Abstract
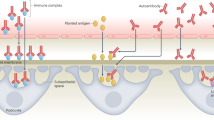
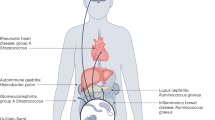
Microorganisms induce strong immune responses, most of which are specific for their encoded antigens. However, microbial infections can also trigger responses against self antigens (autoimmunity), and it has been proposed that this phenomenon could underlie several chronic human diseases, such as type 1 diabetes and multiple sclerosis. Nevertheless, despite intensive efforts, it has proven difficult to identify any single microorganism as the cause of a human autoimmune disease, indicating that the 'one organism–one disease' paradigm that is central to Koch's postulates might not invariably apply to microbially induced autoimmune disease. Here, we review the mechanisms by which microorganisms might induce autoimmunity, and we outline a hypothesis that we call the fertile-field hypothesis to explain how a single autoimmune disease could be induced and exacerbated by many different microbial infections.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Srinivasappa, J. et al. Molecular mimicry: frequency of reactivity of monoclonal antiviral antibodies with normal tissues. J. Virol. 57, 397–401 (1986).
Gebe, J. A. et al. Low-avidity recognition by CD4+ T cells directed to self-antigens. Eur. J. Immunol. 33, 1409–1417 (2003).
Jacobson, D. L., Gange, S. J., Rose, N. R. & Graham, N. M. Epidemiology and estimated population burden of selected autoimmune diseases in the United States. Clin. Immunol. Immunopathol. 84, 223–243 (1997).
Nishioka, K., Nakajima, T., Hasunuma, T. & Sato, K. Rheumatic manifestation of human leukemia virus infection. Rheum. Dis. Clin. North Am. 19, 489–503 (1993).
Zhao, Z. S. et al. Molecular mimicry by herpes simplex virus-type 1: autoimmune disease after viral infection. Science 279, 1344–1347 (1998).
Deshpande, S. P. et al. Herpes simplex virus-induced keratitis: evaluation of the role of molecular mimicry in lesion pathogenesis. J. Virol. 75, 3077–3088 (2001).
Eddy, S. et al. Myasthenia gravis: another autoimmune disease associated with hepatitis C virus infection. Dig. Dis. Sci. 44, 186–189 (1999).
Romero, P. M. et al. Rheumatologic and autoimmune manifestations in patients with chronic hepatitis C virus infection. Rev. Esp. Enferm. Dig. 89, 591–598 (1997).
McMurray, R. W. & Elbourne, K. Hepatitis C virus infection and autoimmunity. Semin. Arthritis Rheum. 26, 689–701 (1997).
James, J. A. & Harley, J. B. B-cell epitope spreading in autoimmunity. Immunol. Rev. 164, 185–200 (1998).
Atkinson, M. A. et al. Cellular immunity to a determinant common to glutamate decarboxylase and coxsackie virus in insulin-dependent diabetes. J. Clin. Invest. 94, 2125–2129 (1994).
Yoon, J. W., Ihm, S. H. & Kim, K. W. Viruses as a triggering factor of type 1 diabetes and genetic markers related to the susceptibility to the virus-associated diabetes. Diabetes Res. Clin. Pract. 7, S47–S58 (1989).
Monteyne, P., Bureau, J. F. & Brahic, M. Viruses and multiple sclerosis. Curr. Opin. Neurobiol. 11, 287–291 (1998).
Panitch, H. S. Influence of infection on exacerbations of multiple sclerosis. Ann. Neurol. 36, S25–S28 (1994).
Cunningham, M. W. Autoimmunity and molecular mimicry in the pathogenesis of post-streptococcal heart disease. Front. Biosci. 8, S533–S543 (2003).
Kirvan, C. A., Swedo, S. E., Heuser, J. S. & Cunningham, M. W. Mimicry and autoantibody-mediated neuronal cell signaling in Sydenham chorea. Nature Med. 9, 914–920 (2003).
Lopez-Larrea, C., Gonzalez, S. & Martinez-Borra, J. The role of HLA-B27 polymorphism and molecular mimicry in spondyloarthropathy. Mol. Med. Today 4, 540–549 (1998).
Taurog, J. D. & Hammer, R. E. Experimental spondyloarthropathy in HLA-B27 transgenic rats. Clin. Rheumatol. 15, 22–27 (1996).
Benoist, C. & Mathis, D. Autoimmunity provoked by infection: how good is the case for T cell epitope mimicry? Nature Immunol. 2, 797–801 (2001).
