Abstract
Visceral leishmaniasis (VL) is a systemic protozoan disease that is transmitted by phlebotomine sandflies. Poor and neglected populations in East Africa and the Indian sub-continent are particularly affected. Early and accurate diagnosis and treatment remain key components of VL control. In addition to improved diagnostic tests, accurate and simple tests are needed to identify treatment failures. Miltefosine, paromomycin and liposomal amphotericin B are gradually replacing pentavalent antimonials and conventional amphotericin B as the preferred treatments in some regions, but in other areas these drugs are still being evaluated in both mono- and combination therapies. New diagnostic tools and new treatment strategies will only have an impact if they are made widely available to patients.
Similar content being viewed by others
Main
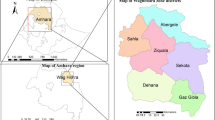
Leishmaniasis, a vector-borne disease that is caused by obligate intra-macrophage protozoa, is endemic in large areas of the tropics, subtropics and the Mediterranean basin (Fig. 1). This disease is characterized by both diversity and complexity1: it is caused by more than 20 leishmanial species and is transmitted to humans by ∼30 different species of phlebotomine sandflies2.
The majority of VL cases occur in just six countries — Bangladesh, Brazil, Ethiopia, India, Nepal and Sudan. Modified, with permission, from Ref. 167 © 2004 Macmillan Publishers Ltd.
Leishmaniasis consists of four main clinical syndromes: cutaneous leishmaniasis; muco-cutaneous leishmaniasis (also known as espundia); visceral leishmaniasis (VL; also known as kala-azar); and post-kala-azar dermal leishmaniasis (PKDL) (Fig. 2). In cutaneous leishmaniasis, the patient generally presents with one or several ulcer(s) or nodule(s) in the skin. Different species of Leishmania can infect the macrophages in the dermis, with variable clinical presentations and prognoses3,4. The ulcers heal spontaneously — although slowly — in immunocompetent individuals, but cause disfiguring scars. In muco-cutaneous leishmaniasis, patients suffer from progressively destructive ulcerations of the mucosa, extending from the nose and mouth to the pharynx and larynx. These lesions are not self-healing and are usually seen months or years after a first episode of cutaneous leishmaniasis, when the macrophages of the naso-oropharyngeal mucosa become colonized. Leishmania braziliensis is responsible for most cases of muco-cutaneous leishmaniasis.
VL is a systemic disease that is fatal if left untreated and is caused by the Leishmania donovani complex — L. donovani sensu stricto in East Africa and the Indian subcontinent and Leishmania infantum in Europe, North Africa and Latin America5,6 (Fig. 1). There are two types of VL, which differ in their transmission characteristics: zoonotic VL is transmitted from animal to vector to human and anthroponotic VL is transmitted from human to vector to human. In the former, humans are occasional hosts and animals, mainly dogs, are the reservoir of the parasite7. Zoonotic VL is found in areas of L. infantum transmission whereas anthroponotic VL is found in areas of L. donovani transmission. PKDL is characterized by a macular, maculo-papular or nodular rash and is a complication of VL that is frequently observed after treatment in Sudan and more rarely in other East African countries and in the Indian subcontinent8. It can also occur in immunosuppressed individuals in L. infantum-endemic areas. The interval between treated VL and PKDL is 0–6 months in Sudan and 6 months to 3 years in India. PKDL cases are highly infectious because the nodular lesions contain many parasites9, and such cases are the putative reservoir for anthroponotic VL between epidemic cycles.
In this Review, the epidemiology, clinical presentation and pathogenesis of VL are reviewed, along with the current control strategies and research challenges.
The epidemiology of VL
As already mentioned, VL is caused by two leishmanial species, L. donovani or L. infantum, depending on the geographical area. L. infantum infects mostly children and immunosuppressed individuals, whereas L. donovani infects all age groups.
There are an estimated 500,000 new cases of VL and more than 50,000 deaths from the disease each year10, a death toll that is surpassed among the parasitic diseases only by malaria11. Both figures are approximations as VL is frequently not recognized or not reported12,13. The majority (>90%) of cases occur in just six countries: Bangladesh, India, Nepal, Sudan, Ethiopia and Brazil. Migration, lack of control measures and HIV–VL co-infection (Box 1) are the three main factors driving the increased incidence of VL14,15. Severe VL epidemics have been reported in the past: in Southern Sudan, in a context of civil war and famine, VL killed an estimated 100,000 people out of a population of 280,000 between 1984 and 1994 (Ref. 16).
VL affects poor communities, generally in remote rural areas. The disease is mostly endemic in countries that are among the least developed in the world (such as Nepal) or in the poorest regions of so-called 'middle-income' countries (such as Bihar State in India). Patients and families affected by VL become poorer because of the high direct costs (for example, the costs of VL diagnosis and treatment) and indirect costs (for example, loss of household income) of the disease17,18,19. As India, Nepal and Bangladesh harbour an estimated 67% of the global VL disease burden20, the commitment of the governments of these countries to launch a regional VL elimination programme is welcome (Box 2). The target of this programme is to eliminate VL as a public health problem in these countries by 2015, by using a local approach to reduce the annual incidence of VL to less than 1 case per 10,000 individuals.
The clinical presentation of VL
Following an incubation period that generally lasts between 2 and 6 months, VL patients present symptoms and signs of persistent systemic infection (including fever, fatigue, weakness, loss of appetite and weight loss) and parasitic invasion of the blood and reticulo-endothelial system (that is, the general phagocytic system), such as enlarged lymph nodes, spleen and liver. Fever is usually associated with rigor and chills and can be intermittent. Fatigue and weakness are worsened by anaemia, which is caused by the persistent inflammatory state, hypersplenism (the peripheral destruction of erythrocytes in the enlarged spleen) and sometimes by bleeding.
The clinical presentation of VL is similar in the various endemic areas but there are some differences. For example, enlarged lymph nodes are rarely found in Indian VL patients but are frequent in Sudanese VL patients21,22. Hyper-pigmentation, which probably led to the name kala-azar (black fever in hindi), has only been described in VL patients from the Indian subcontinent, but today this symptom is uncommon and was perhaps a feature of prolonged illness in the era when effective treatment was not available. As the disease advances, splenomegaly can increase, causing abdominal distension and pain, which is sometimes increased by concomitant hepatomegaly. Symptoms and signs of bacterial co-infections such as pneumonia, diarrhoea or tuberculosis can confuse the clinical picture at the time of initial diagnosis. VL symptoms often persist for several weeks to months before patients either seek medical care or die from bacterial co-infections, massive bleeding or severe anaemia.
The pathogenesis of VL
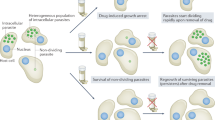
The lifecycle of L. donovani has two distinct forms: a promastigote flagellar form found in the gut of the arthropod vector and an amastigote form, which develops intracellularly in the mammalian host (Fig. 3). Only female phlebotomine sandflies transmit the disease, by inoculation of the promastigote form into the skin. The parasites are internalized by dendritic cells and macrophages in the dermis and transform into amastigotes by losing their flagella. They multiply and survive in phagolysosomes through a complex parasite–host interaction23,24. The parasites disseminate through the lymphatic and vascular systems and infect other monocytes and macrophages in the reticulo-endothelial system, resulting in infiltration of the bone marrow, hepato-splenomegaly and sometimes enlarged lymph nodes (lymphadenopathy).
The promastigote form of L. donovani is transmitted into the skin by female phlebotomine sandflies. Once transmitted, the parasites are internalized by dendritic cells and macrophages in the dermis where they lose their flagella, transforming into the amastigote form. The amastigotes multiply, destroy the host cell and infect other phagocytic cells. The amastigotes disseminate through the lymphatic and vascular systems, eventually infiltrating the bone marrow, liver and spleen. Reproduced, with permission, from Ref. 168 © (2001) American Society for Microbiology.
