Key Points
-
Reverse cholesterol transport is a process by which cholesterol is transferred from peripheral cells, including macrophages, to the liver for excretion. The acute phase response results in suppression of reverse cholesterol transport at multiple steps, which may in turn promote cholesterol accumulation in macrophages and other immune cells. This can lead to a beneficial enhancement of inflammatory responses in the setting of infection, but when inflammation becomes prolonged these changes may worsen conditions such as atherosclerosis and obesity.
-
During the acute phase response, high-density lipoprotein (HDL) levels are decreased and compositional changes in HDL, including myeloperoxidase-mediated modifications of apolipoprotein A1 (APOA1), may convert HDL into a dysfunctional form that cannot efficiently mediate cholesterol efflux and that becomes pro-inflammatory. Although these changes in HDL and APOA1 are probably pro-atherogenic, they may also have a physiological function in the setting of infection by enhancing the inflammatory response.
-
Liver X receptor (LXR) transcription factors promote reverse cholesterol transport by inducing the expression of genes involved in cellular cholesterol efflux, transport in the bloodstream and excretion in the liver. The mechanisms connecting inflammation with decreases in reverse cholesterol transport include the ability of endotoxins to suppress the expression of LXR and its partner retinoid X recceptor (RXR), as well as the suppression of cellular LXR responses via a trans-repression mechanism.
-
The mechanisms of pro-inflammatory effects of cellular cholesterol accumulation include enhanced Toll-like receptor (TLR) signalling and inflammasome activation. Inflammasome activation may be stimulated by cholesterol crystal uptake or formation in macrophages. Conversely, the induction of cholesterol 25-hydroxylase by lipopolysaccharide and type I interferons opposes inflammasome activation, probably because 25-hydroxycholesterol suppresses cellular sterol synthesis.
-
Defective cholesterol efflux promotes monocyte and neutrophil production in the bone marrow and the spleen, involving the proliferation of haematopoietic stem cells (HSCs) and myeloid progenitor cells, mobilization of HSCs and extramedullary haematopoiesis. Although these pathways probably enhance the response to infections, genetic suppression and dietary challenge lead to aberrant responses that promote atherogenesis.
-
Therapeutic interventions such as increased production or infusion of HDL may sever the links between cholesterol accumulation and inflammation with benefits for metabolic diseases. This may involve infusions of cholesterol-poor reconstituted HDL or targeting the APOA1 gene locus to increase endogenous APOA1 production.
Abstract
Hypercholesterolaemia leads to cholesterol accumulation in macrophages and other immune cells, which promotes inflammatory responses, including augmentation of Toll-like receptor (TLR) signalling, inflammasome activation, and the production of monocytes and neutrophils in the bone marrow and spleen. On a cellular level, activation of TLR signalling leads to decreased cholesterol efflux, which results in further cholesterol accumulation and the amplification of inflammatory responses. Although cholesterol accumulation through the promotion of inflammatory responses probably has beneficial effects in the response to infections, it worsens diseases that are associated with chronic metabolic inflammation, including atherosclerosis and obesity. Therapeutic interventions such as increased production or infusion of high-density lipoproteins may sever the links between cholesterol accumulation and inflammation, and have beneficial effects in patients with metabolic diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Moore, K. J. & Tabas, I. Macrophages in the pathogenesis of atherosclerosis. Cell 145, 341–355 (2011).
Stewart, C. R. et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nature Immunol. 11, 155–161 (2010).
Yvan-Charvet, L. et al. Combined deficiency of ABCA1 and ABCG1 promotes foam cell accumulation and accelerates atherosclerosis in mice. J. Clin. Invest. 117, 3900–3908 (2007).
Yvan-Charvet, L. et al. Increased inflammatory gene expression in ABC transporter-deficient macrophages: free cholesterol accumulation, increased signaling via toll-like receptors, and neutrophil infiltration of atherosclerotic lesions. Circulation 118, 1837–1847 (2008).
Fessler, M. B. & Parks, J. S. Intracellular lipid flux and membrane microdomains as organizing principles in inflammatory cell signaling. J. Immunol. 187, 1529–1535 (2011).