Leon, J. S. & Engman, D. M. The significance of autoimmunity in the pathogenesis of Chagas heart disease. Front. Biosci. 8, E315–E322 (2003).
Kierszenbaum, F. Views on the autoimmunity hypothesis for Chagas disease pathogenesis. FEMS Immunol. Med. Microbiol. 37, 1–11 (2003).
Fujinami, R. S. & Oldstone, M. B. A. Amino acid homology between the encephalitogenic site of myelin basic protein and virus: mechanism for autoimmunity. Science 230, 1043–1045 (1985).
Barnett, L. A., Whitton, J. L., Wada, Y. & Fujinami, R. S. Enhancement of autoimmune disease using recombinant vaccinia virus encoding myelin proteolipid protein. J. Neuroimmunol. 44, 15–25 (1993).
Tsunoda, I. et al. Enhancement of experimental allergic encephalomyelitis (EAE) by DNA immunization with myelin proteolipid protein (PLP) plasmid DNA. J. Neuropathol. Exp. Neurol. 57, 758–767 (1998).
Ohashi, P. S. et al. Ablation of tolerance and induction of diabetes by virus infection in viral antigen transgenic mice. Cell 65, 305–318 (1991).
Oldstone, M. B. A. et al. Virus infection triggers insulin-dependent diabetes mellitus in a transgenic model: role of anti-self (virus) immune response. Cell 65, 319–331 (1991).
von Herrath M. G., Dockter, J. & Oldstone, M. B. A. How virus induces a rapid or slow onset insulin-dependent diabetes mellitus in a transgenic model. Immunity 1, 231–242 (1994).
von Herrath M. G. et al. Coexpression of B7-1 and viral ('self') transgenes in pancreatic β cells can break peripheral ignorance and lead to spontaneous autoimmune diabetes. Immunity 3, 727–738 (1995).
Kagi, D. et al. Development of insulitis without diabetes in transgenic mice lacking perforin-dependent cytotoxicity. J. Exp. Med. 183, 2143–2152 (1996).
von Herrath M. G. & Oldstone, M. B. A. Interferon-γ is essential for destruction of β cells and development of insulin-dependent diabetes mellitus. J. Exp. Med. 185, 531–539 (1997).
Wucherpfennig, K. W. Structural basis of molecular mimicry. J. Autoimmun. 16, 293–302 (2001).
Whitton, J. L. et al. Molecular analyses of a five-amino-acid cytotoxic T-lymphocyte (CTL) epitope: an immunodominant region which induces non-reciprocal CTL cross-reactivity. J. Virol. 63, 4303–4310 (1989).
Sevilla, N. et al. Virus-induced diabetes in a transgenic model: role of cross-reacting viruses and quantitation of effector T cells needed to cause disease. J. Virol. 74, 3284–3292 (2000).
Quinn, A. et al. Induction of autoimmune valvular heart disease by recombinant streptococcal M protein. Infect. Immun. 69, 4072–4078 (2001).
Honeyman, M. C. et al. Association between rotavirus infection and pancreatic islet autoimmunity in children at risk of developing type 1 diabetes. Diabetes 49, 1319–1324 (2000).
Theil, D. J. et al. Viruses can silently prime for and trigger central nervous system autoimmune disease. J. Neurovirol. 7, 1–8 (2001).
Olson, J. K. et al. A virus-induced molecular mimicry model of multiple sclerosis. J. Clin. Invest. 108, 311–318 (2001).
Shevach, E. M., Chang, J. T. & Segal, B. M. The critical role of IL-12 and the IL-12R β 2 subunit in the generation of pathogenic autoreactive Th1 cells. Springer Semin. Immunopathol. 21, 249–262 (1999).
Tough, D. F. & Sprent, J. Viruses and T cell turnover: evidence for bystander proliferation. Immunol. Rev. 150, 129–142 (1996).
Tough, D. F., Borrow, P. & Sprent, J. Induction of bystander T cell proliferation by viruses and type I interferon in vivo. Science 272, 1947–1950 (1996).
Murali-Krishna, K. et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8, 177–187 (1998).
Butz, E. A. & Bevan, M. J. Massive expansion of antigen-specific CD8+ T cells during an acute virus infection. Immunity 8, 167–175 (1998).
Hislop, A. D. et al. Epitope-specific evolution of human CD8+ T cell responses from primary to persistent phases of Epstein–Barr virus infection. J. Exp. Med. 195, 893–905 (2002).
