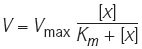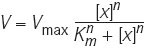Key Points
-
The performance of bacterial transcriptional regulatory networks is often affected by post-transcriptional, post-translational and pleiotropic effects.
-
Despite their importance, non-transcriptional effects are often obscure or difficult to characterize without quantitative analytical techniques.
-
Feedback loops can arise via non-transcriptional interactions, and these loops have important effects on signal processing.
-
Stress-response networks, cell cycle regulators and small RNA-mediated control of gene expression are examples of bacterial signalling networks that depend strongly on non-transcriptional interactions.
-
Mathematical network analysis techniques used in combination with quantitative experimental approaches can reveal how non-transcriptional processes contribute to complex dynamic phenotypes.
-
Synthetic biological networks are a powerful tool for studying the role of non-transcriptional effects in natural networks. Synthetic networks are well defined and easily manipulated. Recent advances in synthetic-network design underscore the importance of non-transcriptional effects.
-
Synthetic-network construction complemented by quantitative network analysis will speed discovery and deepen our understanding of the fundamental organizing principles of biology.
Abstract
Information about the extra- or intracellular environment is often captured as biochemical signals that propagate through regulatory networks. These signals eventually drive phenotypic changes, typically by altering gene expression programmes in the cell. Reconstruction of transcriptional regulatory networks has given a compelling picture of bacterial physiology, but transcriptional network maps alone often fail to describe phenotypes. Cellular response dynamics are ultimately determined by interactions between transcriptional and non-transcriptional networks, with dramatic implications for physiology and evolution. Here, we provide an overview of non-transcriptional interactions that can affect the performance of natural and synthetic bacterial regulatory networks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Cosentino Lagomarsino, M., Jona, P., Bassetti, B. & Isambert, H. Hierarchy and feedback in the evolution of the Escherichia coli transcription network. Proc. Natl Acad. Sci. USA 104, 5516–5520 (2007).
Balázsi, G., Heath, A. P., Shi, L. & Gennaro, M. L. The temporal response of the Mycobacterium tuberculosis gene regulatory network during growth arrest. Mol. Syst. Biol. 4, 225 (2008).
Wall, M. E., Hlavacek, W. S. & Savageau, M. A. Design of gene circuits: lessons from bacteria. Nature Rev. Genet. 5, 34–42 (2004).
Alon, U. An Introduction to Systems Biology: Design Principles of Biological Circuits (Chapman and Hall/CRC, 2006).
Mangan, S. & Alon, U. Structure and function of the feed-forward loop network motif. Proc. Natl Acad. Sci. USA 100, 11980–11985 (2003).
Voigt, C. A., Wolf, D. M. & Arkin, A. P. The Bacillus subtilis SIN operon: an evolvable network motif. Genetics 169, 1187–1202 (2005).
Prill, R. J., Iglesias, P. A. & Levchenko, A. Dynamic properties of network motifs contribute to biological network organization. PLoS Biol. 3, e343 (2005).
Wall, M. E., Dunlop, M. J. & Hlavacek, W. S. Multiple functions of a feed-forward-loop gene circuit. J. Mol. Biol. 349, 501–514 (2005).
Stock, A. M., Robinson, V. L. & Goudreau, P. N. Two-component signal transduction. Annu. Rev. Biochem. 69, 183–215 (2000).
Martínez-Antonio, A., Janga, S. C. & Thieffry, D. Functional organisation of Escherichia coli transcriptional regulatory network. J. Mol. Biol. 381, 238–247 (2008).
Ray, J. C. J. & Igoshin, O. A. Adaptable functionality of transcriptional feedback in bacterial two-component systems. PLoS Comput. Biol. 6, e1000676 (2010).
Shin, D., Lee, E.-J., Huang, H. & Groisman, E. A positive feedback loop promotes transcription surge that jump-starts Salmonella virulence circuit. Science 314, 1607–1609 (2006). This study demonstrates the physiological importance of network dynamics for a virulent microorganism.
Savageau, M. A. Design principles for elementary gene circuits: elements, methods, and examples. Chaos 11, 142–159 (2001).
Chen, W. W., Niepel, M. & Sorger, P. K. Classic and contemporary approaches to modeling biochemical reactions. Genes Dev. 24, 1861–1875 (2010).
