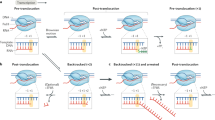
Key Points
-
RNA polymerase II (Pol II) transcribes all eukaryotic protein-coding genes and most non-coding RNA genes. The final step of transcription is termination, which leads to the release of Pol II and RNA from the DNA template through a poorly defined mechanism.
-
Transcription termination serves many vital functions in the cell, such as preventing RNA polymerase interference with neighbouring DNA elements, recycling RNA polymerase, promoting RNA 3′-end processing, and regulating gene expression via premature termination of transcription (that is, attenuation).
-
Termination can be elicited through different pathways depending on the phosphorylation status of the Pol II carboxy-terminal domain (CTD) and the presence of various RNA signals and termination factors. Two of the best-studied termination pathways are the poly(A)-dependent pathway and Sen1-dependent pathway, which are connected to RNA 3′-end processing events for mRNAs and non-coding RNAs, respectively.
-
Three mechanisms are proposed to cause Pol II termination: conformational changes induced by binding of factors to Pol II; collision of an exoribonuclease with Pol II; and/or disruption of the Pol II active site hybrid by an RNA–DNA helicase. However, these molecular models have thus far remained insufficient to fully explain how termination occurs.
-
Pol II is similar both structurally and biochemically to bacterial RNA polymerase, suggesting that there may be some general features of termination that are common to all cellular RNA polymerases. Recent studies investigating the requirements for bacterial termination implicate several regions of Pol II as putative termination effector domains, including the lid, trigger loop, clamp helices, dock and flap.
-
The phosphorylation status of the Pol II CTD residues Ser7, Ser5 and Ser2 is dynamic across the length of a gene. Genome-wide localization studies suggest that a combination of high Ser7-P and Ser5-P and low Ser2-P in the CTD of Pol II at promoter-proximal positions may serve as a signal to trigger Sen1-dependent termination.
-
Genome-wide localization of protein 1 of CFI (Pcf11), Nrd1, and RNA-trafficking protein 1 (Rat1) reveals extensive overlap of these termination factors along both protein-coding and non-coding RNA genes. This pattern is consistent with the idea that the machinery for Sen1-dependent termination and poly(A)-dependent termination is broadly available to target Pol II during transcription of most genes, and in fact may provide a way to ensure fail-safe termination.
Abstract
The pervasiveness of RNA synthesis in eukaryotes is largely the result of RNA polymerase II (Pol II)-mediated transcription, and termination of its activity is necessary to partition the genome and maintain the proper expression of neighbouring genes. Despite its ever-increasing biological significance, transcription termination remains one of the least understood processes in gene expression. However, recent mechanistic studies have revealed a striking convergence among several overlapping models of termination, including the poly(A)- and Sen1-dependent pathways, as well as new insights into the specificity of Pol II termination among its diverse gene targets. Broader knowledge of the role of Pol II carboxy-terminal domain phosphorylation in promoting alternative mechanisms of termination has also been gained.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lee, T. I. & Young, R. A. Transcription of eukaryotic protein-coding genes. Annu. Rev. Genet. 34, 77–137 (2000).
Espinosa, J. M. The meaning of pausing. Mol. Cell 40, 507–508 (2010).
Nechaev, S. & Adelman, K. Pol II waiting in the starting gates: regulating the transition from transcription initiation into productive elongation. Biochim. Biophys. Acta 1809, 34–45 (2011).
Rosonina, E., Kaneko, S. & Manley, J. L. Terminating the transcript: breaking up is hard to do. Genes Dev. 20, 1050–1056 (2006).
Gilmour, D. S. & Fan, R. Derailing the locomotive: transcription termination. J. Biol. Chem. 283, 661–664 (2008).
Richard, P. & Manley, J. L. Transcription termination by nuclear RNA polymerases. Genes Dev. 23, 1247–1269 (2009).
Merino, E. & Yanofsky, C. Transcription attenuation: a highly conserved regulatory strategy used by bacteria. Trends Genet. 21, 260–264 (2005).
Naville, M. & Gautheret, D. Transcription attenuation in bacteria: theme and variations. Brief Funct. Genomics 9, 178–189 (2010).
Dichtl, B. Transcriptional ShortCUTs. Mol. Cell 31, 617–618 (2008).
Kim, K. Y. & Levin, D. E. Mpk1 MAPK association with the Paf1 complex blocks Sen1-mediated premature transcription termination. Cell 144, 745–756 (2011).
Jacquier, A. The complex eukaryotic transcriptome: unexpected pervasive transcription and novel small RNAs. Nature Rev. Genet. 10, 833–844 (2009).
