Key Points
-
Collective cell migration is defined as the coordinated movement of multiple cells that retain cell–cell contacts while coordinating their actin dynamics and intracellular signalling.
-
Because the cells form a structural and functional unit, both active and passive cell translocation occur.
-
The movement of connected cells contributes to morphogenesis, wound healing and cancer invasion and each process underlies homologous but distinct molecular mechanisms of cell–cell interaction and pro-migratory extracellular signalling.
-
The concept of collective movement explains how the body forms and reshapes as well as how cancer cells destructively invade as a 'socially' organized mass.
Abstract
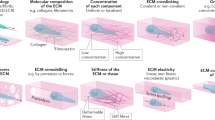
The collective migration of cells as a cohesive group is a hallmark of the tissue remodelling events that underlie embryonic morphogenesis, wound repair and cancer invasion. In such migration, cells move as sheets, strands, clusters or ducts rather than individually, and use similar actin- and myosin-mediated protrusions and guidance by extrinsic chemotactic and mechanical cues as used by single migratory cells. However, cadherin-based junctions between cells additionally maintain 'supracellular' properties, such as collective polarization, force generation, decision making and, eventually, complex tissue organization. Comparing different types of collective migration at the molecular and cellular level reveals a common mechanistic theme between developmental and cancer research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ridley, A. J. et al. Cell migration: integrating signals from front to back. Science 302, 1704–1709 (2003).
Friedl, P. & Weigelin, B. Interstitial leukocyte trafficking and immune function. Nature Immunol. 9, 839–848 (2008).
Vaughan, R. B. & Trinkaus, J. P. Movements of epithelial cell sheets in vitro. J. Cell Sci. 1, 407–413 (1966).
Friedl, P., Hegerfeldt, Y. & Tusch, M. Collective cell migration in morphogenesis and cancer. Int. J. Dev. Biol. 48, 441–449 (2004).
Friedl, P. Prespecification and plasticity: shifting mechanisms of cell migration. Curr. Opin. Cell Biol. 16, 14–23 (2004). Describes the concept of different individual and collective migration strategies in various systems that serve distinct purposes.
Montell, D. J. Morphogenetic cell movements: diversity from modular mechanical properties. Science 322, 1502–1505 (2008). A comprehensive review of recent work on the mechanics of morphogenic tissue rearrangements.
Nobes, C. D. & Hall, A. Rho GTPases control polarity, protrusion, and adhesion during cell movement. J. Cell Biol. 144, 1235–1244 (1999).
Mattila, P. K. & Lappalainen, P. Filopodia: molecular architecture and cellular functions. Nature Rev. Mol. Cell Biol. 9, 446–454 (2008).
Friedl, P. & Wolf, K. Proteolytic interstitial cell migration: a five-step process. Cancer Metastasis Rev. 28, 129–135 (2009).
Carmona-Fontaine, C. et al. Contact inhibition of locomotion in vivo controls neural crest directional migration. Nature 456, 957–961 (2008).
Farooqui, R. & Fenteany, G. Multiple rows of cells behind an epithelial wound edge extend cryptic lamellipodia to collectively drive cell-sheet movement. J. Cell Sci. 118, 51–63 (2005).
Simpson, K. J. et al. Identification of genes that regulate epithelial cell migration using an siRNA screening approach. Nature Cell Biol. 10, 1027–1038 (2008).
Gaggioli, C. et al. Fibroblast-led collective invasion of carcinoma cells with differing roles for Rho GTPases in leading and following cells. Nature Cell Biol. 9, 1392–1400 (2007).
Wolf, K. et al. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nature Cell Biol. 9, 893–904 (2007). References 13 and 14 show how collective cancer cell invasion is facilitated by tip cells; both leading cancer cells or leading stromal fibroblasts can pave the way to enhance invasion.
Lee, G. Y., Kenny, P. A., Lee, E. H. & Bissell, M. J. Three-dimensional culture models of normal and malignant breast epithelial cells. Nature Methods 4, 359–365 (2007).
Friedl, P. et al. Migration of coordinated cell clusters in mesenchymal and epithelial cancer explants in vitro. Cancer Res. 55, 4557–4560 (1995).