Nguyen, K. B. & Biron, C. A. Synergism for cytokine-mediated disease during concurrent endotoxin and viral challenges: roles for NK and T cell IFN-γ production. J. Immunol. 162, 5238–5246 (1999).
Gangappa, S. et al. Virus-induced immunoinflammatory lesions in the absence of viral antigen recognition. J. Immunol. 161, 4289–4300 (1998).
Haring, J. S., Pewe, L. L. & Perlman, S. BystanderCD8 T cell-mediated demyelination after viral infection of the central nervous system. J. Immunol. 169, 1550–1555 (2002).
Horwitz, M. S. et al. Diabetes induced by Coxsackie virus: initiation by bystander damage and not molecular mimicry. Nature Med. 4, 781–785 (1998).
Kim, S. K., Brehm, M. A., Welsh, R. M. & Selin, L. K. Dynamics of memory T cell proliferation under conditions of heterologous immunity and bystander stimulation. J. Immunol. 169, 90–98 (2002).
Ostler, T., Pircher, H. & Ehl, S. 'Bystander' recruitment of systemic memory T cells delays the immune response to respiratory virus infection. Eur. J. Immunol. 33, 1839–1848 (2003).
Murali-Krishna, K. et al. In vivo dynamics of anti-viral CD8 T cell responses to different epitopes. An evaluation of bystander activation in primary and secondary responses to viral infection. Adv. Exp. Med. Biol. 452, 123–142 (1998).
Welsh, R. M. & Selin, L. K. No one is naive: the significance of heterologous T-cell immunity. Nature Rev. Immunol. 2, 417–426 (2002).
Chen, H. D. et al. Memory CD8+ T cells in heterologous antiviral immunity and immunopathology in the lung. Nature Immunol. 2, 1067–1076 (2001).
Lin, M. Y., Selin, L. K. & Welsh, R. M. Evolution of the CD8 T-cell repertoire during infections. Microbes. Infect. 2, 1025–1039 (2000).
Welsh, R. M., McNally, J. M., Brehm, M. A. & Selin, L. K. Consequences of cross-reactive and bystander CTL responses during viral infections. Virology 270, 4–8 (2000).
Selin, L. K. et al. Attrition of T cell memory: selective loss of LCMV epitope-specific memory CD8 T cells following infections with heterologous viruses. Immunity 11, 733–742 (1999).
Ahmed, Z. et al. Myelin/axonal pathology in interleukin-12 induced serial relapses of experimental allergic encephalomyelitis in the Lewis rat. Am. J. Pathol. 158, 2127–2138 (2001).
von Herrath M. G., Allison, J., Miller, J. F. & Oldstone, M. B. Focal expression of interleukin-2 does not break unresponsiveness to 'self' (viral) antigen expressed in β cells but enhances development of autoimmune disease (diabetes) after initiation of an anti-self immune response. J. Clin. Invest. 95, 477–485 (1995).
Holz, A., Brett, K. & Oldstone, M. B. Constitutive β cell expression of IL-12 does not perturb self-tolerance but intensifies established autoimmune diabetes. J. Clin. Invest. 108, 1749–1758 (2001).
Lee, M. S. et al. Sensitization to self (virus) antigen by in situ expression of murine interferon-gamma. J. Clin. Invest. 95, 486–492 (1995).
Seewaldt, S. et al. Virus-induced autoimmune diabetes: most beta-cells die through inflammatory cytokines and not perforin from autoreactive (anti-viral) cytotoxic T-lymphocytes. Diabetes 49, 1801–1809 (2000).
Gale, C. R. & Martyn, C. N. Migrant studies in multiple sclerosis. Prog. Neurobiol. 47, 425–448 (1995).
Schwarze, J. & Gelfand, E. W. The role of viruses in development or exacerbation of atopic asthma. Clin. Chest Med. 21, 279–287 (2000).
Schwarze, J. & Gelfand, E. W. Respiratory viral infections as promoters of allergic sensitization and asthma in animal models. Eur. Respir. J. 19, 341–349 (2002).
Cohen, I. R. Antigenic mimicry, clonal selection and autoimmunity. J. Autoimmun. 16, 337–340 (2001).
Christen, U. et al. A dual role for TNFα in type 1 diabetes: islet-specific expression abrogates the ongoing autoimmune process when induced late but not early during pathogenesis. J. Immunol. 166, 7023–7032 (2001).
Jacob, C. O. et al. Prevention of diabetes in nonobese diabetic mice by tumor necrosis factor (TNF): similarities between TNFα and interleukin 1. Proc. Natl Acad. Sci. USA 87, 968–972 (1990).
