Key Points
-
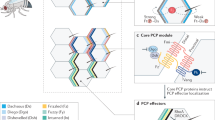
Planar cell polarity (PCP) is a polarity axis that organizes cells in the plane of the tissue. PCP is conserved in metazoans and is essential for proper development and tissue homeostasis.
-
Asymmetric and mutually exclusive subcellular enrichment of key PCP proteins patterns cells in planar-polarized tissues. PCP proteins also coordinate planar polarity between cells and control polarized behaviours by modulating the cytoskeleton.
-
PCP patterns develop gradually from an initially disordered state through dynamic trafficking and various feedback interactions that can influence protein localization and stability.
-
PCP patterns seem to be globally oriented along a pre-defined axis in a given tissue. Notably, multiple mechanistic inputs may have differential influences on PCP patterning depending on developmental timing and tissue context, and may only partially overlap in different contexts.
-
The morphogenetic events governed by PCP signalling are best understood in Drosophila melanogaster, in which the particular orientation of hairs and bristles on the fly body has served to unravel basic principles of PCP-dependent processes. Information obtained from this model has helped to better understand equivalent mechanisms in vertebrates, particularly in the context of the orientation of fluid flow mediated by multiciliated cells and cell rearrangements during convergent extension.
-
Mutations in PCP genes have been implicated in diverse human pathologies, and the body of evidence supporting the involvement of PCP aberrations in human birth defects continues to grow rapidly.
Abstract
Planar cell polarity (PCP) is an essential feature of animal tissues, whereby distinct polarity is established within the plane of a cell sheet. Tissue-wide establishment of PCP is driven by multiple global cues, including gradients of gene expression, gradients of secreted WNT ligands and anisotropic tissue strain. These cues guide the dynamic, subcellular enrichment of PCP proteins, which can self-assemble into mutually exclusive complexes at opposite sides of a cell. Endocytosis, endosomal trafficking and degradation dynamics of PCP components further regulate planar tissue patterning. This polarization propagates throughout the whole tissue, providing a polarity axis that governs collective morphogenetic events such as the orientation of subcellular structures and cell rearrangements. Reflecting the necessity of polarized cellular behaviours for proper development and function of diverse organs, defects in PCP have been implicated in human pathologies, most notably in severe birth defects.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Goodrich, L. V. & Strutt, D. Principles of planar polarity in animal development. Development 138, 1877–1892 (2011).
Adler, P. N. The frizzled/stan pathway and planar cell polarity in the Drosophila wing. Curr. Top. Dev. Biol. 101, 1–31 (2012).
Peng, Y. & Axelrod, J. D. Asymmetric protein localization in planar cell polarity: mechanisms, puzzles and challenges. Curr. Top. Dev. Biol. 101, 33–53 (2012).
Lawrence, P. A. & Casal, J. The mechanisms of planar cell polarity, growth and the Hippo pathway: some known unknowns. Dev. Biol. 377, 1–8 (2013).
Yang, Y. & Mlodzik, M. Wnt-Frizzled/planar cell polarity signalling: cellular orientation by facing the wind (Wnt). Annu. Rev. Cell Dev. Biol. 31, 623–646 (2015).
Usui, T. et al. Flamingo, a seven-pass transmembrane cadherin, regulates planar cell polarity under the control of Frizzled. Cell 98, 585–595 (1999).
Lawrence, P. A., Casal, J. & Struhl, G. Cell interactions and planar polarity in the abdominal epidermis of Drosophila. Development 131, 4651–4664 (2004). This analysis of multiple mutant clones and backgrounds identifies Fmi as the essential component for intercellular PCP signalling and provides a more detailed description of the establishment of global PCP patterns.
Strutt, H. & Strutt, D. Differential stability of Flamingo protein complexes underlies the establishment of planar polarity. Curr. Biol. 18, 1555–1564 (2008). This examination of Fmi binding preferences and dynamics under different conditions provides a fresh perspective on how PCP complexes assemble.
Chen, W.-S. et al. Asymmetric homotypic interactions of the atypical cadherin Flamingo mediate intercellular polarity signaling. Cell 133, 1093–1105 (2008). This detailed analysis of cell–cell signalling through Fmi provides evidence for two different signalling states of Fmi when bound with Fz or when bound with Vang or neither.
Wu, J. & Mlodzik, M. The Frizzled extracellular domain is a ligand for Van Gogh/Stbm during nonautonomous planar cell polarity signaling. Dev. Cell 15, 462–469 (2008).
Strutt, H., Warrington, S. J. & Strutt, D. Dynamics of core planar polarity protein turnover and stable assembly into discrete membrane subdomains. Dev. Cell 20, 511–525 (2011). This first highly dynamic analysis of PCP localization contributes greatly to how we understand and study PCP signalling complex assembly.
