Key Points
-
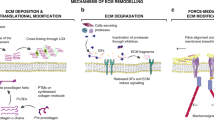
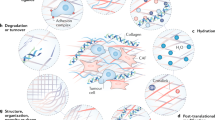
The extracellular matrix (ECM) is a dynamic structure that is constantly remodelled to control tissue homeostasis.
-
The ECM in mammals is composed of around 300 proteins, known as the core matrisome.
-
Metalloproteinases are the main endopeptidases responsible for ECM degradation. These enzymes can also generate ECM fragments with different bioactive properties than their full-length proteins. These matrikines regulate many processes such as migration, adhesion and differentiation.
-
ECM remodelling is important during organogenesis and development of the intestine, mammary and salivary glands and lung.
-
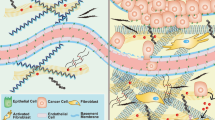
Dysregulation of the ECM composition and structure and mutations in genes that affect ECM remodeling can lead to several severe human conditions, including fibrosis and cancer.
-
ECM components and the proteins that regulate ECM remodelling represent promising therapeutic targets. Additional preclinical and clinical studies are required to fully understand the role of the ECM in human disease.
Abstract
The extracellular matrix (ECM) is a highly dynamic structure that is present in all tissues and continuously undergoes controlled remodelling. This process involves quantitative and qualitative changes in the ECM, mediated by specific enzymes that are responsible for ECM degradation, such as metalloproteinases. The ECM interacts with cells to regulate diverse functions, including proliferation, migration and differentiation. ECM remodelling is crucial for regulating the morphogenesis of the intestine and lungs, as well as of the mammary and submandibular glands. Dysregulation of ECM composition, structure, stiffness and abundance contributes to several pathological conditions, such as fibrosis and invasive cancer. A better understanding of how the ECM regulates organ structure and function and of how ECM remodelling affects disease progression will contribute to the development of new therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hynes, R. O. The extracellular matrix: not just pretty fibrils. Science 326, 1216–1219 (2009).
Jarvelainen, H., Sainio, A., Koulu, M., Wight, T. N. & Penttinen, R. Extracellular matrix molecules: potential targets in pharmacotherapy. Pharmacol. Rev. 61, 198–223 (2009).
Bateman, J. F., Boot-Handford, R. P. & Lamande, S. R. Genetic diseases of connective tissues: cellular and extracellular effects of ECM mutations. Nature Rev. Genet. 10, 173–183 (2009).
Rozario, T. & DeSimone, D. W. The extracellular matrix in development and morphogenesis: a dynamic view. Dev. Biol. 341, 126–140 (2010).
Hynes, R. O. & Naba, A. Overview of the matrisome—an inventory of extracellular matrix constituents and functions. Cold Spring Harb. Perspect. Biol. 4, a004903 (2012). This review gives a complete list of ECM proteins that are part of the matrisome, and describes the ECM structure and function modifiers and the evolution of the matrisome.
Lu, P., Takai, K., Weaver, V. M. & Werb, Z. Extracellular matrix degradation and remodeling in development and disease. Cold Spring Harb. Perspect. Biol. 3, a005058 (2011).
Frantz, C., Stewart, K. M. & Weaver, V. M. The extracellular matrix at a glance. J. Cell Sci. 123, 4195–4200 (2010).
Zhen, G. & Cao, X. Targeting TGFβ signaling in subchondral bone and articular cartilage homeostasis. Trends Pharmacol. Sci. 35, 227–236 (2014).
Gross, J. & Lapiere, C. M. Collagenolytic activity in amphibian tissues: a tissue culture assay. Proc. Natl Acad. Sci. USA 48, 1014–1022 (1962).
Hite, L. A., Shannon, J. D., Bjarnason, J. B. & Fox, J. W. Sequence of a cDNA clone encoding the zinc metalloproteinase hemorrhagic toxin e from Crotalus atrox: evidence for signal, zymogen, and disintegrin-like structures. Biochemistry 31, 6203–6211 (1992).
Kuno, K. et al. Molecular cloning of a gene encoding a new type of metalloproteinase-disintegrin family protein with thrombospondin motifs as an inflammation associated gene. J. Biol. Chem. 272, 556–562 (1997).
Murphy, G. The ADAMs: signalling scissors in the tumour microenvironment. Nature Rev. Cancer 8, 929–941 (2008).
White, J. M. ADAMs: modulators of cell-cell and cell–matrix interactions. Curr. Opin. Cell Biol. 15, 598–606 (2003).
Apte, S. S. A disintegrin-like and metalloprotease (reprolysin-type) with thrombospondin type 1 motif (ADAMTS) superfamily: functions and mechanisms. J. Biol. Chem. 284, 31493–31497 (2009).