Hlavacek, W. S. & Savageau, M. A. Subunit structure of regulator proteins influences the design of gene circuitry: analysis of perfectly coupled and completely uncoupled circuits. J. Mol. Biol. 248, 739–755 (1995).
Perutz, M. F. Mechanisms of cooperativity and allosteric regulation in proteins. Q. Rev. Biophys. 22, 139–237 (1989).
Goldbeter, A. & Koshland, D. E. An amplified sensitivity arising from covalent modification in biological systems. Proc. Natl Acad. Sci. USA 78, 6840–6844 (1981).
Kim, S. Y. & Ferrell, J. E. Substrate competition as a source of ultrasensitivity in the inactivation of Wee1. Cell 128, 1133–1145 (2007).
Palani, S. & Sarkar, C. A. Positive receptor feedback during lineage commitment can generate ultrasensitivity to ligand and confer robustness to a bistable switch. Biophys. J. 95, 1575–1589 (2008).
Wang, L. et al. Bistable switches control memory and plasticity in cellular differentiation. Proc. Natl Acad. Sci. USA 106, 6638–6643 (2009).
Cluzel, P., Surette, M. & Leibler, S. An ultrasensitive bacterial motor revealed by monitoring signaling proteins in single cells. Science 287, 1652–1655 (2000).
LaPorte, D. C. & Koshland, D. E. Phosphorylation of isocitrate dehydrogenase as a demonstration of enhanced sensitivity in covalent regulation. Nature 305, 286–290 (1983).
Buchler, N. E., Gerland, U. & Hwa, T. Nonlinear protein degradation and the function of genetic circuits. Proc. Natl Acad. Sci. USA 102, 9559–9564 (2005).
Buchler, N. E. & Louis, M. Molecular titration and ultrasensitivity in regulatory networks. J. Mol. Biol. 384, 1106–1119 (2008).
Tiwari, A., Balázsi, G., Gennaro, M. L. & Igoshin, O. A. The interplay of multiple feedback loops with post-translational kinetics results in bistability of mycobacterial stress response. Phys. Biol. 7, 036005 (2010).
Levine, E., Zhang, Z., Kuhlman, T. & Hwa, T. Quantitative characteristics of gene regulation by small RNA. PLoS Biol. 5, e229 (2007).
Legewie, S., Dienst, D., Wilde, A., Herzel, H. & Axmann, I. M. Small RNAs establish delays and temporal thresholds in gene expression. Biophys. J. 95, 3232–3238 (2008).
Xiong, W. & Ferrell, J. E. A positive-feedback-based bistable 'memory module' that governs a cell fate decision. Nature 426, 460–465 (2003).
Ghosh, S. et al. Phenotypic heterogeneity in mycobacterial stringent response. BMC Syst. Biol. 5, 18 (2011).
Berg, O. G., Paulsson, J. & Ehrenberg, M. Fluctuations and quality of control in biological cells: zero-order ultrasensitivity reinvestigated. Biophys. J. 79, 1228–1236 (2000).
Igoshin, O. A., Price, C. W. & Savageau, M. A. Signalling network with a bistable hysteretic switch controls developmental activation of the F transcription factor in Bacillus subtilis. Mol. Microbiol. 61, 165–184 (2006).
Igoshin, O. A., Brody, M. S., Price, C. W. & Savageau, M. A. Distinctive topologies of partner-switching signaling networks correlate with their physiological roles. J. Mol. Biol. 369, 1333–1352 (2007).
Craciun, G., Tang, Y. & Feinberg, M. Understanding bistability in complex enzyme-driven reaction networks. Proc. Natl Acad. Sci. USA 103, 8697–8702 (2006).
Thomas, R. & Kaufman, M. Multistationarity, the basis of cell differentiation and memory. I. Structural conditions of multistationarity and other nontrivial behavior. Chaos 11, 170–179 (2001).
Klumpp, S., Zhang, Z. & Hwa, T. Growth rate-dependent global effects on gene expression in bacteria. Cell 139, 1366–1375 (2009). A re-evaluation of classic microbiology data combined with new theory reveals that the growth rate has widespread consequences for bacterial phenotypes.
