatory receptor systems and excitatory amino acid antagonists. Pain 37, 111–123 (1989).
Rexed, B. The cytoarchitectonic organization of the spinal cord in the cat. J. Comp. Neurol. 96, 414–495 (1952).
Woodbury, C. J., Ritter, A. M. & Koerber, H. R. On the problem of lamination in the superficial dorsal horn of mammals: a reappraisal of the substantia gelatinosa in postnatal life. J. Comp. Neurol. 417, 88–102 (2000).
Dhaka, A., Earley, T. J., Watson, J. & Patapoutian, A. Visualizing cold spots: TRPM8-expressing sensory neurons and their projections. J. Neurosci. 28, 566–575 (2008).
Liu, Q. et al. Molecular genetic visualization of a rare subset of unmyelinated sensory neurons that may detect gentle touch. Nature Neurosci. 10, 946–948 (2007).
Seal, R. P. et al. Injury-induced mechanical hypersensitivity requires C-low threshold mechanoreceptors. Nature 462, 651–655 (2009).
Lawson, S. N., Crepps, B. A. & Perl, E. R. Relationship of substance P to afferent characteristics of dorsal root ganglion neurones in guinea-pig. J. Physiol. 505, 177–191 (1997).
Snider, W. D. & McMahon, S. B. Tackling pain at the source: new ideas about nociceptors. Neuron 20, 629–632 (1998).
Taylor, A. M., Peleshok, J. C. & Ribeiro-da-Silva, A. Distribution of P2X(3)-immunoreactive fibers in hairy and glabrous skin of the rat. J. Comp. Neurol. 514, 555–566 (2009).
Bennett, D. L., Dmietrieva, N., Priestley, J. V., Clary, D. & McMahon, S. B. trkA, CGRP and IB4 expression in retrogradely labelled cutaneous and visceral primary sensory neurones in the rat. Neurosci. Lett. 206, 33–36 (1996).
Perry, M. J. & Lawson, S. N. Differences in expression of oligosaccharides, neuropeptides, carbonic anhydrase and neurofilament in rat primary afferent neurons retrogradely labelled via skin, muscle or visceral nerves. Neuroscience 85, 293–310 (1998).
Plenderleith, M. B. & Snow, P. J. The plant lectin Bandeiraea simplicifolia I-B4 identifies a subpopulation of small diameter primary sensory neurones which innervate the skin in the rat. Neurosci. Lett. 159, 17–20 (1993).
Zylka, M. J., Rice, F. L. & Anderson, D. J. Topographically distinct epidermal nociceptive circuits revealed by axonal tracers targeted to Mrgprd. Neuron 45, 17–25 (2005).
Cavanaugh, D. J. et al. Distinct subsets of unmyelinated primary sensory fibers mediate behavioral responses to noxious thermal and mechanical stimuli. Proc. Natl Acad. Sci. USA 106, 9075–9080 (2009).
Lynn, B. Effect of neonatal treatment with capsaicin on the numbers and properties of cutaneous afferent units from the hairy skin of the rat. Brain Res. 322, 255–260 (1984).
Michael, G. J. et al. Nerve growth factor treatment increases brain-derived neurotrophic factor selectively in TrkA-expressing dorsal root ganglion cells and in their central terminations within the spinal cord. J. Neurosci. 17, 8476–8490 (1997).
Rethelyi, M., Light, A. R. & Perl, E. R. Synaptic complexes formed by functionally defined primary afferent units with fine myelinated fibers. J. Comp. Neurol. 207, 381–393 (1982).
Ribeiro-da-Silva, A. & Coimbra, A. Two types of synaptic glomeruli and their distribution in laminae I-III of the rat spinal cord. J. Comp. Neurol. 209, 176–186 (1982).
Ribeiro-da-Silva, A., Tagari, P. & Cuello, A. C. Morphological characterization of substance P-like immunoreactive glomeruli in the superficial dorsal horn of the rat spinal cord and trigeminal subnucleus caudalis: a quantitative study. J. Comp. Neurol. 281, 497–415 (1989).
Zoli, M., Jansson, A., Sykova, E., Agnati, L. F. & Fuxe, K. Volume transmission in the CNS and its relevance for neuropsychopharmacology. Trends Pharmacol. Sci. 20, 142–150 (1999).
Antal, M., Petko, M., Polgar, E., Heizmann, C. W. & Storm-Mathisen, J. Direct evidence of an extensive GABAergic innervation of the spinal dorsal horn by fibres descending from the rostral ventromedial medulla. Neuroscience 73, 509–518 (1996).
Kato, G. et al. Direct GABAergic and glycinergic inhibition of the substantia gelatinosa from the rostral ventromedial medulla revealed by in vivo patch-clamp analysis in rats. J. Neurosci. 26, 1787–1794 (2006).
Polgár, E. et al. Selective loss of spinal GABAergic or glycinergic neurons is not necessary for development of thermal hyperalgesia in the chronic constriction injury model of neuropathic pain. Pain 104, 229–239 (2003).
