Key Points
-
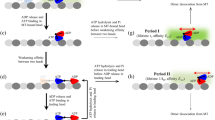
Kinesins are a superfamily of molecular motors that generate force and/or motility along microtubule filaments. Kinesins contain a conserved motor domain for ATP hydrolysis and microtubule binding, and divergent non-motor domains for family-specific regulatory mechanisms and functions.
-
The regulation of kinesin motors ensures their proper spatial and temporal activation. A general model is emerging in which kinesin motors are kept inactive by autoinhibitory mechanisms. Recent work has shown that this autoinhibition can be relieved by cargo binding and/or phosphorylation mechanisms.
-
Some kinesin motors drive intracellular transport events during interphase. The association of motors with specific cargos for transport can be regulated by kinases and Rab GTPases. Kinesin-driven cargo transport can be influenced by the state of the microtubule tracks, specifically by post-translational modifications or microtubule-associated proteins on the tracks. The release of cargo at the destination can be regulated by kinases that are part of signalling pathways, which can also be cargos of the motor.
-
Kinesin motors also function in spindle assembly and chromosome segregation during mitosis. The expression levels of these kinesin motors can be regulated by controlling their synthesis anddegradation at different stages of the cell cycle.
-
Mitotic kinesin motors can also be regulated by the small GTPase Ran and by cyclin-dependent kinase, Aurora and Polo-like kinases. These regulatory mechanisms ensure the proper localization and activation of kinesin motors at specific stages of the mitotic cycle.
Abstract
Kinesins are a family of molecular motors that use the energy of ATP hydrolysis to move along the surface of, or destabilize, microtubule filaments. Much progress has been made in understanding the mechanics and functions of the kinesin motors that play important parts in cell division, cell motility, intracellular trafficking and ciliary function. How kinesins are regulated in cells to ensure the temporal and spatial fidelity of their microtubule-based activities is less well understood. Recent work has revealed molecular mechanisms that control kinesin autoinhibition and subsequent activation, binding to cargos and microtubule tracks, and localization at specific sites of action.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gerdes, J. M., Davis, E. E. & Katsanis, N. The vertebrate primary cilium in development, homeostasis, and disease. Cell 137, 32–45 (2009).
Salinas, S., Bilsland, L. G. & Schiavo, G. Molecular landmarks along the axonal route: axonal transport in health and disease. Curr. Opin. Cell Biol. 20, 445–453 (2008).
Sarli, V. & Giannis, A. Targeting the kinesin spindle protein: basic principles and clinical implications. Clin. Cancer Res. 14, 7583–7587 (2008).
Wood, K. W., Chua, P., Sutton, D. & Jackson, J. R. Centromere-associated protein E: a motor that puts the brakes on the mitotic checkpoint. Clin. Cancer Res. 14, 7588–7592 (2008).
Lawrence, C. J. et al. A standardized kinesin nomenclature. J. Cell Biol. 167, 19–22 (2004).
Wickstead, B. & Gull, K. A “holistic” kinesin phylogeny reveals new kinesin families and predicts protein functions. Mol. Biol. Cell 17, 1734–1743 (2006).
Gupta, M. L. Jr., Carvalho, P., Roof, D. M. & Pellman, D. Plus end-specific depolymerase activity of Kip3, a kinesin-8 protein, explains its role in positioning the yeast mitotic spindle. Nature Cell Biol. 8, 913–923 (2006).
Varga, V. et al. Yeast kinesin-8 depolymerizes microtubules in a length-dependent manner. Nature Cell Biol. 8, 957–962 (2006).
Mayr, M. I. et al. The human kinesin Kif18A is a motile microtubule depolymerase essential for chromosome congression. Curr. Biol. 17, 488–98 (2007).
Okada, Y. & Hirokawa, N. A processive single-headed motor: kinesin superfamily protein KIF1A. Science 283, 1152–7 (1999).
Helenius, J., Brouhard, G., Kalaidzidis, Y., Diez, S. & Howard, J. The depolymerizing kinesin MCAK uses lattice diffusion to rapidly target microtubule ends. Nature 441, 115–119 (2006).
Kwok, B. H. et al. Allosteric inhibition of kinesin-5 modulates its processive directional motility. Nature Chem. Biol. 2, 480–485 (2006).
Kim, Y., Heuser, J. E., Waterman, C. M. & Cleveland, D. W. CENP-E combines a slow, processive motor and a flexible coiled coil to produce an essential motile kinetochore tether. J. Cell Biol. 181, 411–419 (2008).
Furuta, K., Edamatsu, M., Maeda, Y. & Toyoshima, Y. Y. Diffusion and directed movement: in vitro motile properties of fission yeast kinesin-14 Pkl1. J. Biol. Chem. 283, 36465–36473 (2008).
Furuta, K. & Toyoshima, Y. Y. Minus-end-directed motor Ncd exhibits processive movement that is enhanced by microtubule bundling in vitro. Curr. Biol. 18, 152–157 (2008).
Hammond, J. W. et al. Mammalian Kinesin-3 motors are dimeric in vivo and move by processive motility upon release of autoinhibition. PLoS Biology 7, e72 (2009).
Fink, G. et al. The mitotic kinesin-14 Ncd drives directional microtubule–microtubule sliding. Nature Cell Biol. 11, 717–723 (2009).