Tan, C., Marguet, P. & You, L. Emergent bistability by a growth-modulating positive feedback circuit. Nature Chem. Biol. 5, 842–848 (2009). An elegant experimental approach that demonstrates growth-modulated bistability.
Gottesman, S. Proteolysis in bacterial regulatory circuits. Annu. Rev. Cell Dev. Biol. 19, 565–587 (2003).
Rotem, E. et al. Regulation of phenotypic variability by a threshold-based mechanism underlies bacterial persistence. Proc. Natl Acad. Sci. USA 107, 12541–12546 (2010).
Balaban, N. Q., Merrin, J., Chait, R., Kowalik, L. & Leibler, S. Bacterial persistence as a phenotypic switch. Science 305, 1622–1625 (2004).
Reed, M. C., Lieb, A. & Nijhout, F. F. The biological significance of substrate inhibition: a mechanism with diverse functions. Bioessays 32, 422–429 (2010).
Chaudhury, S. & Igoshin, O. A. Dynamic disorder-driven substrate inhibition and bistability in a simple enzymatic reaction. J. Phys. Chem. B 113, 13421–13428 (2009).
Igoshin, O. A., Alves, R. & Savageau, M. A. Hysteretic and graded responses in bacterial two-component signal transduction. Mol. Microbiol. 68, 1196–1215 (2008).
Ishii, N. et al. Multiple high-throughput analyses monitor the response of E. coli to perturbations. Science 316, 593–597 (2007).
Lynch, M. The frailty of adaptive hypotheses for the origins of organismal complexity. Proc. Natl Acad. Sci. USA 104, 8597–8604 (2007).
Rice, S. Evolutionary Theory (Sinauer Associates, Inc., 2004).
Miyashiro, T. & Goulian, M. High stimulus unmasks positive feedback in an autoregulated bacterial signaling circuit. Proc. Natl Acad. Sci. USA 105, 17457–17462 (2008).
Angeli, D., Ferrell, J. E. & Sontag, E. D. Detection of multistability, bifurcations, and hysteresis in a large class of biological positive-feedback systems. Proc. Natl Acad. Sci. USA 101, 1822–1827 (2004).
Eguchi, Y., Ishii, E., Hata, K. & Utsumi, R. Regulation of acid resistance by connectors of two-component signal transduction systems in Escherichia coli. J. Bacteriol. 193, 1222–1228 (2011).
Burton, N. A., Johnson, M. D., Antczak, P., Robinson, A. & Lund, P. A. Novel aspects of the acid response network of E. coli K-12 are revealed by a study of transcriptional dynamics. J. Mol. Biol. 401, 726–742 (2010). These authors take a detail-oriented experimental approach to evaluating the dynamics of gene-regulatory networks without losing sight of the 'big picture'.
Savageau, M. A. Comparison of classical and autogenous systems of regulation in inducible operons. Nature 252, 546–549 (1974).
Traxler, M. F. et al. Discretely calibrated regulatory loops controlled by ppGpp partition gene induction across the 'feast to famine' gradient in Escherichia coli. Mol. Microbiol. 79, 830–845 (2010).
Hoffer, S. M., Westerhoff, H. V., Hellingwerf, K. J., Postma, P. W. & Tommassen, J. Autoamplification of a two-component regulatory system results in “learning” behavior. J. Bacteriol. 183, 4914–4917 (2001).
Chastanet, A. et al. Broadly heterogeneous activation of the master regulator for sporulation in Bacillus subtilis. Proc. Natl Acad. Sci. USA 107, 8486–8491 (2010).
Bischofs, I. B., Hug, J. A., Liu, A. W., Wolf, D. M. & Arkin, A. P. Complexity in bacterial cell–cell communication: quorum signal integration and subpopulation signaling in the Bacillus subtilis phosphorelay. Proc. Natl Acad. Sci. USA 106, 6459–6464 (2009).
Schultz, D., Wolynes, P. G., Jacob, E. & Onuchic, J. N. Deciding fate in adverse times: sporulation and competence in Bacillus subtilis. Proc. Natl Acad. Sci. USA 106, 21027–21034 (2009).
Saini, S., Ellermeier, J. R., Slauch, J. M. & Rao, C. V. The role of coupled positive feedback in the expression of the SPI1 type three secretion system in Salmonella. PLoS Pathog. 6, e1001025 (2010).