Bond, J. S., Rojas, K., Overhauser, J., Zoghbi, H. Y. & Jiang, W. The structural genes, MEP1A and MEP1B, for the α and β subunits of the metalloendopeptidase meprin map to human chromosomes 6p and 18q, respectively. Genomics 25, 300–303 (1995).
Bertenshaw, G. P., Norcum, M. T. & Bond, J. S. Structure of homo- and hetero-oligomeric meprin metalloproteases. Dimers, tetramers, and high molecular mass multimers. J. Biol. Chem. 278, 2522–2532 (2003).
Herzog, C., Haun, R. S., Ludwig, A., Shah, S. V. & Kaushal, G. P. ADAM10 is the major sheddase responsible for the release of membrane-associated meprin A. J. Biol. Chem. 289, 13308–13322 (2014).
Kruse, M. N. et al. Human meprin α and β homo-oligomers: cleavage of basement membrane proteins and sensitivity to metalloprotease inhibitors. Biochem. J. 378, 383–389 (2004).
Broder, C. et al. Metalloproteases meprinα and meprinβ are C- and N-procollagen proteinases important for collagen assembly and tensile strength. Proc. Natl Acad. Sci. USA 110, 14219–14224 (2013).
Jefferson, T. et al. The substrate degradome of meprin metalloproteases reveals an unexpected proteolytic link between meprin β and ADAM10. Cell. Mol. Life Sci. 70, 309–333 (2013).
Geurts, N. et al. Meprins process matrix metalloproteinase-9 (MMP-9)/gelatinase B and enhance the activation kinetics by MMP-3. FEBS Lett. 586, 4264–4269 (2012).
Khokha, R., Murthy, A. & Weiss, A. Metalloproteinases and their natural inhibitors in inflammation and immunity. Nature Rev. Immunol. 13, 649–665 (2013).
Baker, A. H., Edwards, D. R. & Murphy, G. Metalloproteinase inhibitors: biological actions and therapeutic opportunities. J. Cell Sci. 115, 3719–3727 (2002).
Smith, H. W. & Marshall, C. J. Regulation of cell signalling by uPAR. Nature Rev. Mol. Cell. Biol. 11, 23–36 (2010).
Bonnefoy, A. & Legrand, C. Proteolysis of subendothelial adhesive glycoproteins (fibronectin, thrombospondin, and von Willebrand factor) by plasmin, leukocyte cathepsin G, and elastase. Thromb. Res. 98, 323–332 (2000).
Giuffrida, P., Biancheri, P. & MacDonald, T. T. Proteases and small intestinal barrier function in health and disease. Curr. Opin. Gastroenterol. 30, 147–153 (2014).
Mohamed, M. M. & Sloane, B. F. Cysteine cathepsins: multifunctional enzymes in cancer. Nature Rev. Cancer 6, 764–775 (2006).
Fonovic, M. & Turk, B. Cysteine cathepsins and extracellular matrix degradation. Biochim. Biophys. Acta 1840, 2560–2570 (2014).
Uchimura, K. et al. HSulf-2, an extracellular endoglucosamine-6-sulfatase, selectively mobilizes heparin-bound growth factors and chemokines: effects on VEGF, FGF-1, and SDF-1. BMC Biochem. 7, 2 (2006).
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007).
Hasebe, T. et al. Thyroid hormone-induced cell–cell interactions are required for the development of adult intestinal stem cells. Cell Biosci. 3, 18 (2013).
Su, Y., Shi, Y., Stolow, M. A. & Shi, Y. B. Thyroid hormone induces apoptosis in primary cell cultures of tadpole intestine: cell type specificity and effects of extracellular matrix. J. Cell Biol. 139, 1533–1543 (1997).
Patterton, D., Hayes, W. P. & Shi, Y. B. Transcriptional activation of the matrix metalloproteinase gene stromelysin-3 coincides with thyroid hormone-induced cell death during frog metamorphosis. Dev. Biol. 167, 252–262 (1995).
Ishizuya-Oka, A. et al. Requirement for matrix metalloproteinase stromelysin-3 in cell migration and apoptosis during tissue remodeling in Xenopus laevis. J. Cell Biol. 150, 1177–1188 (2000). This paper showed that MMP11 is required for cell fate determination and cell migration during morphogenesis, most probably through ECM remodelling.
Amano, T., Kwak, O., Fu, L., Marshak, A. & Shi, Y. B. The matrix metalloproteinase stromelysin-3 cleaves laminin receptor at two distinct sites between the transmembrane domain and laminin binding sequence within the extracellular domain. Cell Res. 15, 150–159 (2005).
Fujimoto, K., Nakajima, K. & Yaoita, Y. Expression of matrix metalloproteinase genes in regressing or remodeling organs during amphibian metamorphosis. Dev. Growth Differ. 49, 131–143 (2007).