Masson, V. V. et al. Mouse aortic ring assay: a new approach of the molecular genetics of angiogenesis. Biol. Proced. Online 4, 24–31 (2002).
Affolter, M. et al. Tube or not tube: remodeling epithelial tissues by branching morphogenesis. Dev. Cell 4, 11–18 (2003).
Affolter, M. & Caussinus, E. Tracheal branching morphogenesis in Drosophila: new insights into cell behaviour and organ architecture. Development 135, 2055–2064 (2008).
Lecaudey, V. & Gilmour, D. Organizing moving groups during morphogenesis. Curr. Opin. Cell Biol. 18, 102–107 (2006).
Kibbey, M. C., Grant, D. S. & Kleinman, H. K. Role of the SIKVAV site of laminin in promotion of angiogenesis and tumor growth: an in vivo Matrigel model. J. Natl Cancer Inst. 84, 1633–1638 (1992).
Becker, M. D. et al. In vivo fluorescence microscopy of corneal neovascularization. Graefes Arch. Clin. Exp. Ophthalmol. 236, 390–398 (1998).
Hellstrom, M. et al. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 445, 776–780 (2007).
Siekmann, A. F. & Lawson, N. D. Notch signalling limits angiogenic cell behaviour in developing zebrafish arteries. Nature 445, 781–784 (2007). References 23 and 24 show how tip and sprout cell fate is controlled through Notch-based lateral inhibition in mouse and zebrafish, respectively. Together with evidence for tracheal development in D. melanogaster (see references 88 and 89), these findings suggest that this polarity mechanism in branching morphogenesis is highly conserved throughout evolution.
Alexander, S., Koehl, G. E., Hirschberg, M., Geissler, E. K. & Friedl, P. Dynamic imaging of cancer growth and invasion: a modified skin-fold chamber model. Histochem. Cell Biol. 130, 1147–1154 (2008). First in vivo demonstration of collective invasion of cancer cells.
Christiansen, J. J. & Rajasekaran, A. K. Reassessing epithelial to mesenchymal transition as a prerequisite for carcinoma invasion and metastasis. Cancer Res. 66, 8319–8326 (2006).
Niessen, C. M. Tight junctions/adherens junctions: basic structure and function. J. Invest. Dermatol. 127, 2525–2532 (2007).
Kametani, Y. & Takeichi, M. Basal-to-apical cadherin flow at cell junctions. Nature Cell Biol. 9, 92–98 (2007).
Yamada, S. & Nelson, W. J. Localized zones of Rho and Rac activities drive initiation and expansion of epithelial cell–cell adhesion. J. Cell Biol. 178, 517–527 (2007).
van Kempen, L. C. et al. Activated leukocyte cell adhesion molecule/CD166, a marker of tumor progression in primary malignant melanoma of the skin. Am. J. Pathol. 156, 769–774 (2000).
Gavert, N., Ben-Shmuel, A., Raveh, S. & Ben-Ze'ev, A. L1-CAM in cancerous tissues. Expert Opin. Biol. Ther. 8, 1749–1757 (2008).
Ewald, A. J., Brenot, A., Duong, M., Chan, B. S. & Werb, Z. Collective epithelial migration and cell rearrangements drive mammary branching morphogenesis. Dev. Cell 14, 570–581 (2008). Compelling live-cell imaging and immunohistochemistry of 3D mammary gland cultures that show that cells at the tips of growing buds, albeit devoid of lamellopodia and filopodia, protrude efficiently, which suggests a 'pushing' rather than a 'pulling' mechanism.
di Bari, M. G. et al. Msx2 induces epithelial–mesenchymal transition in mouse mammary epithelial cells through upregulation of Cripto-1. J. Cell. Physiol. 219, 659–666 (2009).
Grunert, S., Jechlinger, M. & Beug, H. Diverse cellular and molecular mechanisms contribute to epithelial plasticity and metastasis. Nature Rev. Mol. Cell Biol. 4, 657–665 (2003).
Lee, J. M., Dedhar, S., Kalluri, R. & Thompson, E. W. The epithelial–mesenchymal transition: new insights in signaling, development, and disease. J. Cell Biol. 172, 973–981 (2006).