Strutt, D. I. Asymmetric localization of frizzled and the establishment of cell polarity in the Drosophila wing. Mol. Cell 7, 367–375 (2001).
Bastock, R., Strutt, H. & Strutt, D. Strabismus is asymmetrically localised and binds to Prickle and Dishevelled during Drosophila planar polarity patterning. Development 130, 3007–3014 (2003).
Struhl, G., Casal, J. & Lawrence, P. A. Dissecting the molecular bridges that mediate the function of Frizzled in planar cell polarity. Development 139, 3665–3674 (2012). This comprehensive study of transmembrane core PCP components provides multiple insights into the intricacies of intercellular PCP signalling.
Axelrod, J. D., Miller, J. R., Shulman, J. M., Moon, R. T. & Perrimon, N. Differential recruitment of Dishevelled provides signaling specificity in the planar cell polarity and Wingless signaling pathways. Genes Dev. 12, 2610–2622 (1998).
Axelrod, J. D. Unipolar membrane association of Dishevelled mediates Frizzled planar cell polarity signaling. Genes Dev. 15, 1182–1187 (2001).
Jenny, A., Darken, R. S., Wilson, P. A. & Mlodzik, M. Prickle and Strabismus form a functional complex to generate a correct axis during planar cell polarity signaling. EMBO J. 22, 4409–4420 (2003).
Jenny, A., Reynolds-Kenneally, J., Das, G., Burnett, M. & Mlodzik, M. Diego and Prickle regulate Frizzled planar cell polarity signalling by competing for Dishevelled binding. Nat. Cell Biol. 7, 691–697 (2005).
Strutt, D. & Strutt, H. Differential activities of the core planar polarity proteins during Drosophila wing patterning. Dev. Biol. 302, 181–194 (2007).
Tree, D. R. P. et al. Prickle mediates feedback amplification to generate asymmetric planar cell polarity signaling. Cell 109, 371–381 (2002).
Butler, M. T. & Wallingford, J. B. Control of vertebrate core planar cell polarity protein localization and dynamics by Prickle 2. Development 142, 3429–3439 (2015).
Park, M. & Moon, R. T. The planar cell-polarity gene stbm regulates cell behaviour and cell fate in vertebrate embryos. Nat. Cell Biol. 4, 20–25 (2002).
Das, G., Jenny, A., Klein, T. J., Eaton, S. & Mlodzik, M. Diego interacts with Prickle and Strabismus/Van Gogh to localize planar cell polarity complexes. Development 131, 4467–4476 (2004).
Vladar, E. K., Bayly, R. D., Sangoram, A. M., Scott, M. P. & Axelrod, J. D. Microtubules enable the planar cell polarity of airway cilia. Curr. Biol. 22, 2203–2212 (2012). This exhaustive analysis reveals cell- and tissue-specific localizations of different vertebrate PCP family members and strong links between PCP signalling and microtubule polarity.
Aw, W. Y., Heck, B. W., Joyce, B. & Devenport, D. Transient tissue-scale deformation coordinates alignment of planar cell polarity junctions in the mammalian skin. Curr. Biol. 26, 2090–2100 (2016).
Mottola, G., Classen, A. K., Gonzalez-Gaitan, M., Eaton, S. & Zerial, M. A novel function for the Rab5 effector Rabenosyn-5 in planar cell polarity. Development 137, 2353–2364 (2010).
Classen, A.-K., Anderson, K. I., Marois, E. & Eaton, S. Hexagonal packing of Drosophila wing epithelial cells by the planar cell polarity pathway. Dev. Cell 9, 805–817 (2005).
Cho, B., Pierre-Louis, G., Sagner, A., Eaton, S. & Axelrod, J. D. Clustering and negative feedback by endocytosis in planar cell polarity signaling is modulated by ubiquitinylation of Prickle. PLoS Genet. 11, e1005259 (2015).
Wong, H.-C. et al. Direct binding of the PDZ domain of Dishevelled to a conserved internal sequence in the C-terminal region of Frizzled. Mol. Cell 12, 1251–1260 (2003).
Yu, A. et al. Association of Dishevelled with the clathrin AP-2 adaptor is required for Frizzled endocytosis and planar cell polarity signaling. Dev. Cell 12, 129–141 (2007).
Carvajal-Gonzalez, J. M. et al. The clathrin adaptor AP-1 complex and Arf1 regulate planar cell polarity in vivo. Nat. Commun. 6, 6751 (2015).
Devenport, D., Oristian, D., Heller, E. & Fuchs, E. Mitotic internalization of planar cell polarity proteins preserves tissue polarity. Nat. Cell Biol. 13, 893–902 (2011).
