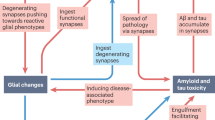
Key Points
-
Certain neurodegenerative disorders are characterized by the progressive accumulation of insoluble deposits of distinct misfolded proteins. In general, mutations causing inherited forms of the respective diseases affect the folding, aggregation and/or solubility of these proteins.
-
In Alzheimer's disease (AD), amyloid β-protein (Aβ) production by regulated intramembrane proteolysis is crucial for pathogenicity. An unusual intramembrane cleavage generates various Aβ species, only one of which (the longest variant) is particularly prone to form potentially toxic oligomers.
-
Soluble oligomeric assemblies of amyloidogenic proteins are increasingly thought to initiate disease-specific cytopathology and subsequent symptoms. Larger deposits, such as compacted Aβ plaques, seem to be relatively inert but might serve as reservoirs of diffusible oligomers.
-
Oligomeric assemblies of amyloidogenic peptides from distinct neurodegenerative diseases — such as Parkinson's, AD, Huntington's and prion disorders — share common structural properties.
-
Small oligomeric assemblies of Aβ have been specifically shown to impair long-term potentiation and dendritic spine structure in the hippocampus and to decrease memory in animals.
-
Oligomeric species of Aβ are active targets for therapeutic intervention. Aβ immunotherapy, Aβ-aggregation inhibitors, allosteric modulators of γ-secretase and Aβ-degrading proteases can all reduce oligomeric Aβ, consequent neuronal degeneration and behavioural deficits in mouse models of AD.
-
The first promising clinical data from AD human trials seem to support the potential of Aβ-directed immunotherapy that can neutralize oligomers.
-
Similar therapeutic approaches based on oligomeric amyloidogenic species are likely to apply to other protein-folding diseases of the nervous system.
Abstract
The distinct protein aggregates that are found in Alzheimer's, Parkinson's, Huntington's and prion diseases seem to cause these disorders. Small intermediates — soluble oligomers — in the aggregation process can confer synaptic dysfunction, whereas large, insoluble deposits might function as reservoirs of the bioactive oligomers. These emerging concepts are exemplified by Alzheimer's disease, in which amyloid β-protein oligomers adversely affect synaptic structure and plasticity. Findings in other neurodegenerative diseases indicate that a broadly similar process of neuronal dysfunction is induced by diffusible oligomers of misfolded proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gasser, T. Genetics of Parkinson's disease. Curr. Opin. Neurol. 18, 363–369 (2005).
Polymeropoulos, M. H. et al. Mutation in the α-synuclein gene identified in families with Parkinson's disease. Science 276, 2045–2047 (1997).
Kahle, P. J., Haass, C., Kretzschmar, H. A. & Neumann, M. Structure/function of α-synuclein in health and disease: rational development of animal models for Parkinson's and related diseases. J. Neurochem. 82, 449–457 (2002).
Spillantini, M. G. et al. α-Synuclein in Lewy bodies. Nature 388, 839–840 (1997).
Baba, M. et al. Aggregation of α-synuclein in Lewy bodies of sporadic Parkinson's disease and dementia with Lewy bodies. Am. J. Pathol. 152, 879–884 (1998).
Rosen, D. R. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 364, 362 (1993).
Deng, H. X. et al. Amyotrophic lateral sclerosis and structural defects in Cu, Zn superoxide dismutase. Science 261, 1047–1051 (1993).
A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. The Huntington's Disease Collaborative Research Group. Cell 72, 971–983 (1993).
Alzheimer, A. Über eine eigenartige Erkrankung der Hirnrinde. Allg. Z. Psychiatrie Psychisch-Gerichtl. Med. 64, 146–148 (1907).
Glenner, G. G. & Wong, C. W. Alzheimer's disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem. Biophys. Res. Commun. 120, 885–890 (1984).
