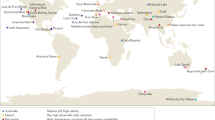
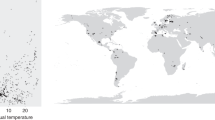
Key Points
-
Since the eighteenth century, biologists have investigated plant and animal biogeography, but only recently have the distributions of microorganisms been examined.
-
We consider microbial biogeography in light of habitats types (the contemporary environment) and provinces (legacies of historical events such as dispersal limitation). This framework is useful for addressing whether the distributions of microbial taxa, like those of macroorganisms, reflect the influences of both contemporary environmental conditions and past events.
-
We review a growing body of literature that suggests that microbial assemblages are not only influenced by their current environment, but that some display a degree of provincialism — evidence that these microbial assemblages have diverged and are maintained by genetic isolation. We also find that the relative influence of historical versus environmental factors appears to be related to the scale of sampling.
-
As a first hypothesis, we suggest that the same processes that influence macroorganism biogeography (colonization, diversification and extinction) also apply to microbial life, but that their rates scale with body size, or for single-celled organisms, cell size. Therefore, we use the idea of allometry as a structure for discussing the rates of biogeographic processes in microorganisms.
-
We conclude that the rates of biogeographic processes probably vary more widely for microorganisms of a given size than for macroorganisms of a given size.
-
To tackle the mechanisms generating microbial biogeographic patterns, we recommend that new microbial biogeography studies should systematically sample and record data from various distances, habitats and environmental conditions.
Abstract
We review the biogeography of microorganisms in light of the biogeography of macroorganisms. A large body of research supports the idea that free-living microbial taxa exhibit biogeographic patterns. Current evidence confirms that, as proposed by the Baas-Becking hypothesis, 'the environment selects' and is, in part, responsible for spatial variation in microbial diversity. However, recent studies also dispute the idea that 'everything is everywhere'. We also consider how the processes that generate and maintain biogeographic patterns in macroorganisms could operate in the microbial world.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Brown, J. H. & Lomolino, M. V. Biogeography (Sinauer, Sunderland, 1998). A definitive textbook on the biogeography of macroorganisms; has just been updated in a 2005 edition.
Ward, D. M., Weller, R. & Bateson, M. M. 16S ribosomal-RNA sequences reveal numerous uncultured microorganisms in a natural community. Nature 345, 63–65 (1990).
Øvreås, L. Population and community level approaches for analysing microbial diversity in natural environments. Ecol. Lett. 3, 236–251 (2000).
Floyd, M. M., Tang, J., Kane, M. & Emerson, D. Captured diversity in a culture collection: case study of the geographic and habitat distributions of environmental isolates held at the American type culture collection. Appl. Environ. Microbiol. 71, 2813–2823 (2005).
Schloss, P. D. & Handelsman, J. Status of the microbial census. Microbiol. Mol. Biol. Rev. 68, 686–691 (2004).
Venter, J. C. et al. Environmental genome shotgun sequencing of the Sargasso Sea. Science 304, 66–74 (2004).
Fenchel, T., Esteban, G. F. & Finlay, B. J. Local versus global diversity of microorganisms: cryptic diversity of ciliated protozoa. Oikos 80, 220–225 (1997).
Staley, J. T. Biodiversity: are microbial species threatened? Curr. Opin. Biotechnol. 8, 340–345 (1997).
Finlay, B. J. Global dispersal of free-living microbial eukaryote species. Science 296, 1061–1063 (2002). A summary of the arguments for why microbial eukaryotes might not be restricted by geographic barriers.
Hedlund, B. P. & Staley, J. T. Microbial endemism and biogeography. In Microbial Diversity and Bioprospecting (ed. Bull, A. T.) (ASM, Washington DC, 2003).
Anagnostakis, S. Chestnut blight: the classical problem of an introduced pathogen. Mycologia 79, 23–27 (1987).
Falush, D. et al. Traces of human migrations in Helicobacter pylori populations. Science 299, 1582–1585 (2003).
Breitbart, M. & Rohwer, F. Here a virus, there a virus, everywhere the same virus? Trends Microbiol. 13, 278–284 (2005).
de Candolle, A. P. Essai Elementaire de Geographie Botanique (F. G. Levrault, Paris, 1820).