Todd, A. J. & Sullivan, A. C. Light microscope study of the coexistence of GABA-like and glycine-like immunoreactivities in the spinal cord of the rat. J. Comp. Neurol. 296, 496–505 (1990).
Keller, A. F., Coull, J. A., Chery, N., Poisbeau, P. & De Koninck, Y. Region-specific developmental specialization of GABA-glycine cosynapses in laminas I-II of the rat spinal dorsal horn. J. Neurosci. 21, 7871–7880 (2001).
Yasaka, T. et al. Cell-type-specific excitatory and inhibitory circuits involving primary afferents in the substantia gelatinosa of the rat spinal dorsal horn in vitro. J. Physiol. 581, 603–618 (2007).
Todd, A. J. et al. The expression of vesicular glutamate transporters VGLUT1 and VGLUT2 in neurochemically defined axonal populations in the rat spinal cord with emphasis on the dorsal horn. Eur. J. Neurosci. 17, 13–27 (2003).
Maxwell, D. J., Belle, M. D., Cheunsuang, O., Stewart, A. & Morris, R. Morphology of inhibitory and excitatory interneurons in superficial laminae of the rat dorsal horn. J. Physiol. 584, 521–533 (2007).
Yasaka, T., Tiong, S. Y. X., Hughes, D. I., Riddell, J. S. & Todd, A. J. Populations of inhibitory and excitatory interneurons in lamina II of the adult rat spinal dorsal horn revealed by a combined electrophysiological and anatomical approach. Pain 151, 475–488 (2010). A recent study that compared physiological and morphological properties of lamina II interneurons with neurotransmitter phenotype and showed that A-type potassium current firing patterns were largely restricted to glutamatergic cells.
Graham, B. A., Brichta, A. M. & Callister, R. J. Moving from an averaged to specific view of spinal cord pain processing circuits. J. Neurophysiol. 98, 1057–1063 (2007). A review that highlights the heterogeneity of neurons in the superficial dorsal horn and emphasizes the need to identify functional populations.
Ruscheweyh, R. & Sandkuhler, J. Lamina-specific membrane and discharge properties of rat spinal dorsal horn neurones in vitro. J. Physiol. 541, 231–244 (2002).
Hu, H. J. et al. The kv4.2 potassium channel subunit is required for pain plasticity. Neuron 50, 89–100 (2006). This study showed that Kv4.2, which is a downstream target for phosphorylation by extracellular signal-regulated kinases, mediates the majority of the A-type potassium currents in the dorsal horn and plays a crucial role in pain plasticity.
Huang, H. Y. et al. Expression of A-type K channel α subunits Kv 4.2 and Kv 4.3 in rat spinal lamina II excitatory interneurons and colocalization with pain-modulating molecules. Eur. J. Neurosci. 22, 1149–1157 (2005).
Grudt, T. J. & Perl, E. R. Correlations between neuronal morphology and electrophysiological features in the rodent superficial dorsal horn. J. Physiol. 540, 189–207 (2002). This combined physiological and morphological study developed the most widely used classification scheme for superficial dorsal horn neurons.
Hantman, A. W., van den Pol., A. N. & Perl, E. R. Morphological and physiological features of a set of spinal substantia gelatinosa neurons defined by green fluorescent protein expression. J. Neurosci. 24, 836–842 (2004).
Lu, Y. & Perl, E. R. A specific inhibitory pathway between substantia gelatinosa neurons receiving direct C-fiber input. J. Neurosci. 23, 8752–8758 (2003).
Lu, Y. & Perl, E. R. Modular organization of excitatory circuits between neurons of the spinal superficial dorsal horn (laminae I and II). J. Neurosci. 25, 3900–3907 (2005). One of a series of papers describing elegant studies in which paired recordings were used to investigate synaptic linkages in the superficial dorsal horn. In this case, a synaptic connection between glutamatergic vertical cells and NK1R-expressing lamina I projection neurons was revealed.
Todd, A. J. & McKenzie, J. GABA-immunoreactive neurons in the dorsal horn of the rat spinal cord. Neuroscience 31, 799–806 (1989).
Albuquerque, C., Lee, C. J., Jackson, A. C. & MacDermott, A. B. Subpopulations of GABAergic and non-GABAergic rat dorsal horn neurons express Ca2+-permeable AMPA receptors. Eur. J. Neurosci. 11, 2758–2766 (1999).
Zheng, J., Lu, Y. & Perl, E. R. Inhibitory neurones of the spinal substantia gelatinosa mediate interaction of signals from primary afferents. J. Physiol. 588, 2065–2075 (2010).
Hantman, A. W. & Perl, E. R. Molecular and genetic features of a labeled class of spinal substantia gelatinosa neurons in a transgenic mouse. J. Comp. Neurol. 492, 90–100 (2005).
Han, Z. S., Zhang, E. T. & Craig, A. D. Nociceptive and thermoreceptive lamina I neurons are anatomically distinct. Nature Neurosci. 1, 218–225 (1998).