Tracy, S. et al. Toward testing the hypothesis that group B coxsackieviruses (CVB) trigger insulin-dependent diabetes: inoculating nonobese diabetic mice with CVB markedly lowers diabetes incidence. J. Virol. 76, 12097–12111 (2002).
Barnett, L. A., Whitton, J. L., Wang, L. Y. & Fujinami, R. S. Virus encoding an encephalitogenic peptide protects mice from experimental allergic encephalomyelitis. J. Neuroimmunol. 64, 163–173 (1996).
Coon, B., An, L. L., Whitton, J. L. & von Herrath M. G., DNA immunization to prevent autoimmune diabetes. J. Clin. Invest. 104, 189–194 (1999).
von Herrath M. G. & Whitton, J. L. DNA vaccination to treat autoimmune diabetes. Ann. Med. 32, 285–292 (2000).
Robinson, W. H., Steinman, L. & Utz, P. J. Protein and peptide array analysis of autoimmune disease. Biotechniques 6, S66–S69 (2002).
Acknowledgements
M. G. V. H. would like to thank D. Frye for her excellent help with this (and many other) manuscripts and his wife B. Ford for her patience with his work. Thanks should also go to C. J. and Chiara, who not only brighten my life but also have certainly primed me with many childcare viruses to hopefully abrogate, but not trigger, autoimmunity. R. S. F. is supported by the National Institutes for Health. The work of J. L. W. on T-cell responses is supported the National Institutes for Health.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Infectious Disease Information
LocusLink
OMIM
FURTHER INFORMATION
Glossary
- ANTIGEN-PRESENTING CELL
-
A cell that is capable of providing co-stimulation through CD80/86, CD40 and other mechanisms, in addition to presenting antigen in the context of MHC molecules. Examples include dendritic cells, B cells and macrophages.
- AUTOAGGRESSIVE
-
A term that is used to describe autoreactive T cells that are harmful to the host.
- AUTOIMMUNE DISEASE
-
A clinically apparent disease that is attributable to the activities of autoaggressive lymphocytes.
- AUTOIMMUNITY
-
An antigen-specific response against host tissues that is equivalent to autoreactivity. Note that many autoimmune and autoreactive lymphocytes seem to be harmless, or even protective to the host.
- AVIDITY
-
The strength with which a T cell binds to a target cell by multiple receptor–ligand interactions.
- BYSTANDER ACTIVATION
-
The activation of autoreactive lymphocytes that do not recognize microbial antigens. This can be mediated through cytokines and/or APCs.
- COGNATE ANTIGEN
-
An antigen for which an antibody, or a T-cell receptor, is specific.
- ISLETS OF LANGERHANS
-
Clusters of cells that are found in the pancreas. There are five types of islet cells, including β cells, which make insulin; α cells, which make glucagon; and δ cells, which make somatostatin.
- MOLECULAR MIMICRY
-
This can occur when lymphocytes that are induced by a foreign (usually microbial) antigen can recognize (crossreact with) host proteins. Usually, the avidity of the lymphocytes for the self antigen is lower than their avidity for the microbial antigen.
- M PROTEIN
-
A streptococcal virulence factor that protects the bacteria from phagocytosis.
- PERFORIN
-
A pore-forming protein that is present in the granules of cytotoxic lymphocytes.
- RANTES
-
A CC-chemokine that binds to, and activates, signal transduction by several chemokine receptors.
Rights and permissions
About this article
Cite this article
von Herrath, M., Fujinami, R. & Whitton, J. Microorganisms and autoimmunity: making the barren field fertile?. Nat Rev Microbiol 1, 151–157 (2003). https://doi.org/10.1038/nrmicro754
Issue Date:
DOI: https://doi.org/10.1038/nrmicro754
This article is cited by
-
Incorporating the Molecular Mimicry of Environmental Antigens into the Causality of Autoimmune Hepatitis
Digestive Diseases and Sciences (2023)
-
Detection of Mycobacterium avium Subspecies paratuberculosis in the Blood of Patients with Rheumatoid Arthritis by Using Serological and Molecular Techniques
Current Microbiology (2023)
-
Probiotic normalization of systemic inflammation in siblings of type 1 diabetes patients: an open-label pilot study
Scientific Reports (2022)
-
Lyme arthritis: linking infection, inflammation and autoimmunity
Nature Reviews Rheumatology (2021)
-
Neural Stem Cell-Based Regenerative Approaches for the Treatment of Multiple Sclerosis
Molecular Neurobiology (2018)