Shearwin, K. E., Callen, B. P. & Egan, J. B. Transcriptional interference — a crash course. Trends Genet. 21, 339–345 (2005).
West, S. & Proudfoot, N. J. Transcriptional termination enhances protein expression in human cells. Mol. Cell 33, 354–364 (2009).
Mapendano, C. K., Lykke-Andersen, S., Kjems, J., Bertrand, E. & Jensen, T. H. Crosstalk between mRNA 3′ end processing and transcription initiation. Mol. Cell 40, 410–422 (2010).
Ardehali, M. B. & Lis, J. T. Tracking rates of transcription and splicing in vivo. Nature Struct. Mol. Biol. 16, 1123–1124 (2009).
Kireeva, M. L., Komissarova, N., Waugh, D. S. & Kashlev, M. The 8-nucleotide-long RNA:DNA hybrid is a primary stability determinant of the RNA polymerase II elongation complex. J. Biol. Chem. 275, 6530–6536 (2000).
Komissarova, N., Becker, J., Solter, S., Kireeva, M. & Kashlev, M. Shortening of RNA:DNA hybrid in the elongation complex of RNA polymerase is a prerequisite for transcription termination. Mol. Cell 10, 1151–1162 (2002). References 16 and 17 implicate the RNA–DNA hybrid of the transcription elongation complex as a primary target of Pol II and bacterial RNA polymerase termination mechanisms.
Lykke-Andersen, S. & Jensen, T. H. Overlapping pathways dictate termination of RNA polymerase II transcription. Biochimie 89, 1177–1182 (2007).
Rondon, A. G., Mischo, H. E. & Proudfoot, N. J. Terminating transcription in yeast: whether to be a 'nerd' or a 'rat'. Nature Struct. Mol. Biol. 15, 775–776 (2008).
Logan, J., Falck-Pedersen, E., Darnell, J. E. Jr & Shenk, T. A poly(A) addition site and a downstream termination region are required for efficient cessation of transcription by RNA polymerase II in the mouse β maj-globin gene. Proc. Natl Acad. Sci. USA 84, 8306–8310 (1987).
Whitelaw, E. & Proudfoot, N. α-thalassaemia caused by a poly(A) site mutation reveals that transcriptional termination is linked to 3′ end processing in the human α 2 globin gene. EMBO J. 5, 2915–2922 (1986).
Edwalds-Gilbert, G., Prescott, J. & Falck-Pedersen, E. 3′ RNA processing efficiency plays a primary role in generating termination-competent RNA polymerase II elongation complexes. Mol. Cell. Biol. 13, 3472–3480 (1993).
Plant, K. E., Dye, M. J., Lafaille, C. & Proudfoot, N. J. Strong polyadenylation and weak pausing combine to cause efficient termination of transcription in the human γ-globin gene. Mol. Cell. Biol. 25, 3276–3285 (2005).
Kim, H. et al. Gene-specific RNA polymerase II phosphorylation and the CTD code. Nature Struct. Mol. Biol. 17, 1279–1286 (2010). Provides genome-wide analysis of the dynamics of Pol II CTD phosphorylation and the recruitment of termination factors Pcf11, Nrd1 and Rat1.
Mandel, C. R., Bai, Y. & Tong, L. Protein factors in pre-mRNA 3′-end processing. Cell. Mol. Life Sci. 65, 1099–1122 (2008).
Millevoi, S. & Vagner, S. Molecular mechanisms of eukaryotic pre-mRNA 3′ end processing regulation. Nucleic Acids Res. 38, 2757–2774 (2009).
Shi, Y. et al. Molecular architecture of the human pre-mRNA 3′ processing complex. Mol. Cell 33, 365–376 (2009).
Egloff, S. & Murphy, S. Cracking the RNA polymerase II CTD code. Trends Genet. 24, 280–288 (2008).
Buratowski, S. Progression through the RNA Polymerase II CTD Cycle. Mol. Cell 36, 541–546 (2009).
Buratowski, S. The CTD code. Nature Struct. Biol. 10, 679–680 (2003).
Gromak, N., West, S. & Proudfoot, N. J. Pause sites promote transcriptional termination of mammalian RNA polymerase II. Mol. Cell. Biol. 26, 3986–3996 (2006). Shows that a pause sequence promotes poly(A)-dependent termination in vivo , and the efficiency of termination is influenced by the strength of the poly(A) site and its proximity to the pause site.
Glover-Cutter, K., Kim, S., Espinosa, J. & Bentley, D. L. RNA polymerase II pauses and associates with pre-mRNA processing factors at both ends of genes. Nature Struct. Mol. Biol. 15, 71–78 (2008).