Thompson, E. W. & Williams, E. D. EMT and MET in carcinoma—clinical observations, regulatory pathways and new models. Clin. Exp. Metastasis 25, 591–592 (2008).
Schreiber, S. C. et al. Polysialylated NCAM represses E-cadherin-mediated cell–cell adhesion in pancreatic tumor cells. Gastroenterology 134, 1555–1566 (2008).
Lehembre, F. et al. NCAM-induced focal adhesion assembly: a functional switch upon loss of E-cadherin. EMBO J. 27, 2603–2615 (2008).
Wei, J., Hortsch, M. & Goode, S. Neuroglian stabilizes epithelial structure during Drosophila oogenesis. Dev. Dyn. 230, 800–808 (2004).
Massoumi, R. et al. Down-regulation of CYLD expression by Snail promotes tumor progression in malignant melanoma. J. Exp. Med. 206, 221–232 (2009).
Salmenpera, P. et al. Formation and activation of fibroblast spheroids depend on fibronectin–integrin interaction. Exp. Cell Res. 314, 3444–3452 (2008).
Belvindrah, R. Hankel, S., Walker, J., Patton, B. L. & Muller, U. β1 integrins control the formation of cell chains in the adult rostral migratory stream. J. Neurosci. 27, 2704–2717 (2007).
Khan, K. et al. Desmocollin switching in colorectal cancer. Br. J. Cancer 95, 1367–1370 (2006).
Chidgey, M. & Dawson, C. Desmosomes: a role in cancer? Br. J. Cancer 96, 1783–1787 (2007).
Langbein, L. et al. Tight junction-related structures in the absence of a lumen: occludin, claudins and tight junction plaque proteins in densely packed cell formations of stratified epithelia and squamous cell carcinomas. Eur. J. Cell Biol. 82, 385–400 (2003).
Smalley, K. S. et al. Up-regulated expression of zonula occludens protein-1 in human melanoma associates with N-cadherin and contributes to invasion and adhesion. Am. J. Pathol. 166, 1541–1554 (2005).
Ito, A. et al. Increased expression of connexin 26 in the invasive component of lung squamous cell carcinoma: significant correlation with poor prognosis. Cancer Lett. 234, 239–248 (2006).
Defranco, B. H. et al. Migrating cells retain gap junction plaque structure and function. Cell Commun. Adhes. 15, 273–288 (2008).
Czyz, J. The stage-specific function of gap junctions during tumourigenesis. Cell. Mol. Biol. Lett. 13, 92–102 (2008).
Vitorino, P. & Meyer, T. Modular control of endothelial sheet migration. Genes Dev. 22, 3268–3281 (2008).
Fischer, R. S., Gardel, M., Ma, X., Adelstein, R. S. & Waterman, C. M. Local cortical cension by myosin II guides 3D endothelial cell branching. Curr. Biol. 19, 260–265 (2009).
Nabeshima, K. et al. Front-cell-specific expression of membrane-type 1 matrix metalloproteinase and gelatinase A during cohort migration of colon carcinoma cells induced by hepatocyte growth factor/scatter factor. Cancer Res. 60, 3364–3369 (2000).
Palmieri, D. et al. Procollagen I COOH-terminal fragment induces VEGF-A and CXCR4 expression in breast carcinoma cells. Exp. Cell Res. 314, 2289–2298 (2008).
Wolf, K. & Friedl, P. Tube travel: the role of proteases in individual and collective cancer cell invasion. Cancer Res. 68, 7247–7249 (2008).
Smola, H. et al. Dynamics of basement membrane formation by keratinocyte–fibroblast interactions in organotypic skin culture. Exp. Cell Res. 239, 399–410 (1998).
Schmidt, M. et al. EGFL7 regulates the collective migration of endothelial cells by restricting their spatial distribution. Development 134, 2913–2923 (2007).
Lecaudey, V., Cakan-Akdogan, G., Norton, W. H. & Gilmour, D. Dynamic Fgf signaling couples morphogenesis and migration in the zebrafish lateral line primordium. Development 135, 2695–2705 (2008).
Aman, A. & Piotrowski, T. Wnt/β-catenin and Fgf signaling control collective cell migration by restricting chemokine receptor expression. Dev. Cell 15, 749–761 (2008).
