Abstract
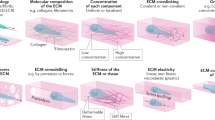
Studies of cell migration in three-dimensional (3D) cell culture systems and in vivo have revealed several differences when compared with cell migration in two dimensions, including their morphology and mechanical and signalling control. Here, researchers assess the contribution of 3D models to our understanding of cell migration, both in terms of the mechanisms used to drive single cell and collective cell migration and how migrating cells respond to a changing environment in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Petrie, R. J., Gavara, N., Chadwick, R. S. & Yamada, K. M. Nonpolarized signaling reveals two distinct modes of 3D cell migration. J. Cell Biol. 197,
Madsen, C. D. & Sahai, E. Cancer dissemination — lessons from leukocytes. Dev. Cell 19, 13–26 (2010).
Friedl, P. & Alexander, S. Cancer invasion and the microenvironment: plasticity and reciprocity. Cell 147, 992–1009 (2011).
Onodera, T. et al. Btbd7 regulates epithelial cell dynamics and branching morphogenesis. Science 329, 562–565 (2010).
Nelson, C. M. & Bissell, M. J. Of extracellular matrix, scaffolds, and signaling: tissue architecture regulates development, homeostasis, and cancer. Annu. Rev. Cell Dev. Biol. 22, 287–309 (2006).
Yamada, K. M. & Cukierman, E. Modeling tissue morphogenesis and cancer in 3D. Cell 130, 601–610 (2007).
Grinnell, F. & Petroll, W. M. Cell motility and mechanics in three-dimensional collagen matrices. Annu. Rev. Cell Dev. Biol. 26, 335–361 (2010).
DuFort, C. C., Paszek, M. J. & Weaver, V. M. Balancing forces: architectural control of mechanotransduction. Nature Rev. Mol. Cell Biol. 12, 308–319 (2011).
Baker, B. M. & Chen, C. S. Deconstructing the third dimension — how 3D culture microenvironments alter cellular cues. J. Cell Sci. 125, 3015–3024 (2012).
Friedl, P., Wolf, K. & Lammerding, J. Nuclear mechanics during cell migration. Curr. Opin. Cell Biol. 23, 55–64 (2011).
Balzer, E. M. et al. Physical confinement alters tumor cell adhesion and migration phenotypes. FASEB J. 26, 4045–4056 (2012).
Rowe, R. G. & Weiss, S. J. Navigating ECM barriers at the invasive front: the cancer cell–stroma interface. Annu. Rev. Cell Dev. Biol. 25, 567–595 (2009).
Wolf, K. et al. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nature Cell Biol. 9, 893–904 (2007).
Friedl, P. & Wolf, K. Plasticity of cell migration: a multiscale tuning model. J. Cell Biol. 188, 11–19 (2010).
Friedl, P. & Gilmour, D. Collective cell migration in morphogenesis, regeneration and cancer. Nature Rev. Mol. Cell Biol. 10, 445–457 (2009).
Friedl, P. & Weigelin, B. Interstitial leukocyte migration and immune function. Nature Immunol. 9, 960–969 (2008).
Lammermann, T. et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 453, 51–55 (2008).
Bailey, J. L. et al. Collagen oligomers modulate physical and biological properties of three-dimensional self-assembled matrices. Biopolymers 95, 77–93 (2011).
Ahlfors, J. E. & Billiar, K. L. Biomechanical and biochemical characteristics of a human fibroblast-produced and remodeled matrix. Biomaterials 28, 2183–2191 (2007).
Abbott, R. D. et al. Cytoskeletal remodeling of connective tissue fibroblasts in response to static stretch is dependent on matrix material properties. J. Cell. Physiol. 2 May 2012 (doi:10.1002/jcp.24102).
Sabeh, F., Shimizu-Hirota, R. & Weiss, S. J. Protease-dependent versus -independent cancer cell invasion programs: three-dimensional amoeboid movement revisited. J. Cell Biol. 185, 11–19 (2009).
Chun, T. H. et al. A pericellular collagenase directs the 3-dimensional development of white adipose tissue. Cell 125, 577–591 (2006).
Wolf, K. & Friedl, P. Extracellular matrix determinants for proteolytic and non-proteolytic cell migration. Trends Cell Biol. 21, 736–744 (2011).
Conklin, M. W. et al. Aligned collagen is a prognostic signature for survival in human breast carcinoma. Am. J. Pathol. 178, 1221–1232 (2011).
Williams, E. H., Carpentier, P. & Misteli, T. The JCB DataViewer scales up. J. Cell Biol. 198, 271–272 (2012).