Importantly, infection does not always equate with clinical illness. The ratio of incident asymptomatic infections to incident clinical cases varies, being 1:2.6 to 11:1 in Sudan25,26, 4:1 in Kenya27, 5.6:1 in Ethiopia28, 13:1 in Iran29, 8:1 to 18:1 in Brazil30,31 and 50:1 in Spain32. It is of utmost importance for vaccine development and disease control to understand the factors that might predispose some individuals to develop the disease or control the infection. The host specific cell-mediated immune (CMI) response has an important role in controlling the infection. In VL patients, the inability to control L. donovani infection is associated with a profound T-cell unresponsiveness to L. donovani antigens33 and the production of interleukin 10 (IL10)34. IL10-producing CD25−Foxp3−T cells were recently implicated in the pathogenesis of human VL in India35. The crucial role of the CMI response is illustrated by the increased risk of developing clinical illness in cases of malnutrition or concomitant immunosuppressive diseases, such as HIV infection36,37,38. Other risk factors for developing clinical illness have been identified and include young age26,29,30, decreased production of interferon γ (IFN-γ)39 and polymorphisms in the promoter of the tumour necrosis factor α (TNF-α) gene40. The gene encoding solute carrier family 11 A1 (SLC11A1; formerly NRAMP1), which regulates macrophage activation, and polymorphisms in the gene encoding IL4 were also associated with underlying susceptibility to VL in Sudan, highlighting the key role of innate immunity in driving the adaptive host immune response41.
So far, VL research has considered only a single parameter or a limited number of parameters, and has mainly been conducted in L. infantum-endemic areas. There is thus an urgent need to conduct more ambitious studies on the environmental, clinical, parasitic, genetic and immunological predictors of VL in L. donovani-endemic areas (East Africa and the Indian subcontinent).
VL control strategies
The current control strategies for VL rely on reservoir and vector control, the use of insecticide-impregnated materials and active case detection and treatment14,42; anti-leishmanial vaccines are still being developed.
Reservoir control. Dogs are the main reservoir of L. infantum in zoonotic VL. Despite evidence from experimental studies showing a decreased incidence of VL in both dogs and children following serological screening of dogs and killing of sero-positive animals43,44, the efficiency and acceptability of this control strategy is increasingly being debated7,45,46. Treating infected dogs is not an effective control strategy as relapses are frequent and dogs can regain infectivity weeks after treatment, despite being clinically cured47. Moreover, the widespread veterinary use of VL drugs might lead to resistance in parasites. A new control approach is the use of deltamethrine-treated collars, which reduced the risk of infection in dogs (by 54%) and children (by 43%) in a study conducted in Iran48. Vaccination of dogs would nevertheless be the best strategy if an efficacious vaccine can be developed.
Vector control. Sandflies are susceptible to the same insecticides as Anopheles mosquitoes, the malaria vector. Residual insecticide spraying of houses and animal shelters was shown to be efficacious in India49, where the vector (Phlebotomus argentipes) is restricted to areas in and around the home. Following the large scale anti-malarial insecticide (dichloro-diphenyl-trichloroethane (DDT)) spraying campaigns that were implemented in the 1950s, VL almost completely disappeared from the Indian subcontinent. Unfortunately, the disease quickly re-emerged when these spraying campaigns were discontinued. Resistance of P. argentipes to DDT remains limited, but has been reported in Bihar50. In Sudan and other endemic countries in East Africa, transmission occurs mainly, but not exclusively51, outside villages, during shepherding for example. Indoor residual spraying for disease control is therefore unlikely to be as efficient in this region.
Insecticide-impregnated materials. The use of insecticide-treated bednets (ITNs) could concomitantly prevent VL and other vector-borne diseases, such as malaria and Japanese encephalitis. There is limited evidence that bednets provide protection against VL. Case-control studies conducted in Bangladesh and Nepal showed that sleeping under a non-impregnated bednet during the warm months was a protective factor against VL (odds ratio = 0.20, p = 0.001; odds ratio = 0.69, p = 0.01, respectively)52,53. Despite low usage, the mass distribution of ITNs in Sudan was accompanied by a 27% reduction in the incidence of VL in an observational study54. A large prospective randomized controlled trial testing the efficacy of long-lasting ITNs to prevent L. donovani infection and VL is underway in Nepal and India (see the Kalanet Project website). Depending on the sleeping traditions of the population and the biting habits of the local vector, other insecticide-impregnated materials such as curtains and blankets should be evaluated for use in VL prevention, as some have been shown to provide efficient protection against cutaneous leishmaniasis55,56.
Early diagnosis and treatment. Early diagnosis and treatment are essential for both individual patients and for the community. The treatment outcome is worse in VL patients in bad general health. Adult patients in Sudan with severe anaemia, malnutrition and long duration of illness were shown to be at an increased risk of death57. Untreated VL patients act as a reservoir for parasites and therefore contribute to disease transmission in anthroponotic VL areas. Early case-finding and treatment is therefore considered an essential component of VL control10,14,58. The concomitant detection and treatment of PKDL patients is also likely to be beneficial and the feasibility, impact and cost of a PKDL management strategy should therefore be properly evaluated.
VL diagnosis
As the clinical presentation of VL lacks specificity, confirmatory tests are required to decide which patients should be treated. Such tests should be highly sensitive (>95%) as VL is a fatal condition, but also highly specific because the current drugs used to treat VL are toxic. Ideally, a test should be able to make the distinction between acute disease and asymptomatic infection, because none of the drugs currently available is safe enough to treat aymptomatic infections. Moreover, such tests should be simple and affordable.
Non-leishmanial tests. A reduction in the number of red and white blood cells and platelets (pancytopenia) was found to be highly specific (98%) for VL in suspected clinical patients in Nepal but the sensitivity was low (16%)59. Marked polyclonal hypergammaglobulinemia (the production of high titres of non-specific antibody), a common finding in VL, can be detected by a formol gel test (FGT; also called the aldehyde test), which is still used in East Africa and Asia because of its simplicity and low cost. However, as the sensitivity of this test is poor (as low as 34%59), some experts have recommended its use be discontinued60.
Parasite detection. The visualization of the amastigote form of the parasite by microscopic examination of aspirates from lymph nodes, bone marrow or spleen is the classical confirmatory test for VL. Although the specificity is high, the sensitivity of microscopy varies, being higher for spleen (93–99%) than for bone marrow (53–86%) or lymph node (53–65%) aspirates61,62,63,64,65. However, spleen aspiration can be complicated by life-threatening haemorrhages in ∼0.1% of individuals and therefore requires considerable technical expertise66, as well as facilities for nursing surveillance, blood transfusion and surgery. Moreover, the accuracy of microscopic examination is influenced by the ability of the laboratory technician and the quality of the reagents used. The detection of parasites in the blood or organs by culture or by using molecular techniques such as PCR is more sensitive than microscopic examination but these techniques remain restricted to referral hospitals and research centres, despite efforts to simplify them67.
Antibody-detection tests. Several tests that detect specific anti-leishmanial antibodies have been developed, but all have two major limitations. First, though serum antibody levels decrease after successful treatment68,69, they remain detectable up to several years after cure53,70,71. Therefore, VL relapse cannot be diagnosed by serology. Second, a significant proportion of healthy individuals living in endemic areas with no history of VL are positive for anti-leishmanial antibodies owing to asymptomatic infections. The seroprevalence in healthy populations varies from <10% in low to moderate endemic areas27,72,73, to >30% in high-transmission foci or in household contacts74,75,76. Antibody-based tests must therefore always be used in combination with a standardized clinical case definition for VL diagnosis.