Zhu, X. et al. Macrophage ABCA1 reduces MyD88-dependent Toll-like receptor trafficking to lipid rafts by reduction of lipid raft cholesterol. J. Lipid Res. 51, 3196–3206 (2010).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010). This study provides the first evidence for cholesterol crystal-induced inflammasome activation in atherosclerotic lesions and shows that genetic deletion of essential inflammasome components results in reduced atherosclerosis.
Sheedy, F. J. et al. CD36 coordinates NLRP3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nature Immunol. 14, 812–820 (2013).
Tall, A. R., Yvan-Charvet, L., Terasaka, N., Pagler, T. & Wang, N. H. D. L. ABC transporters, and cholesterol efflux: implications for the treatment of atherosclerosis. Cell. Metab. 7, 365–375 (2008).
Rader, D. J. & Tall, A. R. The not-so-simple HDL story: Is it time to revise the HDL cholesterol hypothesis? Nature Med. 18, 1344–1346 (2012).
Castrillo, A. et al. Crosstalk between LXR and toll-like receptor signaling mediates bacterial and viral antagonism of cholesterol metabolism. Mol. Cell 12, 805–816 (2003). In this paper, activation of TLRs was shown to suppress LXR-responsive genes, notably those involved in cholesterol efflux from macrophages, providing a molecular mechanism to link the response to infectious organisms to suppression of the RCT pathway.
Feingold, K. R. & Grunfeld, C. The acute phase response inhibits reverse cholesterol transport. J. Lipid Res. 51, 682–684 (2010). This paper is an overview of RCT and the acute phase response showing that the acute phase response suppresses RCT at multiple steps.
Post, W. S. et al. Associations between HIV infection and subclinical coronary atherosclerosis. Ann. Intern. Med. 160, 458–467 (2014).
The Strategies for Management of Antiretroviral Therapy Study Group. CD4+ count-guided interruption of antiretroviral treatment. N. Engl. J. Med. 355, 2283–2296 (2006).
Sherer, Y. & Shoenfeld, Y. Mechanisms of disease: atherosclerosis in autoimmune diseases. Nature Clin. Pract. Rheumatol. 2, 99–106 (2006).
Vandanmagsar, B. et al. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nature Med. 17, 179–188 (2011).
Stienstra, R. et al. Inflammasome is a central player in the induction of obesity and insulin resistance. Proc. Natl Acad. Sci. USA 108, 15324–15329 (2011).
Umemoto, T. et al. Apolipoprotein AI and high-density lipoprotein have anti-inflammatory effects on adipocytes via cholesterol transporters: ATP-binding cassette A-1, ATP-binding cassette G-1, and scavenger receptor B-1. Circ. Res. 112, 1345–1354 (2013).
Chung, S. et al. Dietary cholesterol promotes adipocyte hypertrophy and adipose tissue inflammation in visceral, but not in subcutaneous, fat in monkeys. Arterioscler. Thromb. Vasc. Biol. 34, 1880–1887 (2014).
Norata, G. D., Pirillo, A., Ammirati, E. & Catapano, A. L. Emerging role of high density lipoproteins as a player in the immune system. Atherosclerosis 220, 11–21 (2012).
Ronda, N. et al. Impaired serum cholesterol efflux capacity in rheumatoid arthritis and systemic lupus erythematosus. Ann. Rheum. Dis. 73, 609–615 (2014).
Rosenson, R. S. et al. Cholesterol efflux and atheroprotection: advancing the concept of reverse cholesterol transport. Circulation 125, 1905–1919 (2012).
Wang, X. et al. Macrophage ABCA1 and ABCG1, but not SR-BI, promote macrophage reverse cholesterol transport in vivo. J. Clin. Invest. 117, 2216–2224 (2007). This paper uses mice with deficiency of Abca1 and Abcg1 in macrophages to show that these cholesterol efflux-promoting transporters promote the movement of cholesterol from macrophages to the liver followed by excretion in the faeces — so-called 'macrophage reverse cholesterol transport'.