Park, N. J., Tsao, D. C. & Martinson, H. G. The two steps of poly(A)-dependent termination, pausing and release, can be uncoupled by truncation of the RNA polymerase II carboxyl-terminal repeat domain. Mol. Cell. Biol. 24, 4092–4103 (2004).
Nag, A., Narsinh, K. & Martinson, H. G. The poly(A)-dependent transcriptional pause is mediated by CPSF acting on the body of the polymerase. Nature Struct. Mol. Biol. 14, 662–669 (2007).
Kazerouninia, A., Ngo, B. & Martinson, H. G. Poly(A) signal-dependent degradation of unprocessed nascent transcripts accompanies poly(A) signal-dependent transcriptional pausing in vitro. RNA 16, 197–210 (2010). References 33–35 from the Martinson laboratory show that poly(A)-dependent termination of Pol II can be separated into two steps, pausing and release, which depend on interactions of the cleavage and polyadenylation machinery with the body of Pol II and the Pol II CTD.
Alexander, R. D., Innocente, S. A., Barrass, J. D. & Beggs, J. D. Splicing-dependent RNA polymerase pausing in yeast. Mol. Cell 40, 582–593 (2010).
Carrillo Oesterreich, F., Preibisch, S. & Neugebauer, K. M. Global analysis of nascent RNA reveals transcriptional pausing in terminal exons. Mol. Cell 40, 571–581 (2010).
Kim, M. et al. The yeast Rat1 exonuclease promotes transcription termination by RNA polymerase II. Nature 432, 517–522 (2004).
West, S., Gromak, N. & Proudfoot, N. J. Human 5′–3′ exonuclease Xrn2 promotes transcription termination at co-transcriptional cleavage sites. Nature 432, 522–525 (2004). References 38 and 39 demonstrate that the 5′–3′ exoribonuclease Rat1 (XRN2 in mammals) is important for poly(A)-dependent termination and strengthen support for the torpedo model.
Luo, W., Johnson, A. W. & Bentley, D. L. The role of Rat1 in coupling mRNA 3′-end processing to transcription termination: implications for a unified allosteric-torpedo model. Genes Dev. 20, 954–965 (2006). Reveals that in addition to its exoribonuclease activity, Rat1 helps recruit mRNA 3′-end-processing factors. The authors propose a model for the termination mechanism that incorporates both allosteric and torpedo components.
Lunde, B. M. et al. Cooperative interaction of transcription termination factors with the RNA polymerase II C-terminal domain. Nature Struct. Mol. Biol. 17, 1195–1201 (2010).
Teixeira, A. et al. Autocatalytic RNA cleavage in the human β-globin pre-mRNA promotes transcription termination. Nature 432, 526–530 (2004).
Ghazal, G. et al. Yeast RNase III triggers polyadenylation-independent transcription termination. Mol. Cell 36, 99–109 (2009).
Rondón, A., Mischo, H., Kawauchi, J. & Proudfoot, N. Fail-safe transcriptional termination for protein-coding genes in S. cerevisiae. Mol. Cell 36, 88–98 (2009).
Nabavi, S. & Nazar, R. N. Pac1 endonuclease and Dhp1p 5′–3′ exonuclease are required for U3 snoRNA termination in Schizosaccharomyces pombe. FEBS Lett. 584, 3436–3441 (2010).
Connelly, S. & Manley, J. L. A functional mRNA polyadenylation signal is required for transcription termination by RNA polymerase II. Genes Dev. 2, 440–452 (1988).
Houseley, J. & Tollervey, D. The many pathways of RNA degradation. Cell 136, 763–776 (2009).
Kim, H. D., Choe, J. & Seo, Y. S. The sen1+ gene of Schizosaccharomyces pombe, a homologue of budding yeast SEN1, encodes an RNA and DNA helicase. Biochemistry 38, 14697–14710 (1999).
Steinmetz, E. J. & Brow, D. A. Repression of gene expression by an exogenous sequence element acting in concert with a heterogeneous nuclear ribonucleoprotein-like protein, Nrd1, and the putative helicase Sen1. Mol. Cell. Biol. 16, 6993–7003 (1996).
Steinmetz, E. J., Conrad, N. K., Brow, D. A. & Corden, J. L. RNA-binding protein Nrd1 directs poly(A)-independent 3′-end formation of RNA polymerase II transcripts. Nature 413, 327–331 (2001).
Steinmetz, E. J. et al. Genome-wide distribution of yeast RNA polymerase II and its control by Sen1 helicase. Mol. Cell 24, 735–746 (2006). This paper establishes Sen1 as a general transcription termination factor in yeast for most snRNAs and snoRNAs and some short mRNA transcripts.