Lima, D. & Coimbra, A. A Golgi study of the neuronal population of the marginal zone (lamina I) of the rat spinal cord. J. Comp. Neurol. 244, 53–71 (1986).
Prescott, S. A. & De Koninck, Y. Four cell types with distinctive membrane properties and morphologies in lamina I of the spinal dorsal horn of the adult rat. J. Physiol. 539, 817–836 (2002).
Al Ghamdi, K. S., Polgar, E. & Todd, A. J. Soma size distinguishes projection neurons from neurokinin 1 receptor-expressing interneurons in lamina I of the rat lumbar spinal dorsal horn. Neuroscience 164, 1794–1804 (2009).
Antal, M. et al. Different populations of parvalbumin- and calbindin-D28k-immunoreactive neurons contain GABA and accumulate 3H-D-aspartate in the dorsal horn of the rat spinal cord. J. Comp. Neurol. 314, 114–124 (1991).
Todd, A. J. & Spike, R. C. The localization of classical transmitters and neuropeptides within neurons in laminae I–III of the mammalian spinal dorsal horn. Prog. Neurobiol. 41, 609–645 (1993).
Polgár, E., Furuta, T., Kaneko, T. & Todd, A. Characterization of neurons that express preprotachykinin B in the dorsal horn of the rat spinal cord. Neuroscience 139, 687–697 (2006).
Laing, I., Todd, A. J., Heizmann, C. W. & Schmidt, H. H. Subpopulations of GABAergic neurons in laminae I–III of rat spinal dorsal horn defined by coexistence with classical transmitters, peptides, nitric oxide synthase or parvalbumin. Neuroscience 61, 123–132 (1994).
Mori, M., Kose, A., Tsujino, T. & Tanaka, C. Immunocytochemical localization of protein kinase C subspecies in the rat spinal cord: light and electron microscopic study. J. Comp. Neurol. 299, 167–177 (1990).
Polgár, E., Fowler, J. H., McGill, M. M. & Todd, A. J. The types of neuron which contain protein kinase C gamma in rat spinal cord. Brain Res. 833, 71–80 (1999).
Spike, R. C., Todd, A. J. & Johnston, H. M. Coexistence of NADPH diaphorase with GABA, glycine, and acetylcholine in rat spinal cord. J. Comp. Neurol. 335, 320–333 (1993).
Burstein, R., Dado, R. J. & Giesler, G. J., Jr The cells of origin of the spinothalamic tract of the rat: a quantitative reexamination. Brain Res. 511, 329–337 (1990).
Hylden, J. L., Anton, F. & Nahin, R. L. Spinal lamina I projection neurons in the rat: collateral innervation of parabrachial area and thalamus. Neuroscience 28, 27–37 (1989).
Lima, D. & Coimbra, A. The spinothalamic system of the rat: structural types of retrogradely labelled neurons in the marginal zone (lamina I). Neuroscience 27, 215–230 (1988).
Lima, D., Mendes-Ribeiro, J. A. & Coimbra, A. The spino-latero-reticular system of the rat: projections from the superficial dorsal horn and structural characterization of marginal neurons involved. Neuroscience 45, 137–152 (1991).
Spike, R. C., Puskar, Z., Andrew, D. & Todd, A. J. A quantitative and morphological study of projection neurons in lamina I of the rat lumbar spinal cord. Eur. J. Neurosci. 18, 2433–2448 (2003).
Todd, A. J., McGill, M. M. & Shehab, S. A. Neurokinin 1 receptor expression by neurons in laminae, I., III and IV of the rat spinal dorsal horn that project to the brainstem. Eur. J. Neurosci. 12, 689–700 (2000).
Almarestani, L., Waters, S. M., Krause, J. E., Bennett, G. J. & Ribeiro-da-Silva, A. Morphological characterization of spinal cord dorsal horn lamina I neurons projecting to the parabrachial nucleus in the rat. J. Comp. Neurol. 504, 287–297 (2007).
Bernard, J. F., Dallel, R., Raboisson, P., Villanueva, L. & Le Bars, D. Organization of the efferent projections from the spinal cervical enlargement to the parabrachial area and periaqueductal gray: a PHA-L study in the rat. J. Comp. Neurol. 353, 480–505 (1995).
Feil, K. & Herbert, H. Topographic organization of spinal and trigeminal somatosensory pathways to the rat parabrachial and Kolliker-Fuse nuclei. J. Comp. Neurol. 353, 506–528 (1995).
Gauriau, C. & Bernard, J. F. A comparative reappraisal of projections from the superficial laminae of the dorsal horn in the rat: the forebrain. J. Comp. Neurol. 468, 24–56 (2004).
Slugg, R. M. & Light, A. R. Spinal cord and trigeminal projections to the pontine parabrachial region in the rat as demonstrated with Phaseolus vulgaris leucoagglutinin. J. Comp. Neurol. 339, 49–61 (1994).
Al-Khater, K. M. & Todd, A. J. Collateral projections of neurons in laminae, I., III, and IV of rat spinal cord to thalamus, periaqueductal gray matter, and lateral parabrachial area. J. Comp. Neurol. 515, 629–646 (2009).