Serological tests based on indirect fluorescence antibody (IFA), enzyme-linked immunosorbent assay (ELISA) or western blot have shown high diagnostic accuracy in most studies but are poorly adapted to field settings77,78,79,80. Two serological tests have been specifically developed for field use and have been sufficiently validated — the direct agglutination test (DAT) and the rK39-based immunochromatographic test (ICT) (Fig. 4). The DAT is a semi-quantitative test that uses microtitre plates in which increasing dilutions of patient's serum or blood are mixed with stained killed L. donovani promastigotes81,82. If specific antibodies are present, agglutination is visible after 18 hours with the naked eye. This test has been extensively validated in most endemic areas. Thirty studies were included in a recent meta-analysis, which gave sensitivity and specificity estimates of 94.8% (95% confidence intervals (CI), 92.7–96.4) and 97.1% (95% CI, 93.9–98.7), respectively83. The performance of the DAT was influenced by neither the region nor by the Leishmania species. Freeze-dried antigen is more robust than liquid antigen74,84,85,86. The DAT is simpler than many other tests but it requires equipment such as microtitre plates and micropipettes, well-trained laboratory technicians and regular quality control. The storage of the antigen at 2–8°C once it has been dissolved and the prolonged incubation time are other drawbacks. The fast agglutination screening test (FAST) is a simplified (single serum dilution at a cutoff of 1:800 or 1:1600) and more rapid (2–3 hours) version of the DAT, and its diagnostic accuracy seems comparable87,88,89, but further validation is needed.
rK39 is a 39-amino acid repeat that is part of a kinesin-related protein in Leishmania chagasi and which is conserved within the L. donovani complex90. An rK39-based ELISA showed excellent sensitivity (93–100%) and specificity (97–98%) in many VL-endemic countries69,90,91,92,93,94,95,96,97,98. The test was then developed into an ICT, or dipstick, format that was more suitable for field use. A meta-analysis that included 13 validation studies of the rK39 ICT showed sensitivity and specificity estimates of 93.9% (95% CI, 87.7–97.1) and 95.3% (95% CI, 88.8–98.1), respectively83. Recently, the excellent diagnostic performance of rK39 ICT was confirmed in India and Nepal74,75,99,100. However, this test has been shown to be less accurate in East Africa92,101,102,103,104. For reasons that remain unclear, Sudanese patients seem to develop lower titres of antibodies against rK39 than do Indian patients, although the format of the test might be a factor, as other brands of ICT performed better in this region101,105.
rK39 ICTs are easy to perform, rapid (10–20 minutes), cheap (around US$1 per test) and give reproducible results. They are currently the best available diagnostic tool for VL for use in remote areas, and their wide distribution and use within an appropriate VL diagnostic algorithm should be promoted. The case-management strategy of the VL elimination programme planned for the Indian subcontinent (Box 2), which is based on the treatment of suspected clinically infected individuals who have positive rK39 ICT results, is supported by solid scientific evidence. Given that several counterfeit VL ICTs have already been found in the Indian subcontinent, the need for rigorous quality standards and regulation of diagnostics should be addressed at the same time.
Antigen-detection tests. In theory, antigen-detection tests should be more specific than antibody-detection tests as they avoid cross-reactivity and can distinguish active from past infections. A latex agglutination test detecting a heat-stable, low-molecular-weight carbohydrate antigen in the urine of VL patients has shown promising initial results106,107. Several studies conducted in East Africa and the Indian subcontinent showed good specificity but only low to moderate (48–87%) sensitivity99,100,108,109. The latex agglutination test correlated well with cure in a high proportion (97–100%) of patients during anti-leishmanial treatment109,110. Apart from its low sensitivity, there are two practical limitations: the urine must be boiled to avoid false-positive reactions and it is difficult to distinguish weakly positive from negative results, which affects the reproducibility of the test99,108. Work to improve the format of this test is ongoing.
VL treatment strategies
Treatment of VL relies on specific anti-leishmanial drugs and the aggressive management of any concomitant bacterial or parasitic infections, anaemia, hypovolemia (decreased blood volume) and malnutrition. The pentavalent antimonials sodium stibogluconate and meglumine antimoniate have been the first-line treatment for VL in many areas for more than 70 years. Cheaper generic forms of these drugs are available that have been shown to be equivalent to the branded products111,112,113. Antimonials are toxic drugs with frequent, sometimes life-threatening, adverse side effects, including cardiac arrhythmia and acute pancreatitis. Patients under the age of 2 or aged 45 or over with signs of advanced disease and/or severe malnutrition are at higher risk of death during antimonial therapy owing to drug toxicity, slowness of drug action, VL complications or a combination of these factors57,114.
Conventional amphotericin B has replaced antimonials as the first-line treatment for VL in some areas of the Bihar State of India where treatment failure rates for antimonials reached >60% (Ref. 115). In reality, antimonials are still in use in many peripheral public health facilities in these areas (F. C., unpublished data). Infusion-related fever, chills and rigor are almost universal side effects following treatment with conventional amphotericin B, and life-threatening adverse side effects such as hypokalemia (low potassium levels in the blood), nephrotoxicity and first-dose anaphylaxis are not uncommon. Moreover, this drug is costly and requires a complicated regimen (15 slow infusions on alternate days).
Although there is still a need for more research and development (R&D) to improve the drug pipeline for VL116, some alternatives have become available in recent years. Liposomal amphoterin B is considered by many experts as the best existing drug against VL, and is used as first-line treatment in Europe and the United States. Until recently, its use in developing countries was precluded by its high market price (US$2,800 per treatment)117,118. This situation might change as the World Health Organization (WHO) announced a drastic price reduction in May 2007, with the cost of an average course for the public health sector in VL-endemic countries reduced to US$200.
Miltefosine, which was initially developed as an anti-cancer drug, is the first effective oral drug for VL119. In a recent Phase IV trial, the final cure rate was 82% by intention to treat and 95% by per protocol analysis, with only three deaths out of 1,132 patients. In this study, common toxicity criteria grade 3 adverse events were found in 3% of patients, including severe gastrointestinal toxicity and significant increases in the levels of serum aspartate aminotransferase, alanine aminotransferase and creatinine120. Data on miltefosine use in East Africa are restricted to one study that was conducted in northern Ethiopia, in which it was found to be as safe and effective as sodium stibogluconate in HIV-negative patients and safer, but less effective, in HIV co-infected patients121. Miltefosine is a teratogenic drug and its use is therefore strictly forbidden in pregnant women or in women who could become pregnant within two months of treatment. Miltefosine has been registered in India since 2002 and so far it has only been available on the private market, at a retail cost of US$125–200 per treatment course120,122. For large orders, miltefosine can be ordered through WHO at a preferential price (for example, €54.9 EUR (∼$75) per pack for adults for an order of 3,000–9,000 packs). This drug has a long half life (∼150 hours) and parasite resistance is easily induced in vitro123. Non-adherence to the recommended regimen could lead to widespread parasite resistance124. Therefore, a strictly supervised public distribution system, preferably taking the form of directly observed therapy (DOT), must be urgently implemented125. The increasing use of miltefosine for canine leishmaniasis in Europe might also increase the development of miltefosine resistance in L. infantum.
Paromomycin (formerly known as aminosidine), is an aminoglycoside antibiotic with good anti-leishmanial activity125. The results from initial studies in India and Africa were promising126,127, but the original manufacturer abandoned production. The results of Phase III trials recently conducted in India showed excellent efficacy and safety. No nephrotoxicity was observed, reversible high-tone ototoxicity (damage to the inner ear) was found in 2% of patients and 1.8% of patients showed a significant increase (>fivefold) in hepatic transaminases128. Mild injection pain was reported by over 50% of patients. Paromomycin was registered in India in August 2006. In East Africa, paromomycin is currently being evaluated in mono- and combination (with sodium stibogluconate) therapy. Other advantages of paromomycin include the fact that it is active against a wide variety of pathogens, including bacteria, and its low cost (US$5–10 per treatment).
Sitamaquine is an oral 8-aminoquinoline drug that showed evidence of efficacy against VL more than 20 years ago129. Phase II studies were conducted in Brazil, Kenya and India and obtained cure rates ranging from 27% to 87%, but there were also several cases of serious renal adverse events130,131,132. Phase IIb and III studies are ongoing and planned, respectively, in India.
Combination therapy is the suggested way forward to increase treatment efficacy, prevent the development of drug resistance, reduce treatment duration and perhaps decrease treatment cost133. The association of sodium stibogluconate and paromomycin was found to be safe and effective in early trials conducted in India and East Africa126,134,135, and has been successfully used by Médecins san Frontieres in more than 4,000 Sudanese patients136. Drug combinations including liposomal amphotericin B and miltefosine are currently being studied in India.