Hirokawa, N. et al. Submolecular domains of bovine brain kinesin identified by electron microscopy and monoclonal antibody decoration. Cell 56, 867–878 (1989).
Hackney, D. D., Levitt, J. D. & Suhan, J. Kinesin undergoes a 9S to 6S conformational transition. J. Biol. Chem. 267, 8696–8701 (1992).
Verhey, K. J. et al. Light chain-dependent regulation of Kinesin's interaction with microtubules. J. Cell Biol. 143, 1053–1066 (1998).
Stock, M. F. et al. Formation of the compact confomer of kinesin requires a COOH-terminal heavy chain domain and inhibits microtubule-stimulated ATPase activity. J. Biol. Chem. 274, 14617–14623 (1999).
Coy, D. L., Hancock, W. O., Wagenbach, M. & Howard, J. Kinesin's tail domain is an inhibitory regulator of the motor domain. Nature Cell Biol. 1, 288–292 (1999).
Friedman, D. S. & Vale, R. D. Single-molecule analysis of kinesin motility reveals regulation by the cargo-binding tail domain. Nature Cell Biol. 1, 293–297 (1999).
Seiler, S. et al. Cargo binding and regulatory sites in the tail of fungal conventional kinesin. Nature Cell Biol. 2, 333–338 (2000).
Cai, D., Hoppe, A. D., Swanson, J. A. & Verhey, K. J. Kinesin-1 structural organization and conformational changes revealed by FRET stoichiometry in live cells. J. Cell Biol. 176, 51–63 (2007).
Okada, Y., Yamazaki, H., Sekine-Aizawa, Y. & Hirokawa, N. The neuron-specific kinesin superfamily protein KIF1A is a unique monomeric motor for anterograde axonal transport of synaptic vesicle precursors. Cell 81, 769–80 (1995).
Imanishi, M., Endres, N. F., Gennerich, A. & Vale, R. D. Autoinhibition regulates the motility of the C. elegans intraflagellar transport motor OSM-3. J. Cell Biol. 174, 931–937 (2006).
Espeut, J. et al. Phosphorylation relieves autoinhibition of the kinetochore motor Cenp-E. Mol. Cell 29, 637–643 (2008).
Wong, Y.L., Dietrich, K.A., Naber, N., Cooke, R. & Rice, S.E. The Kinesin-1 tail conformationally restricts the nucleotide pocket. Biophys. J. 96, 2799–2807 (2009).
Hackney, D. D., Baek, N. & Snyder, A. C. Half-site inhibition of dimeric kinesin head domains by monomeric tail domains. Biochemistry 48, 3448–3456 (2009).
Hackney, D. D. & Stock, M. F. Kinesin's IAK tail domain inhibits initial microtubule-stimulated ADP release. Nature Cell Biol. 2, 257–260 (2000).
Dietrich, K. A. et al. The kinesin-1 motor protein is regulated by a direct interaction of its head and tail. Proc. Natl Acad. Sci. USA 105, 8938–8943 (2008). Identifies a direct interaction between the IAK segment in the KHC tail and the enzymatically crucial Switch I helix in the KHC motor of a Kinesin-1. This work provides a detailed molecular mechanism for Kinesin-1 autoinhibition and is a further example of the mechanistic homology between kinesins and small GTPases.
Hackney, D. D. & Stock, M. F. Kinesin tail domains and Mg2+ directly inhibit release of ADP from head domains in the absence of microtubules. Biochemistry 47, 7770–7778 (2008).
Vale, R. D. Switches, latches, and amplifiers: common themes of G proteins and molecular motors. J. Cell Biol. 135, 291–302 (1996).
Lee, J. R. et al. An intramolecular interaction between the FHA domain and a coiled coil negatively regulates the kinesin motor KIF1A. EMBO J. 23, 1506–1515 (2004).
Yamada, K. H., Hanada, T. & Chishti, A. H. The effector domain of human Dlg tumor suppressor acts as a switch that relieves autoinhibition of kinesin-3 motor GAKIN/KIF13B. Biochemistry 46, 10039–10045 (2007).
Bathe, F. et al. The complex interplay between the neck and hinge domains in kinesin-1 dimerization and motor activity. Mol. Biol. Cell 16, 3529–3537 (2005).
Blasius, T. L., Cai, D., Jih, G. T., Toret, C. P. & Verhey, K. J. Two binding partners cooperate to activate the molecular motor Kinesin-1. J. Cell Biol. 176, 11–17 (2007). Together with reference 28, this paper provides the first demonstration of how autoinhibition of a kinesin motor is released for activation of motility. Reference 28 shows that autoinhibition of the kinesin-7 motor CENPE is relieved by phosphorylation of the tail domain, whereas reference 38 shows that binding partners of both the KHC and the KLC inhibitory regions are required for activation of a Kinesin-1.
Cho, K. I. et al. RANBP2 is an allosteric activator of the conventional kinesin-1 motor protein, KIF5B, in a minimal cell-free system. EMBO Rep. 10, 480–486 (2009).
Klopfenstein, D. R., Tomishige, M., Stuurman, N. & Vale, R. D. Role of phosphatidylinositol(4, 5)bisphosphate organization in membrane transport by the Unc104 kinesin motor. Cell 109, 347–358 (2002).