Al-Khater, K. M., Kerr, R. & Todd, A. J. A quantitative study of spinothalamic neurons in laminae, I, III, and IV in lumbar and cervical segments of the rat spinal cord. J. Comp. Neurol. 511, 1–18 (2008).
Polgár, E., Wright, L. L. & Todd, A. J. A quantitative study of brainstem projections from lamina I neurons in the cervical and lumbar enlargement of the rat. Brain Res. 1308, 58–67 (2010).
Zhang, E. T. & Craig, A. D. Morphology and distribution of spinothalamic lamina I neurons in the monkey. J. Neurosci. 17, 3274–3284 (1997).
Zhang, E. T., Han, Z. S. & Craig, A. D. Morphological classes of spinothalamic lamina I neurons in the cat. J. Comp. Neurol. 367, 537–549 (1996).
Andrew, D. Sensitization of lamina I spinoparabrachial neurons parallels heat hyperalgesia in the chronic constriction injury model of neuropathic pain. J. Physiol. 587, 2005–2017 (2009).
Bester, H., Chapman, V., Besson, J. M. & Bernard, J. F. Physiological properties of the lamina I spinoparabrachial neurons in the rat. J. Neurophysiol. 83, 2239–2259 (2000).
Ruscheweyh, R., Ikeda, H., Heinke, B. & Sandkuhler, J. Distinctive membrane and discharge properties of rat spinal lamina I projection neurones in vitro. J. Physiol. 555, 527–543 (2004).
Zhang, X. & Giesler, G. J. Jr. Response characterstics of spinothalamic tract neurons that project to the posterior thalamus in rats. J. Neurophysiol. 93, 2552–2564 (2005).
Willis, W. D., Trevino, D. L., Coulter, J. D. & Maunz, R. A. Responses of primate spinothalamic tract neurons to natural stimulation of hindlimb. J. Neurophysiol. 37, 358–372 (1974).
Salter, M. W. & Henry, J. L. Responses of functionally identified neurones in the dorsal horn of the cat spinal cord to substance P, neurokinin A and physalaemin. Neuroscience 43, 601–610 (1991).
Mantyh, P. W. et al. Inhibition of hyperalgesia by ablation of lamina I spinal neurons expressing the substance P receptor. Science 278, 275–279 (1997). By selectively ablating NK1R-expressing dorsal horn neurons in vivo , the authors demonstrated that these cells play a pivotal part in the development of hyperalgesia.
Nichols, M. L. et al. Transmission of chronic nociception by spinal neurons expressing the substance P receptor. Science 286, 1558–1561 (1999).
Littlewood, N. K., Todd, A. J., Spike, R. C., Watt, C. & Shehab, S. A. The types of neuron in spinal dorsal horn which possess neurokinin-1 receptors. Neuroscience 66, 597–608 (1995).
Yu, X. H. et al. NK-1 receptor immunoreactivity in distinct morphological types of lamina I neurons of the primate spinal cord. J. Neurosci. 19, 3545–3555 (1999).
Todd, A. J. et al. Projection neurons in lamina I of rat spinal cord with the neurokinin 1 receptor are selectively innervated by substance P-containing afferents and respond to noxious stimulation. J. Neurosci. 22, 4103–4113 (2002). This article demonstrated a strong monosynaptic input from substance P-containing (nociceptive) primary afferents to lamina I projection neurons that express the NK1R.
Polgár, E., Al Ghamdi, K. S. & Todd, A. J. Two populations of neurokinin 1 receptor-expressing projection neurons in lamina I of the rat spinal cord that differ in AMPA receptor subunit composition and density of excitatory synaptic input. Neuroscience 167, 1192–1204 (2010). This recent report identifies two populations of NK1R-expressing lamina I projection neurons that differ in their AMPAR subunit expression and in the density of excitatory synapses that they receive. It also provides evidence that GluA-containing receptors in the superficial dorsal horn are largely restricted to projection neurons.
Polgár, E., Al-Khater, K. M., Shehab, S., Watanabe, M. & Todd, A. J. Large projection neurons in lamina I of the rat spinal cord that lack the neurokinin 1 receptor are densely innervated by VGLUT2-containing axons and possess GluR4-containing AMPA receptors. J. Neurosci. 28, 13150–13160 (2008).
Puskár, Z., Polgár, E. & Todd, A. J. A population of large lamina I projection neurons with selective inhibitory input in rat spinal cord. Neuroscience 102, 167–176 (2001).
Naim, M., Spike, R. C., Watt, C., Shehab, S. A. & Todd, A. J. Cells in laminae III and IV of the rat spinal cord that possess the neurokinin-1 receptor and have dorsally directed dendrites receive a major synaptic input from tachykinin-containing primary afferents. J. Neurosci. 17, 5536–5548 (1997).
Polgár, E., Campbell, A. D., MacIntyre, L. M., Watanabe, M. & Todd, A. J. Phosphorylation of ERK in neurokinin 1 receptor-expressing neurons in laminae III and IV of the rat spinal dorsal horn following noxious stimulation. Mol. Pain 3, 4 (2007).