Hasebe, T., Hartman, R., Fu, L., Amano, T. & Shi, Y. B. Evidence for a cooperative role of gelatinase A and membrane type-1 matrix metalloproteinase during Xenopus laevis development. Mech. Dev. 124, 11–22 (2007).
Simon-Assmann, P., Kedinger, M., De Arcangelis, A., Rousseau, V. & Simo, P. Extracellular matrix components in intestinal development. Experientia 51, 883–900 (1995).
Simon-Assmann, P., Kedinger, M. & Haffen, K. Immunocytochemical localization of extracellular-matrix proteins in relation to rat intestinal morphogenesis. Differentiation 32, 59–66 (1986).
Simon-Assmann, P., Bouziges, F., Freund, J. N., Perrin-Schmitt, F. & Kedinger, M. Type IV collagen mRNA accumulates in the mesenchymal compartment at early stages of murine developing intestine. J. Cell Biol. 110, 849–857 (1990).
Simon-Assmann, P. et al. The laminins: role in intestinal morphogenesis and differentiation. Ann. NY Acad. Sci. 859, 46–64 (1998).
Mahoney, Z. X., Stappenbeck, T. S. & Miner, J. H. Laminin α 5 influences the architecture of the mouse small intestine mucosa. J. Cell Sci. 121, 2493–2502 (2008).
Beaulieu, J. F. Integrins and human intestinal cell functions. Front. Biosci. 4, D310–D321 (1999).
Benoit, Y. D. et al. Integrin α8β1 regulates adhesion, migration and proliferation of human intestinal crypt cells via a predominant RhoA/ROCK-dependent mechanism. Biol. Cell 101, 695–708 (2009).
Benoit, Y. D. et al. Integrin α8β1 confers anoikis susceptibility to human intestinal epithelial crypt cells. Biochem. Biophys. Res. Commun. 399, 434–439 (2010).
Groulx, J. F. et al. Collagen VI is a basement membrane component that regulates epithelial cell-fibronectin interactions. Matrix Biol. 30, 195–206 (2011).
Henning, S. J. et al. Meprin mRNA in rat intestine during normal and glucocorticoid-induced maturation: divergent patterns of expression of α and β subunits. FEBS Lett. 462, 368–372 (1999).
Sato, T. & Clevers, H. Growing self-organizing mini-guts from a single intestinal stem cell: mechanism and applications. Science 340, 1190–1194 (2013).
Kim, H. Y. & Nelson, C. M. Extracellular matrix and cytoskeletal dynamics during branching morphogenesis. Organogenesis 8, 56–64 (2012).
Grobstein, C. & Cohen, J. Collagenase: effect on the morphogenesis of embryonic salivary epithelium in vitro. Science 150, 626–628 (1965).
Nakanishi, Y., Sugiura, F., Kishi, J. & Hayakawa, T. Collagenase inhibitor stimulates cleft formation during early morphogenesis of mouse salivary gland. Dev. Biol. 113, 201–206 (1986).
Keely, P. J., Wu, J. E. & Santoro, S. A. The spatial and temporal expression of the α 2 β 1 integrin and its ligands, collagen I, collagen IV, and laminin, suggest important roles in mouse mammary morphogenesis. Differentiation 59, 1–13 (1995).
Brownfield, D. G. et al. Patterned collagen fibers orient branching mammary epithelium through distinct signaling modules. Curr. Biol. 23, 703–709 (2013).
Sakai, T., Larsen, M. & Yamada, K. M. Fibronectin requirement in branching morphogenesis. Nature 423, 876–881 (2003). This paper is the first to show that fibronectin expression is required for cleft formation in branching morphogenesis.
Onodera, T. et al. Btbd7 regulates epithelial cell dynamics and branching morphogenesis. Science 329, 562–565 (2010). This paper is the first to give molecular mechanisms for cleft formation in branching morphogenesis, showing that BTBD7 is a regulatory gene that promotes epithelial tissue remodelling and formation of branched organs.
Daley, W. P., Gulfo, K. M., Sequeira, S. J. & Larsen, M. Identification of a mechanochemical checkpoint and negative feedback loop regulating branching morphogenesis. Dev. Biol. 336, 169–182 (2009).
Daley, W. P., Kohn, J. M. & Larsen, M. A focal adhesion protein-based mechanochemical checkpoint regulates cleft progression during branching morphogenesis. Dev. Dyn. 240, 2069–2083 (2011).
Joo, E. E. & Yamada, K. M. MYPT1 regulates contractility and microtubule acetylation to modulate integrin adhesions and matrix assembly. Nature Commun. 5, 3510 (2014).