Shrestha, R. et al. Mitotic control of planar cell polarity by Polo-like kinase 1. Dev. Cell 33, 522–534 (2015).
Eaton, S., Wepf, R. & Simons, K. Roles for Rac1 and Cdc42 in planar polarization and hair outgrowth in the wing of Drosophila. J. Cell Biol. 135, 1277–1289 (1996).
Hannus, M., Feiguin, F., Heisenberg, C.-P. & Eaton, S. Planar cell polarization requires Widerborst, a B' regulatory subunit of protein phosphatase 2A. Development 129, 3493–3503 (2002).
Shimada, Y., Yonemura, S., Ohkura, H., Strutt, D. & Uemura, T. Polarized transport of Frizzled along the planar microtubule arrays in Drosophila wing epithelium. Dev. Cell 10, 209–222 (2006).
Sepich, D. S., Usmani, M., Pawlicki, S. & Solnica-Krezel, L. Wnt/PCP signaling controls intracellular position of MTOCs during gastrulation convergence and extension movements. Development 138, 543–552 (2011).
Olofsson, J., Sharp, K. A., Matis, M., Cho, B. & Axelrod, J. D. Prickle/Spiny-legs isoforms control the polarity of the apical microtubule network in planar cell polarity. Development 141, 2866–2874 (2014).
Matis, M., Russler-Germain, D. A., Hu, Q., Tomlin, C. J. & Axelrod, J. D. Microtubules provide directional information for core PCP function. eLife 3, e02893 (2014).
Shi, D. et al. Dynamics of planar cell polarity protein Vangl2 in the mouse oviduct epithelium. Mech. Dev. 141, 78–89 (2016).
Chien, Y.-H., Keller, R., Kintner, C. & Shook, D. R. Mechanical strain determines the axis of planar polarity in ciliated epithelia. Curr. Biol. 25, 2774–2784 (2015).
Cervenka, I. et al. Dishevelled is a NEK2 kinase substrate controlling dynamics of centrosomal linker proteins. Proc. Natl Acad. Sci. USA 113, 9304–9309 (2016).
Narimatsu, M. et al. Regulation of planar cell polarity by Smurf ubiquitin ligases. Cell 137, 295–307 (2009). This paper reveals ubiquitylation and degradation as PCP signalling feedback mechanisms.
Gao, B. et al. Wnt signaling gradients establish planar cell polarity by inducing Vangl2 phosphorylation through Ror2. Dev. Cell 20, 163–176 (2011). This examination of Wnt and its directional influence on cell behaviour provides insight into the aetiology of Robinow syndrome and related phenotypes that can result from mutant PCP components.
Daulat, A. M. et al. Mink1 regulates β-catenin-independent Wnt signalling via Prickle phosphorylation. Mol. Cell. Biol. 32, 173–185 (2012).
Shafer, B., Onishi, K., Lo, C., Colakoglu, G. & Zou, Y. Vangl2 promotes Wnt/planar cell polarity-like signaling by antagonizing Dvl1-mediated feedback inhibition in growth cone guidance. Dev. Cell 20, 177–191 (2011).
Strutt, H. & Strutt, D. EGF signaling and ommatidial rotation in the Drosophila eye. Curr. Biol. 13, 1451–1457 (2003).
Strutt, H., Searle, E., Thomas-MacArthur, V., Brookfield, R. & Strutt, D. A. Cul-3-BTB ubiquitylation pathway regulates junctional levels and asymmetry of core planar polarity proteins. Development 140, 1693–1702 (2013).
Strutt, H., Thomas-MacArthur, V. & Strutt, D. Strabismus promotes recruitment and degradation of farnesylated Prickle in Drosophila melanogaster planar polarity specification. PLoS Genet. 9, e1003654 (2013).
Thomas, C. & Strutt, D. The roles of the cadherins Fat and Dachsous in planar polarity specification in Drosophila. Dev. Dyn. 241, 27–39 (2011).
Matakatsu, H. & Blair, S. S. Interactions between Fat and Dachsous and the regulation of planar cell polarity in the Drosophila wing. Development 131, 3785–3794 (2004).
Casal, J., Lawrence, P. A. & Struhl, G. Two separate molecular systems, Dachsous/Fat and Starry night/Frizzled, act independently to confer planar cell polarity. Development 133, 4561–4572 (2006).
Yang, C.-H., Axelrod, J. D. & Simon, M. A. Regulation of Frizzled by fat-like cadherins during planar polarity signaling in the Drosophila compound eye. Cell 108, 675–688 (2002).