Wang, N., Lan, D., Chen, W., Matsuura, F. & Tall, A. R. ATP-binding cassette transporters G1 and G4 mediate cellular cholesterol efflux to high-density lipoproteins. Proc. Natl Acad. Sci. USA 101, 9774–9779 (2004).
Rader, D. J., Alexander, E. T., Weibel, G. L., Billheimer, J. & Rothblat, G. H. The role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis. J. Lipid Res. 50, S189–S194 (2009).
Martel, C. et al. Lymphatic vasculature mediates macrophage reverse cholesterol transport in mice. J. Clin. Invest. 123, 1571–1579 (2013).
Lim, H. Y. et al. Lymphatic vessels are essential for the removal of cholesterol from peripheral tissues by SR-BI-mediated transport of HDL. Cell. Metab. 17, 671–684 (2013).
Acton, S. et al. Identification of scavenger receptor SR-BI as a high density lipoprotein receptor. Science 271, 518–520 (1996).
Inazu, A. et al. Increased high-density lipoprotein levels caused by a common cholesteryl-ester transfer protein gene mutation. N. Engl. J. Med. 323, 1234–1238 (1990).
Yu, L. et al. Overexpression of ABCG5 and ABCG8 promotes biliary cholesterol secretion and reduces fractional absorption of dietary cholesterol. J. Clin. Invest. 110, 671–680 (2002).
Zhao, G. J. et al. Antagonism of betulinic acid on LPS-mediated inhibition of ABCA1 and cholesterol efflux through inhibiting nuclear factor-κB signaling pathway and miR-33 expression. PLoS ONE 8, e74782 (2013).
Masucci-Magoulas, L. et al. Decreased cholesteryl ester transfer protein (CETP) mRNA and protein and increased high density lipoprotein following lipopolysaccharide administration in human CETP transgenic mice. J. Clin. Invest. 95, 1587–1594 (1995).
McGillicuddy, F. C. et al. Inflammation impairs reverse cholesterol transport in vivo. Circulation 119, 1135–1145 (2009). This paper shows that in humans, the infusion of reconstituted HDL particles suppresses the inflammatory cytokine response to LPS, providing direct evidence for an anti-inflammatory effect of HDL in vivo.
Pajkrt, D. et al. Antiinflammatory effects of reconstituted high-density lipoprotein during human endotoxemia. J. Exp. Med. 184, 1601–1608 (1996).
Harris, H. W., Gosnell, J. E. & Kumwenda, Z. L. The lipemia of sepsis: triglyceride-rich lipoproteins as agents of innate immunity. J. Endotoxin Res. 6, 421–430 (2000).
Van Lenten, B. J. et al. Anti-inflammatory HDL becomes pro-inflammatory during the acute phase response. Loss of protective effect of HDL against LDL oxidation in aortic wall cell cocultures. J. Clin. Invest. 96, 2758–2767 (1995). This paper is an early study to show that HDL becomes pro-inflammatory during the acute phase response.
Watson, A. D. et al. Protective effect of high density lipoprotein associated paraoxonase. Inhibition of the biological activity of minimally oxidized low density lipoprotein. J. Clin. Invest. 96, 2882–2891 (1995).
Annema, W. et al. Myeloperoxidase and serum amyloid A contribute to impaired in vivo reverse cholesterol transport during the acute phase response but not group IIA secretory phospholipase A2 . J. Lipid Res. 51, 743–754 (2010).
de la Llera Moya, M. et al. Inflammation modulates human HDL composition and function in vivo. Atherosclerosis 222, 390–394 (2012).
Bergt, C. et al. The myeloperoxidase product hypochlorous acid oxidizes HDL in the human artery wall and impairs ABCA1-dependent cholesterol transport. Proc. Natl Acad. Sci. USA 101, 13032–13037 (2004).
Zheng, L. et al. Apolipoprotein A-I is a selective target for myeloperoxidase-catalyzed oxidation and functional impairment in subjects with cardiovascular disease. J. Clin. Invest. 114, 529–541 (2004).