Beijerinck, M. W. De infusies en de ontdekking der backteriën. In Jaarboek van de Koninklijke Akademie van Wetenschappen (Müller, Amsterdam, 1913).
Baas-Becking, L. G. M. Geobiologie of Inleiding Tot de Milieukunde (Van Stockkum & Zoon, The Hague, 1934).
Zhang, N. & Blackwell, M. Population structure of dogwood anthracnose fungus. Phytopathology 92, 1276–1283 (2002).
Bala, A., Murphy, P. & Giller, K. E. Distribution and diversity of rhizobia nodulating agroforestry legumes in soils from three continents in the tropics. Mol. Ecol. 12, 917–929 (2003).
Papke, R. T. & Ward, D M. The importance of physical isolation to microbial diversification. FEMS Microbiol. Ecol. 48, 293–303 (2004). A recent review discussing how physical isolation might affect prokaryote evolution.
Cho, J. C. & Tiedje, J. M. Biogeography and degree of endemicity of fluorescent Pseudomonas strains in soil. Appl. Environ. Microbiol. 66, 5448–5456 (2000). One of the first examples of relating the genetic similarity of a free-living bacterial assemblage with geographic distance, using fluorescent Pseudomonas isolates.
Oda, Y., Star, B., Huisman, L. A., Gottschal, J. C. & Forney, L. J. Biogeography of the purple nonsulfur bacterium Rhodopseudomonas palustris. Appl. Environ. Microbiol. 69, 5186–5191 (2003).
Crump, B. C., Hopkinson, C. S., Sogin, M. L. & Hobbie, J. E. Microbial biogeography along an estuarine salinity gradient: combined influences of bacterial growth and residence time. Appl. Environ. Microbiol. 70, 1494–1505 (2004).
Øvreås, L., Forney, L., Daae, R. L. & Torsvik, V. Distribution of bacterioplankton in meromictic Lake Sælenvannet, as determined by denaturing gradient gel electrophoresis of PCR-amplified gene fragments coding for 16S rRNA. Appl. Environ. Microbiol. 63, 3367–3373 (1997).
Schwalbach, M. S., Hewson, I. & Fuhrman, J. A. Viral effects on bacterial community composition in marine plankton microcosms. Aquat. Microb. Ecol. 34, 117–127 (2004).
Rosenzweig, M. L. Species Diversity in Space and Time. (Cambridge University Press, Cambridge, 1995).
Green, J. L. et al. Spatial scaling of microbial eukaryote diversity. Nature 432, 747–750 (2004). Along with reference 27, this study uses distance–decay curves to demonstrate non-random bacterial-assemblage distributions; the studies then test whether the non-random distributions are due to isolation by distance and/or local environmental conditions.
Horner-Devine, M. C., Lage, M., Hughes, J. B. & Bohannan, B. J. M. A taxa–area relationship for bacteria. Nature 432, 750–753 (2004).
Noguez, A. M. et al. Microbial macroecology: highly structured prokaryotic soil assemblages in a tropical deciduous forest. Glob. Ecol. Biogeogr. 14, 241–248 (2005).
Bell, T. et al. Larger islands house more bacterial taxa. Science 308, 1884 (2005).
Smith, V. H. et al. Phytoplankton species richness scales consistently from laboratory microcosms to the world's oceans. Proc. Natl Acad. Sci. USA 102, 4393–4396 (2005).
Cam, E. et al. Disentangling sampling and ecological explanations underlying species–area relationships. Ecology 84, 1118–1130 (2002).
Colwell, R. K., Mao, C. X. & Chang, J. Interpolating, extrapolating, and comparing incidence-based species accumulation curves. Ecology 85, 2717–2727 (2004).
Kuske, C. R. et al. Comparison of soil bacterial communities in rhizospheres of three plant species and the interspaces in an arid grassland. Appl. Environ. Microbiol. 68, 1854–1863 (2002).
Yannarell, A. C. & Triplett, E. W. Within- and between-lake variability in the composition of bacterioplankton communities: investigations using multiple spatial scales. Appl. Environ. Microbiol. 70, 214–223 (2004).