Thattai, M. & van Oudenaarden, A. Intrinsic noise in gene regulatory networks. Proc. Natl Acad. Sci. USA 98, 8614–8619 (2001).
Nguyen, L. K. & Kulasiri, D. On the functional diversity of dynamical behaviour in genetic and metabolic feedback systems. BMC Syst. Biol. 3, 51 (2009).
Stekel, D. J. & Jenkins, D. J. Strong negative self regulation of Prokaryotic transcription factors increases the intrinsic noise of protein expression. BMC Syst. Biol. 2, 6 (2008).
Goyal, S. & Wingreen, N. S. Growth-induced instability in metabolic networks. Phys. Rev. Lett. 98, 138105 (2007).
Bhartiya, S., Chaudhary, N., Venkatesh, K. V. & Doyle, F. J. Multiple feedback loop design in the tryptophan regulatory network of Escherichia coli suggests a paradigm for robust regulation of processes in series. J. R. Soc. Interface 3, 383–391 (2006).
Curtis, P. D. & Brun, Y. V. Getting in the loop: regulation of development in Caulobacter crescentus. Microbiol. Mol. Biol. Rev. 74, 13–41 (2010).
Jenal, U. The role of proteolysis in the Caulobacter crescentus cell cycle and development. Res. Microbiol. 160, 687–695 (2009).
Thanbichler, M. & Shapiro, L. Chromosome organization and segregation in bacteria. J. Struct. Biol. 156, 292–303 (2006).
Biondi, E. G. et al. Regulation of the bacterial cell cycle by an integrated genetic circuit. Nature 444, 899–904 (2006).
Paul, R. et al. Allosteric regulation of histidine kinases by their cognate response regulator determines cell fate. Cell 133, 452–461 (2008).
Chen, Y. E. et al. Spatial gradient of protein phosphorylation underlies replicative asymmetry in a bacterium. Proc. Natl Acad. Sci. USA 108, 1052–1057 (2011). An experimentally driven study of the C. crescentus cell cycle, making use of mathematical modelling and simulation to circumvent experimental constraints and arrive at a compelling conceptual model.
Hallez, R., Bellefontaine, A.-F., Letesson, J.-J. & De Bolle, X. Morphological and functional asymmetry in α-proteobacteria. Trends Microbiol. 12, 361–365 (2004).
Ackermann, M., Stearns, S. C. & Jenal, U. Senescence in a bacterium with asymmetric division. Science 300, 1920 (2003).
Stewart, E. J., Madden, R., Paul, G. & Taddei, F. Aging and death in an organism that reproduces by morphologically symmetric division. PLoS Biol. 3, e45 (2005).
Sprinzak, D. & Elowitz, M. B. Reconstruction of genetic circuits. Nature 438, 443–448 (2005).
Gardner, T. S., Cantor, C. R. & Collins, J. J. Construction of a genetic toggle switch in Escherichia coli. Nature 403, 339–342 (2000).
Weiss, R. Cellular Computation and Communications Using Engineered Genetic Regulatory Networks. Thesis, Massachussets Institute of Technology (2001).
Atkinson, M. R., Savageau, M. A., Myers, J. T. & Ninfa, A. J. Development of genetic circuitry exhibiting toggle switch or oscillatory behavior in Escherichia coli. Cell 113, 597–607 (2003).
Elowitz, M. B. & Leibler, S. A synthetic oscillatory network of transcriptional regulators. Nature 403, 335–338 (2000).
Yokobayashi, Y., Weiss, R. & Arnold, F. H. Directed evolution of a genetic circuit. Proc. Natl Acad. Sci. USA 99, 16587–16591 (2002).
Golding, I., Paulsson, J., Zawilski, S. M. & Cox, E. C. Real-time kinetics of gene activity in individual bacteria. Cell 123, 1025–1036 (2005).
Pedraza, J. M. & Paulsson, J. Effects of molecular memory and bursting on fluctuations in gene expression. Science 319, 339–343 (2008).
Rosenfeld, N., Young, J. W., Alon, U., Swain, P. S. & Elowitz, M. B. Gene regulation at the single-cell level. Science 307, 1962–1965 (2005).
Pedraza, J. M. & van Oudenaarden, A. Noise propagation in gene networks. Science 307, 1965–1969 (2005).