Woodward, T. L., Mienaltowski, A. S., Modi, R. R., Bennett, J. M. & Haslam, S. Z. Fibronectin and the α5β1 integrin are under developmental and ovarian steroid regulation in the normal mouse mammary gland. Endocrinology 142, 3214–3222 (2001).
Liu, S., Young, S. M. & Varisco, B. M. Dynamic expression of chymotrypsin-like elastase 1 over the course of murine lung development. Am. J. Physiol. Lung Cell. Mol. Physiol. 306, L1104–L1116 (2014).
Mammoto, T., Jiang, E., Jiang, A. & Mammoto, A. Extracellular matrix structure and tissue stiffness control postnatal lung development through the lipoprotein receptor-related protein 5/Tie2 signaling system. Am. J. Respir. Cell. Mol. Biol. 49, 1009–1018 (2013).
Daley, W. P. & Yamada, K. M. ECM-modulated cellular dynamics as a driving force for tissue morphogenesis. Curr. Opin. Genet. Dev. 23, 408–414 (2013).
Tan, J. et al. Stromal matrix metalloproteinase-11 is involved in the mammary gland postnatal development. Oncogene 33, 4050–4059 (2013).
Mori, H. et al. Transmembrane/cytoplasmic, rather than catalytic, domains of Mmp14 signal to MAPK activation and mammary branching morphogenesis via binding to integrin β1. Development 140, 343–352 (2013).
Wiseman, B. S. et al. Site-specific inductive and inhibitory activities of MMP-2 and MMP-3 orchestrate mammary gland branching morphogenesis. J. Cell Biol. 162, 1123–1133 (2003).
Kessenbrock, K. et al. A role for matrix metalloproteinases in regulating mammary stem cell function via the Wnt signaling pathway. Cell Stem Cell 13, 300–313 (2013). This paper shows that MMP3 is a regulator of WNT signaling and mammary stem cell activity; through its haemopexin domain, MMP3 specifically binds and inactivates WNT5B, a noncanonical WNT ligand that inhibits canonical WNT signalling and mammary epithelial outgrowth in vivo.
Correia, A. L., Mori, H., Chen, E. I., Schmitt, F. C. & Bissell, M. J. The hemopexin domain of MMP3 is responsible for mammary epithelial invasion and morphogenesis through extracellular interaction with HSP90β. Genes Dev. 27, 805–817 (2013). The authors show that the haemopexin domain of MMP3, and not its proteolytic activity, is essential for mammary epithelial cells invasion; this domain interacts with the intracellular chaperone heat-shock protein 90β to promote invasion.
Goetz, R. & Mohammadi, M. Exploring mechanisms of FGF signalling through the lens of structural biology. Nature Rev. Mol. Cell. Biol. 14, 166–180 (2013).
Makarenkova, H. P. et al. Differential interactions of FGFs with heparan sulfate control gradient formation and branching morphogenesis. Sci. Signal. 2, ra55 (2009).
Patel, V. N. et al. Heparanase cleavage of perlecan heparan sulfate modulates FGF10 activity during ex vivo submandibular gland branching morphogenesis. Development 134, 4177–4186 (2007).
Kim, H. E. et al. Disruption of the myocardial extracellular matrix leads to cardiac dysfunction. J. Clin. Invest. 106, 857–866 (2000).
Bondeson, J., Wainwright, S., Hughes, C. & Caterson, B. The regulation of the ADAMTS4 and ADAMTS5 aggrecanases in osteoarthritis: a review. Clin. Exp. Rheumatol. 26, 139–145 (2008).
Echtermeyer, F. et al. Syndecan-4 regulates ADAMTS-5 activation and cartilage breakdown in osteoarthritis. Nature Med. 15, 1072–1076 (2009).
Martignetti, J. A. et al. Mutation of the matrix metalloproteinase 2 gene (MMP2) causes a multicentric osteolysis and arthritis syndrome. Nature Genet. 28, 261–265 (2001).
El-Serag, H. B. Hepatocellular carcinoma. N. Engl. J. Med. 365, 1118–1127 (2011).
Boyd, N. F., Martin, L. J., Yaffe, M. J. & Minkin, S. Mammographic density and breast cancer risk: current understanding and future prospects. Breast Cancer Res. 13, 223 (2011).
Biancheri, P. et al. The role of transforming growth factor (TGF)-β in modulating the immune response and fibrogenesis in the gut. Cytokine Growth Factor Rev. 25, 45–55 (2013).
Verrecchia, F., Chu, M. L. & Mauviel, A. Identification of novel TGF-β /Smad gene targets in dermal fibroblasts using a combined cDNA microarray/promoter transactivation approach. J. Biol. Chem. 276, 17058–17062 (2001).