Vaccine development
The simple nature of the parasite lifecycle and the fact that healing and recovery protects individuals from re-infection indicate that it should be possible to develop a vaccine against VL. The extensive knowledge that has been gathered from animal models has shown that protection against live challenge could be achieved using parasite-specific proteins, DNA137 or genetically attenuated parasites138,139,140; advances in our understanding of Leishmania–host interactions, Leishmania pathogenesis, protective immunity141 and the availability of the complete Leishmania major genome sequence142, could take this a step further.
So far, however, progress in developing a protective vaccine against the different human leishmaniases has been limited. 'Leishmanization', which simulates live infection by inoculation of live L. major as a vaccine for protection against cutaneous leishmaniasis, has been carried out in Uzbekistan, Iran and Israel143,144. However, this technique is not suitable for large-scale use or for use in HIV-endemic areas, and has been discontinued. Although studies in a high-prevalence endemic site in Eastern Sudan showed that previous natural L. major cutaneous disease or exposure to it can protect from VL26, a trial of a killed L. major vaccine that was administared with the tuberculosis vaccine bacille Calmette–Guérin (BCG) failed145. In fact, none of the preparations of killed parasite (which are crude preparations and difficult to define and standardize) with or without adjuvants has shown significant prophylactic efficacy146,147,148. A second-generation vaccine is now in clinical development as a therapeutic or prophylactic vaccine against VL; it contains a recombinant protein comprising three leishmanial antigens (Leish-111f)149 and a defined adjuvant (MPL-SE)150.
Interest in developing immunochemotherapeutic vaccines, which combine a vaccine with drug treatment and which have the potential to become a practical and affordable treatment for PKDL and other persistent forms of leishmaniasis, is gaining momentum. Encouraging results have been obtained using alum-precipitated autoclaved L. major plus BCG together with pentavalent antimonials to treat persistent PKDL in Sudan151. The use of such a therapeutic vaccine could reduce the dose and duration of chemotherapy and be an important control tool. Further studies should focus on exploring the L. major genome sequence for candidate protective antigens, testing these candidates in animal models and translating these studies to humans using novel antigen and adjuvant combinations. Clinical trials should be performed in well-defined sites where there is a high prevalence of the target leishmaniasis.
Discussion and conclusions
Recent breakthroughs such as the development of miltefosine and paromomycin as novel drugs, the reduction in the cost of liposomal amphotericin B and the development of the rK39 rapid diagnostic test have allowed the formulation of an ambitious VL elimination programme in the Indian subcontinent (Box 2). The elimination initiative itself raises several pressing research and operational questions related to its implementation that must be addressed in the short term (Box 3).
More generally, although much can be achieved with the existing tools, there is a definite need for continued investment in diagnostics, treatment and prevention of VL. rK39 ICTs perform well for the primary diagnosis of VL in the Indian subcontinent, but further test development and evaluation is required in East Africa, with specific attention to the diagnostic accuracy in HIV co-infected patients. The development of a sensitive and easy to use non-invasive antigen-detection test for the diagnosis of primary VL (particularly in HIV co-infected patients), for the diagnosis of relapses and to assess treatment efficacy also remains an important challenge. Further investment in drug development is still badly needed to fill the pipeline with novel compounds, as all of the current drugs have one or more drawbacks. In the meantime, the evaluation of combination therapies with existing drugs remains a priority. Immunochemotherapy is another approach that should be considered. The development of improved therapies should go hand-in-hand with the development of practical tools to monitor drug resistance. Last but not least, an effective vaccine would significantly improve VL control, as would new methods to control the animal reservoir in L. infantum-endemic areas (for example dog collars) or to prevent human infection (such as insecticide impregnated bednets or blankets).
The translation of the extensive knowledge we now have of leishmanial biology into effective diagnostic, treatment and control tools and their rigorous field validation are essential steps to remove VL from the list of the most neglected diseases. Nevertheless, such efforts will have only limited impact if these tools are not made available to all patients. An increased and sustained commitment from all implementing and funding partners therefore remains an urgent priority.
References
Herwaldt, B. L. Leishmaniasis. Lancet 354, 1191–1199 (1999).
Pearson, R. D. & Sousa, A. Q. Clinical spectrum of Leishmaniasis. Clin. Infect. Dis. 22, 1–13 (1996).
Arevalo, J. et al. Influence of leishmania (viannia) species on the response to antimonial treatment in patients with american tegumentary leishmaniasis. J. Infect. Dis. 195, 1846–1851 (2007).
Dedet, J. P. & Pratlong, F. in Manson's Tropical Diseases (eds Cook, G. C. & Zumla, A. I.) 1339–1364 (Elsevier, London, 2003).
Lukes, J. et al. Evolutionary and geographical history of the Leishmania donovani complex with a revision of current taxonomy. Proc. Natl Acad. Sci. USA 104, 9375–9380 (2007).
Mauricio, I. L., Stothard, J. R. & Miles, M. A. The strange case of Leishmania chagasi. Parasitol. Today 16, 188–189 (2000).
Alvar, J., Canavate, C., Molina, R., Moreno, J. & Nieto, J. Canine leishmaniasis. Adv. Parasitol. 57, 1–88 (2004).
Zijlstra, E. E., Musa, A. M., Khalil, E. A., el-Hassan, I. M. & el-Hassan, A. M. Post-kala-azar dermal leishmaniasis. Lancet Infect. Dis. 3, 87–98 (2003).
Addy, M. & Nandy, A. Ten years of kala-azar in west Bengal, Part I. Did post-kala-azar dermal leishmaniasis initiate the outbreak in 24-Parganas? Bull. World Health Organ. 70, 341–346 (1992).
Desjeux, P. Leishmaniasis: current situation and new perspectives. Comp. Immunol. Microbiol. Infect. Dis. 27, 305–318 (2004).
WHO. The World Health Report, [online] (WHO, Geneva, Switzerland, 2002).
Collin, S. M., Coleman, P. G., Ritmeijer, K. & Davidson, R. N. Unseen Kala-azar deaths in south Sudan (1999–2002). Trop. Med. Int. Health 11, 509–512 (2006).
Singh, S. P., Reddy, D. C., Rai, M. & Sundar, S. Serious underreporting of visceral leishmaniasis through passive case reporting in Bihar, India. Trop. Med. Int. Health 11, 899–905 (2006).
Boelaert, M. et al. Visceral leishmaniasis control: a public health perspective. Trans. R. Soc. Trop. Med. Hyg. 94, 465–471 (2000).
Desjeux, P. The increase in risk factors for leishmaniasis worldwide. Trans. R. Soc. Trop. Med. Hyg. 95, 239–243 (2001)
Seaman, J., Mercer, A. J. & Sondorp, E. The epidemic of visceral leishmaniasis in western Upper Nile, southern Sudan: course and impact from 1984 to 1994. Int. J. Epidemiol. 25, 862–871 (1996).
Alvar, J., Yactayo, S. & Bern, C. Leishmaniasis and poverty. Trends Parasitol. 22, 552–557 (2006).
Ahluwalia, I. B. et al. Visceral leishmaniasis: consequences of a neglected disease in a Bangladeshi community. Am. J. Trop. Med. Hyg. 69, 624–628 (2003).
Rijal, S., Koirala, S., Van der Stuyft, P. & Boelaert, M. The economic burden of visceral leishmaniasis for households in Nepal. Trans. R. Soc. Trop. Med. Hyg. 100, 838–841 (2006).
Hotez, P. J. et al. Combating tropical infectious diseases: report of the Disease Control Priorities in Developing Countries Project. Clin. Infect. Dis. 38, 871–878 (2004).
Siddig, M., Ghalib, H., Shillington, D. C., Petersen, E. A. & Khidir, S. Visceral leishmaniasis in Sudan. Clinical features. Trop. Geogr. Med. 42, 107–112 (1990).
Zijlstra, E. E. et al. Kala-azar in displaced people from southern Sudan: epidemiological, clinical and therapeutic findings. Trans. R. Soc. Trop. Med. Hyg. 85, 365–369 (1991).