Ursic, D., Chinchilla, K., Finkel, J. S. & Culbertson, M. R. Multiple protein/protein and protein/RNA interactions suggest roles for yeast DNA/RNA helicase Sen1p in transcription, transcription-coupled DNA repair and RNA processing. Nucleic Acids Res. 32, 2441–2452 (2004).
Finkel, J. S., Chinchilla, K., Ursic, D. & Culbertson, M. R. Sen1p performs two genetically separable functions in transcription and processing of U5 small nuclear RNA in Saccharomyces cerevisiae. Genetics 184, 107–118 (2010).
Mischo, H. E. et al. Yeast Sen1 helicase protects the genome from transcription-associated instability. Mol. Cell 41, 21–32 (2011).
Arigo, J. T., Eyler, D. E., Carroll, K. L. & Corden, J. L. Termination of cryptic unstable transcripts is directed by yeast RNA-binding proteins Nrd1 and Nab3. Mol. Cell 23, 841–851 (2006).
Thiebaut, M., Kisseleva-Romanova, E., Rougemaille, M., Boulay, J. & Libri, D. Transcription termination and nuclear degradation of cryptic unstable transcripts: a role for the Nrd1-Nab3 pathway in genome surveillance. Mol. Cell 23, 853–864 (2006).
Banerjee, S., Chalissery, J., Bandey, I. & Sen, R. Rho-dependent transcription termination: more questions than answers. J. Microbiol. 44, 11–22 (2006).
Kawauchi, J., Mischo, H., Braglia, P., Rondon, A. & Proudfoot, N. J. Budding yeast RNA polymerases I and II employ parallel mechanisms of transcriptional termination. Genes Dev. 22, 1082–1092 (2008).
Banerjee, A., Sammarco, M. C., Ditch, S., Wang, J. & Grabczyk, E. A novel tandem reporter quantifies RNA polymerase II termination in mammalian cells. PLoS ONE 4, e6193 (2009).
Suraweera, A. et al. Functional role for senataxin, defective in ataxia oculomotor apraxia type 2, in transcriptional regulation. Hum. Mol. Genet. 18, 3384–3396 (2009).
Matera, A. G., Terns, R. M. & Terns, M. P. Non-coding RNAs: lessons from the small nuclear and small nucleolar RNAs. Nature Rev. Mol. Cell Biol. 8, 209–220 (2007).
Egloff, S. et al. Serine-7 of the RNA polymerase II CTD is specifically required for snRNA gene expression. Science 318, 1777–1779 (2007). This study reveals that Ser7-P Pol II CTD helps recruit the integrator complex, which is required for human snRNA 3′-end processing.
Ezzeddine, N. et al. A subset of Drosophila integrator proteins is essential for efficient U7 snRNA and spliceosomal snRNA 3′ end formation. Mol. Cell. Biol. 31, 328–341 (2011).
Baillat, D. et al. Integrator, a multiprotein mediator of small nuclear RNA processing, associates with the C-terminal repeat of RNA polymerase II. Cell 123, 265–276 (2005).
Dominski, Z., Yang, X.-C., Purdy, M., Wagner, E. J. & Marzluff, W. F. A CPSF-73 homologue is required for cell cycle progression but not cell growth and interacts with a protein having features of CPSF-100. Mol. Cell. Biol. 25, 1489–1500 (2005).
Egloff, S., Al-Rawaf, H., O'Reilly, D. & Murphy, S. Chromatin structure is implicated in “late” elongation checkpoints on the U2 snRNA and β-actin genes. Mol. Cell. Biol. 29, 4002–4013 (2009).
Ballarino, M. et al. Coupled RNA processing and transcription of intergenic primary microRNAs. Mol. Cell. Biol. 29, 5632–5638 (2009).
Carninci, P. Molecular biology: the long and short of RNAs. Nature 457, 974–975 (2009).
Dengl, S. & Cramer, P. Torpedo nuclease Rat1 is insufficient to terminate RNA polymerase II in vitro. J. Biol. Chem. 284, 21270–21279 (2009).
Saeki, H. & Svejstrup, J. Q. Stability, flexibility, and dynamic interactions of colliding RNA polymerase II elongation complexes. Mol. Cell 35, 191–205 (2009).
Xiang, S. et al. Structure and function of the 5′–3′ exoribonuclease Rat1 and its activating partner Rail. Nature 458, 784–788 (2009).
Chang, J. H. & Xiang, S. Structural and biochemical studies of the 5′–3′ exoribonuclease Xrnl. Nature Cell Biol. 18, 270–276 (2011).
Epshtein, V., Dutta, D., Wade, J. & Nudler, E. An allosteric mechanism of Rho-dependent transcription termination. Nature 463, 245–249 (2010). Demonstrates that the Rho termination factor associates directly with E. coli RNA polymerase and promotes termination by inducing an allosteric rearrangement of the RNA polymerase active site. Transduction of the termination signal is dependent on the lid and trigger loop domains of the RNA polymerase β′-subunit.