Wynn, T. A. & Ramalingam, T. R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nature Med. 18, 1028–1040 (2012). This review summarizes our current understanding of the molecular and cellular pathways controlling fibrosis, with special focus on the immune response, as well as the key pathways for therapeutic targeting.
Giannandrea, M. & Parks, W. C. Diverse functions of matrix metalloproteinases during fibrosis. Dis. Model. Mech. 7, 193–203 (2014).
Duffield, J. S., Lupher, M., Thannickal, V. J. & Wynn, T. A. Host responses in tissue repair and fibrosis. Annu. Rev. Pathol. 8, 241–276 (2013).
McHedlidze, T. et al. Interleukin-33-dependent innate lymphoid cells mediate hepatic fibrosis. Immunity 39, 357–371 (2013).
Bailey, J. R. et al. IL-13 promotes collagen accumulation in Crohn's disease fibrosis by down-regulation of fibroblast MMP synthesis: a role for innate lymphoid cells? PLoS ONE 7, e52332 (2013).
Clarke, D. L., Carruthers, A. M., Mustelin, T. & Murray, L. A. Matrix regulation of idiopathic pulmonary fibrosis: the role of enzymes. Fibrogen. Tissue Repair 6, 20 (2013).
Rieder, F. The gut microbiome in intestinal fibrosis: environmental protector or provocateur? Sci. Transl. Med. 5, 190ps10 (2013).
Parker, M. W. et al. Fibrotic extracellular matrix activates a profibrotic positive feedback loop. J. Clin. Invest. 124, 1622–1635 (2014). This paper demonstrates that fibrotic ECM stimulates fibroblasts to produce more ECM through downregulation of miR-29.
Bhattacharyya, S. et al. FibronectinEDA promotes chronic cutaneous fibrosis through Toll-like receptor signaling. Sci. Transl. Med. 6, 232ra50 (2014).
Ding, N. et al. A vitamin D receptor/SMAD genomic circuit gates hepatic fibrotic response. Cell 153, 601–613 (2013).
Chan, M. F. et al. Protective effects of matrix metalloproteinase-12 following corneal injury. J. Cell Sci. 126, 3948–3960 (2013).
Jiang, D. et al. Inhibition of pulmonary fibrosis in mice by CXCL10 requires glycosaminoglycan binding and syndecan-4. J. Clin. Invest. 120, 2049–2057 (2010).
Ding, B. S. et al. Divergent angiocrine signals from vascular niche balance liver regeneration and fibrosis. Nature 505, 97–102 (2014).
Atabai, K. et al. Mfge8 diminishes the severity of tissue fibrosis in mice by binding and targeting collagen for uptake by macrophages. J. Clin. Invest. 119, 3713–3722 (2009).
Bissell, M. J. & Hines, W. C. Why don't we get more cancer? A proposed role of the microenvironment in restraining cancer progression. Nature Med. 17, 320–329 (2011).
Tian, X. et al. High-molecular-mass hyaluronan mediates the cancer resistance of the naked mole rat. Nature 499, 346–349 (2013).
Lu, P., Weaver, V. M. & Werb, Z. The extracellular matrix: a dynamic niche in cancer progression. J. Cell Biol. 196, 395–406 (2012).
Burnier, J. V. et al. Type IV collagen-initiated signals provide survival and growth cues required for liver metastasis. Oncogene 30, 3766–3783 (2011).
Bergamaschi, A. et al. Extracellular matrix signature identifies breast cancer subgroups with different clinical outcome. J. Pathol. 214, 357–367 (2008).
Poola, I. et al. Identification of MMP-1 as a putative breast cancer predictive marker by global gene expression analysis. Nature Med. 11, 481–483 (2005).
Paszek, M. J. et al. Tensional homeostasis and the malignant phenotype. Cancer Cell 8, 241–254 (2005).
Erler, J. T. et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature 440, 1222–1226 (2006).
Levental, K. R. et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139, 891–906 (2009). This study describes how ECM stiffening through LOX drives tumour progression and shows that inhibiting collagen crosslinking delays tumorigenesis.
Erler, J. T. et al. Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell 15, 35–44 (2009).
Mouw, J. K. et al. Tissue mechanics modulate microRNA-dependent PTEN expression to regulate malignant progression. Nature Med. 20, 360–367 (2014).
Valiathan, R. R., Marco, M., Leitinger, B., Kleer, C. G. & Fridman, R. Discoidin domain receptor tyrosine kinases: new players in cancer progression. Cancer Metastasis Rev. 31, 295–321 (2012).
Hidalgo-Carcedo, C. et al. Collective cell migration requires suppression of actomyosin at cell-cell contacts mediated by DDR1 and the cell polarity regulators Par3 and Par6. Nature Cell Biol. 13, 49–58 (2011).