Masters, C. L. et al. Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc. Natl Acad. Sci. USA 82, 4245–4249 (1985).
Grundke-Iqbal, I. et al. Abnormal phosphorylation of the microtubule-associated protein τ (tau) in Alzheimer cytoskeletal pathology. Proc. Natl Acad. Sci. USA 83, 4913–4917 (1986).
Kosik, K. S., Joachim, C. L. & Selkoe, D. J. Microtubule-associated protein τ (tau) is a major antigenic component of paired helical filaments in Alzheimer disease. Proc. Natl Acad. Sci. USA 83, 4044–4048 (1986).
Nukina, N. & Ihara, Y. One of the antigenic determinants of paired helical filaments is related to τ protein. J. Biochem. 99, 1541–1544 (1986).
Glenner, G. G. & Wong, C. W. Alzheimer's disease and Down's syndrome: sharing of a unique cerebrovascular amyloid fibril protein. Biochem. Biophys. Res. Commun. 122, 1131–1135 (1984).
Olson, M. I. & Shaw, C. M. Presenile dementia and Alzheimer's disease in mongolism. Brain 92, 147–156 (1969).
Kang, J. et al. The precursor of Alzheimer's disease amyloid A4 protein resembles a cell-surface receptor. Nature 325, 733–736 (1987).
Levy, E. et al. Mutation of the Alzheimer's disease amyloid gene in hereditary cerebral hemorrhage, Dutch type. Science 248, 1124–1126 (1990).
Goate, A. et al. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer's disease. Nature 349, 704–706 (1991).
Chartier-Harlin, M. C. et al. Early-onset Alzheimer's disease caused by mutations at codon 717 of the β-amyloid precursor protein gene. Nature 353, 844–846 (1991).
Mullan, M. et al. A pathogenic mutation for probable Alzheimer's disease in the APP gene at the N-terminus of β-amyloid. Nature Genet. 1, 345–347 (1992).
Selkoe, D. J. The molecular pathology of Alzheimer's disease. Neuron 6, 487–498 (1991).
Hardy, J. A. & Higgins, G. A. Alzheimer's disease: the amyloid cascade hypothesis. Science 256, 184–185 (1992). References 22 and 23 set out the amyloid cascade hypothesis, for which strong experimental evidence is now accumulating.
Hardy, J. & Selkoe, D. J. The amyloid hypothesis of Alzheimer's disease: progress and problems on the road to therapeutics. Science 297, 353–356 (2002).
Haass, C. et al. Amyloid β-peptide is produced by cultured cells during normal metabolism. Nature 359, 322–325 (1992).
Seubert, P. et al. Isolation and quantification of soluble Alzheimer's β-peptide from biological fluids. Nature 359, 325–327 (1992).
Shoji, M. et al. Production of the Alzheimer amyloid β-protein by normal proteolytic processing. Science 258, 126–129 (1992).
Busciglio, J., Gabuzda, D. H., Matsudaira, P. & Yankner, B. A. Generation of β-amyloid in the secretory pathway in neuronal and nonneuronal cells. Proc. Natl Acad. Sci. USA 90, 2092–2096 (1993).
Haass, C. Take five-BACE and the γ-secretase quartet conduct Alzheimer's amyloid β-peptide generation. EMBO J. 23, 483–488 (2004). A review on APP processing by β- and γ-secretase.
Brown, M. S., Ye, J., Rawson, R. B. & Goldstein, J. L. Regulated intramembrane proteolysis: a control mechanism conserved from bacteria to humans. Cell 100, 391–398 (2000).
Weihofen, A. & Martoglio, B. Intramembrane-cleaving proteases: controlled liberation of functional proteins and peptides from membranes. Trends Cell Biol. 13, 71–78 (2003).
Mumm, J. S. & Kopan, R. Notch signaling: from the outside in. Dev. Biol. 228, 151–165 (2000).