Tomishige, M., Klopfenstein, D. R. & Vale, R. D. Conversion of Unc104/KIF1A kinesin into a processive motor after dimerization. Science 297, 2263–2267 (2002).
Adio, S. et al. Kinetic and mechanistic basis of the nonprocessive Kinesin-3 motor NcKin3. J. Biol. Chem. 281, 37782–37793 (2006).
Dorner, C., Ullrich, A., Haring, H. U. & Lammers, R. The kinesin-like motor protein KIF1C occurs in intact cells as a dimer and associates with proteins of the 14-3-3 family. J. Biol. Chem. 274, 33654–33660 (1999).
Pollock, N., de Hostos, E. L., Turck, C. W. & Vale, R. D. Reconstitution of membrane transport powered by a novel dimeric kinesin motor of the Unc104/KIF1A family purified from Dictyostelium. J. Cell Biol. 147, 493–506 (1999).
Wozniak, M. J. & Allan, V. J. Cargo selection by specific kinesin light chain 1 isoforms. EMBO J. 25, 5457–5468 (2006).
Cahu, J. et al. Phosphorylation by Cdk1 increases the binding of Eg5 to microtubules in vitro and in Xenopus egg extract spindles. PLoS ONE 3, e3936 (2008).
Kapitein, L. C. et al. Microtubule cross-linking triggers the directional motility of kinesin-5. J. Cell Biol. 182, 421–428 (2008).
Hirokawa, N., Noda, Y., Tanaka, Y. & Niwa, S. Kinesin superfamily motor proteins and intracellular transport. Nature Rev. Mol. Cell Biol. 10, 682–696 (2009).
Hoepfner, S. et al. Modulation of receptor recycling and degradation by the endosomal kinesin KIF16B. Cell 121, 437–50 (2005).
Toda, H. et al. UNC-51/ATG1 kinase regulates axonal transport by mediating motor-cargo assembly. Genes Dev. 22, 3292–3307 (2008).
Niwa, S., Tanaka, Y. & Hirokawa, N. KIF1Bβ- and KIF1A-mediated axonal transport of presynaptic regulator Rab3 occurs in a GTP-dependent manner through DENN/MADD. Nature Cell Biol. 10, 1269–1279 (2008).
Caviston, J. P. & Holzbaur, E. L. Microtubule motors at the intersection of trafficking and transport. Trends Cell Biol. 16, 530–537 (2006).
Bowman, A. B. et al. Kinesin-dependent axonal transport is mediated by the sunday driver (SYD) protein. Cell 103, 583–594 (2000).
Verhey, K. J. et al. Cargo of kinesin identified as JIP scaffolding proteins and associated signaling molecules. J. Cell Biol. 152, 959–970 (2001).
Byrd, D. T. et al. UNC-16, a JNK-signaling scaffold protein, regulates vesicle transport in C. elegans. Neuron 32, 787–800 (2001).
Horiuchi, D. et al. Control of a kinesin-cargo linkage mechanism by JNK pathway kinases. Curr. Biol. 17, 1313–1317 (2007).
Cavalli, V., Kujala, P., Klumperman, J. & Goldstein, L. S. Sunday Driver links axonal transport to damage signaling. J. Cell Biol. 168, 775–787 (2005).
Morfini, G. et al. JNK mediates pathogenic effects of polyglutamine-expanded androgen receptor on fast axonal transport. Nature Neurosci. 907, 907–916 (2006).
Stagi, M., Gorlovoy, P., Larionov, S., Takahashi, K. & Neumann, H. Unloading kinesin transported cargoes from the tubulin track via the inflammatory c-Jun N-terminal kinase pathway. Faseb J. 20, 2573–2575 (2006).
Morfini, G. A. et al. Pathogenic huntingtin inhibits fast axonal transport by activating JNK3 and phosphorylating kinesin. Nature Neurosci. 12, 864–871 (2009).
Bengs, F., Scholz, A., Kuhn, D. & Wiese, M. LmxMPK9, a mitogen-activated protein kinase homologue affects flagellar length in Leishmania mexicana. Mol. Microbiol. 55, 1606–1615 (2005).
Berman, S. A., Wilson, N. F., Haas, N. A. & Lefebvre, P. A. A novel MAP kinase regulates flagellar length in Chlamydomonas. Curr. Biol. 13, 1145–1149 (2003).
Burghoorn, J. et al. Mutation of the MAP kinase DYF-5 affects docking and undocking of kinesin-2 motors and reduces their speed in the cilia of Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 104, 7157–7162 (2007).
Guillaud, L., Wong, R. & Hirokawa, N. Disruption of KIF17-Mint1 interaction by CaMKII-dependent phosphorylation: a molecular model of kinesin-cargo release. Nature Cell Biol. 10, 19–29 (2008). Together with reference 56, this paper provides the first direct evidence of a mechanism for the release of a cargo from its motor at its destination. In both cases, a signalling cascade results in release of the cargo from its motor. In reference 56, components of the regulatory signalling pathway are also cargos of the motor they regulate, whereas in reference 64, the signalling cascade is localized at the motor–cargo's destination.
Midorikawa, R., Takei, Y. & Hirokawa, N. KIF4 motor regulates activity-dependent neuronal survival by suppressing PARP-1 enzymatic activity. Cell 125, 371–383 (2006).