Zhang, K. et al. The collagen receptor discoidin domain receptor 2 stabilizes SNAIL1 to facilitate breast cancer metastasis. Nature Cell Biol. 15, 677–687 (2013).
Sengupta, S. et al. MicroRNA 29c is down-regulated in nasopharyngeal carcinomas, up-regulating mRNAs encoding extracellular matrix proteins. Proc. Natl Acad. Sci. USA 105, 5874–5878 (2008).
Chou, J. et al. GATA3 suppresses metastasis and modulates the tumour microenvironment by regulating microRNA-29b expression. Nature Cell Biol. 15, 201–213 (2013).
Enerly, E. et al. miRNA-mRNA integrated analysis reveals roles for miRNAs in primary breast tumors. PLoS ONE 6, e16915 (2011).
Cheresh, D. A. & Stupack, D. G. Regulation of angiogenesis: apoptotic cues from the ECM. Oncogene 27, 6285–6298 (2008).
Saupe, F. et al. Tenascin-C downregulates wnt inhibitor dickkopf-1, promoting tumorigenesis in a neuroendocrine tumor model. Cell Rep. 5, 482–492 (2013).
Mott, J. D. & Werb, Z. Regulation of matrix biology by matrix metalloproteinases. Curr. Opin. Cell Biol. 16, 558–564 (2004).
Burgess, J. K. & Weckmann, M. Matrikines and the lungs. Pharmacol. Ther. 134, 317–337 (2012).
Sudhakar, A. et al. Human α1 type IV collagen NC1 domain exhibits distinct antiangiogenic activity mediated by α1β1 integrin. J. Clin. Invest. 115, 2801–2810 (2005).
Ray, S. J. et al. The collagen binding α1β1 integrin VLA-1 regulates CD8 T cell-mediated immune protection against heterologous influenza infection. Immunity 20, 167–179 (2004).
Song, J. et al. Extracellular matrix of secondary lymphoid organs impacts on B-cell fate and survival. Proc. Natl Acad. Sci. USA 110, E2915–E2924 (2013).
Sorokin, L. The impact of the extracellular matrix on inflammation. Nature Rev. Immunol. 10, 712–723 (2010).
Monboisse, J. C., Oudart, J. B., Ramont, L., Brassart-Pasco, S. & Maquart, F. X. Matrikines from basement membrane collagens: a new anti-cancer strategy. Biochim. Biophys. Acta 1840, 2589–2598 (2014). This review summarizes the involvement of collagen-derived matrikines in cancer, particularly matrikines derived from the NC1 domains of the different constitutive chains of basement membrane-associated collagens, mainly collagen IV.
Gaggar, A. et al. A novel proteolytic cascade generates an extracellular matrix-derived chemoattractant in chronic neutrophilic inflammation. J. Immunol. 180, 5662–5669 (2008).
Weathington, N. M. et al. A novel peptide CXCR ligand derived from extracellular matrix degradation during airway inflammation. Nature Med. 12, 317–323 (2006).
Muto, J. et al. Hyaluronan digestion controls DC migration from the skin. J. Clin. Invest. 124, 1309–1319 (2014).
Salmon, H. et al. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J. Clin. Invest. 122, 899–910 (2012).
Kim, S. et al. Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature 457, 102–106 (2009).
Wolf, K. & Friedl, P. Extracellular matrix determinants of proteolytic and non-proteolytic cell migration. Trends Cell Biol. 21, 736–744 (2011).
Tozluoglu, M. et al. Matrix geometry determines optimal cancer cell migration strategy and modulates response to interventions. Nature Cell Biol. 15, 751–762 (2013).
Bergers, G. et al. Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nature Cell Biol. 2, 737–744 (2000).
Lee, S., Jilani, S. M., Nikolova, G. V., Carpizo, D. & Iruela-Arispe, M. L. Processing of VEGF-A by matrix metalloproteinases regulates bioavailability and vascular patterning in tumors. J. Cell Biol. 169, 681–691 (2005).
Ferrara, N. Binding to the extracellular matrix and proteolytic processing: two key mechanisms regulating vascular endothelial growth factor action. Mol. Biol. Cell 21, 687–690 (2010).
Chen, T. T. et al. Anchorage of VEGF to the extracellular matrix conveys differential signaling responses to endothelial cells. J. Cell Biol. 188, 595–609 (2010).
Phillips, J. J. et al. Heparan sulfate sulfatase SULF2 regulates PDGFRα signaling and growth in human and mouse malignant glioma. J. Clin. Invest. 122, 911–922 (2012).
Grafe, I. et al. Excessive transforming growth factor-β signaling is a common mechanism in osteogenesis imperfecta. Nature Med. 20, 670–675 (2014).
Alcaraz, L. B. et al. Tenascin-X promotes epithelial-to-mesenchymal transition by activating latent TGF-β. J. Cell Biol. 205, 409–428 (2014).