Ma, D., Yang, C.-H., McNeill, H., Simon, M. A. & Axelrod, J. D. Fidelity in planar cell polarity signalling. Nature 421, 543–547 (2003).
Gubb, D. et al. The balance between isoforms of the prickle LIM domain protein is critical for planar polarity in Drosophila imaginal discs. Genes Dev. 13, 2315–2327 (1999).
Ayukawa, T. et al. Dachsous-dependent asymmetric localization of Spiny-legs determines planar cell polarity orientation in Drosophila. Cell Rep. 8, 610–621 (2014).
Ambegaonkar, A. A. & Irvine, K. D. Coordination of planar cell polarity pathways through Spiny legs. eLife 27, e09946 (2015).
Merkel, M. et al. The balance of Prickle/Spiny-legs isoforms controls the amount of coupling between Core and Fat PCP systems. Curr. Biol. 24, 2111–2123 (2014).
Sokol, S. Y. Spatial and temporal aspects of Wnt signaling and planar cell polarity during vertebrate embryonic development. Semin. Cell Dev. Biol. 42, 78–85 (2015).
Wu, J., Roman, A.-C., Carvajal-Gonzalez, J. M. & Mlodzik, M. Wg and Wnt4 provide long-range directional input to planar cell polarity orientation in Drosophilas. Nat. Cell Biol. 15, 1–13 (2013).
Chu, C.-W. & Sokol, S. Y. Wnt proteins can direct planar cell polarity in vertebrate ectoderm. eLife 5, e16463 (2016). This study utilizes dual mosaic embryos to provide solid evidence for an instructive role of Wnt proteins in determining PCP pattern orientation.
Mitchell, B., Jacobs, R., Li, J., Chien, S. & Kintner, C. A positive feedback mechanism governs the polarity and motion of motile cilia. Nature 447, 97–101 (2007).
Mitchell, B. et al. The PCP pathway instructs the planar orientation of ciliated cells in the Xenopus larval skin. Curr. Biol. 19, 924–929 (2009).
Guirao, B. et al. Coupling between hydrodynamic forces and planar cell polarity orients mammalian motile cilia. Nat. Cell Biol. 12, 341–350 (2010).
Aigouy, B. et al. Cell flow reorients the axis of planar polarity in the wing epithelium of Drosophila. Cell 142, 773–786 (2010). This tissue-wide analysis of PCP orientation over time shows shifts along the axis of dramatic growth and cell rearrangements, suggesting that forces can contribute to PCP alignment.
Mahaffey, J. P., Grego-Bessa, J., Liem, K. F. & Anderson, K. V. Cofilin and Vangl2 cooperate in the initiation of planar cell polarity in the mouse embryo. Development 140, 1262–1271 (2013).
Luxenburg, C. et al. Wdr1-mediated cell shape dynamics and cortical tension are essential for epidermal planar cell polarity. Nat. Cell Biol. 17, 592–604 (2015).
Ossipova, O. et al. Role of Rab11 in planar cell polarity and apical constriction during vertebrate neural tube closure. Nat. Commun. 5, 1–8 (2014).
Hooke, R. Micrographia or Some Physiological Descriptions of Minute Bodies Made by Magnifying Glasses (J. Martyn & J. Allestry, 1665).
Vinson, C. R. & Adler, P. N. Directional non-cell autonomy and the transmission of polarity information by the frizzled gene of Drosophila. Nature 329, 549–551 (1987). This report uncovers the directional, non-autonomous effects of Fz perturbation on neighbouring cells and thus reveals the coordinated nature of planar tissue polarity.
Taylor, J., Abramova, N., Charlton, J. & Adler, P. N. Van Gogh: a new Drosophila tissue polarity gene. Genetics 150, 199–210 (1998).
Jenny, A. Planar cell polarity signaling in the Drosophila eye. Curr. Top. Dev. Biol. 93, 189 (2010).
Tada, M. & Smith, J. C. Xwnt11 is a target of Xenopus Brachyury: regulation of gastrulation movements via Dishevelled, but not through the canonical Wnt pathway. Development 127, 2227–2238 (2000).
Heisenberg, C. P. et al. Silberblick/Wnt11 mediates convergent extension movements during zebrafish gastrulation. Nature 405, 76–81 (2000).
Wallingford, J. B. et al. Dishevelled controls cell polarity during Xenopus gastrulation. Nature 405, 81–85 (2000). References 73–75 are pioneering reports of PCP signalling in vertebrates and provide the foundation for understanding how PCP perturbations might lead to NTDs.
Tada, M. & Heisenberg, C. P. Convergent extension: using collective cell migration and cell intercalation to shape embryos. Development 139, 3897–3904 (2012).