Legendre, P. & Legendre, L. Numerical Ecology 2nd edn (Elsevier, Amsterdam, 1998).
Papke, R. T., Ramsing, N. B., Bateson, M. M. & Ward, D. M. Geographical isolation in hot spring cyanobacteria. Environ. Microbiol. 5, 650–659 (2003).
Whitaker, R. J., Grogan, D. W. & Taylor, J. W. Geographic barriers isolate endemic populations of hyperthermophilic Archaea. Science 301, 976–978 (2003). Already a classic microbial biogeography study, it considers both the effects of spatial isolation and contemporary environmental parameters on hotspring Sulfolobus assemblages within and across continents.
Reche, I., Pulido-Villena, E., Morales-Baquero, R. & Casamayor, E. O. Does ecosystem size determine aquatic bacterial richness? Ecology 86, 1715–1722 (2005).
Brown, J. H., West, G. B. & Enquist, B. J. Scaling in biology: patterns and processes, causes and consequences. In Scaling in Biology (eds Brown, J. H. & West, G. B.) 1–24 (Oxford University Press, Oxford, 2000). A comprehensive introduction to allometric patterns in biology.
Bell, G. The distribution of abundance in neutral communities. Am. Nat. 155, 606–617 (2000).
Hubbell, S. P. The Unified Neutral Theory of Biodiversity and Biogeography (Princeton University Press, Princeton, 2001).
Clark, J. S., Silman, M., Kern, R., Macklin, E. & HilleRisLambers, J. Seed dispersal near and far: patterns across temperate and tropical forests. Ecology 80, 1475–1494 (1999).
Brown, J. H. & Maurer, B. A. Evolution of species assemblages: effects of energetic constraints and species dynamics on the diversification of the North American avifauna. Am. Nat. 130, 1–17 (1987).
Morse, D. R., Stork, N. E. & Lawton, J. H. Species number, species abundance and body length relationships of arboreal beetles in Bornean lowland rain forest trees. Ecol. Entomol. 13, 25–37 (1988).
Lawton, J. H. Species richness and population dynamics of animal assemblages. Patterns in body size: abundance space. Philos. Trans. R. Soc. Lond. B Biol. Sci. 330, 283–291 (1990).
Siemann, E., Tilman, D. & Haarstad, J. Insect species diversity, abundance and body size relationships. Nature 380, 704–706 (1996).
Brown, J. H. Macroecology (Chicago University Press, Chicago, 1995).
Li, W. K. W. Macroecological patterns of phytoplankton in the northwestern North Atlantic Ocean. Nature 419, 154–157 (2002).
Roberts, M. S. & Cohan, F. M. Recombination and migration rates in natural populations of Bacillus subtilis and Bacillus mojavensis. Evolution 49, 1081–1094 (1995).
Stanley, S. M. Macroevolution: Pattern and Process (W. H. Freeman, San Francisco, 1979).
Paradis, E. Statistical analysis of diversification with species traits. Evolution 59, 1–12 (2005).
Lenski, R. E. & Travisano, M. Dynamics of adaptation and diversification: a 10,000-generation experiment with bacterial-populations. Proc. Natl Acad. Sci. USA 91, 6808–6814 (1994).
Rainey, P. B. & Travisano, M. Adaptive radiation in a heterogeneous environment. Nature 394, 69–72 (1998).
Manne, L. L., Brooks, T. M. & Pimm, S. L. Relative risk of extinction of passerine birds on continents and islands. Nature 399, 258–261 (1999).
Purvis, A., Gittleman, J. L., Cowlishaw, G. & Mace, G. M. Predicting extinction risk in declining species. Proc. R. Soc. Lond. B Biol. Sci. 267, 1947–1952 (2000).
Lawton, J. H. Range, Population abundance and conservation. Trends Ecol. Evol. 8, 409–413 (1993).
Silva, M. & Downing, J. A. Allometric scaling of minimal mammal densities. Conserv. Biol. 8, 732–743 (1994).
Meretsky, V. J., Snyder, N. F. R., Beissinger, S. R., Clendenen, D. A. & Wiley, J. W. Demography of the California condor: implications for reestablishment. Conserv. Biol. 14, 957–967 (2000).