Watt, F. M. & Huck, W. T. Role of the extracellular matrix in regulating stem cell fate. Nature Rev. Mol. Cell. Biol. 14, 467–473 (2013).
Debnath, J. & Brugge, J. S. Modelling glandular epithelial cancers in three-dimensional cultures. Nature Rev. Cancer 5, 675–688 (2005).
Ridky, T. W., Chow, J. M., Wong, D. J. & Khavari, P. A. Invasive three-dimensional organotypic neoplasia from multiple normal human epithelia. Nature Med. 16, 1450–1455 (2010).
Yurchenco, P. D. Basement membranes: cell scaffoldings and signaling platforms. Cold Spring Harb. Perspect. Biol. 3, a004911 (2011).
Seif-Naraghi, S. B. et al. Safety and efficacy of an injectable extracellular matrix hydrogel for treating myocardial infarction. Sci. Transl. Med. 5, 173ra25 (2013).
Lee, B. et al. Perlecan domain V is neuroprotective and proangiogenic following ischemic stroke in rodents. J. Clin. Invest. 121, 3005–3023 (2011).
Martino, M. M. et al. Growth factors engineered for super-affinity to the extracellular matrix enhance tissue healing. Science 343, 885–888 (2014).
Giger, R. J., Hollis, E. R. 2nd & Tuszynski, M. H. Guidance molecules in axon regeneration. Cold Spring Harb. Perspect. Biol. 2, a001867 (2010).
Busch, S. A. & Silver, J. The role of extracellular matrix in CNS regeneration. Curr. Opin. Neurobiol. 17, 120–127 (2007).
Alilain, W. J., Horn, K. P., Hu, H., Dick, T. E. & Silver, J. Functional regeneration of respiratory pathways after spinal cord injury. Nature 475, 196–200 (2011).
Back, S. A. et al. Hyaluronan accumulates in demyelinated lesions and inhibits oligodendrocyte progenitor maturation. Nature Med. 11, 966–972 (2005).
Barry-Hamilton, V. et al. Allosteric inhibition of lysyl oxidase-like-2 impedes the development of a pathologic microenvironment. Nature Med. 16, 1009–1017 (2010). This study demonstrates that inhibiting LOXL2 using a monoclonal antibody decreases fibrosis in models of lung and liver fibrosis and reduces metastasis in xenografted tumours.
Coussens, L. M., Fingleton, B. & Matrisian, L. M. Matrix metalloproteinase inhibitors and cancer: trials and tribulations. Science 295, 2387–2392 (2002).
Devy, L. & Dransfield, D. T. New strategies for the next generation of matrix-metalloproteinase inhibitors: selectively targeting membrane-anchored MMPs with therapeutic antibodies. Biochem. Res. Int. 2011, 191670 (2011).
Zeisberg, M. et al. Stage-specific action of matrix metalloproteinases influences progressive hereditary kidney disease. PLoS Med. 3, e100 (2006).
Junttila, M. R. & de Sauvage, F. J. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature 501, 346–354 (2013).
Miyamoto, H. et al. Tumor-stroma interaction of human pancreatic cancer: acquired resistance to anticancer drugs and proliferation regulation is dependent on extracellular matrix proteins. Pancreas 28, 38–44 (2004).
Sethi, T. et al. Extracellular matrix proteins protect small cell lung cancer cells against apoptosis: a mechanism for small cell lung cancer growth and drug resistance in vivo. Nature Med. 5, 662–668 (1999).
Eke, I., Storch, K., Krause, M. & Cordes, N. Cetuximab attenuates its cytotoxic and radiosensitizing potential by inducing fibronectin biosynthesis. Cancer Res. 73, 5869–5879 (2013).
Henderson, N. C. et al. Targeting of αv integrin identifies a core molecular pathway that regulates fibrosis in several organs. Nature Med. 19, 1617–1624 (2013).
Psaila, B. & Lyden, D. The metastatic niche: adapting the foreign soil. Nature Rev. Cancer 9, 285–293 (2009).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Oskarsson, T. et al. Breast cancer cells produce tenascin C as a metastatic niche component to colonize the lungs. Nature Med. 17, 867–874 (2011).
Malanchi, I. et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 481, 85–89 (2011). References 156 and 157 show that ECM components are crucial for providing proliferative, survival and stemness signals during tumour metastasis.
Ghajar, C. M. et al. The perivascular niche regulates breast tumour dormancy. Nature Cell Biol. 15, 807–817 (2013).
Pietras, A. et al. Osteopontin-CD44 signaling in the glioma perivascular niche enhances cancer stem cell phenotypes and promotes aggressive tumor growth. Cell Stem Cell 14, 357–369 (2014).