Lang, W. H., Platt, T. & Reeder, R. H. Escherichia coli Rho factor induces release of yeast RNA polymerase II but not polymerase I or III. Proc. Natl Acad. Sci. USA 95, 4900–4905 (1998).
Schmidt, M. C. & Chamberlin, M. J. nusA protein of Escherichia coli is an efficient transcription termination factor for certain terminator sites. J. Mol. Biol. 195, 809–818 (1987).
Sullivan, S. L. & Gottesman, M. E. Requirement for E. coli NusG protein in factor-dependent transcription termination. Cell 68, 989–994 (1992).
Mason, S. W., Li, J. & Greenblatt, J. Host factor requirements for processive antitermination of transcription and suppression of pausing by the N protein of bacteriophage λ. J. Biol. Chem. 267, 19418–19426 (1992).
Torres, M., Condon, C., Balada, J. M., Squires, C. & Squires, C. L. Ribosomal protein S4 is a transcription factor with properties remarkably similar to NusA, a protein involved in both non-ribosomal and ribosomal RNA antitermination. EMBO J. 20, 3811–3820 (2001).
Shankar, S., Hatoum, A. & Roberts, J. W. A transcription antiterminator constructs a NusA-dependent shield to the emerging transcript. Mol. Cell 27, 914–927 (2007).
Ha, K. S., Toulokhonov, I., Vassylyev, D. G. & Landick, R. The NusA N-terminal domain is necessary and sufficient for enhancement of transcriptional pausing via interaction with the RNA exit channel of RNA polymerase. J. Mol. Biol. 401, 708–725 (2010). Findings from this study show that interaction of the NusA termination factor with regions of bacterial RNA polymerase near the RNA exit channel (such as the β-flap and β′-dock) stimulates pausing and release.
Toulokhonov, I., Artsimovitch, I. & Landick, R. Allosteric control of RNA polymerase by a site that contacts nascent RNA hairpins. Science 292, 730–733 (2001).
Deighan, P., Diez, C. M., Leibman, M., Hochschild, A. & Nickels, B. E. The bacteriophage λQ antiterminator protein contacts the β-flap domain of RNA polymerase. Proc. Natl Acad. Sci. USA 105, 15305–15310 (2008).
Mooney, R. A., Schweimer, K., Rösch, P., Gottesman, M. & Landick, R. Two structurally independent domains of E. coli NusG create regulatory plasticity via distinct interactions with RNA polymerase and regulators. J. Mol. Biol. 391, 341–358 (2009). Reveals how interactions with two separate protein domains of NusG contribute to its RNA polymerase termination and antitermination activities. The NusG NTD contacts the RNA polymerase β′-clamp helices while its CTD binds Rho or other transcriptional regulators.
Nickels, B. E. Genetic assays to define and characterize protein–protein interactions involved in gene regulation. Methods 47, 53–62 (2009).
Belogurov, G. A., Mooney, R. A., Svetlov, V., Landick, R. & Artsimovitch, I. Functional specialization of transcription elongation factors. EMBO J. 28, 112–122 (2009).
Belogurov, G. A., Sevostyanova, A., Svetlov, V. & Artsimovitch, I. Functional regions of the N-terminal domain of the antiterminator RfaH. Mol. Microbiol. 76, 286–301 (2010).
Komarnitsky, P., Cho, E. J. & Buratowski, S. Different phosphorylated forms of RNA polymerase II and associated mRNA processing factors during transcription. Genes Dev. 14, 2452–2460 (2000). Shows that dynamic phosphorylation of the Pol II CTD contributes to differential recruitment of mRNA processing factors, a discovery that forms the basis of the CTD code hypothesis.
Licatalosi, D. D. et al. Functional interaction of yeast pre-mRNA 3′ end processing factors with RNA polymerase II. Mol. Cell 9, 1101–1111 (2002).
Ahn, S. H., Kim, M. & Buratowski, S. Phosphorylation of serine 2 within the RNA polymerase II C-terminal domain couples transcription and 3′ end processing. Mol. Cell 13, 67–76 (2004).
Chapman, R. D. et al. Transcribing RNA polymerase II is phosphorylated at CTD residue serine-7. Science 318, 1780–1782 (2007). Identifies dynamic phosphorylation of the Pol II CTD at Ser 7 as a new component of the CTD code.
Guo, J., Garrett, M., Micklem, G. & Brogna, S. Poly(A) signals located near the 5′ end of genes are silenced by a general mechanism that prevents premature 3′-end processing. Mol. Cell. Biol. 31, 639–651 (2011).