Morfini, G., Szebenyi, G., Elluru, R., Ratner, N. & Brady, S. T. Glycogen synthase kinase 3 phosphorylates kinesin light chains and negatively regulates kinesin-based motility. EMBO J. 21, 281–293 (2002).
Nakata, T. & Hirokawa, N. Microtubules provide directional cues for polarized axonal transport through interaction with kinesin motor head. J. Cell Biol. 162, 1045–1055 (2003).
Jacobson, C., Schnapp, B. & Banker, G. A. A change in the selective translocation of the Kinesin-1 motor domain marks the initial specification of the axon. Neuron 49, 797–804 (2006).
Cai, D., McEwen, D. P., Martens, J. R., Meyhofer, E. & Verhey, K. J. Single molecule imaging reveals differences in microtubule track selection between kinesin motors. PLoS Biol. 13 Oct 2009 (doi:10.1371/journal.pbio.1000216).
Dunn, S. et al. Differential trafficking of Kif5c on tyrosinated and detyrosinated microtubules in live cells. J. Cell Sci. 121, 1085–1095 (2008).
Reed, N. A. et al. Microtubule acetylation promotes kinesin-1 binding and transport. Curr. Biol. 16, 2166–2172 (2006).
Konishi, Y. & Setou, M. Tubulin tyrosination navigates the kinesin-1 motor domain to axons. Nature Neurosci. 12, 559–567 (2009). References 69–72 show that Kinesin-1 motors can be influenced by post-translational modifications of tubulin subunits in microtubule tracks. Kinesin-1 motors prefer microtubules marked by detyrosination and/or acetylation in unpolarized cells. Detyrosination provides a polarity cue that directs Kinesin-1 motors to axons in neurons.
Lin, S. X., Gundersen, G. G. & Maxfield, F. R. Export from pericentriolar endocytic recycling compartment to cell surface depends on stable, detyrosinated (glu) microtubules and kinesin. Mol. Biol. Cell 13, 96–109 (2002).
Dompierre, J. P. et al. Histone deacetylase 6 inhibition compensates for the transport deficit in Huntington's disease by increasing tubulin acetylation. J. Neurosci. 27, 3571–3583 (2007).
Hammond, J. W., Cai, D & Verhey, K.J. Tubulin modifications and their cellular functions. Curr. Opin. Cell. Biol. 20, 71–76 (2009).
Ikegami, K. et al. Loss of α-tubulin polyglutamylation in ROSA22 mice is associated with abnormal targeting of KIF1A and modulated synaptic function. Proc. Natl Acad. Sci. USA 104, 3213–8 (2007).
Verhey, K. J. & Gaertig, J. The tubulin code. Cell Cycle 6, 2152–2160 (2007).
Ebneth, A. et al. Overexpression of tau protein inhibits kinesin-dependent trafficking of vesicles, mitochondria, and endoplasmic reticulum: implications for Alzheimer's disease. J. Cell Biol. 143, 777–794 (1998).
Trinczek, B., Ebneth, A., Mandelkow, E. M. & Mandelkow, E. Tau regulates the attachment/detachment but not the speed of motors in microtubule-dependent transport of single vesicles and organelles. J. Cell Sci. 112, 2355–2367 (1999).
Stamer, K., Vogel, R., Thies, E., Mandelkow, E. & Mandelkow, E. M. Tau blocks traffic of organelles, neurofilaments, and APP vesicles in neurons and enhances oxidative stress. J. Cell Biol. 156, 1051–1063 (2002).
Vershinin, M., Carter, B. C., Razafsky, D. S., King, S. J. & Gross, S. P. Multiple-motor based transport and its regulation by Tau. Proc. Natl Acad. Sci. USA 104, 87–92 (2007).
LaPointe, N. E. et al. The amino terminus of tau inhibits kinesin-dependent axonal transport: implications for filament toxicity. J. Neurosci. Res. 87, 440–451 (2009).
Seitz, A. et al. Single-molecule investigation of the interference between kinesin, tau and MAP2c. EMBO J. 21, 4896–4905 (2002).
Dixit, R., Ross, J. L., Goldman, Y. E. & Holzbaur, E. L. Differential regulation of dynein and kinesin motor proteins by tau. Science 319, 1086–1089 (2008).
Sung, H. H. et al. Drosophila Ensconsin promotes productive recruitment of Kinesin-1 to microtubules. Dev. Cell 15, 866–876 (2008).
Stowers, R. S., Megeath, L. J., Gorska-Andrzejak, J., Meinertzhagen, I. A. & Schwarz, T. L. Axonal transport of mitochondria to synapses depends on Milton, a novel Drosophila protein. Neuron 36, 1063–1077 (2002).
Brickley, K., Smith, M. J., Beck, M. & Stephenson, F. A. GRIF-1 and OIP106, members of a novel gene family of coiled-coil domain proteins: association in vivo and in vitro with kinesin. J. Biol. Chem. 280, 14723–14732 (2005).
Guo, X. et al. The GTPase dMiro is required for axonal transport of mitochondria to Drosophila synapses. Neuron 47, 379–393 (2005).
Glater, E. E., Megeath, L. J., Stowers, R. S. & Schwarz, T. L. Axonal transport of mitochondria requires Milton to recruit kinesin heavy chain and is light chain independent. J. Cell Biol. 173, 545–557 (2006).