Friedl, P., Locker, J., Sahai, E. & Segall, J. E. Classifying collective cancer cell invasion. Nature Cell Biol. 14, 777–783 (2012).
Germain, R. N. et al. Making friends in out-of-the-way places: how cells of the immune system get together and how they conduct their business as revealed by intravital imaging. Immunol. Rev. 221, 163–181 (2008).
Alexander, S., Koehl, G. E., Hirschberg, M., Geissler, E. K. & Friedl, P. Dynamic imaging of cancer growth and invasion: a modified skin-fold chamber model. Histochem. Cell Biol. 130, 1147–1154 (2008).
Weigelin, B., Bakker, G.-J. & Friedl, P. Intravital third harmonic generation microscopy of collective melanoma cell invasion: principles of interface guidance and microvesicle dynamics. Intravital 1, 32–43 (2012).
Alexander, S. & Friedl, P. Cancer invasion and resistance: interconnected processes of disease progression and therapy failure. Trends Mol. Med. 18, 13–26 (2012).
Acknowledgements
P.F is supported by the Dutch Cancer Foundation (KWF 2008-4031) and the Netherlands Science Foundation (NWO-VICI 918.11.626). E.S. thanks Cancer Research UK for funding. K.M.Y. is supported by the Intramural Research Program of the NIDCR, NIH.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- β1 integrin
-
A cell surface, transmembrane protein that forms a heterodimer with distinct α-chains to generate adhesive structures that are capable of binding to subsets of extracellular matrix macromolecules (for example, type I collagen).
- Blebs
-
Rounded protrusions driven by hydrostatic pressure. Blebs initially lack filamentous actin (F-actin) and later incoporate cortical F-actin.
- Elastic modulus
-
The amount of force per unit area required to deform a material by a defined amount.
- Fibrillogenesis
-
The formation of fibrillar arrays of extracellular matrix molecules by head-to-head and side-to-side interactions between protomer subunits.
- Filopodia
-
Finger-like protrusions driven by actin polymerization.
- Hydrogel
-
A three-dimensional structure that 'behaves' like a solid owing to its crosslinked structure despite the fact that the bulkof its mass is comprised of water.
- Lamellipodia
-
Planar protrusions driven by actin polymerization.
- Linear and nonlinear elasticity
-
A mechanical property of compliant materials in which the elastic modulus is either independent of applied strain (linear elasticity) or increases dramatically as applied strain increases (nonlinear elasticity).
- Lobopodia
-
Blunt protrusions driven by contractility and intracellular pressure.
- Matrix metalloproteinase
-
(MMP). A major class of proteolytic enzymes that contain a metal ion, commonly Zn2+, within the catalytic site.
- Organotypic
-
A culture system that aims to recreate the organization of real tissue. For example, skin organotypic cultures include a lower collagen layer that mimics the dermis and an upper keratinocyte layer that is exposed to the air in a similar manner to skin. In some cases, these culture use matrices directly derived from animal or human tissue.
- Pseudopods
-
A broadly used term to describe protrusions that are formedby actin polymerization but lack the defined geometry of lamellipodia or filopodia.
- Second harmonic generation
-
(SHG). An imaging process that allows for the visualization of type I collagen fibrils. In this process two photons of incident light interact with the non-centrosymmetric structure of collagen to generate photons that are half the original wavelength.
- Stromal cell
-
Non-tumour cell that is found within tumours, such as leukocytes, fibroblasts and endothelial cells.
- Telopeptides
-
Non-helical portions of type I collagen fibrils that are located at both the amino- and carboxy-terminal ends of the fibril.Telopeptides support covalent crosslink formation.
Rights and permissions
About this article
Cite this article
Friedl, P., Sahai, E., Weiss, S. et al. New dimensions in cell migration. Nat Rev Mol Cell Biol 13, 743–747 (2012). https://doi.org/10.1038/nrm3459
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3459
This article is cited by
-
Tumor spheroids accelerate persistently invading cancer cells
Scientific Reports (2022)
-
Role of RhoC in cancer cell migration
Cancer Cell International (2021)
-
Development of a model for fibroblast-led collective migration from breast cancer cell spheroids to study radiation effects on invasiveness
Radiation Oncology (2021)
-
Force-FAK signaling coupling at individual focal adhesions coordinates mechanosensing and microtissue repair
Nature Communications (2021)
-
Mechanical homeostasis in tissue equivalents: a review
Biomechanics and Modeling in Mechanobiology (2021)