Shintani, Y. et al. Collagen I-mediated up-regulation of N-cadherin requires cooperative signals from integrins and discoidin domain receptor 1. J. Cell Biol. 180, 1277–1289 (2008).
Orimo, A. et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335–348 (2005).
Valentin, G., Haas, P. & Gilmour, D. The chemokine SDF1a coordinates tissue migration through the spatially restricted activation of Cxcr7 and Cxcr4b. Curr. Biol. 17, 1026–1031 (2007).
Hegerfeldt, Y., Tusch, M., Brocker, E. B. & Friedl, P. Collective cell movement in primary melanoma explants: plasticity of cell–cell interaction, β1-integrin function, and migration strategies. Cancer Res. 62, 2125–2130 (2002).
Haas, P. & Gilmour, D. Chemokine signaling mediates self-organizing tissue migration in the zebrafish lateral line. Dev. Cell 10, 673–680 (2006). By combining zebrafish genetics and in vivo imaging, this work shows that a few chemokine-sensing leader cells direct the migration of many non-responsive followers during lateral line development.
Kolega, J. The movement of cell clusters in vitro: morphology and directionality. J. Cell Sci. 49, 15–32 (1981).
Zaidel-Bar, R., Itzkovitz, S., Ma'ayan, A., Iyengar, R. & Geiger, B. Functional atlas of the integrin adhesome. Nature Cell Biol. 9, 858–867 (2007).
Boguslavsky, S. et al. p120 catenin regulates lamellipodial dynamics and cell adhesion in cooperation with cortactin. Proc. Natl Acad. Sci. USA 104, 10882–10887 (2007).
le Noble, F., Klein, C., Tintu, A., Pries, A. & Buschmann, I. Neural guidance molecules, tip cells, and mechanical factors in vascular development. Cardiovasc. Res. 78, 232–241 (2008).
Ribeiro, C., Ebner, A. & Affolter, M. In vivo imaging reveals different cellular functions for FGF and Dpp signaling in tracheal branching morphogenesis. Dev. Cell 2, 677–683 (2002).
Caussinus, E., Colombelli, J. & Affolter, M. Tip-cell migration controls stalk-cell intercalation during Drosophila tracheal tube elongation. Curr. Biol. 18, 1727–1734 (2008).
Vasioukhin, V., Bauer, C., Yin, M. & Fuchs, E. Directed actin polymerization is the driving force for epithelial cell–cell adhesion. Cell 100, 209–219 (2000).
Geisbrecht, E. R. & Montell, D. J. Myosin VI is required for E-cadherin-mediated border cell migration. Nature Cell Biol. 4, 616–620 (2002).
Yana, I. et al. Crosstalk between neovessels and mural cells directs the site-specific expression of MT1-MMP to endothelial tip cells. J. Cell Sci. 120, 1607–1614 (2007).
Hendrix, M. J., Seftor, E. A., Hess, A. R. & Seftor, R. E. Molecular plasticity of human melanoma cells. Oncogene 22, 3070–3075 (2003).
Gudjonsson, T. et al. Normal and tumor-derived myoepithelial cells differ in their ability to interact with luminal breast epithelial cells for polarity and basement membrane deposition. J. Cell Sci. 115, 39–50 (2002).
Brachvogel, B. et al. Isolated Anxa5+/Sca-1+ perivascular cells from mouse meningeal vasculature retain their perivascular phenotype in vitro and in vivo. Exp. Cell Res. 313, 2730–2743 (2007).
Nischt, R. et al. Lack of nidogen-1 and -2 prevents basement membrane assembly in skin-organotypic coculture. J. Invest. Dermatol. 127, 545–554 (2007).
Rorth, P. Collective guidance of collective cell migration. Trends Cell Biol. 17, 575–579 (2007).
Murphy, A. M. & Montell, D. J. Cell type-specific roles for Cdc42, Rac, and RhoL in Drosophila oogenesis. J. Cell Biol. 133, 617–630 (1996).
Fulga, T. A. & Rorth, P. Invasive cell migration is initiated by guided growth of long cellular extensions. Nature Cell Biol. 4, 715–719 (2002).