Naim, M. M., Shehab, S. A. & Todd, A. J. Cells in laminae III and IV of the rat spinal cord which possess the neurokinin-1 receptor receive monosynaptic input from myelinated primary afferents. Eur. J. Neurosci. 10, 3012–3019 (1998).
Sakamoto, H., Spike, R. C. & Todd, A. J. Neurons in laminae III and IV of the rat spinal cord with the neurokinin-1 receptor receive few contacts from unmyelinated primary afferents which do not contain substance P. Neuroscience 94, 903–908 (1999).
Polgár, E., Shehab, S. A., Watt, C. & Todd, A. J. GABAergic neurons that contain neuropeptide Y selectively target cells with the neurokinin 1 receptor in laminae III and IV of the rat spinal cord. J. Neurosci. 19, 2637–2646 (1999).
Uta, D. et al. TRPA1-expressing primary afferents synapse with a morphologically identified subclass of substantia gelatinosa neurons in the adult rat spinal cord. Eur. J. Neurosci. 31, 1960–1973 (2010).
Neumann, S., Braz, J. M., Skinner, K., Llewellyn-Smith, I. J. & Basbaum, A. I. Innocuous, not noxious, input activates PKCγ interneurons of the spinal dorsal horn via myelinated afferent fibers. J. Neurosci. 28, 7936–7944 (2008).
Hughes, D. I., Scott, D. T., Todd, A. J. & Riddell, J. S. Lack of evidence for sprouting of Abeta afferents into the superficial laminas of the spinal cord dorsal horn after nerve section. J. Neurosci. 23, 9491–9499 (2003).
Santos, S. F., Rebelo, S., Derkach, V. A. & Safronov, B. V. Excitatory interneurons dominate sensory processing in the spinal substantia gelatinosa of rat. J. Physiol. 581, 241–254 (2007).
Hughes, D. I. et al. P boutons in lamina IX of the rodent spinal cord express high levels of glutamic acid decarboxylase-65 and originate from cells in deep medial dorsal horn. Proc. Natl Acad. Sci. USA 102, 9038–9043 (2005).
Todd, A. J. GABA and glycine in synaptic glomeruli of the rat spinal dorsal horn. Eur. J. Neurosci. 8, 2492–2498 (1996).
Watson, A. H., Hughes, D. I. & Bazzaz, A. A. Synaptic relationships between hair follicle afferents and neurones expressing GABA and glycine-like immunoreactivity in the spinal cord of the rat. J. Comp. Neurol. 452, 367–380 (2002).
Mantyh, P. W. et al. Receptor endocytosis and dendrite reshaping in spinal neurons after somatosensory stimulation. Science 268, 1629–1632 (1995).
Ding, Y. Q. et al. Two major distinct subpopulations of neurokinin-3 receptor-expressing neurons in the superficial dorsal horn of the rat spinal cord. Eur. J. Neurosci. 16, 551–556 (2002).
Seybold, V. S. et al. Relationship of NK3 receptor-immunoreactivity to subpopulations of neurons in rat spinal cord. J. Comp. Neurol. 381, 439–448 (1997).
Todd, A. J., Spike, R. C. & Polgar, E. A quantitative study of neurons which express neurokinin-1 or somatostatin sst2a receptor in rat spinal dorsal horn. Neuroscience 85, 459–473 (1998).
Kemp, T., Spike, R. C., Watt, C. & Todd, A. J. The mu-opioid receptor (MOR1) is mainly restricted to neurons that do not contain GABA or glycine in the superficial dorsal horn of the rat spinal cord. Neuroscience 75, 1231–1238 (1996).
Brumovsky, P. et al. The neuropeptide tyrosine Y1R is expressed in interneurons and projection neurons in the dorsal horn and area X of the rat spinal cord. Neuroscience 138, 1361–1376 (2006).
Zhang, X., Tong, Y. G., Bao, L. & Hokfelt, T. The neuropeptide Y Y1 receptor is a somatic receptor on dorsal root ganglion neurons and a postsynaptic receptor on somatostatin dorsal horn neurons. Eur. J. Neurosci. 11, 2211–2225 (1999).
Abe, K. et al. Responses to 5-HT in morphologically identified neurons in the rat substantia gelatinosa in vitro. Neuroscience 159, 316–324 (2009).
Lu, Y. & Perl, E. R. Selective action of noradrenaline and serotonin on neurones of the spinal superficial dorsal horn in the rat. J. Physiol. 582, 127–136 (2007).
Gassner, M., Ruscheweyh, R. & Sandkuhler, J. Direct excitation of spinal GABAergic interneurons by noradrenaline. Pain 145, 204–210 (2009).
Sandkuhler, J. Models and mechanisms of hyperalgesia and allodynia. Physiol. Rev. 89, 707–758 (2009). This review provides a detailed and systematic account of the mechanisms that have been proposed to underlie abnormal pain states.