Wolfe, M. S. et al. Two transmembrane aspartates in presenilin-1 required for presenilin endoproteolysis and γ-secretase activity. Nature 398, 513–517 (1999).
Steiner, H. et al. Glycine 384 is required for presenilin-1 function and is conserved in polytopic bacterial aspartyl proteases. Nature Cell Biol. 2, 848–851 (2000).
Kimberly, W. T. et al. γ-Secretase is a membrane protein complex comprised of presenilin, nicastrin, Aph-1, and Pen-2. Proc. Natl Acad. Sci. USA 100, 6382–6387 (2003).
Takasugi, N. et al. The role of presenilin cofactors in the γ-secretase complex. Nature 422, 438–441 (2003).
Edbauer, D. et al. Reconstitution of γ-secretase activity. Nature Cell Biol. 5, 486–488 (2003).
Sastre, M. et al. Presenilin-dependent γ-secretase processing of β-amyloid precursor protein at a site corresponding to the S3 cleavage of Notch. EMBO Rep. 2, 835–841 (2001).
Weidemann, A. et al. A novel ε-cleavage within the transmembrane domain of the Alzheimer amyloid precursor protein demonstrates homology with Notch processing. Biochemistry 41, 2825–2835 (2002).
Gu, Y. et al. Distinct intramembrane cleavage of the β-amyloid precursor protein family resembling γ-secretase-like cleavage of Notch. J. Biol. Chem. 276, 35235–35238 (2001).
Qi-Takahara, Y. et al. Longer forms of amyloid β protein: implications for the mechanism of intramembrane cleavage by γ-secretase. J. Neurosci. 25, 436–445 (2005).
Zhao, G. et al. γ-cleavage is dependent on ζ-cleavage during the proteolytic processing of amyloid precursor protein within its transmembrane domain. J. Biol. Chem. 280, 37689–37697 (2005).
Dickson, D. W. The pathogenesis of senile plaques. J. Neuropathol. Exp. Neurol. 56, 321–339 (1997).
Naslund, J. et al. Correlation between elevated levels of amyloid β-peptide in the brain and cognitive decline. JAMA 283, 1571–1577 (2000).
Lue, L. F. et al. Soluble amyloid β peptide concentration as a predictor of synaptic change in Alzheimer's disease. Am. J. Pathol. 155, 853–862 (1999).
McLean, C. A. et al. Soluble pool of Aβ amyloid as a determinant of severity of neurodegeneration in Alzheimer's disease. Ann. Neurol. 46, 860–866 (1999).
Wang, J., Dickson, D. W., Trojanowski, J. Q. & Lee, V. M. The levels of soluble versus insoluble brain Aβ distinguish Alzheimer's disease from normal and pathologic aging. Exp. Neurol. 158, 328–337 (1999).
Arrasate, M., Mitra, S., Schweitzer, E. S., Segal, M. R. & Finkbeiner, S. Inclusion body formation reduces levels of mutant huntingtin and the risk of neuronal death. Nature 431, 805–810 (2004). Evidence that small, diffusible aggregates of intracellular huntingtin can confer neurotoxicity, perhaps analogously to soluble Aβ oligomers.
Schaffar, G. et al. Cellular toxicity of polyglutamine expansion proteins: mechanism of transcription factor deactivation. Mol. Cell 15, 95–105 (2004).
Cummings, C. J. et al. Mutation of the E6-AP ubiquitin ligase reduces nuclear inclusion frequency while accelerating polyglutamine-induced pathology in SCA1 mice. Neuron 24, 879–892 (1999).
Tsai, J., Grutzendler, J., Duff, K. & Gan, W. B. Fibrillar amyloid deposition leads to local synaptic abnormalities and breakage of neuronal branches. Nature Neurosci. 7, 1181–1183 (2004).
Caughey, B. & Lansbury, P. T. Protofibrils, pores, fibrils, and neurodegeneration: separating the responsible protein aggregates from the innocent bystanders. Annu. Rev. Neurosci. 26, 267–298 (2003).