Finlay, B. J. & Clarke, K. J. Apparent global ubiquity of species in the protist genus Paraphysomonas. Protist 150, 419–430 (1999).
Massana, R., DeLong, E. F. & Pedrós-Alió, C. A few cosmopolitan phylotypes dominate planktonic archaeal assemblages in widely different oceanic provinces. Appl. Environ. Microbiol. 66, 1777–1787 (2000).
Lindström, E. S. & Leskinen, E. Do neighboring lakes share common taxa of bacterioplankton? Comparison of 16S rDNA fingerprints and sequences from three geographic regions. Microb. Ecol. 44, 1–9 (2002).
Brown, J. H., Stevens, G. C. & Kaufman, D. M. The geographic range: size, shape, boundaries, and internal structure. Annu. Rev. Ecol. Syst. 27, 597–623 (1996).
May, R. M. How many species are there on Earth? Science 241, 1441–1449 (1988).
Gaston, K. J., Chown, S. L. & Mercer, R. D. The animal species-body size distribution of Marion Island. Proc. Natl Acad. Sci. USA 98, 14493–14496 (2001).
Dykhuizen, D. E. Santa Rosalia revisited: why are there so many species of bacteria? Antonie Van Leeuwenhoek 73, 25–33 (1998).
Avise, J. C. & Aquadro, C. F. A comparative summary of genetic distances in the vertebrates. Evol. Biol. 15, 151–185 (1982).
de Vargas, C., Norris, R., Zaninetti, L., Gibb, S. W. & Pawlowski, J. Molecular evidence of cryptic speciation in planktonic foraminifers and their relation to oceanic provinces. Proc. Natl Acad. Sci. USA 96, 2864–2868 (1999).
Cohan, F. M. What are bacterial species? Annu. Rev. Microbiol. 56, 457–487 (2002).
Coyne, J. A., Orr, H. A. & Futuyma, D. J. Do we need a new species concept? Syst. Zool. 37, 190–200 (1988).
Green, J. L. & Bohannan, B. J. M. Spatial scaling of microbial biodiversity in Scaling Biodiversity (eds Storch, D. & Marquet, P. A. & Brown, J. H.) (Cambridge University Press, Cambridge, 2006).
Martiny, J. B. H. & Field, D. Ecological perspectives on the sequenced genome collection. Ecol. Lett. (in the press).
Naeem, S., Thompson, L. J., Lawler, S. P., Lawton, J. H. & Woodfin, R. M. Declining biodiversity can alter the performance of ecosystems. Nature 368, 734–737 (1994).
Hooper, D. U. &. Vitousek, P. M. The effects of plant composition and diversity on ecosystem processes. Science 277, 1302–1305 (1997).
Dukes, J. S. Biodiversity and invasibility in grassland microcosms. Oecologia 126, 563–568 (2001).
Treseder, K. K. & Vitousek, P. M. Potential ecosystem-level effects of genetic variation among populations of Metrosideros polymorpha from a soil fertility gradient in Hawaii. Oecologia 126, 266–275 (2001).
McGrady-Steed, J., Harris, P. M. & Morin, P. J. Biodiversity regulates ecosystem predictability. Nature 390, 162–165 (1997).
Bell, T., Newman, J. A., Silverman, B. W., Turner, S. L. & Lilley, A. K. The contribution of species richness and composition to bacterial services. Nature 436, 1157–1160 (2005).
Naeem, S. & Li, S. B. Biodiversity enhances ecosystem reliability. Nature 390, 507–509 (1997).
van der Heijden, M. G. A. et al. Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396, 69–72 (1998).
Cavigelli, M. A. & Robertson, G. P. The functional significance of denitrifier community composition in a terrestrial ecosystem. Ecology 81, 1402–1414 (2000).
Horz, H. -P., Barbrook, A., Field, C. B. & Bohannan, B. J. M. Ammonia-oxidizing bacteria respond to multifactorial global change. Proc. Natl Acad. Sci. USA 101, 15136–15141 (2004).