Rittig, M.G. & Bogdan, C. Leishmania–host-cell interaction: complexities and alternative views. Parasitol. Today 16, 292–297 (2000).
Lodge, R., Diallo, T.O. & Descoteaux, A. Leishmania donovani lipophosphoglycan blocks NADPH oxidase assembly at the phagosome membrane. Cell. Microbiol. 8, 1922–1931 (2006).
Khalil, E. A., Zijlstra, E. E., Kager, P. A. & El Hassan, A. M. Epidemiology and clinical manifestations of Leishmania donovani infection in two villages in an endemic area in eastern Sudan. Trop. Med. Int. Health 7, 35–44 (2002).
Zijlstra, E. E., el-Hassan, A. M., Ismael, A. & Ghalib, H. W. Endemic kala-azar in eastern Sudan: a longitudinal study on the incidence of clinical and subclinical infection and post-kala-azar dermal leishmaniasis. Am. J. Trop. Med. Hyg. 51, 826–836 (1994).
Schaefer, K. U., Kurtzhals, J. A., Gachihi, G. S., Muller, A. S. & Kager, P. A. A prospective sero-epidemiological study of visceral leishmaniasis in Baringo District, Rift Valley Province, Kenya. Trans. R. Soc. Trop. Med. Hyg. 89, 471–475 (1995).
Ali, A. & Ashford, R. W. Visceral leishmaniasis in Ethiopia. IV. Prevalence, incidence and relation of infection to disease in an endemic area. Ann. Trop. Med. Parasitol. 88, 289–293 (1994).
Davies, C. R. & Mazloumi Gavgani, A. S. Age, acquired immunity and the risk of visceral leishmaniasis: a prospective study in Iran. Parasitology 119, 247–257 (1999).
Evans, T. G. et al. Epidemiology of visceral leishmaniasis in northeast Brazil. J. Infect. Dis. 166, 1124–1132 (1992).
Badaro, R. et al. New perspectives on a subclinical form of visceral leishmaniasis. J. Infect. Dis. 154, 1003–1011 (1986).
Moral, L., Rubio, E. M. & Moya, M. A leishmanin skin test survey in the human population of l'Alacanti region (Spain): implications for the epidemiology of Leishmania infantum infection in southern Europe. Trans. R. Soc. Trop. Med. Hyg. 96, 129–132 (2002).
Sacks, D. L., Lal, S. L., Shrivastava, S. N., Blackwell, J. & Neva, F. A. An analysis of T cell responsiveness in Indian kala-azar. J. Immunol. 138, 908–913 (1987).
Ghalib, H. W. et al. Interleukin 10 production correlates with pathology in human Leishmania donovani infections. J. Clin. Invest. 92, 324–329 (1993).
Nylen, S. et al. Splenic accumulation of IL-10 mRNA in T cells distinct from CD4+CD25+ (Foxp3) regulatory T cells in human visceral leishmaniasis. J. Exp. Med. 204, 805–817 (2007).
Cerf, B. J. et al. Malnutrition as a risk factor for severe visceral leishmaniasis. J. Infect. Dis. 156, 1030–1033 (1987).
Murray, H. W. Kala-azar as an AIDS-related opportunistic infection. AIDS Patient Care STDs 13, 459–465 (1999).
Alvar, J. et al. Leishmania and human immunodeficiency virus coinfection: the first 10 years. Clin. Microbiol. Rev. 10, 298–319 (1997).
Carvalho, E. M. et al. Immunologic markers of clinical evolution in children recently infected with Leishmania donovani chagasi. J. Infect. Dis. 165, 535–540 (1992).
Karplus, T. M. et al. Association between the tumor necrosis factor locus and the clinical outcome of Leishmania chagasi infection. Infect. Immun. 70, 6919–6925 (2002).
Blackwell, J. M., Mohamed, H. S. & Ibrahim, M. E. Genetics and visceral leishmaniasis in the Sudan: seeking a link. Trends Parasitol. 20, 268–274 (2004).
Davies, C. R., Kaye, P., Croft, S. L. & Sundar, S. Leishmaniasis: new approaches to disease control. BMJ 326, 377–382 (2003).
Ashford, D. A. et al. Studies on control of visceral leishmaniasis: impact of dog control on canine and human visceral leishmaniasis in Jacobina, Bahia, Brazil. Am. J. Trop. Med. Hyg. 59, 53–57 (1998).
Palatnik-de-Sousa, C. B. et al. Impact of canine control on the epidemiology of canine and human visceral leishmaniasis in Brazil. Am. J. Trop. Med. Hyg. 65, 510–517 (2001).
Reithinger, R. & Davies, C. R. Canine leishmaniasis: novel strategies for control. Trends Parasitol. 18, 289–290 (2002).
Tesh, R. B. Control of zoonotic visceral leishmaniasis: is it time to change strategies? Am. J. Trop. Med. Hyg. 52, 287–292 (1995).
Alvar, J. et al. Canine leishmaniasis: clinical, parasitological and entomological follow-up after chemotherapy. Ann. Trop. Med. Parasitol. 88, 371–378 (1994).
Gavgani, A. S., Hodjati, M. H., Mohite, H. & Davies, C. R. Effect of insecticide-impregnated dog collars on incidence of zoonotic visceral leishmaniasis in Iranian children: a matched-cluster randomised trial. Lancet 360, 374–379 (2002).
Kaul, S. M., Sharma, R. S., Dey, K. P., Rai, R. N. & Verghese, T. Impact of DDT indoor residual spraying on Phlebotomus argentipes in a kala-azar endemic village in eastern Uttar Pradesh. Bull. World Health Organ. 72, 79–81 (1994).
Singh, R., Das, R. K. & Sharma, S. K. Resistance of sandflies to DDT in Kala-azar endemic districts of Bihar, India. Bull. World Health Organ. 79, 793 (2001).
Hassan, M. M., Elraba'a, F. M., Ward, R. D., Maingon, R. D. & Elnaiem, D. A. Detection of high rates of in-village transmission of Leishmania donovani in eastern Sudan. Acta Trop. 92, 77–82 (2004).
Bern, C. et al. Factors associated with visceral leishmaniasis in Nepal: bed-net use is strongly protective. Am. J. Trop. Med. Hyg. 63, 184–188 (2000).
Bern, C. et al. Risk factors for kala-azar in Bangladesh. Emerg. Infect. Dis. 11, 655–662 (2005).
Ritmeijer, K. et al. Evaluation of a mass distribution programme for fine-mesh impregnated bednets against visceral leishmaniasis in eastern Sudan. Trop. Med. Int. Health 12, 404–414 (2007).
Reyburn, H., Ashford, R., Mohsen, M., Hewitt, S. & Rowland, M. A randomized controlled trial of insecticide-treated bednets and chaddars or top sheets, and residual spraying of interior rooms for the prevention of cutaneous leishmaniasis in Kabul, Afghanistan. Trans. R. Soc. Trop. Med. Hyg. 94, 361–366 (2000).
Kroeger, A., Avila, E. V. & Morison, L. Insecticide impregnated curtains to control domestic transmission of cutaneous leishmaniasis in Venezuela: cluster randomised trial. BMJ 325, 810–813 (2002).
Collin, S. et al. Conflict and kala-azar: determinants of adverse outcomes of kala-azar among patients in southern Sudan. Clin. Infect. Dis. 38, 612–619 (2004).
Guerin, P. J. et al. Visceral leishmaniasis: current status of control, diagnosis, and treatment, and a proposed research and development agenda. Lancet Infect. Dis. 2, 494–501 (2002).
Boelaert, M. et al. A comparative study of the effectiveness of diagnostic tests for visceral leishmaniasis. Am. J. Trop. Med. Hyg. 70, 72–77 (2004).
Sundar, S. Diagnosis of kala-azar — an important stride. J. Assoc. Physicians India 51, 753–755 (2003).
Zijlstra, E. E. et al. Kala-azar: a comparative study of parasitological methods and the direct agglutination test in diagnosis. Trans. R. Soc. Trop. Med. Hyg. 86, 505–507 (1992).