Lienkamp, S. S. et al. Vertebrate kidney tubules elongate using a planar cell polarity-dependent, rosette-based mechanism of convergent extension. Nat. Genet. 44, 1382–1387 (2012).
Jiang, D., Munro, E. M. & Smith, W. C. Ascidian prickle regulates both mediolateral and anterior–posterior cell polarity of notochord cells. Curr. Biol. 15, 79–85 (2005).
Ciruna, B., Jenny, A., Lee, D., Mlodzik, M. & Schier, A. F. Planar cell polarity signalling couples cell division and morphogenesis during neurulation. Nat. Cell Biol. 439, 220–224 (2006).
Yin, C., Kiskowski, M., Pouille, P.-A., Farge, E. & Solnica-Krezel, L. Cooperation of polarized cell intercalations drives convergence and extension of presomitic mesoderm during zebrafish gastrulation. J. Cell Biol. 180, 221–232 (2008).
Ossipova, O., Kim, K. & Sokol, S. Y. Planar polarization of Vangl2 in the vertebrate neural plate is controlled by Wnt and Myosin II signaling. Biol. Open 4, 722–730 (2015).
Roszko, I. S. Sepich, D., Jessen, J. R., Chandrasekhar, A. & Solnica-Krezel, L. A dynamic intracellular distribution of Vangl2 accompanies cell polarization during zebrafish gastrulation. Development 142, 2508–2520 (2015).
Rida, P. C. G. & Chen, P. Line up and listen: planar cell polarity regulation in the mammalian inner ear. Semin. Cell Dev. Biol. 20, 978–985 (2009).
Wallingford, J. B. Planar cell polarity and the developmental control of cell behavior in vertebrate embryos. Annu. Rev. Cell Dev. Biol. 28, 627–653 (2012).
Devenport, D. Tissue morphodynamics: translating planar polarity cues into polarized cell behaviors. Semin. Cell Dev. Biol. 55, 99–110 (2016).
Sharpey, W. in Cyclopædia of Anatomy and Physiology Vol. 1 (ed. Todd, R. B) 606–637 (Sherwood, Gilbert and Piper, 1836).
Park, T. J., Mitchell, B. J., Abitua, P. B., Kintner, C. & Wallingford, J. B. Dishevelled controls apical docking and planar polarization of basal bodies in ciliated epithelial cells. Nat. Genet. 40, 871–879 (2008).
Tissir, F. et al. Lack of cadherins Celsr2 and Celsr3 impairs ependymal ciliogenesis, leading to fatal hydrocephalus. Nat. Neurosci. 13, 700–707 (2010).
Ohata, S. et al. Loss of Dishevelleds disrupts planar polarity in ependymal motile cilia and results in hydrocephalus. Neuron 83, 558–571 (2014).
Boutin, C. et al. A dual role for planar cell polarity genes in ciliated cells. Proc. Natl Acad. Sci. USA 111, E3129–E3138 (2014).
Shi, D. et al. Celsr1 is required for the generation of polarity at multiple levels of the mouse oviduct. Development 141, 4558–4568 (2014).
Hegan, P. S., Ostertag, E., Geurts, A. M. & Mooseker, M. S. Myosin Id is required for planar cell polarity in ciliated tracheal and ependymal epithelial cells. Cytoskeleton (Hoboken) 72, 503–516 (2015).
Teves, M. E. et al. Sperm-associated antigen 6 (SPAG6) deficiency and defects in ciliogenesis and cilia function: polarity, density, and beat. PLoS ONE 9, e107271 (2014).
Jaffe, K. M. et al. c21orf59/kurly controls both cilia motility and polarization. Cell Rep. 14, 1841–1849 (2016).
Kunimoto, K. et al. Coordinated ciliary beating requires Odf2-mediated polarization of basal bodies via basal feet. Cell 148, 189–200 (2012).
Ying, G. et al. Centrin 2 is required for mouse olfactory ciliary trafficking and development of ependymal cilia planar polarity. J. Neurosci. 34, 6377–6388 (2014).
Turk, E. et al. Zeta-tubulin is a member of a conserved tubulin module and is a component of the centriolar basal foot in multiciliated cells. Curr. Biol. 25, 2177–2183 (2015).
Shih, J. & Keller, R. Cell motility driving mediolateral intercalation in explants of Xenopus laevis. Development 116, 901–914 (1992).
Shindo, A. & Wallingford, J. B. PCP and septins compartmentalize cortical actomyosin to direct collective cell movement. Science 343, 649–652 (2014).
Yen, W. W. et al. PTK7 is essential for polarized cell motility and convergent extension during mouse gastrulation. Development 136, 2039–2048 (2009).