Bull, A. T., ed. Microbial Diversity and Bioprospecting. (ASM Press, Washington DC, 2003).
Finlay, B. J., Esteban, G. F., Olmo, J. L. & Tyler, P. A. Global distribution of free-living microbial species. Ecography 22, 138–144 (1999).
Magurran, A. E. Ecological Diversity and Its Measurement (Princeton University Press, Princeton, 1988).
Chao, A., Chazdon, R. L., Colwell, R. K. & Shen, T. J. A new statistical approach for assessing similarity of species composition with incidence and abundance data. Ecol. Lett. 8, 148–159 (2005).
Ricketts, T. H. The matrix matters: effective isolation in fragmented landscapes. Am. Nat. 158, 87–99 (2001).
Mantel, N. The detection of disease clustering and a generalized regression approach. Cancer Res. 27, 209–220 (1967).
Borcard, D., Legendre, P. & Drapeau, P. Partialling out the spatial component of ecological variation. Ecology 73, 1045–1055 (1992).
Smouse, P. E., Long, J. C. & Sokal, R. R. Multiple regression and correlation extensions of the Mantel test of matrix correspondence. Syst. Zool. 35, 627–632 (1986).
Fulthorpe, R. R., Rhodes, A. N. & Tiedje, J. M. High levels of endemicity of 3-chlorobenzoate-degrading soil bacteria. Appl. Environ. Microbiol. 64, 1620–1627 (1998).
Schwalbach, M. S. & Fuhrman, J. A. Wide-ranging abundances of aerobic anoxygenic phototrophic bacteria in the world ocean revealed by epifluorescence microscopy and quantitative PCR. Limnol. Oceanogr. 50, 620–628 (2005).
Garcia-Martinez, J. & Rodriguez-Valera, F. Microdiversity of uncultured marine prokaryotes: the SAR11 cluster and the marine Archaea of Group I. Mol. Ecol. 9, 935–948 (2000).
Glaeser, J. & Overmann, J. Biogeography, evolution, and diversity of epibionts in phototrophic consortia. Appl. Environ. Microbiol. 70, 4821–4830 (2004).
Yeager, C. M. et al. Diazotrophic community structure and function in two successional stages of biological soil crusts from the Colorado plateau and Chihuahuan desert. Appl. Environ. Microbiol. 70, 973–983 (2004).
Sliwinski, M. K. & Goodman, R. M. Spatial heterogeneity of Crenarchaeal assemblages within mesophilic soil ecosystems as revealed by PCR-single-stranded conformation polymorphism profiling. Appl. Environ. Microbiol. 70, 1811–1820 (2004).
Sliwinski, M. K. & Goodman, R. M. Comparison of Crenarchaeal consortia inhabiting the rhizosphere of diverse terrestrial plants with those in bulk soil in native environments. Appl. Environ. Microbiol. 70, 1821–1826 (2004).
Riemann, L. & Middelboe, M. Stability of bacterial and viral community compositions in Danish coastal waters as depicted by DNA fingerprinting techniques. Aquat. Microb. Ecol. 27, 219–232 (2002).
Pinhassi, J. et al. Spatial variability in bacterioplankton community composition at the Skagerrak–Kattegat Front. Mar. Ecol. Prog. Ser. 255, 1–13 (2003).
Troussellier, M. et al. Bacterial activity and genetic richness along an estuarine gradient (Rhone River plume, France). Aquat. Microb. Ecol. 28, 13–24 (2002).
Casamayor, E. O. et al. Changes in archaeal, bacterial and eukaryal assemblages along a salinity gradient by comparison of genetic fingerprinting methods in a multipond solar saltern. Environ. Microbiol. 4, 338–348 (2002).
McArthur, J. V., Kovacic, D. A. & Smith, M. H. Genetic diversity in natural populations of a soil bacterium across a landscape gradient. Proc. Natl Acad. Sci. USA 85, 9621–9624 (1988).
Buckley, D. H. & Schmidt, T. M. Diversity and dynamics of microbial communities in soils from agro-ecosystems. Environ. Microbiol. 5, 441–452 (2003).
Staddon, W. J., Trevors, J. T., Duchesne, L. C. & Colombo, C. A. Soil microbial diversity and community structure across a climatic gradient in western Canada. Biodivers. Conserv. 7, 1081–1092 (1998).