Elowitz, M. B., Levine, A. J., Siggia, E. D. & Swain, P. S. Stochastic gene expression in a single cell. Science 297, 1183–1186 (2002).
Anderson, J. C., Clarke, E. J., Arkin, A. P. & Voigt, C. A. Environmentally controlled invasion of cancer cells by engineered bacteria. J. Mol. Biol. 355, 619–627 (2006).
Basu, S., Mehreja, R., Thiberge, S., Chen, M.-T. & Weiss, R. Spatiotemporal control of gene expression with pulse-generating networks. Proc. Natl Acad. Sci. USA 101, 6355–6360 (2004).
Salis, H. M., Mirsky, E. A. & Voigt, C. A. Automated design of synthetic ribosome binding sites to control protein expression. Nature Biotech. 27, 946–950 (2009).
Na, D., Lee, S. & Lee, D. Mathematical modeling of translation initiation for the estimation of its efficiency to computationally design mRNA sequences with desired expression levels in prokaryotes. BMC Syst. Biol. 4, 71 (2010).
Miyazaki, K. Creating random mutagenesis libraries by megaprimer PCR of whole plasmid (MEGAWHOP). Methods Mol. Biol. 231, 23–28 (2003).
Stricker, J. et al. A fast, robust and tunable synthetic gene oscillator. Nature 456, 516–519 (2008). This article describes the engineering of a robust, tunable synthetic oscillator. The results illustrate the importance of post-transcriptional delays for the dynamic functionality of gene-regulatory networks.
Danino, T., Mondragon-Palomino, O., Tsimring, L. & Hasty, J. A synchronized quorum of genetic clocks. Nature 463, 326–330 (2010).
Lim, W. A. Designing customized cell signalling circuits. Nature Rev. Mol. Cell Biol. 11, 393–403 (2010).
Martin, V. J. J., Pitera, D. J., Withers, S. T., Newman, J. D. & Keasling, J. D. Engineering a mevalonate pathway in Escherichia coli for production of terpenoids. Nature Biotech. 21, 796–802 (2003).
Dueber, J. E. et al. Synthetic protein scaffolds provide modular control over metabolic flux. Nature Biotech. 27, 753–759 (2009). A clever non-transcriptional-modification scheme is shown to greatly boost efficiency in a synthetic metabolic pathway, laying fundamental groundwork for mechanistic synthetic biology.
Keasling, J. D. Synthetic biology for synthetic chemistry. ACS Chem. Biol. 3, 64–76 (2008).
Marles-Wright, J. & Lewis, R. J. The stressosome: molecular architecture of a signalling hub. Biochem. Soc. Trans. 38, 928–933 (2010).
Marles-Wright, J. et al. Molecular architecture of the “stressosome,” a signal integration and transduction hub. Science 322, 92–96 (2008).
Løvdok, L. et al. Role of translational coupling in robustness of bacterial chemotaxis pathway. PLoS Biol. 7, e1000171 (2009).
Saiz, L. & Vilar, J. M. J. Ab initio thermodynamic modeling of distal multisite transcription regulation. Nucleic Acids Res. 36, 726–731 (2008).
Long, T. et al. Quantifying the integration of quorum-sensing signals with single-cell resolution. PLoS Biol. 7, e1000068 (2009).
Feinberg, M. The existence and uniqueness of steady states for a class of chemical reaction networks. Arch. Rational Mech. Anal. 132, 311–370 (1995).
Shinar, G. & Feinberg, M. Structural sources of robustness in biochemical reaction networks. Science 327, 1389–1391 (2010).
Batchelor, E. & Goulian, M. Robustness and the cycle of phosphorylation and dephosphorylation in a two-component regulatory system. Proc. Natl Acad. Sci. USA 100, 691–696 (2003).
Shinar, G., Milo, R., Martínez, M. R. & Alon, U. Input output robustness in simple bacterial signaling systems. Proc. Natl Acad. Sci. USA 104, 19931–19935 (2007).
Acknowledgements
The authors thank G. Balázsi, M. Laub, M. Bennett and M. Gennaro for useful comments on manuscript drafts and P. Lund for sharing his data for figure 4. This work is supported by grant R01-GM096189-01 from the US National Institutes of Health (O.A.I.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (box)
(PDF 212 kb)
Related links
Glossary
- Networks
-
Sets of biochemical reactions or interactions that are employed for information processing in the cell. The term network can refer to either interactions on the whole-cell level or smaller circuits (subsystems) within the larger network.