Teplow, D. B. Structural and kinetic features of amyloid β-protein fibrillogenesis. Amyloid 5, 121–142 (1998).
Harper, J. D., Wong, S. S., Lieber, C. M. & Lansbury, P. T. Observation of metastable Aβ amyloid protofibrils by atomic force microscopy. Chem. Biol. 4, 119–125 (1997).
Hartley, D. M. et al. Protofibrillar intermediates of amyloid β-protein induce acute electrophysiological changes and progressive neurotoxicity in cortical neurons. J. Neurosci. 19, 8876–8884 (1999).
Walsh, D. M., Lomakin, A., Benedek, G. B., Condron, M. M. & Teplow, D. B. Amyloid β-protein fibrillogenesis. Detection of a protofibrillar intermediate. J. Biol. Chem. 272, 22364–22372 (1997).
Walsh, D. M. et al. Amyloid β-protein fibrillogenesis. Structure and biological activity of protofibrillar intermediates. J. Biol. Chem. 274, 25945–25952 (1999).
Bitan, G. et al. Amyloid β-protein (Aβ) assembly: Aβ40 and Aβ42 oligomerize through distinct pathways. Proc. Natl Acad. Sci. USA 100, 330–335 (2003).
Lashuel, H. A., Hartley, D., Petre, B. M., Walz, T. & Lansbury, P. T. Jr. Neurodegenerative disease: amyloid pores from pathogenic mutations. Nature 418, 291 (2002).
Lambert, M. P. et al. Diffusible, nonfibrillar ligands derived from Aβ1–42 are potent central nervous system neurotoxins. Proc. Natl Acad. Sci. USA 95, 6448–6453 (1998).
Gong, Y. et al. Alzheimer's disease-affected brain: presence of oligomeric Aβ ligands (ADDLs) suggests a molecular basis for reversible memory loss. Proc. Natl Acad. Sci. USA 100, 10417–10422 (2003).
Lesne, S. et al. A specific amyloid-β protein assembly in the brain impairs memory. Nature 440, 352–357 (2006). Identification of a brain-derived Aβ oligomeric assembly, which impairs memory.
Podlisny, M. B. et al. Aggregation of secreted amyloid β-protein into sodium dodecyl sulfate-stable oligomers in cell culture. J. Biol. Chem. 270, 9564–9570 (1995).
Walsh, D. M., Tseng, B. P., Rydel, R. E., Podlisny, M. B. & Selkoe, D. J. The oligomerization of amyloid β-protein begins intracellularly in cells derived from human brain. Biochemistry 39, 10831–10839 (2000).
Funato, H., Enya, M., Yoshimura, M., Morishima-Kawashima, M. & Ihara, Y. Presence of sodium dodecyl sulfate-stable amyloid β-protein dimers in the hippocampus CA1 not exhibiting neurofibrillary tangle formation. Am. J. Pathol. 155, 23–28 (1999).
Enya, M. et al. Appearance of sodium dodecyl sulfate-stable amyloid β-protein (Aβ) dimer in the cortex during aging. Am. J. Pathol. 154, 271–279 (1999).
Kawarabayashi, T. et al. Dimeric amyloid β protein rapidly accumulates in lipid rafts followed by apolipoprotein E and phosphorylated τ accumulation in the Tg2576 mouse model of Alzheimer's disease. J. Neurosci. 24, 3801–3809 (2004).
Roher, A. E. et al. Morphology and toxicity of Aβ-(1–42) dimer derived from neuritic and vascular amyloid deposits of Alzheimer's disease. J. Biol. Chem. 271, 20631–20635 (1996).
Walsh, D. M. et al. Naturally secreted oligomers of amyloid β protein potently inhibit hippocampal long-term potentiation in vivo. Nature 416, 535–539 (2002). Defines a synaptotoxic function for small, soluble oligomers of secreted Aβ in vivo.