Siddig, M., Ghalib, H., Shillington, D. C. & Petersen, E. A. Visceral leishmaniasis in the Sudan: comparative parasitological methods of diagnosis. Trans. R. Soc. Trop. Med. Hyg. 82, 66–68 (1988).
Young, S. Kala-azar in Pi-Hsien District, Kiangsu Province, China. II. Findings in films of spleen and liver puncture juice and some other observations in kala-azar. J. Shanghai Sci. Inst. 4, 265–272 (1939).
Ho, E. A., Soong, T. H. & Li, Y. Comparative merits of sternum, spleen and liver punctures in the study of human leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 41, 629–636 (1948).
Babiker, Z. O., Davidson, R., Mazinda, C., Kipngetich, S. & Ritmeijer, K. Utility of lymph node aspiration in the diagnosis of visceral leishmaniasis in Sudan. Am. J. Trop. Med. Hyg. 76, 689–693 (2007).
Kager, P. A. & Rees, P. H. Splenic aspiration. Review of the literature. Trop. Geogr. Med. 35, 111–124 (1983).
Reithinger, R. & Dujardin, J. C. Molecular diagnosis of leishmaniasis: current status and future applications. J. Clin. Microbiol. 45, 21–25 (2007).
Kumar, R., Pai, K., Pathak, K. & Sundar, S. Enzyme-linked immunosorbent assay for recombinant K39 antigen in diagnosis and prognosis of Indian visceral leishmaniasis. Clin. Diagn. Lab. Immunol. 8, 1220–1224 (2001).
Braz, R. F. et al. The sensitivity and specificity of Leishmania chagasi recombinant K39 antigen in the diagnosis of American visceral leishmaniasis and in differentiating active from subclinical infection. Am. J. Trop. Med. Hyg. 67, 344–348 (2002).
De Almeida Silva, L. et al. Immunologic tests in patients after clinical cure of visceral leishmaniasis. Am. J. Trop. Med. Hyg. 75, 739–743 (2006).
Hailu, A. Pre- and post-treatment antibody levels in visceral leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 84, 673–675 (1990).
Koirala, S., Karki, P., Das, M. L., Parija, S. C. & Karki, B. M. Epidemiological study of kala-azar by direct agglutination test in two rural communities of eastern Nepal. Trop. Med. Int. Health 9, 533–537 (2004).
Schenkel, K. et al. Visceral leishmaniasis in southeastern Nepal: a cross-sectional survey on Leishmania donovani infection and its risk factors. Trop. Med. Int. Health 11, 1792–1799 (2006).
Sundar, S. et al. Serological diagnosis of Indian visceral leishmaniasis: direct agglutination test versus rK39 strip test. Trans. R. Soc. Trop. Med. Hyg. 100, 533–537 (2006).
Sundar, S. et al. Rapid, noninvasive diagnosis of visceral leishmaniasis in India: comparison of two immunochromatographic strip tests for detection of anti-K39 antibody. J. Clin. Microbiol. 44, 251–253 (2006).
Ibrahim, M. E. et al. Kala-azar in a high transmission focus: an ethnic and geographic dimension. Am. J. Trop. Med. Hyg. 61, 941–944 (1999).
Ho, M., Leeuwenburg, J., Mbugua, G., Wamachi, A. & Voller, A. An enzyme-linked immunosorbent assay (ELISA) for field diagnosis of visceral leishmaniasis. Am. J. Trop. Med. Hyg. 32, 943–946 (1983).
Iqbal, J. et al. Imported visceral leishmaniasis: diagnostic dilemmas and comparative analysis of three assays. J. Clin. Microbiol. 40, 475–479 (2002).
Sinha, R. & Sehgal, S. Comparative evaluation of serological tests in Indian kala-azar. J. Trop. Med. Hyg. 97, 333–340 (1994).
Sreenivas, G. et al. Diagnosis of visceral leishmaniasis: comparative potential of amastigote antigen, recombinant antigen and PCR. Br. J. Biomed. Sci. 59, 218–222 (2002).
Harith, A. E. et al. A simple and economical direct agglutination test for serodiagnosis and sero-epidemiological studies of visceral leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 80, 583–536 (1986).
Harith, A. E. et al. Evaluation of a newly developed direct agglutination test (DAT) for serodiagnosis and sero-epidemiological studies of visceral leishmaniasis: comparison with IFAT and ELISA. Trans. R. Soc. Trop. Med. Hyg. 81, 603–606 (1987).
Chappuis, F., Rijal, S., Soto, A., Menten, J. & Boelaert, M. A meta-analysis of the diagnostic performance of the direct agglutination test and rK39 dipstick for visceral leishmaniasis. BMJ 333, 723 (2006).
Boelaert, M. et al. Multi-centre evaluation of repeatability and reproducibility of the direct agglutination test for visceral leishmaniasis. Trop. Med. Int. Health 4, 31–37 (1999).
Jacquet, D. et al. Comparative evaluation of freeze-dried and liquid antigens in the direct agglutination test for serodiagnosis of visceral leishmaniasis (ITMA-DAT/VL). Trop. Med. Int. Health 11, 1777–1784 (2006).
Meredith, S. E. et al. Leish-KIT, a stable direct agglutination test based on freeze-dried antigen for serodiagnosis of visceral leishmaniasis. J. Clin. Microbiol. 33, 1742–1745 (1995).
Schoone, G. J. et al. A fast agglutination screening test (FAST) for the detection of anti-Leishmania antibodies. Trans. R. Soc. Trop. Med. Hyg. 95, 400–401 (2001).
Hailu, A. et al. Field evaluation of a fast anti-Leishmania antibody detection assay in Ethiopia. Trans. R. Soc. Trop. Med. Hyg. 100, 48–52 (2006).
Silva, E. S. et al. Application of direct agglutination test (DAT) and fast agglutination screening test (FAST) for sero-diagnosis of visceral leishmaniasis in endemic area of Minas Gerais, Brazil. Kinetoplastid Biol. Dis. 4, 4 (2005).
Burns, J. M., Jr et al. Molecular characterization of a kinesin-related antigen of Leishmania chagasi that detects specific antibody in African and American visceral leishmaniasis. Proc. Natl Acad. Sci. USA 90, 775–779 (1993).
Zijlstra, E. E. et al. rK39 enzyme-linked immunosorbent assay for diagnosis of Leishmania donovani infection. Clin. Diagn. Lab. Immunol. 5, 717–720 (1998).
Zijlstra, E. E. et al. Diagnosing visceral leishmaniasis with the recombinant K39 strip test: experience from the Sudan. Trop. Med. Int. Health 6, 108–113 (2001).
Ozensoy, S. et al. Serodiagnosis and epidemiology of visceral leishmaniasis in Turkey. Am. J. Trop. Med. Hyg. 59, 363–369 (1998).
Maalej, I. A. et al. Comparative evaluation of ELISAs based on ten recombinant or purified Leishmania antigens for the serodiagnosis of Mediterranean visceral leishmaniasis. Am. J. Trop. Med. Hyg. 68, 312–320 (2003).
Kurkjian, K. M. et al. Application of an improved method for the recombinant k39 enzyme-linked immunosorbent assay to detect visceral leishmaniasis disease and infection in Bangladesh. Clin. Diagn. Lab. Immunol. 12, 1410–1415 (2005).
Singh, S., Gilman-Sachs, A., Chang, K. P. & Reed, S. G. Diagnostic and prognostic value of K39 recombinant antigen in Indian leishmaniasis. J. Parasitol. 81, 1000–1003 (1995).
Qu, J. Q. et al. Serodiagnosis of Asian leishmaniasis with a recombinant antigen from the repetitive domain of a Leishmania kinesin. Trans. R. Soc. Trop. Med. Hyg. 88, 543–545 (1994).
Badaro, R. et al. rK39: a cloned antigen of Leishmania chagasi that predicts active visceral leishmaniasis. J. Infect. Dis. 173, 758–761 (1996).