Gudipati, R. K., Villa, T., Boulay, J. & Libri, D. Phosphorylation of the RNA polymerase II C-terminal domain dictates transcription termination choice. Nature Struct. Mol. Biol. 15, 786–794 (2008). Shows that the selection of the poly(A)-dependent versus Sen1-dependent termination pathway is influenced by the phosphorylation status of the Pol II CTD, which is in turn influenced by the distance that Pol II travels from the transcription start site.
Jenks, M. H., O'Rourke, T. W. & Reines, D. Properties of an intergenic terminator and start site switch that regulate IMD2 transcription in yeast. Mol. Cell. Biol. 28, 3883–3893 (2008).
Vasiljeva, L., Kim, M., Mutschler, H., Buratowski, S. & Meinhart, A. The Nrd1–Nab3–Sen1 termination complex interacts with the Ser5-phosphorylated RNA polymerase II C-terminal domain. Nature Struct. Mol. Biol. 15, 795–804 (2008).
Mayer, A. et al. Uniform transitions of the general RNA polymerase II transcription complex. Nature Struct. Mol. Biol. 17, 1272–1278 (2010).
Tietjen, J. R. et al. Chemical-genomic dissection of the CTD code. Nature Struct. Mol. Biol. 17, 1154–1161 (2010). References 95 and 96 provide genome-wide analyses of the dynamics of Pol II CTD-phosphorylation and identify gene-specific patterns of CTD marks or transcription factor recruitment, respectively.
Kim, M. et al. Distinct pathways for snoRNA and mRNA termination. Mol. Cell 24, 723–734 (2006). Reveals that many poly(A)-dependent and Sen1-dependent termination factors can be localized to both mRNA and snoRNA-encoding genes, but their requirements for termination are gene-specific.
Egloff, S. et al. The integrator complex recognizes a new double mark on the RNA polymerase II carboxyl-terminal domain. J. Biol. Chem. 285, 20564–20569 (2010).
Singh, N. et al. The Ess1 prolyl isomerase is required for transcription termination of small noncoding RNAs via the Nrd1 pathway. Mol. Cell 36, 255–266 (2009).
Werner-Allen, J. W. et al. cis-proline-mediated Ser(P)5 dephosphorylation by the RNA polymerase II C-terminal domain phosphatase Ssu72. J. Biol. Chem. 286, 5717–5726 (2011).
Selth, L. A., Sigurdsson, S. & Svejstrup, J. Q. Transcript Elongation by RNA Polymerase II. Annu. Rev. Biochem. 79, 271–293 (2010).
Kim, M., Ahn, S. H., Krogan, N. J., Greenblatt, J. F. & Buratowski, S. Transitions in RNA polymerase II elongation complexes at the 3′ ends of genes. EMBO J. 23, 354–364 (2004).
Jaehning, J. A. The Paf1 complex: platform or player in RNA polymerase II transcription? Biochim. Biophys. Acta 1799, 379–388 (2010).
Alén, C. et al. A role for chromatin remodeling in transcriptional termination by RNA polymerase II. Mol. Cell 10, 1441–1452 (2002).
Wood, A. J. et al. Regulation of alternative polyadenylation by genomic imprinting. Genes Dev. 22, 1141–1146 (2008).
Spies, N., Nielsen, C. B., Padgett, R. A. & Burge, C. B. Biased chromatin signatures around polyadenylation sites and exons. Mol. Cell 36, 245–254 (2009).
Alló, M. & Kornblihtt, A. R. Gene silencing: small RNAs control RNA polymerase II elongation. Curr. Biol. 20, R704–R707 (2010).
Fan, X. et al. Nucleosome depletion at yeast terminators is not intrinsic and can occur by a transcriptional mechanism linked to 3′-end formation. Proc. Natl Acad. Sci. USA 107, 17945–17950 (2010).
Mayr, C. & Bartel, D. P. Widespread shortening of 3′UTRs by alternative cleavage and polyadenylation activates oncogenes in cancer cells. Cell 138, 673–684 (2009).
Calvo, O. & Manley, J. L. Strange bedfellows: polyadenylation factors at the promoter. Genes Dev. 17, 1321–1327 (2003).
Venters, B. J. & Pugh, B. F. How eukaryotic genes are transcribed. Crit. Rev. Biochem. Mol. Biol. 44, 117–141 (2009).
Hampsey, M., Singh, B. N., Ansari, A., Lainé, J.-P. & Krishnamurthy, S. Control of eukaryotic gene expression: gene loops and transcriptional memory. Adv. Enzyme Regul. 29 Oct 2010 (doi:10.1016/j.advenzreg.2010.10.001).