Kamenetz, F. et al. APP processing and synaptic function. Neuron 37, 925–937 (2003). Demonstrates the effects of Aβ on synaptic function upon the stimulation of neuronal activity.
Townsend, M., Shankar, G. M., Mehta, T., Walsh, D. M. & Selkoe, D. J. Effects of secreted oligomers of amyloid β-protein on hippocampal synaptic plasticity: a potent role for trimers. J. Physiol. 572, 477–492 (2006).
Klyubin, I. et al. Amyloid β protein immunotherapy neutralizes Aβ oligomers that disrupt synaptic plasticity in vivo. Nature Med. 11, 556–561 (2005). LTP inhibition by soluble oligomers of human Aβ is prevented by active and passive Aβ immunotherapy.
Cleary, J. P. et al. Natural oligomers of the amyloid-β protein specifically disrupt cognitive function. Nature Neurosci. 8, 79–84 (2005).
Cirrito, J. R. et al. Synaptic activity regulates interstitial fluid amyloid-β levels in vivo. Neuron 48, 913–922 (2005). An in vivo demonstration of the effects of synaptic activity on Aβ levels.
Snyder, E. M. et al. Regulation of NMDA receptor trafficking by amyloid-β. Nature Neurosci. 8, 1051–1058 (2005). A cellular mechanism that describes how Aβ lowers NMDA-evoked currents.
Gong, B. et al. Ubiquitin hydrolase Uch-L1 rescues β-amyloid-induced decreases in synaptic function and contextual memory. Cell 126, 775–788 (2006).
Meyer-Luehmann, M. et al. Exogenous induction of cerebral β-amyloidogenesis is governed by agent and host. Science 313, 1781–1784 (2006).
Glabe, C. G. & Kayed, R. Common structure and toxic function of amyloid oligomers implies a common mechanism of pathogenesis. Neurology 66, S74–S78 (2006).
Kayed, R. et al. Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 300, 486–489 (2003). Describes common conformational epitopes on oligomers of completely distinct amyloidogenic proteins.
Lashuel, H. A. et al. α-Synuclein, especially the Parkinson's disease-associated mutants, forms pore-like annular and tubular protofibrils. J. Mol. Biol. 322, 1089–1102 (2002).
Conway, K. A., Harper, J. D. & Lansbury, P. T. Jr. Fibrils formed in vitro from α-synuclein and two mutant forms linked to Parkinson's disease are typical amyloid. Biochemistry 39, 2552–2563 (2000).
Conway, K. A., Harper, J. D. & Lansbury, P. T. Accelerated in vitro fibril formation by a mutant α-synuclein linked to early-onset Parkinson disease. Nature Med. 4, 1318–1320 (1998).
Conway, K. A. et al. Acceleration of oligomerization, not fibrillization, is a shared property of both α-synuclein mutations linked to early-onset Parkinson's disease: implications for pathogenesis and therapy. Proc. Natl Acad. Sci. USA 97, 571–576 (2000).
Marchut, A. J. & Hall, C. K. Spontaneous formation of annular structures observed in molecular dynamics simulations of polyglutamine peptides. Comput. Biol. Chem. 30, 215–218 (2006).
Srinivasan, R., Marchant, R. E. & Zagorski, M. G. ABri peptide associated with familial British dementia forms annular and ring-like protofibrillar structures. Amyloid 11, 10–13 (2004).
Wille, H., Drewes, G., Biernat, J., Mandelkow, E. M. & Mandelkow, E. Alzheimer-like paired helical filaments and antiparallel dimers formed from microtubule-associated protein τ in vitro. J. Cell. Biol. 118, 573–584 (1992).
Friedhoff, P., von Bergen, M., Mandelkow, E. M., Davies, P. & Mandelkow, E. A nucleated assembly mechanism of Alzheimer paired helical filaments. Proc. Natl Acad. Sci. USA 95, 15712–15717 (1998).