Niewiadomska, P., Godt, D. & Tepass, U. DE-Cadherin is required for intercellular motility during Drosophila oogenesis. J. Cell Biol. 144, 533–547 (1999).
Pacquelet, A. & Rorth, P. Regulatory mechanisms required for DE-cadherin function in cell migration and other types of adhesion. J. Cell Biol. 170, 803–812 (2005).
Edwards, K. A. & Kiehart, D. P. Drosophila nonmuscle myosin II has multiple essential roles in imaginal disc and egg chamber morphogenesis. Development 122, 1499–1511 (1996).
Bianco, A. et al. Two distinct modes of guidance signalling during collective migration of border cells. Nature 448, 362–365 (2007). Elegant genetic mosaic experiments that show how border cell migration results from two genetically and mechanistically distinct guidance mechanisms.
Duchek, P., Somogyi, K., Jekely, G., Beccari, S. & Rorth, P. Guidance of cell migration by the Drosophila PDGF/VEGF receptor. Cell 107, 17–26 (2001).
Llense, F. & Martin-Blanco, E. JNK signaling controls border cell cluster integrity and collective cell migration. Curr. Biol. 18, 538–544 (2008).
Melani, M., Simpson, K. J., Brugge, J. S. & Montell, D. Regulation of cell adhesion and collective cell migration by hindsight and its human homolog RREB1. Curr. Biol. 18, 532–537 (2008).
Sutherland, D., Samakovlis, C. & Krasnow, M. A. branchless encodes a Drosophila FGF homolog that controls tracheal cell migration and the pattern of branching. Cell 87, 1091–1101 (1996). Seminal work on the instructive role of the FGF ligand Branchless in the pattern of branching in the D. melanogaster tracheal system.
Llimargas, M. The Notch pathway helps to pattern the tips of the Drosophila tracheal branches by selecting cell fates. Development 126, 2355–2364 (1999).
Ghabrial, A. S. & Krasnow, M. A. Social interactions among epithelial cells during tracheal branching morphogenesis. Nature 441, 746–749 (2006).
Lu, P., Sternlicht, M. D. & Werb, Z. Comparative mechanisms of branching morphogenesis in diverse systems. J. Mammary Gland Biol. Neoplasia 11, 213–228 (2006).
Lu, P. & Werb, Z. Patterning mechanisms of branched organs. Science 322, 1506–1509 (2008).
Ghysen, A. & Dambly-Chaudiere, C. The lateral line microcosmos. Genes Dev. 21, 2118–2130 (2007).
David, N. B. et al. Molecular basis of cell migration in the fish lateral line: role of the chemokine receptor CXCR4 and of its ligand, SDF1. Proc. Natl Acad. Sci. USA 99, 16297–16302 (2002).
Nechiporuk, A. & Raible, D. W. FGF-dependent mechanosensory organ patterning in zebrafish. Science 320, 1774–1777 (2008).
Dambly-Chaudiere, C., Cubedo, N. & Ghysen, A. Control of cell migration in the development of the posterior lateral line: antagonistic interactions between the chemokine receptors CXCR4 and CXCR7/RDC1. BMC Dev. Biol. 7, 23 (2007).
Boldajipour, B. et al. Control of chemokine-guided cell migration by ligand sequestration. Cell 132, 463–473 (2008).
Sainson, R. C. et al. Cell-autonomous notch signaling regulates endothelial cell branching and proliferation during vascular tubulogenesis. FASEB J. 19, 1027–1029 (2005).
Gerhardt, H. et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J. Cell Biol. 161, 1163–1177 (2003).
Gerhardt, H. VEGF and endothelial guidance in angiogenic sprouting. Organogenesis 4, 241–246 (2008).
Poujade, M. et al. Collective migration of an epithelial monolayer in response to a model wound. Proc. Natl Acad. Sci. USA 104, 15988–15993 (2007).
Zelenka, P. S. & Arpitha, P. Coordinating cell proliferation and migration in the lens and cornea. Semin. Cell Dev. Biol. 19, 113–124 (2008).
Grose, R. et al. A crucial role of β1 integrins for keratinocyte migration in vitro and during cutaneous wound repair. Development 129, 2303–2315 (2002).
Cowin, A. J. et al. Wound healing is defective in mice lacking tetraspanin CD151. J. Invest. Dermatol. 126, 680–689 (2006).