Nishimura, T., Honda, H. & Takeichi, M. Planar cell polarity links axes of spatial dynamics in neural-tube closure. Cell 149, 1084–1097 (2012). This study shows that PCP signalling controls the spatially restricted contractile actomyosin machinery, providing a mechanism by which PCP guides neuroepithelial cell rearrangements.
Williams, M., Yen, W., Lu, X. & Sutherland, A. Distinct apical and basolateral mechanisms drive planar cell polarity-dependent convergent extension of the mouse neural plate. Dev. Cell 29, 34–46 (2014).
Strutt, D. I., Weber, U. & Mlodzik, M. The role of RhoA in tissue polarity and Frizzled signalling. Nature 387, 292–295 (1997).
Winter, C. G. et al. Drosophila Rho-associated kinase (Drok) links Frizzled-mediated planar cell polarity signaling to the actin cytoskeleton. Cell 105, 81–91 (2001).
Bosveld, F. et al. Mechanical control of morphogenesis by Fat/Dachsous/Four-jointed planar cell polarity pathway. Science 336, 724–727 (2012). This paper shows that asymmetric Ds polarizes the myosin Dachs as a mechanism to generate the anisotropic junctional tension essential for proper tissue morphogenesis.
Lu, X. et al. PTK7/CCK-4 is a novel regulator of planar cell polarity in vertebrates. Nature 430, 93–98 (2004).
Hayes, M., Naito, M., Daulat, A., Angers, S. & Ciruna, B. Ptk7 promotes non-canonical Wnt/PCP-mediated morphogenesis and inhibits Wnt/β-catenin-dependent cell fate decisions during vertebrate development. Development 140, 1807–1818 (2013).
Lee, J. et al. PTK7 regulates myosin II activity to orient planar polarity in the mammalian auditory epithelium. Curr. Biol. 22, 956–966 (2012).
Andreeva, A. et al. PTK7-Src signaling at epithelial cell contacts mediates spatial organization of actomyosin and planar cell polarity. Dev. Cell 29, 20–33 (2014).
Kim, S. K. et al. Planar cell polarity acts through septins to control collective cell movement and ciliogenesis. Science 329, 1337–1340 (2010).
Kibar, Z. et al. Ltap, a mammalian homolog of Drosophila Strabismus/Van Gogh, is altered in the mouse neural tube mutant Loop-tail. Nat. Genet. 28, 251–255 (2001).
Murdoch, J. N., Doudney, K., Paternotte, C., Copp, A. J. & Stanier, P. Severe neural tube defects in the loop-tail mouse result from mutation of Lpp1, a novel gene involved in floor plate specification. Hum. Mol. Genet. 10, 2593–2601 (2001).
Wallingford, J. B. & Harland, R. M. Neural tube closure requires Dishevelled-dependent convergent extension of the midline. Development 129, 5815–5825 (2002).
Wang, J. et al. Dishevelled genes mediate a conserved mammalian PCP pathway to regulate convergent extension during neurulation. Development 133, 1767–1778 (2006).
Ybot-Gonzalez, P. et al. Convergent extension, planar-cell-polarity signalling and initiation of mouse neural tube closure. Development 134, 789–799 (2007).
Kibar, Z. et al. Mutations in VANGL1 associated with neural-tube defects. N. Engl. J. Med. 356, 1432–1437 (2007).
Bosoi, C. M. et al. Identification and characterization of novel rare mutations in the planar cell polarity gene PRICKLE1 in human neural tube defects. Hum. Mutat. 32, 1371–1375 (2011).
Seo, J. H. et al. Mutations in the planar cell polarity gene, Fuzzy, are associated with neural tube defects in humans. Hum. Mol. Genet. 20, 4324–4333 (2011).
Brzoska, H. L. et al. Planar cell polarity genes Celsr1 and Vangl2 are necessary for kidney growth, differentiation, and rostrocaudal patterning. Kidney Int. 90, 1274–1284 (2016).
Kochanek, K. D., Murphy, S. L., Xu, J. & Tejada-Vera, B. Deaths: final data for 2014. Natl Vital Stat. Rep. 65, 1–122 (2016).
Wallingford, J. B., Niswander, L. A., Shaw, G. M. & Finnell, R. H. The continuing challenge of understanding, preventing, and treating neural tube defects. Science 339, 1222002 (2013).
Juriloff, D. M. & Harris, M. J. A consideration of the evidence that genetic defects in planar cell polarity contribute to the etiology of human neural tube defects. Birth Defects Res. A Clin. Mol. Teratol. 94, 824–840 (2012).
De Marco, P. et al. Planar cell polarity gene mutations contribute to the etiology of human neural tube defects in our population. Birth Defects Res. A Clin. Mol. Teratol. 100, 633–641 (2014).