Neumann, M. et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314, 130–133 (2006).
Willem, M. et al. Control of peripheral nerve myelination by the β-secretase BACE1. Science 314, 664–666 (2006).
Schenk, D. et al. Immunization with amyloid-β attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature 400, 173–177 (1999). Initial report of the beneficial effects of Aβ immunotherapy in a transgenic mouse model of AD.
Bard, F. et al. Peripherally administered antibodies against amyloid β-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nature Med. 6, 916–919 (2000).
Orgogozo, J. M. et al. Subacute meningoencephalitis in a subset of patients with AD after Aβ42 immunization. Neurology 61, 46–54 (2003).
Hock, C. et al. Antibodies against β-amyloid slow cognitive decline in Alzheimer's disease. Neuron 38, 547–554 (2003). First report of the beneficial effects of Aβ immunotherapy in a small cohort of vaccinated patients with AD.
Gilman, S. et al. Clinical effects of Aβ immunization (AN1792) in patients with AD in an interrupted trial. Neurology 64, 1553–1562 (2005).
Nicoll, J. A. et al. Neuropathology of human Alzheimer disease after immunization with amyloid-β peptide: a case report. Nature Med. 9, 448–452 (2003). First report of the apparent removal of Aβ deposits in humans by a therapeutic agent.
Patton, R. L. et al. Amyloid-β peptide remnants in AN-1792-immunized Alzheimer's disease patients: a biochemical analysis. Am. J. Pathol. 169, 1048–1063 (2006).
Oddo, S., Billings, L., Kesslak, J. P., Cribbs, D. H. & LaFerla, F. M. Aβ immunotherapy leads to clearance of early, but not late, hyperphosphorylated τ aggregates via the proteasome. Neuron 43, 321–332 (2004). Further evidence of a linear connection between Aβ deposition and tau hyperphosphorylation in an animal model.
DeMattos, R. B., Bales, K. R., Cummins, D. J., Paul, S. M. & Holtzman, D. M. Brain to plasma amyloid-β efflux: a measure of brain amyloid burden in a mouse model of Alzheimer's disease. Science 295, 2264–2267 (2002).
Dodart, J. C. et al. Immunization reverses memory deficits without reducing brain Aβ burden in Alzheimer's disease model. Nature Neurosci. 5, 452–457 (2002).
Janus, C. et al. A β peptide immunization reduces behavioural impairment and plaques in a model of Alzheimer's disease. Nature 408, 979–982 (2000).
Gelinas, D. S., DaSilva, K., Fenili, D., St George-Hyslop, P. & McLaurin, J. Immunotherapy for Alzheimer's disease. Proc. Natl Acad. Sci. USA 101, 14657–14662 (2004).
Selkoe, D. J. & Schenk, D. Alzheimer's disease: molecular understanding predicts amyloid-based therapeutics. Annu. Rev. Pharmacol. Toxicol. 43, 545–584 (2003).
McLaurin, J. et al. Therapeutically effective antibodies against amyloid-β peptide target amyloid-β residues 4–10 and inhibit cytotoxicity and fibrillogenesis. Nature Med. 8, 1263–1269 (2002). Important mechanistic insights about how Aβ immunotherapy can prevent oligomerization and cytotoxicity.
Solomon, B., Koppel, R., Hanan, E. & Katzav, T. Monoclonal antibodies inhibit in vitro fibrillar aggregation of the Alzheimer β-amyloid peptide. Proc. Natl Acad. Sci. USA 93, 452–455 (1996).
Qiu, W. Q. et al. Insulin-degrading enzyme regulates extracellular levels of amyloid β- protein by degradation. J. Biol. Chem. 273, 32730–32738 (1998).
Tucker, H. M. et al. The plasmin system is induced by and degrades amyloid-β aggregates. J. Neurosci. 20, 3937–3946 (2000).