Macaskill, A. F. et al. Miro1 is a calcium sensor for glutamate receptor-dependent localization of mitochondria at synapses. Neuron 61, 541–555 (2009).
Wang, X. & Schwarz, T. L. The mechanism of Ca2+ -dependent regulation of kinesin-mediated mitochondrial motility. Cell 136, 163–174 (2009). References 90 and 91 show that the motor–microtubule interaction can be regulated in order to position a cargo, in this case mitochondria, at specific subcellular positions. Signalling pathways that locally increase intracellular Ca2+ levels work through the mitochondrial membrane protein Miro to release Miro-associated kinesin motors from their microtubule tracks.
Saotome, M. et al. Bidirectional Ca2+-dependent control of mitochondrial dynamics by the Miro GTPase. Proc. Natl Acad. Sci. USA 105, 20728–20733 (2008).
Deavours, B. E., Reddy, A. S. & Walker, R. A. Ca2+/calmodulin regulation of the Arabidopsis kinesin-like calmodulin-binding protein. Cell. Motil. Cytoskeleton 40, 408–416 (1998).
Vinogradova, M. V., Malanina, G. G., Reddy, V. S., Reddy, A. S. & Fletterick, R. J. Structural dynamics of the microtubule binding and regulatory elements in the kinesin-like calmodulin binding protein. J. Struct. Biol. 163, 76–83 (2008).
Brown, K. D., Coulson, R. M., Yen, T. J. & Cleveland, D. W. Cyclin-like accumulation and loss of the putative kinetochore motor CENP-E results from coupling continuous synthesis with specific degradation at the end of mitosis. J. Cell Biol. 125, 1303–1312 (1994).
Funabiki, H. & Murray, A. W. The Xenopus chromokinesin Xkid is essential for metaphase chromosome alignment and must be degraded to allow anaphase chromosome movement. Cell 102, 411–424 (2000).
Fontijn, R. D. et al. The human kinesin-like protein RB6K is under tight cell cycle control and is essential for cytokinesis. Mol. Cell. Biol. 21, 2944–2955 (2001).
Levesque, A. A. & Compton, D. A. The chromokinesin Kid is necessary for chromosome arm orientation and oscillation, but not congression, on mitotic spindles. J. Cell Biol. 154, 1135–1146 (2001).
Carvalho, P., Gupta, M. L. Jr., Hoyt, M. A. & Pellman, D. Cell cycle control of kinesin-mediated transport of Bik1 (CLIP-170) regulates microtubule stability and dynein activation. Dev. Cell 6, 815–829 (2004).
Feine, O., Zur, A., Mahbubani, H. & Brandeis, M. Human Kid is degraded by the APC/C(Cdh1) but not by the APC/C(Cdc20). Cell Cycle 6, 2516–2523 (2007).
Ganguly, A., Bhattacharya, R. & Cabral, F. Cell cycle dependent degradation of MCAK: evidence against a role in anaphase chromosome movement. Cell Cycle 7, 3187–3193 (2008).
Seguin, L. et al. CUX1 and E2F1 regulate coordinated expression of the mitotic complex genes Ect2, MgcRacGAP, and MKLP1 in S phase. Mol. Cell. Biol. 29, 570–81 (2009).
Antonio, C. et al. Xkid, a chromokinesin required for chromosome alignment on the metaphase plate. Cell 102, 425–435 (2000).
Goshima, G. & Vale, R. D. Cell cycle-dependent dynamics and regulation of mitotic kinesins in Drosophila S2 cells. Mol. Biol. Cell 16, 3896–3907 (2005). An RNA interference based rescue strategy was used to examine the localization and function of mitotic kinesins in D. melanogaster S2 cells. Shows that Kinesin-8 and Kinesin-14 motors are sequestered in an active form in the nucleus during interphase and engage their microtubule targets on nuclear envelope breakdown, that the interactions of Kinesin-5 and Kinesin-6 motors with microtubules are regulated by CDC2 phosphorylation, and that Kinesin-8 and Kinesin-13 motors undergo regulated changes in subcellular localization throughout the cell cycle.
Liu, X. & Erikson, R. L. The nuclear localization signal of mitotic kinesin-like protein Mklp-1: effect on Mklp-1 function during cytokinesis. Biochem. Biophys. Res. Commun. 353, 960–964 (2007).
Cai, S., Weaver, L. N., Ems-McClung, S. C. & Walczak, C. E. Kinesin-14 family proteins HSET/XCTK2 control spindle length by cross-linking and sliding microtubules. Mol. Biol. Cell 20, 1348–1359 (2009).
Brown, K. D., Wood, K. W. & Cleveland, D. W. The kinesin-like protein CENP-E is kinetochore-associated throughout poleward chromosome segregation during anaphase-A. J. Cell Sci. 109, 961–969 (1996).
Unsworth, A., Masuda, H., Dhut, S. & Toda, T. Fission yeast kinesin-8 Klp5 and Klp6 are interdependent for mitotic nuclear retention and required for proper microtubule dynamics. Mol. Biol. Cell 19, 5104–5115 (2008).
Clarke, P. R. & Zhang, C. Spatial and temporal coordination of mitosis by Ran GTPase. Nature Rev. Mol. Cell Biol. 9, 464–477 (2008).
Tahara, K. et al. Importin-β and the small guanosine triphosphatase Ran mediate chromosome loading of the human chromokinesin Kid. J. Cell Biol. 180, 493–506 (2008).