Moll, I., Houdek, P., Schafer, S., Nuber, U. & Moll, R. Diversity of desmosomal proteins in regenerating epidermis: immunohistochemical study using a human skin organ culture model. Arch. Dermatol. Res. 291, 437–446 (1999).
Vaezi, A., Bauer, C., Vasioukhin, V. & Fuchs, E. Actin cable dynamics and Rho/Rock orchestrate a polarized cytoskeletal architecture in the early steps of assembling a stratified epithelium. Dev. Cell 3, 367–381 (2002).
Nikolic, D. L., Boettiger, A. N., Bar-Sagi, D., Carbeck, J. D. & Shvartsman, S. Y. Role of boundary conditions in an experimental model of epithelial wound healing. Am J. Physiol. Cell Physiol. 291, C68–C75 (2006).
Werner, S., Krieg, T. & Smola, H. Keratinocyte–fibroblast interactions in wound healing. J. Invest. Dermatol. 127, 998–1008 (2007).
Nabeshima, K., Inoue, T., Shimao, Y., Kataoka, H. & Koono, M. Cohort migration of carcinoma cells: differentiated colorectal carcinoma cells move as coherent cell clusters or sheets. Histol. Histopathol. 14, 1183–1197 (1999).
Hsu, M., Andl, T., Li, G., Meinkoth, J. L. & Herlyn, M. Cadherin repertoire determines partner-specific gap junctional communication during melanoma progression. J. Cell Sci. 113, 1535–1542 (2000).
Radunsky, G. S. & van Golen, K. L. The current understanding of the molecular determinants of inflammatory breast cancer metastasis. Clin. Exp. Metastasis 22, 615–620 (2005).
Lahlou, H., Fanjul, M., Pradayrol, L., Susini, C. & Pyronnet, S. Restoration of functional gap junctions through internal ribosome entry site-dependent synthesis of endogenous connexins in density-inhibited cancer cells. Mol. Cell. Biol. 25, 4034–4045 (2005).
Daly, A. J., McIlreavey, L. & Irwin, C. R. Regulation of HGF and SDF-1 expression by oral fibroblasts—implications for invasion of oral cancer. Oral Oncol. 44, 646–651 (2008).
Liotta, L. A., Kleinerman, J. & Saidel, G. M. Quantitative relationships of intravascular tumor cells, tumor vessels, and pulmonary metastases following tumor implantation. Cancer Res. 34, 997–1004 (1974).
Kusters, B. et al. Micronodular transformation as a novel mechanism of VEGF-A-induced metastasis. Oncogene 26, 5808–5815 (2007).
Le, M. G. et al. Dermal lymphatic emboli in inflammatory and noninflammatory breast cancer: a French–Tunisian joint study in 337 patients. Clin. Breast Cancer 6, 439–445 (2005).
Sternlicht, M. D. et al. The stromal proteinase MMP3/stromelysin-1 promotes mammary carcinogenesis. Cell 98, 137–146 (1999).
Friedl, P. & Wolf, K. Proteolytic and non-proteolytic migration in tumor cells and leukocytes. Biochem. Soc. Symp. 70 277–285 (2003).
Friedl, P. & Wolf, K. Tumour-cell invasion and migration: diversity and escape mechanisms. Nature Rev. Cancer 3, 362–374 (2003).
Toyama, Y., Peralta, X. G., Wells, A. R., Kiehart, D. P. & Edwards, G. S. Apoptotic force and tissue dynamics during Drosophila embryogenesis. Science 321, 1683–1686 (2008).
Bertet, C., Sulak, L. & Lecuit, T. Myosin-dependent junction remodelling controls planar cell intercalation and axis elongation. Nature 429, 667–671 (2004).
Kopfstein, L. et al. Distinct roles of vascular endothelial growth factor-D in lymphangiogenesis and metastasis. Am. J. Pathol. 170, 1348–1361 (2007).
Teddy, J. M. & Kulesa, P. M. In vivo evidence for short- and long-range cell communication in cranial neural crest cells. Development 131, 6141–6151 (2004).
Weijer, C. J. Dictyostelium morphogenesis. Curr. Opin. Genet. Dev. 14, 392–398 (2004).