El Kaderi, B., Medler, S., Raghunayakula, S. & Ansari, A. Gene looping is conferred by activator-dependent interaction of transcription initiation and termination machineries. J. Biol. Chem. 284, 25015–25025 (2009).
Wang, Y., Fairley, J. A. & Roberts, S. G. E. Phosphorylation of TFIIB links transcription initiation and termination. Curr. Biol. 20, 548–553 (2010).
Moore, M. J. & Proudfoot, N. J. Pre-mRNA processing reaches back to transcription and ahead to translation. Cell 136, 688–700 (2009).
Lykke-Andersen, S., Mapendano, C. K. & Jensen, T. H. An ending is a new beginning: transcription termination supports re-initiation. Cell Cycle 10, 863–865 (2011).
Glover-Cutter, K. et al. TFIIH-associated Cdk7 kinase functions in phosphorylation of C-terminal domain Ser7 residues, promoter-proximal pausing, and termination by RNA polymerase II. Mol. Cell. Biol. 29, 5455–5464 (2009).
Jimeno-González, S., Haaning, L. L., Malagon, F. & Jensen, T. H. The yeast 5′-3′ exonuclease Rat1p functions during transcription elongation by RNA polymerase II. Mol. Cell 37, 580–587 (2010).
Opalka, N. et al. Complete structural model of Escherichia coli RNA polymerase from a hybrid approach. PLoS Biol. 8, e1000483 (2010).
Cramer, P., Bushnell, D. A. & Kornberg, R. D. Structural basis of transcription: RNA polymerase II at 2.8 angstrom resolution. Science 292, 1863–1876 (2001).
Kettenberger, H., Armache, K. J. & Cramer, P. Complete RNA polymerase II elongation complex structure and its interactions with NTP and TFIIS. Mol. Cell 16, 955–965 (2004).
Birse, C. E., Minvielle-Sebastia, L., Lee, B. A., Keller, W. & Proudfoot, N. J. Coupling termination of transcription to messenger RNA maturation in yeast. Science 280, 298–301 (1998).
Sadowski, M., Dichtl, B., Hübner, W. & Keller, W. Independent functions of yeast Pcf11p in pre-mRNA 3′ end processing and in transcription termination. EMBO J. 22, 2167–2177 (2003).
Zhang, Z., Fu, J. & Gilmour, D. S. CTD-dependent dismantling of the RNA polymerase II elongation complex by the pre-mRNA 3′-end processing factor, Pcf11. Genes Dev. 19, 1572–1580 (2005).
Dichtl, B. et al. Yhh1p/Cft1p directly links poly(A) site recognition and RNA polymerase II transcription termination. EMBO J. 21, 4125–4135 (2002).
Garas, M., Dichtl, B. & Keller, W. The role of the putative 3′ end processing endonuclease Ysh1p in mRNA and snoRNA synthesis. RNA 14, 2671–2684 (2008).
Nedea, E. et al. Organization and function of APT, a subcomplex of the yeast cleavage and polyadenylation factor involved in the formation of mRNA and small nucleolar RNA 3′-ends. J. Biol. Chem. 278, 33000–33010 (2003).
Nedea, E. et al. The Glc7 phosphatase subunit of the cleavage and polyadenylation factor is essential for transcription termination on snoRNA genes. Mol. Cell 29, 577–587 (2008).
Ghazy, M. A., He, X., Singh, B. N., Hampsey, M. & Moore, C. The essential N terminus of the Pta1 scaffold protein is required for snoRNA transcription termination and Ssu72 function but is dispensable for pre-mRNA 3′-end processing. Mol. Cell. Biol. 29, 2296–2307 (2009).
Ganem, C. et al. Ssu72 is a phosphatase essential for transcription termination of snoRNAs and specific mRNAs in yeast. EMBO J. 22, 1588–1598 (2003).
Steinmetz, E. J. & Brow, D. A. Ssu72 protein mediates both poly(A)-coupled and poly(A)-independent termination of RNA polymerase II transcription. Mol. Cell. Biol. 23, 6339–6349 (2003).
Xue, Y. et al. Saccharomyces cerevisiae RAI1 (YGL246c) is homologous to human DOM3Z and encodes a protein that binds the nuclear exoribonuclease Rat1p. Mol. Cell. Biol. 20, 4006–4015 (2000).
Kaneko, S., Rozenblatt-Rosen, O., Meyerson, M. & Manley, J. L. The multifunctional protein p54nrb/PSF recruits the exonuclease XRN2 to facilitate pre-mRNA 3′ processing and transcription termination. Genes Dev. 21, 1779–1789 (2007).