Mueller-Steiner, S. et al. Antiamyloidogenic and neuroprotective functions of cathepsin B: implications for Alzheimer's disease. Neuron 51, 703–714 (2006).
Iwata, N. et al. Identification of the major Aβ1–42-degrading catabolic pathway in brain parenchyma: suppression leads to biochemical and pathological deposition. Nature Med. 6, 143–150 (2000).
Leissring, M. A. et al. Enhanced proteolysis of β-amyloid in APP transgenic mice prevents plaque formation, secondary pathology, and premature death. Neuron 40, 1087–1093 (2003).
Huang, S. M. et al. Neprilysin-sensitive synapse-associated Aβ oligomers impair neuronal plasticity and cognitive function. J. Biol. Chem. 281, 17941–17951 (2006).
McLaurin, J., Franklin, T., Zhang, X., Deng, J. & Fraser, P. E. Interactions of Alzheimer amyloid-β peptides with glycosaminoglycans effects on fibril nucleation and growth. Eur. J. Biochem. 266, 1101–1110 (1999).
McLaurin, J., Golomb, R., Jurewicz, A., Antel, J. P. & Fraser, P. E. Inositol stereoisomers stabilize an oligomeric aggregate of Alzheimer amyloid β peptide and inhibit aβ-induced toxicity. J. Biol. Chem. 275, 18495–18502 (2000).
McLaurin, J. et al. Cyclohexanehexol inhibitors of Aβ aggregation prevent and reverse Alzheimer phenotype in a mouse model. Nature Med. 12, 801–808 (2006). New Aβ-aggregation inhibitors show beneficial effects on plaque burden and behaviour in mice.
Weggen, S. et al. A subset of NSAIDs lower amyloidogenic Aβ42 independently of cyclooxygenase activity. Nature 414, 212–216 (2001). Discovery of certain NSAIDs as γ-secretase modulators: they lead to shorter, less amyloidogenic Aβ species.
Doerfler, P., Shearman, M. S. & Perlmutter, R. M. Presenilin-dependent γ-secretase activity modulates thymocyte development. Proc. Natl Acad. Sci. USA 98, 9312–9317 (2001).
Geling, A., Steiner, H., Willem, M., Bally-Cuif, L. & Haass, C. A γ-secretase inhibitor blocks Notch signaling in vivo and causes a severe neurogenic phenotype in zebrafish. EMBO Rep. 3, 688–694 (2002).
Scheuner, D. et al. Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer's disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer's disease. Nature Med. 2, 864–870 (1996).
Suzuki, N. et al. An increased percentage of long amyloid β-protein secreted by familial amyloid β-protein precursor (βAPP717) mutants. Science 264, 1336–1340 (1994).
Burdick, D. et al. Assembly and aggregation properties of synthetic Alzheimer's A4/β amyloid peptide analogs. J. Biol. Chem. 267, 546–554 (1992).
Jarrett, J. T., Berger, E. P. & Lansbury, P. T. Jr. The carboxy terminus of the β-amyloid protein is critical for the seeding of amyloid formation: implications for the pathogenesis of Alzheimer's disease. Biochemistry 32, 4693–4697 (1993).
Chen, Y. R. & Glabe, C. G. Distinct early folding and aggregation properties of Alzheimer amyloid-β peptides Aβ40 and Aβ42: stable trimer or tetramer formation by Aβ42. J. Biol. Chem. 281, 24414–24422 (2006).
Nilsberth, C. et al. The 'Arctic' APP mutation (E693G) causes Alzheimer's disease by enhanced Aβ protofibril formation. Nature Neurosci. 4, 887–893 (2001). A genetic explanation for the development of rare forms of AD that is strongly supportive of the amyloid hypothesis.
Iwatsubo, T. et al. Visualization of Aβ42(43) and Aβ40 in senile plaques with end-specific A β monoclonals: evidence that an initially deposited species is Aβ42(43). Neuron 13, 45–53 (1994).