Chappuis, F. et al. Field validity, reproducibility and feasibility of diagnostic tests for visceral leishmaniasis in rural Nepal. Trop. Med. Int. Health 11, 31–40 (2006).
Sundar, S. et al. Comparative evaluation of parasitology and serological tests in the diagnosis of visceral leishmaniasis in India: a phase III diagnostic accuracy study. Trop. Med. Int. Health 12, 284–289 (2007).
Ritmeijer, K. et al. Evaluation of a new recombinant K39 rapid diagnostic test for Sudanese visceral leishmaniasis. Am. J. Trop. Med. Hyg. 74, 76–80 (2006).
Veeken, H., Ritmeijer, K., Seaman, J. & Davidson, R. Comparison of an rK39 dipstick rapid test with direct agglutination test and splenic aspiration for the diagnosis of kala-azar in Sudan. Trop. Med. Int. Health 8, 164–167 (2003).
Diro, E. et al. Field evaluation of FD-DAT, rk-39 dipstick and KATEX (urine latex agglutination) for diagnosis of visceral leishmaniasis in northwest Ethiopia. Trans. R. Soc. Trop. Med. Hyg. (in the press).
Boelaert, M. et al. Diagnostic tests for kala-azar: a multi-centre study of the freeze-dried DAT, rK39 strip test and KAtex in East-Africa and the Indian subcontinent. Trans. R. Soc. Trop. Med. Hyg. (in the press).
Chappuis, F. et al. Diagnostic accuracy of two rK39 antigen-based dipsticks and the formol gel test for rapid diagnosis of visceral leishmaniasis in northeastern Uganda. J. Clin. Microbiol. 43, 5973–5977 (2005).
Attar, Z. J. et al. Latex agglutination test for the detection of urinary antigens in visceral leishmaniasis. Acta Trop. 78, 11–16 (2001).
Sarkari, B., Chance, M. & Hommel, M. Antigenuria in visceral leishmaniasis: detection and partial characterisation of a carbohydrate antigen. Acta Trop. 82, 339–348 (2002).
Rijal, S. et al. Evaluation of a urinary antigen-based latex agglutination test in the diagnosis of kala-azar in eastern Nepal. Trop. Med. Int. Health 9, 724–729 (2004).
Sundar, S., Agrawal, S., Pai, K., Chance, M. & Hommel, M. Detection of leishmanial antigen in the urine of patients with visceral leishmaniasis by a latex agglutination test. Am. J. Trop. Med. Hyg. 73, 269–271 (2005).
El-Safi, S. H. et al. Field evaluation of latex agglutination test for detecting urinary antigens in visceral leishmaniasis in Sudan. East Mediterr. Health J. 9, 844–855 (2003).
Veeken, H., Ritmeijer, K., Seaman, J. & Davidson, R. A randomized comparison of branded sodium stibogluconate and generic sodium stibogluconate for the treatment of visceral leishmaniasis under field conditions in Sudan. Trop. Med. Int. Health 5, 312–317 (2000).
Moore, E. et al. Comparison of generic and proprietary sodium stibogluconate for the treatment of visceral leishmaniasis in Kenya. Bull. World Health Organ. 79, 388–393 (2001).
Ritmeijer, K. et al. Ethiopian visceral leishmaniasis: generic and proprietary sodium stibogluconate are equivalent; HIV co-infected patients have a poor outcome. Trans. R. Soc. Trop. Med. Hyg. 95, 668–672 (2001).
Seaman, J., Mercer, A. J., Sondorp, H. E. & Herwaldt, B. L. Epidemic visceral leishmaniasis in southern Sudan: treatment of severely debilitated patients under wartime conditions and with limited resources. Ann. Intern. Med. 124, 664–672 (1996).
Sundar, S. et al. Failure of pentavalent antimony in visceral leishmaniasis in India: report from the center of the Indian epidemic. Clin. Infect. Dis. 31, 1104–1107 (2000).
Yamey, G. & Torreele, E. The world's most neglected diseases. BMJ 325, 176–177 (2002).
Gradoni, L., Gramiccia, M. & Scalone, A. Visceral leishmaniasis treatment, Italy. Emerg. Infect. Dis. 9, 1617–1620 (2003).
Bern, C. et al. Liposomal amphotericin B for the treatment of visceral leishmaniasis. Clin. Infect. Dis. 43, 917–924 (2006).
Berman, J. Miltefosine to treat leishmaniasis. Expert Opin. Pharmacother. 6, 1381–1388 (2005).
Bhattacharya, S. K. et al. Phase IV trial of miltefosine in the treatment of Indian visceral leishmaniasis. J. Infect. Dis. 196, 591–598 (2007).
Ritmeijer, K. et al. A comparison of miltefosine and sodium stibogluconate for treatment of visceral leishmaniasis in an Ethiopian population with high prevalence of HIV infection. Clin. Infect. Dis. 43, 357–364 (2006).
Sundar, S. & Murray, H.W. Availability of miltefosine for the treatment of kala-azar in India. Bull. World Health Organ. 83, 394–395 (2005).
Perez-Victoria, F. J. et al. Mechanisms of experimental resistance of Leishmania to miltefosine: implications for clinical use. Drug Resist. Update 9, 26–39 (2006).
Thakur, C.P. Socio-economics of visceral leishmaniasis in Bihar (India). Trans. R. Soc. Trop. Med. Hyg. 94, 156–157 (2000).
den Boer, M. & Davidson, R. N. Treatment options for visceral leishmaniasis. Expert Rev. Anti Infect. Ther. 4, 187–197 (2006).
Chunge, C. N., Owate, J., Pamba, H. O. & Donno, L. Treatment of visceral leishmaniasis in Kenya by aminosidine alone or combined with sodium stibogluconate. Trans. R. Soc. Trop. Med. Hyg. 84, 221–225 (1990).
Jha, T. K. et al. Randomised controlled trial of aminosidine (paromomycin) v sodium stibogluconate for treating visceral leishmaniasis in North Bihar, India. BMJ 316, 1200–1205 (1998).
Sundar, S. et al. Injectable paromomycin for visceral leishmaniasis in India. N. Engl. J. Med. 356, 2571–2581 (2007).
Sherwood, J. A. et al. Phase 2 efficacy trial of an oral 8-aminoquinoline (WR6026) for treatment of visceral leishmaniasis. Clin. Infect. Dis. 19, 1034–1039 (1994).
Jha, T. K. et al. A phase II dose-ranging study of sitamaquine for the treatment of visceral leishmaniasis in India. Am. J. Trop. Med. Hyg. 73, 1005–1011 (2005).
Dietze, R. et al. Phase 2 trial of WR6026, an orally administered 8-aminoquinoline, in the treatment of visceral leishmaniasis caused by Leishmania chagasi. Am. J. Trop. Med. Hyg. 65, 685–689 (2001).
Wasunna, M. K. et al. A phase II dose-increasing study of sitamaquine for the treatment of visceral leishmaniasis in Kenya. Am. J. Trop. Med. Hyg. 73, 871–876 (2005).
Bryceson, A. A policy for leishmaniasis with respect to the prevention and control of drug resistance. Trop. Med. Int. Health 6, 928–934 (2001).
Seaman, J. et al. Epidemic visceral leishmaniasis in Sudan: a randomized trial of aminosidine plus sodium stibogluconate versus sodium stibogluconate alone. J. Infect. Dis. 168, 715–720 (1993).
Thakur, C. P. et al. A prospective randomized, comparative, open-label trial of the safety and efficacy of paromomycin (aminosidine) plus sodium stibogluconate versus sodium stibogluconate alone for the treatment of visceral leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 94, 429–431 (2000).
Melaku, Y. et al. Treatment of kala-azar in southern Sudan using a 17-day regimen of sodium stibogluconate combined with paromomycin: a retrospective comparison with 30-day sodium stibogluconate monotherapy. Am. J. Trop. Med. Hyg. 77, 89–94 (2007).
Kedzierski, L., Zhu, Y. & Handman, E. Leishmania vaccines: progress and problems. Parasitology 133, S87–S112 (2006).