Franklin, R. B., Taylor, D. R. & Mills, A. L. The distribution of microbial communities in anaerobic and aerobic zones of a shallow coastal plain aquifer. Microb. Ecol. 38, 377–386 (1999).
Franklin, R. B. & Mills, A. L. Multi-scale variation in spatial heterogeneity for microbial community structure in an eastern Virginia agricultural field. FEMS Microbiol. Ecol. 44, 335–346 (2003).
Martiny, A. C., Jorgensen, T. M., Albrechtsen, H. J., Arvin, E. & Molin, S. Long-term succession of structure and diversity of a biofilm formed in a model drinking water distribution system. Appl. Environ. Microbiol. 69, 6899–6907 (2003).
Franklin, R. B., Blum, L. K., McComb, A. C. & Mills, A. L. A geostatistical analysis of small-scale spatial variability in bacterial abundance and community structure in salt marsh creek bank sediments. FEMS Microbiol. Ecol. 42, 71–80 (2002).
Rohwer, F., Seguritan, V., Azam, F. & Knowlton, N. Diversity and distribution of coral-associated bacteria. Mar. Ecol. Prog. Ser. 243, 1–10 (2002).
Yannarell, A. C. & Triplett, E. W. Geographic and environmental sources of variation in lake bacterial community composition. Appl. Environ. Microbiol. 71, 227–239 (2005).
Hewson, I. & Fuhrman, J. A. Richness and diversity of bacterioplankton species along an estuarine gradient in Moreton Bay, Australia. Appl. Environ. Microbiol. 70, 3425–3433 (2004).
Acknowledgements
This work was conducted as part of the Patterns in Microbial Biodiversity Working Group supported by the National Center for Ecological Analysis and Synthesis, a centre funded by the National Science Foundation, the University of California at Santa Barbara and the State of California. We thank M. Liebold, G. Muyzer, O. Petchey and D. Ward for useful and lively discussions, and C. van der Gast for comments on the manuscript. Any opinions, findings and conclusions or recommendations expressed in this study are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Province
-
A region the biotic composition of which reflects the legacies of historical events.
- Habitat type
-
An environment defined by the suite of its abiotic and biotic characteristics.
- Beta diversity
-
Taxonomic diversity due to turnover in composition between assemblages.
- Distance effect
-
The influence of isolation on biotic composition after controlling for the influence of the contemporary environment.
- Genetic drift
-
Changes in gene frequencies in a population caused solely by chance.
- Allometry
-
The relationship between organismal attributes and body size of the form Y = Y0 Mb, in which Y is a variable such as metabolic rate, lifespan or population density, Y0 is a normalization constant (the y-intercept on a logarithmic graph), M is body mass (or other measure of body size) and b is the scaling exponent (the slope on the graph).
- Ecological drift
-
The influence of random demographic variability (such as birth, death and migration rates) on biotic composition.
- Propagule
-
The smallest unit of dispersal that is necessary to colonize a new population.
- Geographic range
-
The area encompassing the extent of a taxon's distribution.
Rights and permissions
About this article
Cite this article
Martiny, J., Bohannan, B., Brown, J. et al. Microbial biogeography: putting microorganisms on the map. Nat Rev Microbiol 4, 102–112 (2006). https://doi.org/10.1038/nrmicro1341
Issue Date:
DOI: https://doi.org/10.1038/nrmicro1341
This article is cited by
-
Biotic homogenization, lower soil fungal diversity and fewer rare taxa in arable soils across Europe
Nature Communications (2024)
-
Functional and taxonomic approaches differently highlight local and spatial processes in phytoplankton metacommunities
Hydrobiologia (2024)
-
Variation Among Japanese Miso Breweries in Indoor Microbiomes is Mainly Ascribed to Variation in Type of Indoor Surface
Current Microbiology (2024)
-
Response of particle-attached and free-living bacterial communities to Microcystis blooms
Applied Microbiology and Biotechnology (2024)
-
Beta diversity of freshwater algal communities: influence of different dispersal mechanisms
Aquatic Sciences (2024)