Robinson, A. et al. Mutations in the planar cell polarity genes CELSR1 and SCRIB are associated with the severe neural tube defect craniorachischisis. Hum. Mutat. 33, 440–447 (2012).
Iliescu, A., Gravel, M., Horth, C. & Gros, P. Independent mutations at Arg181 and Arg274 of Vangl proteins that are associated with neural tube defects in humans decrease protein stability and impair membrane targeting. Biochemistry 53, 5356–5364 (2014).
Lei, Y. et al. Identification of novel CELSR1 mutations in spina bifida. PLoS ONE 9, e92207 (2014).
Afzal, A. R. et al. Recessive Robinow syndrome, allelic to dominant brachydactyly type B, is caused by mutation of ROR2. Nat. Genet. 25, 419–422 (2000).
van Bokhoven, H. et al. Mutation of the gene encoding the ROR2 tyrosine kinase causes autosomal recessive Robinow syndrome. Nat. Genet. 25, 423–426 (2000).
White, J. et al. DVL1 frameshift mutations clustering in the penultimate exon cause autosomal-dominant Robinow syndrome. Am. J. Hum. Genet. 96, 612–622 (2015).
Bunn, K. J. et al. Mutations in DVL1 cause an osteosclerotic form of Robinow syndrome. Am. J. Hum. Genet. 96, 623–630 (2015).
White, J. J. et al. DVL3 alleles resulting in a −1 frameshift of the last exon mediate autosomal-dominant Robinow syndrome. Am. J. Hum. Genet. 98, 553–561 (2016).
Person, A. D. et al. WNT5A mutations in patients with autosomal dominant Robinow syndrome. Dev. Dyn. 239, 327–337 (2010).
Hayes, M. et al. ptk7 mutant zebrafish models of congenital and idiopathic scoliosis implicate dysregulated Wnt signalling in disease. Nat. Commun. 5, 4777 (2014).
Grimes, D. T. et al. Zebrafish models of idiopathic scoliosis link cerebrospinal fluid flow defects to spine curvature. Science 352, 1341–1344 (2016).
Andersen, M. R. et al. Mutation of the planar cell polarity gene VANGL1 in adolescent idiopathic scoliosis. Spine http://dx.doi.org/10.1097/BRS.0000000000001927 (2016).
Matis, M. & Axelrod, J. D. Regulation of PCP by the Fat signaling pathway. Genes Dev. 27, 2207–2220 (2013).
Rock, R., Schrauth, S. & Gessler, M. Expression of mouse dchs1, fjx1, and fat-j suggests conservation of the planar cell polarity pathway identified in Drosophila. Dev. Dyn. 234, 747–755 (2005).
Saburi, S. et al. Loss of Fat4 disrupts PCP signaling and oriented cell division and leads to cystic kidney disease. Nat. Genet. 40, 1010–1015 (2008).
Mao, Y. et al. Characterization of a Dchs1 mutant mouse reveals requirements for Dchs1–Fat4 signaling during mammalian development. Development 138, 947–957 (2011).
Zakaria, S. et al. Regulation of neuronal migration by Dchs1–Fat4 planar cell polarity. Curr. Biol. 24, 1620–1627 (2014).
Mao, Y. et al. Dchs1–Fat4 regulation of polarized cell behaviours during skeletal morphogenesis. Nat. Commun. 7, 11469 (2016).
Strutt, H. & Strutt, D. Nonautonomous planar polarity patterning in Drosophila: dishevelled-independent functions of frizzled. Dev. Cell 3, 851–863 (2002).
Ishikawa, H. O., Takeuchi, H., Haltiwanger, R. S. & Irvine, K. D. Four-jointed is a Golgi kinase that phosphorylates a subset of cadherin domains. Science 321, 401–404 (2008).
Simon, M. A., Xu, A., Ishikawa, H. O. & Irvine, K. D. Modulation of Fat:Dachsous binding by the cadherin domain kinase Four-jointed. Curr. Biol. 20, 811–817 (2010).
Brittle, A. L., Repiso, A., Casal, J., Lawrence, P. A. & Strutt, D. Four-jointed modulates growth and planar polarity by reducing the affinity of Dachsous for Fat. Curr. Biol. 20, 803–810 (2010).
Hale, R., Brittle, A. L., Fisher, K. H., Monk, N. A. M. & Strutt, D. Cellular interpretation of the long-range gradient of Four-jointed activity in the Drosophila wing. eLife 4, e05789 (2015).
Brittle, A., Thomas, C. & Strutt, D. Planar polarity specification through asymmetric subcellular localization of Fat and Dachsous. Curr. Biol. 22, 907–914 (2012).