Amaral, V. F. et al. Study of the safety, immunogenicity and efficacy of attenuated and killed Leishmania (Leishmania) major vaccines in a rhesus monkey (Macaca mulatta) model of the human disease. Mem. Inst. Oswaldo Cruz 97, 1041–1048 (2002).
Uzonna, J. E., Spath, G. F., Beverley, S. M. & Scott, P. Vaccination with phosphoglycan-deficient Leishmania major protects highly susceptible mice from virulent challenge without inducing a strong Th1 response. J. Immunol. 172, 3793–3797 (2004).
Alexander, J., Coombs, G. H. & Mottram, J. C. Leishmania mexicana cysteine proteinase-deficient mutants have attenuated virulence for mice and potentiate a Th1 response. J. Immunol. 161, 6794–6801 (1998).
Saha, S. et al. Immune responses in kala-azar. Indian J. Med. Res. 123, 245–266 (2006).
Ivens, A. C. et al. The genome of the kinetoplastid parasite, Leishmania major. Science 309, 436–442 (2005).
Nadim, A., Javadian, E., Tahvildar-Bidruni, G. & Ghorbani, M. Effectiveness of leishmanization in the control of cutaneous leishmaniasis. Bull. Soc. Pathol. Exot. Filiales 76, 377–383 (1983).
Greenblatt, C. L. Cutaneous leishmaniasis: the prospects for a killed vaccine. Parasitol. Today 4, 53–54 (1988).
Khalil, E. A. et al. Autoclaved Leishmania major vaccine for prevention of visceral leishmaniasis: a randomised, double-blind, BCG-controlled trial in Sudan. Lancet 356, 1565–1569 (2000).
Khamesipour, A. et al. Leishmanization: use of an old method for evaluation of candidate vaccines against leishmaniasis. Vaccine 23, 3642–3648 (2005).
Velez, I.D. et al. Failure of a killed Leishmania amazonensis vaccine against American cutaneous leishmaniasis in Colombia. Trans. R. Soc. Trop. Med. Hyg. 99, 593–598 (2005).
Modabber, F., Campos-Neto, A. & Reed, S. in New Generation Vaccines (eds Levine, M. et al.) (Marcel Dekker, New York, 2004).
Skeiky, Y. A. et al. Protective efficacy of a tandemly linked, multi-subunit recombinant leishmanial vaccine (Leish-111f) formulated in MPL adjuvant. Vaccine 20, 3292–3303 (2002).
Reed, S. G., Coler, R. N. & Campos-Neto, A. Development of a leishmaniasis vaccine: the importance of MPL. Expert Rev. Vaccines 2, 239–252 (2003).
Musa, A. M. et al. Safety, immunogenicity and possible efficacy of immunochemotherapy of persistent post kala-azar dermal leishmaniasis. Sudanese J. Dermatol. 3, 63–72 (2005).
Moreno, J., Canavate, C., Chamizo, C., Laguna, F. & Alvar, J. HIV–Leishmania infantum co-infection: humoral and cellular immune responses to the parasite after chemotherapy. Trans. R. Soc. Trop. Med. Hyg. 94, 328–332 (2000).
Deniau, M., Canavate, C., Faraut-Gambarelli, F. & Marty, P. The biological diagnosis of leishmaniasis in HIV-infected patients. Ann. Trop. Med. Parasitol. 97 (Suppl. 1), 115–133 (2003).
Rosenthal, E. et al. HIV and Leishmania coinfection: a review of 91 cases with focus on atypical locations of Leishmania. Clin. Infect. Dis. 31, 1093–1095 (2000).
Hailu, A. & Berhe, N. The performance of direct agglutination tests (DAT) in the diagnosis of visceral leishmaniasis among Ethiopian patients with HIV co-infection. Ann. Trop. Med. Parasitol. 96, 25–30 (2002).
Riera, C. et al. Evaluation of a latex agglutination test (KAtex) for detection of Leishmania antigen in urine of patients with HIV–Leishmania coinfection: value in diagnosis and post-treatment follow-up. Eur. J. Clin. Microbiol. Infect. Dis. 23, 899–904 (2004).
Sindermann, H., Engel, K. R., Fischer, C. & Bommer, W. Oral miltefosine for leishmaniasis in immunocompromised patients: compassionate use in 39 patients with HIV infection. Clin. Infect. Dis. 39, 1520–1523 (2004).
Davidson, R. N. et al. Liposomal amphotericin B (AmBisome) in Mediterranean visceral leishmaniasis: a multi-centre trial. Q. J. Med. 87, 75–81 (1994).
Laguna, F. et al. Treatment of visceral leishmaniasis in HIV-infected patients: a randomized trial comparing meglumine antimoniate with amphotericin B. Spanish HIV-Leishmania Study Group. AIDS 13, 1063–1069 (1999).
Pasquau, F. et al. Leishmaniasis as an opportunistic infection in HIV-infected patients: determinants of relapse and mortality in a collaborative study of 228 episodes in a Mediterreanean region. Eur. J. Clin. Microbiol. Infect. Dis. 24, 411–418 (2005).
Lopez-Velez, R. et al. Clinicoepidemiologic characteristics, prognostic factors, and survival analysis of patients coinfected with human immunodeficiency virus and Leishmania in an area of Madrid, Spain. Am. J. Trop. Med. Hyg. 58, 436–443 (1998).
Mira, J. A. et al. Frequency of visceral leishmaniasis relapses in human immunodeficiency virus-infected patients receiving highly active antiretroviral therapy. Am. J. Trop. Med. Hyg. 70, 298–301 (2004).
Fernandez Cotarelo, M. J. et al. Effect of highly active antiretroviral therapy on the incidence and clinical manifestations of visceral leishmaniasis in human immunodeficiency virus-infected patients. Clin. Infect. Dis. 37, 973–977 (2003).
de la Rosa, R. et al. Influence of highly active antiretroviral therapy on the outcome of subclinical visceral leishmaniasis in human immunodeficiency virus-infected patients. Clin. Infect. Dis. 32, 633–635 (2001).
Lopez-Velez, R. The impact of highly active antiretroviral therapy (HAART) on visceral leishmaniasis in Spanish patients who are co-infected with HIV. Ann. Trop. Med. Parasitol. 97 (Suppl. 1), 143–147 (2003).
Rosenthal, E. et al. Declining incidence of visceral leishmaniasis in HIV-infected individuals in the era of highly active antiretroviral therapy. AIDS 15, 1184–1185 (2001).
Desjeux, P. Disease Watch Focus: Leishmaniasis. Nature Rev. Microbiol. 2, 692–693 (2004).
Handman, E. Leishmaniasis: current status of vaccine development. Clin. Microbiol. Rev. 14, 229–243 (2001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Entrez Genome Project
Entrez Protein
FURTHER INFORMATION
Rights and permissions
About this article
Cite this article
Chappuis, F., Sundar, S., Hailu, A. et al. Visceral leishmaniasis: what are the needs for diagnosis, treatment and control?. Nat Rev Microbiol 5, 873–882 (2007). https://doi.org/10.1038/nrmicro1748
Issue Date:
DOI: https://doi.org/10.1038/nrmicro1748
This article is cited by
-
Recognition of Immunoreactive Proteins in Leishmania infantum Amastigote-Like and Promastigote Using Sera of Visceral Leishmaniasis Patients: a Preliminary Study
Acta Parasitologica (2024)
-
Assessment of knowledge, attitude, and practices towards canine visceral leishmaniasis based on the one health concept in Weliso and Ejaji Towns, Oromia, Ethiopia
Scientific Reports (2023)
-
Gasdermin-D activation promotes NLRP3 activation and host resistance to Leishmania infection
Nature Communications (2023)
-
Identification of potential antileishmanial derivatives of 2,3-dihydroquinazolin-4(1H)-one synthesized using AAIL catalyst and molecular docking studies
Research on Chemical Intermediates (2023)
-
1,2,3-triazenes and 1,2,3-triazoles as antileishmanial, antitrypanosomal, and antiplasmodial agents
Medicinal Chemistry Research (2023)