Reticker-Flynn, N. E. et al. A combinatorial extracellular matrix platform identifies cell-extracellular matrix interactions that correlate with metastasis. Nature Commun. 3, 1122 (2012).
Kessenbrock, K., Plaks, V. & Werb, Z. Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 141, 52–67 (2010).
Ishizuya-Oka, A. & Hasebe, T. Establishment of intestinal stem cell niche during amphibian metamorphosis. Curr. Top. Dev. Biol. 103, 305–327 (2013).
Acknowledgements
This study was supported by funds from the National Cancer Institute (grant numbers CA057621 and CA138818), a Department of Defense (DOD) Era of Hope Scholar Expansion grant (BC122990) and Institut National de la Santé et de la Recherche Médicale (INSERM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Basement membrane
-
The ECM layer that is located basolaterally to all epithelium and endothelium in the body. It provides structural support to the tissue and modulates epithelial and endothelial cell functions. It is mostly composed of collagen IV and laminins.
- TGFβ
-
(Transforming growth factor-β). The TGFβ isoforms (TGFβ1–TGFβ3) are synthesized as latent precursors in complex with both latency-associated peptide (LAP) and latent TGFβ-binding proteins (LTBPs). Proteinases such as plasmin and MMPs catalyse the release of active TGFβ from the complex.
- Zymogens
-
Proenzymes that are inactive enzyme precursors. Zymogens need to be biochemically modified to become active enzymes.
- Integrins
-
Heterodimeric cell surface receptors that mediate cell–cell and cell–ECM interactions and orchestrate cell attachment, movement, growth, differentiation and survival.
- von Willebrand factor
-
A blood glycoprotein that is involved in haemostasis and is defective in von Willebrand disease. This factor is also increased in the plasma of cardiovascular, neoplastic and connective tissue diseases and can contribute to an increased risk of thrombosis.
- Villi
-
A finger-like projection that protrudes from the epithelium of the intestinal wall. Intestinal villi increase the epithelium surface area to promote the absorption of nutrients.
- Intestinal crypts
-
(Also known as crypts of Lieberkühn). Proliferative compartments found in the small intestine and the colon; they contain intestinal stem cells and other specialized cells such as Paneth cells (only in the small intestine) and goblet cells.
- Intestinal stem cells
-
Undifferentiated cells that can self-renew and differentiate into all of the specialized cell types of the tissue or organ. The main role of stem cells is to maintain and repair the tissue in which they reside.
- RGD-dependent integrins
-
A group of integrins that specifically recognize the RGD motif, a sequence of three amino acids (Arg-Gly-Asp) found in many ECM molecules such as fibronectin and osteopontin. Collectively, these interactions are termed the RGD-dependent adhesion system.
- Anoikis
-
A programmed cell death that is induced by lack of correct cell–ECM interactions. Invasive tumour cells may escape from anoikis to target different metastatic organs.
- Organoids
-
Organ epithelial fragments that resemble the whole organ in structure and function during three-dimensional culture.
- Stem cell niche
-
A specialized microenvironment that interacts with cells such as stem cells or tumour cells to regulate their fate.
- E-cadherin
-
A calcium-dependent cell–cell adhesion molecule with pivotal roles in epithelial cell behaviour, tissue formation and suppression of cancer.
- Matrikines
-
ECM fragments released from the ECM by proteolysis or by cryptic site exposure; they have biological activities that are different from those of the full-length protein.
- Matrigel
-
A gelatinous protein mixture secreted by Engelbreth–Holm–Swarm (EHS) mouse sarcoma cells. It contains structural proteins such as laminin, entactin, collagen and HSPGs. Growth factors such as TGFβ and EGF are also present in Matrigel, although growth factor-reduced formulations are also available.
Rights and permissions
About this article
Cite this article
Bonnans, C., Chou, J. & Werb, Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol 15, 786–801 (2014). https://doi.org/10.1038/nrm3904
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3904
This article is cited by
-
Effects and mechanisms of Porphyromonas gingivalis outer membrane vesicles induced cardiovascular injury
BMC Oral Health (2024)
-
Collagen in the central nervous system: contributions to neurodegeneration and promise as a therapeutic target
Molecular Neurodegeneration (2024)
-
Effects of alendronate on cartilage lesions and micro-architecture deterioration of subchondral bone in patellofemoral osteoarthritic ovariectomized rats with patella-baja
Journal of Orthopaedic Surgery and Research (2024)
-
Association between circulating ECM-associated molecules and cardiovascular outcomes in hemodialysis patients: a multicenter prospective cohort study
Biomarker Research (2024)
-
Nasopharyngeal carcinoma: current views on the tumor microenvironment's impact on drug resistance and clinical outcomes
Molecular Cancer (2024)