Torsney, C. & MacDermott, A. B. Disinhibition opens the gate to pathological pain signaling in superficial neurokinin 1 receptor-expressing neurons in rat spinal cord. J. Neurosci. 26, 1833–1843 (2006).
Moore, K. A. et al. Partial peripheral nerve injury promotes a selective loss of GABAergic inhibition in the superficial dorsal horn of the spinal cord. J. Neurosci. 22, 6724–6731 (2002).
Coull, J. A. et al. Trans-synaptic shift in anion gradient in spinal lamina I neurons as a mechanism of neuropathic pain. Nature 424, 938–942 (2003).
Eaton, M. J., Plunkett, J. A., Karmally, S., Martinez, M. A. & Montanez, K. Changes in GAD- and GABA- immunoreactivity in the spinal dorsal horn after peripheral nerve injury and promotion of recovery by lumbar transplant of immortalized serotonergic precursors. J. Chem. Neuroanat. 16, 57–72 (1998).
Ibuki, T., Hama, A. T., Wang, X. T., Pappas, G. D. & Sagen, J. Loss of GABA-immunoreactivity in the spinal dorsal horn of rats with peripheral nerve injury and promotion of recovery by adrenal medullary grafts. Neuroscience 76, 845–858 (1997).
Azkue, J. J., Zimmermann, M., Hsieh, T. F. & Herdegen, T. Peripheral nerve insult induces NMDA receptor-mediated, delayed degeneration in spinal neurons. Eur. J. Neurosci. 10, 2204–2206 (1998).
Scholz, J. et al. Blocking caspase activity prevents transsynaptic neuronal apoptosis and the loss of inhibition in lamina II of the dorsal horn after peripheral nerve injury. J. Neurosci. 25, 7317–7323 (2005).
Whiteside, G. T. & Munglani, R. Cell death in the superficial dorsal horn in a model of neuropathic pain. J. Neurosci. Res. 64, 168–173 (2001).
Polgár, E., Hughes, D. I., Arham, A. Z. & Todd, A. J. Loss of neurons from laminas I–III of the spinal dorsal horn is not required for development of tactile allodynia in the spared nerve injury model of neuropathic pain. J. Neurosci. 25, 6658–6666 (2005).
Polgár, E., Gray, S., Riddell, J. S. & Todd, A. J. Lack of evidence for significant neuronal loss in laminae I–III of the spinal dorsal horn of the rat in the chronic constriction injury model. Pain 111, 144–150 (2004).
Polgár, E. & Todd, A. J. Tactile allodynia can occur in the spared nerve injury model in the rat without selective loss of GABA or GABA(A) receptors from synapses in laminae I–II of the ipsilateral spinal dorsal horn. Neuroscience 156, 193–202 (2008).
Hwang, J. H. & Yaksh, T. L. The effect of spinal GABA receptor agonists on tactile allodynia in a surgically-induced neuropathic pain model in the rat. Pain 70, 15–22 (1997).
Malan, T. P., Mata, H. P. & Porreca, F. Spinal GABA(A) and GABA(B) receptor pharmacology in a rat model of neuropathic pain. Anesthesiology 96, 1161–1167 (2002).
Schoffnegger, D., Heinke, B., Sommer, C. & Sandkuhler, J. Physiological properties of spinal lamina II GABAergic neurons in mice following peripheral nerve injury. J. Physiol. 577, 869–878 (2006).
Bailey, A. L. & Ribeiro-da-Silva, A. Transient loss of terminals from non-peptidergic nociceptive fibers in the substantia gelatinosa of spinal cord following chronic constriction injury of the sciatic nerve. Neuroscience 138, 675–690 (2006).
Castro-Lopes, J. M., Coimbra, A., Grant, G. & Arvidsson, J. Ultrastructural changes of the central scalloped (C1) primary afferent endings of synaptic glomeruli in the substantia gelatinosa Rolandi of the rat after peripheral neurotomy. J. Neurocytol 19, 329–337 (1990).
Ikeda, H., Heinke, B., Ruscheweyh, R. & Sandkuhler, J. Synaptic plasticity in spinal lamina I projection neurons that mediate hyperalgesia. Science 299, 1237–1240 (2003).
Ikeda, H. et al. Synaptic amplifier of inflammatory pain in the spinal dorsal horn. Science 312, 1659–1662 (2006). The second of two studies from these authors that demonstrates a form of LTP in lamina I projection neurons — in this case, induced by activation of C fibres at a rate that occurs in physiological conditions.
Bredt, D. S. & Nicoll, R. A. AMPA receptor trafficking at excitatory synapses. Neuron 40, 361–379 (2003).
Esteban, J. A. et al. PKA phosphorylation of AMPA receptor subunits controls synaptic trafficking underlying plasticity. Nature Neurosci. 6, 136–143 (2003).
Nagy, G. G. et al. Widespread expression of the AMPA receptor GluR2 subunit at glutamatergic synapses in the rat spinal cord and phosphorylation of GluR1 in response to noxious stimulation revealed with an antigen-unmasking method. J. Neurosci. 24, 5766–5777 (2004).