Kriebel, P. W., Barr, V. A., Rericha, E. C., Zhang, G. & Parent, C. A. Collective cell migration requires vesicular trafficking for chemoattractant delivery at the trailing edge. J. Cell Biol. 183, 949–961 (2008).
Siu, C. H., Lam, T. Y. & Choi, A. H. Inhibition of cell–cell binding at the aggregation stage of Dictyostelium discoideum development by monoclonal antibodies directed against an 80,000-dalton surface glycoprotein. J. Biol. Chem. 260, 16030–16036 (1985).
Dormann, D. & Weijer, C. J. Propagating chemoattractant waves coordinate periodic cell movement in Dictyostelium slugs. Development 128, 4535–4543 (2001).
Dormann, D., Weijer, G., Parent, C. A., Devreotes, P. N. & Weijer, C. J. Visualizing PI3 kinase-mediated cell–cell signaling during Dictyostelium development. Curr. Biol. 12, 1178–1188 (2002).
Hafner, C. et al. Ephrin-B2 is differentially expressed in the intestinal epithelium in Crohn's disease and contributes to accelerated epithelial wound healing in vitro. World J. Gastroenterol. 11, 4024–4031 (2005).
Korff, T. & Augustin, H. G. Tensional forces in fibrillar extracellular matrices control directional capillary sprouting. J. Cell Sci. 112, 3249–3258 (1999).
Montell, D. J., Rorth, P. & Spradling, A. C. Slow border cells, a locus required for a developmentally regulated cell migration during oogenesis, encodes Drosophila C/EBP. Cell 71, 51–62 (1992).
Gerharz, M. et al. Morphometric analysis of murine skin wound healing: standardization of experimental procedures and impact of an advanced multitissue array technique. Wound Repair Regen. 15, 105–112 (2007).
Acknowledgements
We thank O. Ilina and C. Rose for providing immunofluorescence and histological images. This work was supported by grants to P.F. from the Deutsche Krebshilfe (106950), Deutsche Forschungsgemeinschaft (FR 1155/8-2) and European Union (European Molecular Imaging Laboratories LSHC-CT-2004-503569).
Author information
Authors and Affiliations
Related links
Related links
DATABASES
OMIM
FURTHER INFORMATION
Glossary
- Invasion
-
A hallmark of cancer, measured as cells breaking away from their origin through the basement membrane. We use this term to mean all forms of cell movement through three-dimensional tissue that involve a change in tissue structure and, eventually, tissue destruction.
- Basement membrane
-
A sheet-like layer of interwoven macromolecules, including laminin, collagen IV and link proteins, that structurally anchor an epithelium or endothelium to the adjacent interstitial tissue. Epithelial or endothelial cells and stromal cells cooperate and deposit the macromolecules from either side.
- Border cell
-
One of a small cluster of cells that delaminate from the follicular epithelium of the Drosophila melanogaster egg chamber and migrate in a stereotypical pattern towards the developing oocyte. Ablation studies suggest that the function of border cells is to generate the micropyle, a structure at the dorso-anterior side of the oocyte that allows sperm entry.
- Neural crest
-
A population of migrating, pluripotent cells that appears transiently in the dorsal neuroectoderm. In the chick embryo, neural crest cells move as loosely associated strands or streams throughout the entire embryo and give rise to different tissues, including craniofacial bones and cartilage, the enteric and peripheral nervous systems and pigment cells.
- Stroma
-
Interstitial tissue consisting of extracellular matrix and mesenchymal cells. The interface between stroma and adjacent epithelia and vessels is formed by a basement membrane layer.
- Lateral line
-
A series of mechanosensory hair cell organs along the skin in fish and amphibia that detect changes in the surrounding water. Its precursor consists of neurogenic placodes, which migrate along defined paths and deposit clusters of cells behind them. These clusters differentiate into sensory hair cells that are analogous to those of the mammalian inner ear.
- Matrigel plug assay
-
An experiment in which tumour cells are suspended in matrigel solution and injected into a mammal, usually a mouse or rat. Because of the avascular matrigel barrier, vessels from the host sprout into the transplant and generate a de novo vessel network.