Trieselmann, N., Armstrong, S., Rauw, J. & Wilde, A. Ran modulates spindle assembly by regulating a subset of TPX2 and Kid activities including Aurora A activation. J. Cell Sci. 116, 4791–4798 (2003).
Ems-McClung, S. C., Zheng, Y. & Walczak, C. E. Importin α/β and Ran-GTP regulate XCTK2 microtubule binding through a bipartite nuclear localization signal. Mol. Biol. Cell 15, 46–57 (2004). Together with reference 110, this paper provides biochemical and functional evidence that the GTPase Ran regulates kinesin motors during spindle assembly. Reference 110 shows that importin-α, importin-β and RanGTP facilitate the chromosome loading of Kinesin-10 motors, and reference 112 shows that this pathway also controls the microtubule-binding activity of Kinesin-14 motors.
Manning, B. D., Barrett, J. G., Wallace, J. A., Granok, H. & Snyder, M. Differential regulation of the Kar3p kinesin-related protein by two associated proteins, Cik1p and Vik1p. J. Cell Biol. 144, 1219–1233 (1999).
Tanaka, K. et al. Molecular mechanisms of kinetochore capture by spindle microtubules. Nature 434, 987–994 (2005).
Allingham, J. S., Sproul, L. R., Rayment, I. & Gilbert, S. P. Vik1 modulates microtubule-Kar3 interactions through a motor domain that lacks an active site. Cell 128, 1161–1172 (2007).
Gardner, M. K. et al. The microtubule-based motor Kar3 and plus end-binding protein Bim1 provide structural support for the anaphase spindle. J. Cell Biol. 180, 91–100 (2008).
Benanti, J. A., Matyskiela, M. E., Morgan, D. O. & Toczyski, D. P. Functionally distinct isoforms of Cik1 are differentially regulated by APC/C-mediated proteolysis. Mol. Cell 33, 581–590 (2009).
Sproul, L. R., Anderson, D. J., Mackey, A. T., Saunders, W. S. & Gilbert, S. P. Cik1 targets the minus-end kinesin depolymerase kar3 to microtubule plus ends. Curr. Biol. 15, 1420–1427 (2005). Together with reference 113, this paper describes the regulation of kinesin localization and function in yeast. Reference 113 shows that Kar3 forms separate complexes with Cik1 and Vik1 and that these accessory proteins target Kar3 to different subcellular locales. Reference 118 also provides important insight into the mechanochemistry of Kar3 by showing that Cik1 targets Kar3 to microtubule plus ends, where minus end-directed motility is coupled to microtubule depolymerization.
Blangy, A. et al. Phosphorylation by p34cdc2 regulates spindle association of human Eg5, a kinesin-related motor essential for bipolar spindle formation in vivo. Cell 83, 1159–1169 (1995).
Sawin, K. E. & Mitchison, T. Mutations in the kinesin-like protein Eg5 disrupting localization to the mitotic spindle. Proc. Natl Acad. Sci. USA 92, 4289–4293 (1995).
Sharp, D. J. et al. The bipolar kinesin, KLP61F, cross-links microtubules within interpolar microtubule bundles of Drosophila embryonic mitotic spindles. J. Cell Biol. 144, 125–38 (1999).
Giet, R., Uzbekov, R., Cubizolles, F., Le Guellec, K. & Prigent, C. The Xenopus laevis aurora-related protein kinase pEg2 associates with and phosphorylates the kinesin-related protein XlEg5. J. Biol. Chem. 274, 15005–15013 (1999).
Bishop, J. D., Han, Z. & Schumacher, J. M. The Caenorhabditis elegans Aurora B kinase AIR-2 phosphorylates and is required for the localization of a BimC kinesin to meiotic and mitotic spindles. Mol. Biol. Cell 16, 742–756 (2005).
Rapley, J. et al. The NIMA-family kinase Nek6 phosphorylates the kinesin Eg5 at a novel site necessary for mitotic spindle formation. J. Cell Sci. 121, 3912–3921 (2008).
Boleti, H., Karsenti, E. & Vernos, I. Xklp2, a novel Xenopus centrosomal kinesin-like protein required for centrosome separation during mitosis. Cell 84, 49–59 (1996).
Wittmann, T., Boleti, H., Antony, C., Karsenti, E. & Vernos, I. Localization of the kinesin-like protein Xklp2 to spindle poles requires a leucine zipper, a microtubule-associated protein, and dynein. J. Cell Biol. 143, 673–685 (1998).
Wittmann, T., Wilm, M., Karsenti, E. & Vernos, I. TPX2, A novel Xenopus MAP involved in spindle pole organization. J. Cell Biol. 149, 1405–1418 (2000).
Liu, D. et al. Human NUF2 interacts with centromere-associated protein E and is essential for a stable spindle microtubule-kinetochore attachment. J. Biol. Chem. 282, 21415–21424 (2007).
Zhang, X. D. et al. SUMO-2/3 modification and binding regulate the association of CENP-E with kinetochores and progression through mitosis. Mol. Cell 29, 729–741 (2008).
Zhu, M. et al. Septin 7 interacts with centromere-associated protein E and is required for its kinetochore localization. J. Biol. Chem. 283, 18916–18925 (2008).