Larsson, M. & Broman, J. Translocation of GluR1-containing AMPA receptors to a spinal nociceptive synapse during acute noxious stimulation. J. Neurosci. 28, 7084–7090 (2008).
Randic, M., Jiang, M. C. & Cerne, R. Long-term potentiation and long-term depression of primary afferent neurotransmission in the rat spinal cord. J. Neurosci. 13, 5228–5241 (1993).
Ji, R. R., Baba, H., Brenner, G. J. & Woolf, C. J. Nociceptive-specific activation of ERK in spinal neurons contributes to pain hypersensitivity. Nature Neurosci. 2, 1114–1119 (1999).
Woolf, C. J., Shortland, P. & Coggeshall, R. E. Peripheral nerve injury triggers central sprouting of myelinated afferents. Nature 355, 75–78 (1992).
Tong, Y. G. et al. Increased uptake and transport of cholera toxin B-subunit in dorsal root ganglion neurons after peripheral axotomy: possible implications for sensory sprouting. J. Comp. Neurol. 404, 143–158 (1999).
Shehab, S. A., Spike, R. C. & Todd, A. J. Evidence against cholera toxin B subunit as a reliable tracer for sprouting of primary afferents following peripheral nerve injury. Brain Res. 964, 218–227 (2003).
Woodbury, C. J., Kullmann, F. A., McIlwrath, S. L. & Koerber, H. R. Identity of myelinated cutaneous sensory neurons projecting to nocireceptive laminae following nerve injury in adult mice. J. Comp. Neurol. 508, 500–509 (2008).
Neumann, S., Doubell, T. P., Leslie, T. & Woolf, C. J. Inflammatory pain hypersensitivity mediated by phenotypic switch in myelinated primary sensory neurons. Nature 384, 360–364 (1996).
Noguchi, K., Dubner, R., De Leon, M., Senba, E. & Ruda, M. A. Axotomy induces preprotachykinin gene expression in a subpopulation of dorsal root ganglion neurons. J. Neurosci. Res. 37, 596–603 (1994).
Malcangio, M., Ramer, M. S., Jones, M. G. & McMahon, S. B. Abnormal substance P release from the spinal cord following injury to primary sensory neurons. Eur. J. Neurosci. 12, 397–399 (2000).
Hughes, D. I., Scott, D. T., Riddell, J. S. & Todd, A. J. Upregulation of substance P in low-threshold myelinated afferents is not required for tactile allodynia in the chronic constriction injury and spinal nerve ligation models. J. Neurosci. 27, 2035–2044 (2007).
Baba, H., Doubell, T. P. & Woolf, C. J. Peripheral inflammation facilitates Abeta fiber-mediated synaptic input to the substantia gelatinosa of the adult rat spinal cord. J. Neurosci. 19, 859–867 (1999).
Schoffnegger, D., Ruscheweyh, R. & Sandkuhler, J. Spread of excitation across modality borders in spinal dorsal horn of neuropathic rats. Pain 135, 300–310 (2008).
Okamoto, M. et al. Functional reorganization of sensory pathways in the rat spinal dorsal horn following peripheral nerve injury. J. Physiol. 532, 241–250 (2001).
Kohno, T., Moore, K. A., Baba, H. & Woolf, C. J. Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the rat dorsal horn. J. Physiol. 548, 131–138 (2003).
Schneider, S. P. Functional properties and axon terminations of interneurons in laminae III-V of the mammalian spinal dorsal horn in vitro. J. Neurophysiol. 68, 1746–1759 (1992).
Lima, D., Albino-Teixeira, A. & Tavares, I. The caudal medullary ventrolateral reticular formation in nociceptive-cardiovascular integration. An experimental study in the rat. Exp. Physiol. 87, 267–274 (2002).
Boscan, P., Pickering, A. E. & Paton, J. F. The nucleus of the solitary tract: an integrating station for nociceptive and cardiorespiratory afferents. Exp. Physiol. 87, 259–266 (2002).
Gauriau, C. & Bernard, J. F. Pain pathways and parabrachial circuits in the rat. Exp. Physiol. 87, 251–258 (2002).
Heinricher, M. M., Tavares, I., Leith, J. L. & Lumb, B. M. Descending control of nociception: Specificity, recruitment and plasticity. Brain Res. Rev. 60, 214–225 (2009).
Gauriau, C. & Bernard, J. F. Posterior triangular thalamic neurons convey nociceptive messages to the secondary somatosensory and insular cortices in the rat. J. Neurosci. 24, 752–761 (2004).
Wall, P. D. et al. Autotomy following peripheral nerve lesions: experimental anaesthesia dolorosa. Pain 7, 103–111 (1979).
Decosterd, I. & Woolf, C. J. Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87, 149–158 (2000).
Bennett, G. J. & Xie, Y. K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33, 87–107 (1988).
Kim, S. H. & Chung, J. M. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 50, 355–363 (1992).