- Adherens junction
-
A punctated or linear cell–cell adhesion that contains cadherins and nectin, which are coupled to the actomyosin cytoskeleton by the adaptors α-, β- and γ-catenin and afadin (also known as AF6), respectively. Adherens junctions are dynamic structures that undergo continuous remodelling and provide cell–cell adhesion and signalling.
- Integrin
-
A heterodimeric protein that consists of an α- and a β-chain that both mediate extracellular ligand binding and intracellular engagement of cytoskeletal and signalling proteins. Integrins provide adhesion and mechanotransduction as well as intracellular signal transduction.
- Epithelial–mesenchymal transition
-
(EMT). The detachment of individual cells from an epithelium after downmodulation of cell–cell junctions, followed by single cell migration. The concept of EMT was established for morphogenic delamination of single cells into the mesenchyme and is discussed here in the context of early steps of cancer invasion.
- Desmosomal protein
-
Desmoglein 1–4 and desmocollin 1–3 connect through desmosomal adaptor proteins (plakoglobin, plakophilin, desmoplakin and desmocollin) to the intermediate filament cytoskeleton. These cadherins form homophilic adhesions and provide mechanically strong intercellular junctions between epithelial cells.
- Tight junction
-
A linear cell–cell adhesion complex in polarized epithelial and endothelial cells. Mediated by homophilic adhesion proteins, junction adhesion molecules, occludin and claudins, tight junctions form a tight barrier for the regulation of liquid, ion and nutrient flow across the epithelial barrier and contribute to cell polarity and signalling.
- Pseudopodium
-
A morphologically dynamic cylindrical cell protrusion of <3 μm thickness. Pseudopodia are controlled by the small GTPase Rac and CDC42, result from rapid filamentous actin polymerization, and allow cells to elongate, probe and adhere to other cells and to the extracellular matrix.
- Lamellipodium
-
A flat, cellular protrusion that is rich in branched actin filaments. Filament formation and branching are controlled by the small GTPase Rac and downstream effectors, including the actin-related protein (Arp)2/3 complex and formins, including mammalian diaphanous 1 (mDIA1; also known as DIAPH1) and mDIA2 (also known as DIAPH3).
- Filopodium
-
A finger-like and highly dynamic cell protrusion, 1 μm in diameter and up to 5 μm in length. Filopodia are formed by anterograde polymerization of actin bundles in parallel and lack microtubules. Their formation is controlled by the small GTPase CDC42.
- Mesenchymal–epithelial transition
-
An experimentally induced aggregation of moving individual cells to form a multicellular complex that maintains cell–cell junctions. Its role in physiological contexts is unclear.
- Gap junction
-
An intercellular hexameric channel between directly adjacent cells that transfers ions, small compounds and messengers between the cytosol of both cells and provides adhesive coupling independent of channel function. Gap junctions synchronize mechanical and metabolic cell functions in multicellular tissues.
- Collective amoeboid transition
-
The detachment of amoeboid cells from a multicellular complex as a consequence of loosened cell–cell junctions. Detached cells then use a leukocyte-like amoeboid migration mode because of the low adhesion and traction force generated.
Rights and permissions
About this article
Cite this article
Friedl, P., Gilmour, D. Collective cell migration in morphogenesis, regeneration and cancer. Nat Rev Mol Cell Biol 10, 445–457 (2009). https://doi.org/10.1038/nrm2720
Issue Date:
DOI: https://doi.org/10.1038/nrm2720
This article is cited by
-
ZO-1 regulates the migration of mesenchymal stem cells in cooperation with α-catenin in response to breast tumor cells
Cell Death Discovery (2024)
-
Vangl-dependent Wnt/planar cell polarity signaling mediates collective breast carcinoma motility and distant metastasis
Breast Cancer Research (2023)
-
Transcriptome analysis reveals temporally regulated genetic networks during Drosophila border cell collective migration
BMC Genomics (2023)
-
A comprehensive analysis of different types of databases reveals that CDH1 mRNA and E-cadherin protein are not downregulated in most carcinoma tissues and carcinoma cell lines
BMC Cancer (2023)
-
CNK2 promotes cancer cell motility by mediating ARF6 activation downstream of AXL signalling
Nature Communications (2023)