Ashar, H. R. et al. Farnesyl transferase inhibitors block the farnesylation of CENP-E and CENP-F and alter the association of CENP-E with the microtubules. J. Biol. Chem. 275, 30451–30457 (2000).
Schafer-Hales, K. et al. Farnesyl transferase inhibitors impair chromosomal maintenance in cell lines and human tumors by compromising CENP-E and CENP-F function. Mol. Cancer Ther. 6, 1317–1328 (2007).
Sampath, S. C. et al. The chromosomal passenger complex is required for chromatin-induced microtubule stabilization and spindle assembly. Cell 118, 187–202 (2004).
Tulu, U. S., Fagerstrom, C., Ferenz, N. P. & Wadsworth, P. Molecular requirements for kinetochore-associated microtubule formation in mammalian cells. Curr. Biol. 16, 536–541 (2006).
Andrews, P. D. et al. Aurora B regulates MCAK at the mitotic centromere. Dev. Cell 6, 253–268 (2004).
Lan, W. et al. Aurora B phosphorylates centromeric MCAK and regulates its localization and microtubule depolymerization activity. Curr. Biol. 14, 273–286 (2004).
Ohi, R., Sapra, T., Howard, J. & Mitchison, T. J. Differentiation of cytoplasmic and meiotic spindle assembly MCAK functions by Aurora B-dependent phosphorylation. Mol. Biol. Cell 15, 2895–2906 (2004). References 135–137 identify Aurora B sites of phosphorylation on the Kinesin-13 family member MCAK and show that Aurora B regulates the localization and activity of MCAK.
Knowlton, A. L., Lan, W. & Stukenberg, P. T. Aurora B is enriched at merotelic attachment sites, where it regulates MCAK. Curr. Biol. 16, 1705–1710 (2006).
Zhang, X., Lan, W., Ems-McClung, S. C., Stukenberg, P. T. & Walczak, C. E. Aurora B phosphorylates multiple sites on mitotic centromere-associated kinesin to spatially and temporally regulate its function. Mol. Biol. Cell 18, 3264–3276 (2007).
Ohi, R., Coughlin, M. L., Lane, W. S. & Mitchison, T. J. An inner centromere protein that stimulates the microtubule depolymerizing activity of a KinI kinesin. Dev. Cell 5, 309–321 (2003).
Knowlton, A. L., Vorozhko, V. V., Lan, W., Gorbsky, G. J. & Stukenberg, P. T. ICIS and Aurora B coregulate the microtubule depolymerase Kif2a. Curr. Biol. 19, 758–763 (2009).
Jang, C. Y., Coppinger, J. A., Seki, A., Yates, J. R., & Fang, G. Plk1 and Aurora A regulate the depolymerase activity and the cellular localization of Kif2a. J. Cell Sci. 122, 1334–1341 (2009).
Mishima, M., Pavicic, V., Gruneberg, U., Nigg, E. A. & Glotzer, M. Cell cycle regulation of central spindle assembly. Nature 430, 908–913 (2004). Shows that CDK1–cyclin B phosphorylates the motor domain of Kinesin-6 motors and reduces their affinity for microtubules. Degradation of CDK1–cyclin B at anaphase ensures the proper spatial and temporal activation of Kinesin-6 motors.
Severson, A. F., Hamill, D. R., Carter, J. C., Schumacher, J. & Bowerman, B. The aurora-related kinase AIR-2 recruits ZEN-4/CeMKLP1 to the mitotic spindle at metaphase and is required for cytokinesis. Curr. Biol. 10, 1162–1171 (2000).
Giet, R. & Glover, D. M. Drosophila aurora B kinase is required for histone H3 phosphorylation and condensin recruitment during chromosome condensation and to organize the central spindle during cytokinesis. J. Cell Biol. 152, 669–682 (2001).
Petronczki, M., Glotzer, M., Kraut, N. & Peters, J. M. Polo-like kinase 1 triggers the initiation of cytokinesis in human cells by promoting recruitment of the RhoGEF Ect2 to the central spindle. Dev. Cell 12, 713–725 (2007).
Santamaria, A. et al. Use of the novel Plk1 inhibitor ZK-thiazolidinone to elucidate functions of Plk1 in early and late stages of mitosis. Mol. Biol. Cell 18, 4024–4036 (2007).
Guse, A., Mishima, M. & Glotzer, M. Phosphorylation of ZEN-4/MKLP1 by Aurora B regulates completion of cytokinesis. Curr. Biol. 15, 778–786 (2005).
Neef, R. et al. Phosphorylation of mitotic kinesin-like protein 2 by polo-like kinase 1 is required for cytokinesis. J. Cell Biol. 162, 863–875 (2003).
Glotzer, M. The 3Ms of central spindle assembly: microtubules, motors and MAPs. Nature Rev. Mol. Cell Biol. 10, 9–20 (2009).
Walczak, C. E. & Heald, R. Mechanisms of mitotic spindle assembly and function. Int. Rev. Cytol. 265, 111–158 (2008).
Gennerich, A. & Vale, R. D. Walking the walk: how kinesin and dynein coordinate their steps. Curr. Opin. Cell Biol. 21, 59–67 (2009).
Moores, C. A. & Milligan, R. A. Lucky 13-microtubule depolymerisation by kinesin-13 motors. J. Cell Sci. 119, 3905–3913 (2006).