Ambegaonkar, A. A., Pan, G., Mani, M., Feng, Y. & Irvine, K. D. Propagation of Dachsous–Fat planar cell polarity. Curr. Biol. 22, 1302–1308 (2012).
Rovira, M., Saavedra, P., Casal, J. & Lawrence, P. A. Regions within a single epidermal cell of Drosophila can be planar polarised independently. eLife 4, e06303 (2015).
Simons, M. et al. Electrochemical cues regulate assembly of the Frizzled/Dishevelled complex at the plasma membrane during planar epithelial polarization. Nat. Cell Biol. 11, 286–294 (2009).
Acknowledgements
J.B.W. is supported by grants from the National Heart, Lung, and Blood Institute (R01HL117164), the National Institute of General Medical Sciences (R01 GM104853-02) and the National Institute of Child Health and Human Development (1R01HD085901, 5R21HD084072).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information S1 (box)
Apico-basal versus Planar Cell Polarity (PDF 496 kb)
Supplementary information S2 (box)
Planar cell polarity and the actin cytoskeleton (PDF 99 kb)
Glossary
- Notum
-
The dorsal region of the thorax of a mature insect.
- Trichome
-
An actin-based hair found on each cell of the fly wing blade that emanates from the apical, distal side of the cells and points distally — properties that are controlled by planar cell polarity signalling.
- Endocytic flux
-
The constant endocytosis and subsequent recycling of unstable protein components associated with the plasma membrane.
- Rab GTPase family
-
A large family of small GTPases central to membrane trafficking regulation.
- Dynamin
-
A GTPase protein critical for driving membrane fission that facilitates endocytic events.
- Clathrin
-
A vesicular coating component that has an important structural role in mediating endocytosis of membrane-associated proteins.
- Polo-like kinase 1
-
(PLK1). A Ser/Thr kinase that serves as an important cell cycle regulator, playing a notable part during the G2–M transition.
- Protocadherins
-
The largest subgroup of cadherins with variable extracellular domains and diverse cytoplasmic domains that distinguish their variety of potential functions from those of classical cadherins.
- Mutant clones
-
Groups of cells in a tissue that have perturbed gene expression and are surrounded by or interspersed between otherwise wild-type cells.
- Ommatidia
-
Optical units of insect compound eyes containing groups of polarized photoreceptive cells.
- Central fold
-
Midline of the neural plate that divides the left and right half; it comprises cells that apically constrict to facilitate neural plate folding and neural tube closure.
- Neural plate
-
A sheet of neuroepithelial cells that undergo convergent extension, apical constriction and regional folding to form the vertebrate neural tube.
- Kinocilium
-
A microtubule-based protrusion on the apical surface of hair cells in tissues of the inner ear that guides the polarized orientation of an actin-based stereociliary bundle.
- Basal bodies
-
Modified centrioles and accessory proteins that serve as specialized microtubule-organizing centres at the base of the cilium, which they nucleate.
- Ependymal cells
-
Multiciliated cells that line the interface between the central nervous system and cerebrospinal fluid, which propel cerebrospinal flow through the brain ventricles and central spinal canal.
- Non-canonical tubulins
-
Tubulin superfamily members that are not conserved in all eukaryotes as α-, β-, and γ-tubulin are; non-canonical tubulins often function as centriole accessory structures.
- Traction force
-
The forces exerted across the junction between two cells that facilitate cell rearrangements and tissue morphogenesis.
- Septins
-
Cytoskeletal components that upon binding to GTP can polymerize into ordered structures such as rings and filaments, which can function as scaffolds or diffusion barriers, for example.
- Spina bifida
-
A neural tube defect associated with a regional, incomplete closure of the spinal cord and associated tissues.
Rights and permissions
About this article
Cite this article
Butler, M., Wallingford, J. Planar cell polarity in development and disease. Nat Rev Mol Cell Biol 18, 375–388 (2017). https://doi.org/10.1038/nrm.2017.11
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm.2017.11
This article is cited by
-
Frizzled receptors (FZDs) in Wnt signaling: potential therapeutic targets for human cancers
Acta Pharmacologica Sinica (2024)
-
Recent insights into the therapeutic strategies targeting the pseudokinase PTK7 in cancer
Oncogene (2024)
-
Helicobacter pylori and Epstein-Barr virus infection in cell polarity alterations
Folia Microbiologica (2024)
-
The role of prickle proteins in vertebrate development and pathology
Molecular and Cellular Biochemistry (2024)
-
Research Progress on the Mechanism of the SFRP-Mediated Wnt Signalling Pathway Involved in Bone Metabolism in Osteoporosis
Molecular Biotechnology (2024)