Cai, X. D., Golde, T. E. & Younkin, S. G. Release of excess amyloid β protein from a mutant amyloid β protein precursor. Science 259, 514–516 (1993).
Citron, M. et al. Mutation of the β-amyloid precursor protein in familial Alzheimer's disease increases β-protein production. Nature 360, 672–674 (1992).
Rovelet-Lecrux, A. et al. APP locus duplication causes autosomal dominant early-onset Alzheimer disease with cerebral amyloid angiopathy. Nature Genet. 38, 24–26 (2006).
Singleton, A. B. et al. α-Synuclein locus triplication causes Parkinson's disease. Science 302, 841 (2003).
McGowan, E. et al. Aβ42 is essential for parenchymal and vascular amyloid deposition in mice. Neuron 47, 191–199 (2005).
Bentahir, M. et al. Presenilin clinical mutations can affect γ-secretase activity by different mechanisms. J. Neurochem. 96, 732–742 (2006).
Deng, Y. et al. Deletion of presenilin 1 hydrophilic loop sequence leads to impaired γ-secretase activity and exacerbated amyloid pathology. J. Neurosci. 26, 3845–3854 (2006).
Acknowledgements
We thank D. Teplow, G. Multhaup, E. Mandelkow and D. Walsh as well as the members of our laboratories for helpful discussions. We also thank M. Neumann for providing the figures that are included in Box 1.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
Christian Haass is a consultant for Boehringer Ingelheim Pharma KG. Dennis J. Selkoe is a consultant for Elan, plc.
Related links
Related links
DATABASES
OMIM
FURTHER INFORMATION
Glossary
- Amyloid
-
Tissue deposits of insoluble, proteinaceous fibrils that are rich in β-pleated sheet structure and therefore bind to the histochemical dye Congo red in a polarized manner.
- Lewy body
-
(LB). A deposit of α-synuclein typically found in neuronal cell bodies of patients with Parkinson's disease or related disorders.
- Lewy neurite
-
(LN). A deposit of α-synuclein typically found in swollen neuronal processes of patients with Parkinson's disease or related disorders.
- Amyloid plaques
-
Spherical extracellular deposits predominantly composed of the amyloid β-peptide and found in the brains of all cases of Alzheimer's disease.
- Neurofibrillary tangle
-
An intraneuronal filamentous aggregate composed of abnormally phosphorylated tau protein found in several human neurodegenerative disorders, including Alzheimer's disease.
- Regulated intramembrane proteolysis
-
(RIP). Regulated cleavage of the luminal domain of certain membrane proteins is followed by a constitutive cleavage in the transmembrane domain. At least in some cases this process is involved in signalling pathways.
- Intracellular domain
-
(ICD). One of the cleavage products of intramembrane proteolysis in the RIP pathway, it is liberated into the cytosol and in some cases targeted to the nucleus.
Rights and permissions
About this article
Cite this article
Haass, C., Selkoe, D. Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer's amyloid β-peptide. Nat Rev Mol Cell Biol 8, 101–112 (2007). https://doi.org/10.1038/nrm2101
Issue Date:
DOI: https://doi.org/10.1038/nrm2101
This article is cited by
-
A single-domain antibody detects and neutralises toxic Aβ42 oligomers in the Alzheimer’s disease CSF
Alzheimer's Research & Therapy (2024)
-
Early onset diagnosis in Alzheimer’s disease patients via amyloid-β oligomers-sensing probe in cerebrospinal fluid
Nature Communications (2024)
-
Combination strategy employing BACE1 inhibitor and memantine to boost cognitive benefits in Alzheimer’s disease therapy
Psychopharmacology (2024)
-
Liquid–liquid phase separation in Alzheimer’s disease
Journal of Molecular Medicine (2024)
-
Disaggregation of amyloid-beta fibrils via natural metabolites using long timescale replica exchange molecular dynamics simulation studies
Journal of Molecular Modeling (2024)