Brown, A. G., Fyffe, R. E., Rose, P. K. & Snow, P. J. Spinal cord collaterals from axons of type II slowly adapting units in the cat. J. Physiol. 316, 469–480 (1981).
Light, A. R. & Perl, E. R. Spinal termination of functionally identified primary afferent neurons with slowly conducting myelinated fibers. J. Comp. Neurol. 186, 133–150 (1979).
Shortland, P., Woolf, C. J. & Fitzgerald, M. Morphology and somatotopic organization of the central terminals of hindlimb hair follicle afferents in the rat lumbar spinal cord. J. Comp. Neurol. 289, 416–433 (1989).
Lorenzo, L. E., Ramien, M., St. Louis, M., De Koninck, Y. & Ribeiro-da-Silva, A. Postnatal changes in the Rexed lamination and markers of nociceptive afferents in the superficial dorsal horn of the rat. J. Comp. Neurol. 508, 592–604 (2008).
Todd, A. J. & Koerber, H. R. in Wall and Melzack's Textbook of Pain 5th Edition (Eds McMahon, S. & Koltzenburg, M.) 73–90 (Elsevier, Edinburgh, 2005).
Todd, A. J. in Handbook of Clinical Neurology 3rd Series, Pain. (Eds Cervero, F. and Jensen, T. S.) 61–76 (Elsevier, Edinburgh, 2006).
Todd, A. J. in Current Topics in Pain: 12th World Congress on Pain (Ed. Castro-Lopes, J.) 25–51 (IASP Press, Seattle, 2009).
Acknowledgements
Support from the Wellcome Trust is gratefully acknowledged. I also thank T. Yasaka, D.I. Hughes and E. Polgár for helpful discussion and advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Related links
Glossary
- Nocifensive reflex
-
A protective reflex generated in response to a damaging (or potentially damaging) stimulus.
- Allodynia
-
Pain following a normally non-painful tactile or thermal stimulus.
- Neuropathic pain
-
Pain resulting from pathology of the nervous system. Most commonly this is caused by conditions affecting peripheral nerves.
- Nociceptive information
-
Stimuli through which we perceive damage (or potential damage) caused to the body by excessive heat, cold or physical injury, for example.
- Laminae of Rexed
-
A system of 10 layers, described by Rexed, to divide the grey matter in the spinal cord.
- Synaptic glomerulus
-
A complex structure in which a central axonal bouton (of primary afferent origin) is in synaptic contact with several surrounding profiles, including dendrites and peripheral axons.
- Nucleus raphe magnus
-
The main source of descending serotonergic axons that innervate the dorsal horn.
- Locus coeruleus
-
The major source of noradrenergic axons to the spinal cord.
- Volume transmission
-
A form of neurotransmission in which a neurotransmitter is released directly into the non-synaptic extracellular space to activate nearby receptors.
- Rostral ventromedial medulla
-
A region of the brainstem that includes the nucleus raphe magnus and gives rise to many descending axons that innervate the dorsal horn.
- Delayed firing pattern
-
A response to injected depolarizing current in which a neuron generates action potentials after a delay.
- Gap firing pattern
-
A response to injected depolarizing current in which an initial action potential is followed by a long inter-spike interval and then regular firing.
- Reluctant firing pattern
-
This term is used to describe neurons that are resistant to action potential firing during injection of depolarizing current.
- Hyperalgesia
-
Exaggerated pain in response to a noxious stimulus.
- Transient receptor potential A1
-
(Often abbreviated to TRPA1.) A non-selective cation channel that is activated by cold and by various chemical irritants (including mustard oil), and that is expressed by certain nociceptive primary afferents (a subset of those that express transient receptor potential V1).
- Transient receptor potential vanilloid1
-
(Often abbreviated to TRPV1.) A non-selective cation channel that can be activated by various noxious stimuli (including heat and application of capsaicin) and that is expressed by many nociceptive primary afferents.
- Long-term potentiation
-
(Often abbreviated to LTP.) A form of synaptic plasticity that results in a long-lasting increase in the strength of synaptic transmission.
Rights and permissions
About this article
Cite this article
Todd, A. Neuronal circuitry for pain processing in the dorsal horn. Nat Rev Neurosci 11, 823–836 (2010). https://doi.org/10.1038/nrn2947
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn2947
This article is cited by
-
Alteration of serotonin release response in the central nucleus of the amygdala to noxious and non-noxious mechanical stimulation in a neuropathic pain model rat
The Journal of Physiological Sciences (2024)
-
Neurochemical mechanism of muscular pain: Insight from the study on delayed onset muscle soreness
The Journal of Physiological Sciences (2024)
-
Rab11a in the spinal cord: an essential contributor to complete Freund’s adjuvant-induced inflammatory pain in mice
Molecular Brain (2023)
-
Central medial thalamic nucleus dynamically participates in acute itch sensation and chronic itch-induced anxiety-like behavior in male mice
Nature Communications (2023)
-
Pathogenic mechanisms of human immunodeficiency virus (HIV)-associated pain
Molecular Psychiatry (2023)