Valentine, M. T. & Gilbert, S. P. To step or not to step? How biochemistry and mechanics influence processivity in Kinesin and Eg5. Curr. Opin. Cell Biol. 19, 75–81 (2007).
Mazumdar, M. & Misteli, T. Chromokinesins: multitalented players in mitosis. Trends Cell Biol. 15, 349–355 (2005).
Hirokawa, N. & Noda, Y. Intracellular transport and kinesin superfamily proteins, KIFs: structure, function, and dynamics. Physiol. Rev. 88, 1089–1118 (2008).
Goldstein, A. Y., Wang, X. & Schwarz, T. L. Axonal transport and the delivery of pre-synaptic components. Curr. Opin. Neurobiol. 18, 495–503 (2008).
Aspengren, S., Hedberg, D., Skold, H. N. & Wallin, M. New insights into melanosome transport in vertebrate pigment cells. Int. Rev. Cell. Mol. Biol. 272, 245–302 (2009).
Shubeita, G. T. et al. Consequences of motor copy number on the intracellular transport of kinesin-1-driven lipid droplets. Cell 135, 1098–1107 (2008).
Scholey, J. M. Intraflagellar transport motors in cilia: moving along the cell's antenna. J. Cell Biol. 180, 23–29 (2008).
Acknowledgements
We are grateful to D. Cai, J. Howard, C. Walczak and S. Gilbert for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Ciliopathies
-
A collection of human diseases that have a common underlying genetic defect in the assembly or function of cilia.
- Coiled coil
-
A structural motif in proteins, often used to control oligomerization, in which two or more coils — α-helical seven amino acid (heptad) repeats — wrap around each other.
- Kinesin heavy chain
-
The catalytic subunit of a Kinesin-1 motor, the domain organization of which consists of a kinesin motor domain, a coiled-coil stalk and a globular tail.
- Kinesin light chain
-
The accessory subunit of a Kinesin-1 motor that contributes to autoinhibition and is important for binding to some cargos.
- MAPK family
-
(Mitogen-activated protein kinase family). A family of Ser/Thr-specific protein kinases that respond to extracellular signals and regulate a range of intracellular events. The MAPK is the third kinase in a cascade and is activated by a MAPK kinase (MAPKK), which is activated by a MAPKK kinase (MAPKKK).
- Intraflagellar transport
-
The system for bidirectional movement of ciliary components along the ciliary axoneme by Kinesin-2 and cytoplasmic dynein motors.
- EF hand
-
A structural domain in proteins that binds to Ca2+ ions.
- Guanine nucleotide exchange factor
-
A protein that facilitates the exchange of GDP for GTP in the nucleotide-binding pocket of a G protein.
- Karyogamy
-
The process during mating in which two haploid nuclei come together and fuse to form a diploid nucleus.
- Centrosome
-
A microtubule organizing centre that contains centrioles and the pericentriolar material that nucleates microtubule polymerization. It duplicates during S phase to create the spindle poles for mitosis.
- Metaphase plate
-
The plane at mid-spindle, perpendicular to the spindle microtubules, where the chromosomes are positioned during metaphase.
- Kinetochore
-
A multiprotein complex that assembles on the centromeric region of chromosomes and attaches to spindle microtubules. The inner kinetochore contains proteins that are tightly and persistently associated with centromeric DNA, whereas the outer kinetochore contains dynamic protein components that interact with microtubules during mitosis.
- Mitotic checkpoint
-
A control mechanism in metaphase that ensures that all chromosomes are properly attached to both spindle poles before the cell can proceed into anaphase.
- Chromosome congression
-
The process by which chromosomes attach to spindle microtubules and align or 'congress' to the metaphase plate. It requires both microtubule dynamics and kinesin motor activities.
- Prenylation
-
A post-translational modification of a protein by the attachment of prenyl moieties (geranyl, farnesyl or geranylgeranyl groups) to a C-terminal Cys residue.
- Chromosomal passenger complex
-
A multiprotein complex that localizes to the kinetochore until early anaphase, when it switches to the central spindle. It consists of the Aurora B kinase, inner centromere protein, survivin and Borealin (also known as Dasra and CDCA8).
- Merotelic
-
Pertains to an incorrect spindle attachment in which a single kinetochore is attached to microtubules from both spindle poles.
- Amphitelic
-
Pertains to a proper spindle attachment in which each sister kinetochore is attached to the microtubules from its nearest facing spindle pole.
Rights and permissions
About this article
Cite this article
Verhey, K., Hammond, J. Traffic control: regulation of kinesin motors. Nat Rev Mol Cell Biol 10, 765–777 (2009). https://doi.org/10.1038/nrm2782
Issue Date:
DOI: https://doi.org/10.1038/nrm2782
This article is cited by
-
Cargo specificity, regulation, and therapeutic potential of cytoplasmic dynein
Experimental & Molecular Medicine (2024)
-
Motor generated torque drives coupled yawing and orbital rotations of kinesin coated gold nanorods
Communications Biology (2022)
-
The architecture of kinesin-3 KLP-6 reveals a multilevel-lockdown mechanism for autoinhibition
Nature Communications (2022)
-
Absence of DEATH kinesin is fatal for Leishmania mexicana amastigotes
Scientific Reports (2022)
-
Kinesin-1 activity recorded in living cells with a precipitating dye
Nature Communications (2021)