Arigo, J. T., Carroll, K. L., Ames, J. M. & Corden, J. L. Regulation of yeast NRD1 expression by premature transcription termination. Mol. Cell 21, 641–651 (2006).
Barillà, D., Lee, B. A. & Proudfoot, N. J. Cleavage/polyadenylation factor IA associates with the carboxyl-terminal domain of RNA polymerase II in Saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 98, 445–450 (2001).
McCracken, S. et al. The C-terminal domain of RNA polymerase II couples mRNA processing to transcription. Nature 385, 357–361 (1997).
Steinmetz, E. J., Ng, S. B., Cloute, J. P. & Brow, D. A. cis- and trans-acting determinants of transcription termination by yeast RNA polymerase II. Mol. Cell. Biol. 26, 2688–2696 (2006).
Mueller, C. L., Porter, S. E., Hoffman, M. G. & Jaehning, J. A. The Paf1 complex has functions independent of actively transcribing RNA polymerase II. Mol. Cell 14, 447–456 (2004).
Sheldon, K. E., Mauger, D. M. & Arndt, K. M. A requirement for the Saccharomyces cerevisiae Paf1 complex in snoRNA 3′ end formation. Mol. Cell 20, 225–236 (2005).
Acknowledgements
Research in the laboratory of C.M. is supported by grants from the US National Institutes of Health National Institute of General Medical Sciences: award numbers K12GM074869 (J.N.K.), R01GM041752 (C.M.) and R01GM068887 (C.M.). We thank G. Meinke for assistance in preparing PyMOL images.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Related links
Related links
DATABASES
Protein Data Bank
FURTHER INFORMATION
Glossary
- Cryptic unstable transcripts
-
Non-coding RNAs (∼200–600 nucleotides long) discovered in yeast that are typically transcribed from intergenic regions of the genome (such as promoters) and are rapidly degraded by the exosome.
- Stable unannotated transcripts
-
Non-coding RNAs discovered in yeast that are generally longer and more stable than cryptic unstable transcripts.
- Promoter-proximal pausing
-
Halting of an early RNA polymerase elongation complex that remains competent to eventually resume transcription.
- Genomic partitioning
-
Separation of adjacent DNA functional units from one another by transcription termination and/or DNA-binding proteins (such as chromatin) in order to prevent transcriptional interference.
- Cleavage and polyadenylation specificity factor
-
A mammalian protein complex containing an endo-ribonuclease that is required for efficient mRNA 3′-end processing and RNA polymerase II (Pol II) transcription termination. Homologous to yeast cleavage and polyadenylation factor, which contains additional subunits required for efficient Pol II termination at small nuclear RNA-encoding and small nucleolar RNA-encoding genes.
- Cleavage stimulatory factor
-
A mammalian protein complex that is required for efficient mRNA 3′-end processing and transcription termination. Homologous to yeast cleavage factor IA, which is also required for efficient RNA polymerase II termination at genes encoding small nuclear RNAs and small nucleolar RNAs.
- Exosome
-
A protein complex that targets various types of RNA for degradation primarily via its 3′–5′ exoribonuclease activity.
- TRAMP
-
A polyadenylation complex that enhances exosome- mediated degradation of aberrant RNAs.
- Drosha
-
A ribonuclease III enzyme that initiates processing of microRNAs.
- Tower domain
-
A pronounced and conserved α-helix near the active site of the yeast nuclear 5′–3′ exoribonuclease RNA-trafficking protein 1 (Rat1; XRN2 in mammals) but not its cytoplasmic orthologue 5′–3′ exoribonuclease 1 (Xrn1).
- ChIP–chip
-
A technique that combines chromatin immunoprecipitation (ChIP) with microarray technology (chip) to investigate genome-wide protein–DNA interactions.
- Prolyl isomerases
-
Enzymes that catalyse the interconversion of cis and trans isomers of peptide bonds with the amino acid Pro.
Rights and permissions
About this article
Cite this article
Kuehner, J., Pearson, E. & Moore, C. Unravelling the means to an end: RNA polymerase II transcription termination. Nat Rev Mol Cell Biol 12, 283–294 (2011). https://doi.org/10.1038/nrm3098
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3098
This article is cited by
-
Synergising single-cell resolution and 4sU labelling boosts inference of transcriptional bursting
Genome Biology (2023)
-
ORC1 binds to cis-transcribed RNAs for efficient activation of replication origins
Nature Communications (2023)
-
Context-specific regulation and function of mRNA alternative polyadenylation
Nature Reviews Molecular Cell Biology (2022)
-
Beyond the canonical role of TFIIB in eukaryotic transcription
Current Genetics (2022)
-
Avian influenza viruses suppress innate immunity by inducing trans-transcriptional readthrough via SSU72
Cellular & Molecular Immunology (2022)