- Signal
-
In the context of this Review, the information that flows through a biological network. In a wider context, biological signals can take a variety of forms.
- Nodes
-
Molecular entities, such as transcription factors or allosterically regulated enzymes, that take in a signal and then output a signal in response. When a node is described as upstream or downstream, this refers to its order in the information flow.
- Pleiotropic
-
Of an interaction: in which one component or effect simultaneously affects many targets. In this Review, we refer to effects originating from coupling with global physiological processes in the cell.
- Ultrasensitivity
-
A type of signal–response curve characterized by a high slope in the responsive range.
- Michaelis–Menten kinetics
-
A model of enzyme kinetics that is often used to mathematically represent first-order saturation processes, in which the flux (V) is determined by the equation:
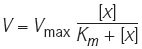
(in which [x] is the concentration of substrate or regulator x, Vmax is the maximum flux rate and Km is the Michaelis–Menten constant).
- Hill kinetics
-
A generalization of Michaelis–Menten kinetics that allows a mathematical representation of higher-order, or cooperative, processes in which the flux
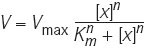
has nth-order effective cooperativity ([x] is the concentration of substrate or regulator x, Vmax is the maximum flux rate, Km is the Michaelis–Menten constant and n is the Hill coefficient).
- Effective cooperativity
-
A measure of sensitivity: how much one molecular species affects the production of another.
- Bistable switch
-
A system in which there are two stable steady states under the same conditions, as reflected in the signal–response curve. Which state the system adopts in practice depends on the initial conditions and noise.
- Bet hedging
-
An evolved phenotype that employs heterogeneity to ensure that distinct subsets of a cellular population are adapted to different outcomes of an unpredictable future environment.
- Noise
-
Variability in signals and responses from cell to cell that arises either intrinsically, from the nature of the physicochemical processes, or from extrinsic variability such as randomness in ribosome inheritance.
- Jacobian matrix
-
A matrix for which the entries quantitate the sensitivity of each variable (often corresponding to chemical species) to each other variable.
- Implicit feedback loop
-
A feedback loop for which its existence is not obvious, but which emerges from non-transcriptional interactions.
- Toxin–antitoxin system
-
A small gene network that typically includes one gene encoding a toxin and another encoding a neutralizing antitoxin.
- Coupled feedback loops
-
Multiple feedback loops that interact in some way, such as being nested or resulting from a single regulatory event that modulates multiple transcriptionally coupled genes.
- Dynamic performance
-
The characteristics of a response to a signal over time.
- Biphasic
-
Of a response: composed of two distinct, characteristic types of dynamics that are separated in time, such as an initial transient phase and a long-term persistent phase.
- Robustness
-
Insensitivity of a dynamic performance to small parameter perturbations that would arise from intrinsic or extrinic noise, slight environmental variations, and so on (for the purposes of this Review; the term has many subtly different meanings in systems biology).
- Oscillator
-
A network architecture that results in periodic oscillations of an output.
- Signal matching
-
Adjusting the amount of signal produced by an upstream node so that it is within the range to which a downstream node is responsive (unsaturated).
Rights and permissions
About this article
Cite this article
Ray, J., Tabor, J. & Igoshin, O. Non-transcriptional regulatory processes shape transcriptional network dynamics. Nat Rev Microbiol 9, 817–828 (2011). https://doi.org/10.1038/nrmicro2667
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2667
This article is cited by
-
Connectivity mapping of angiotensin-PPAR interactions involved in the amelioration of non-alcoholic steatohepatitis by Telmisartan
Scientific Reports (2019)
-
Characterizing bacterial gene circuit dynamics with optically programmed gene expression signals
Nature Methods (2014)
-
Turning Oscillations Into Opportunities: Lessons from a Bacterial Decision Gate
Scientific Reports (2013)
-
Sequestration‐based bistability enables tuning of the switching boundaries and design of a latch
Molecular Systems Biology (2012)
-
Positive allosteric feedback regulation of the stringent response enzyme RelA by its product
EMBO reports (2012)