McMillen, T. S., Heinecke, J. W. & LeBoeuf, R. C. Expression of human myeloperoxidase by macrophages promotes atherosclerosis in mice. Circulation 111, 2798–2804 (2005).
Hewing, B. et al. Effects of native and myeloperoxidase-modified apolipoprotein a-I on reverse cholesterol transport and atherosclerosis in mice. Arterioscler. Thromb. Vasc. Biol. 34, 779–789 (2014).
Shao, B. et al. Humans with atherosclerosis have impaired ABCA1 cholesterol efflux and enhanced high-density lipoprotein oxidation by myeloperoxidase. Circ. Res. 114, 1733–1742 (2014).
Huang, Y. et al. An abundant dysfunctional apolipoprotein A1 in human atheroma. Nature Med. 20, 193–203 (2014).
Zhu, X. et al. Myeloid cell-specific ABCA1 deletion protects mice from bacterial infection. Circ. Res. 111, 1398–1409 (2012).
Brown, M. S. & Goldstein, J. L. Suppression of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity and inhibition of growth of human fibroblasts by 7-ketocholesterol. J. Biol. Chem. 249, 7306–7314 (1974).
Chen, H. W., Kandutsch, A. A. & Waymouth, C. Inhibition of cell growth by oxygenated derivatives of cholesterol. Nature 251, 419–421 (1974).
Beigneux, A. P., Moser, A. H., Shigenaga, J. K., Grunfeld, C. & Feingold, K. R. The acute phase response is associated with retinoid X receptor repression in rodent liver. J. Biol. Chem. 275, 16390–16399 (2000).
Bjorkbacka, H. et al. Reduced atherosclerosis in MyD88-null mice links elevated serum cholesterol levels to activation of innate immunity signaling pathways. Nature Med. 10, 416–421 (2004). This paper links the TLR signalling pathway to inflammatory cytokine and chemokine production in macrophages in atherosclerotic lesions and to accelerated atherosclerosis in the Apoe−/− mouse model.
Mullick, A. E., Tobias, P. S. & Curtiss, L. K. Modulation of atherosclerosis in mice by Toll-like receptor 2. J. Clin. Invest. 115, 3149–3156 (2005).
Mullick, A. E. et al. Increased endothelial expression of Toll-like receptor 2 at sites of disturbed blood flow exacerbates early atherogenic events. J. Exp. Med. 205, 373–383 (2008).
Feig, J. E. et al. HDL promotes rapid atherosclerosis regression in mice and alters inflammatory properties of plaque monocyte-derived cells. Proc. Natl Acad. Sci. USA 108, 7166–7171 (2011).
Monaco, C. et al. Toll-like receptor-2 mediates inflammation and matrix degradation in human atherosclerosis. Circulation 120, 2462–2469 (2009).
Zhu, X. et al. Increased cellular free cholesterol in macrophage-specific Abca1-knock-out mice enhances pro-inflammatory response of macrophages. J. Biol. Chem. 283, 22930–22941 (2008). This is an early paper showing that ABCA1 deficiency in macrophages results in increased responses to TLR4 ligands.
Westerterp, M. et al. Deficiency of ATP-binding cassette transporters A1 and G1 in macrophages increases inflammation and accelerates atherosclerosis in mice. Circ. Res. 112, 1456–1465 (2013).
Spann, N. J. et al. Regulated accumulation of desmosterol integrates macrophage lipid metabolism and inflammatory responses. Cell 151, 138–152 (2012).
Yang, C. et al. Sterol intermediates from cholesterol biosynthetic pathway as liver X receptor ligands. J. Biol. Chem. 281, 27816–27826 (2006).
Triantafilou, M., Miyake, K., Golenbock, D. T. & Triantafilou, K. Mediators of innate immune recognition of bacteria concentrate in lipid rafts and facilitate lipopolysaccharide-induced cell activation. J. Cell Sci. 115, 2603–2611 (2002).
Pagler, T. A. et al. Deletion of ABCA1 and ABCG1 impairs macrophage migration because of increased Rac1 signaling. Circ. Res. 108, 194–200 (2011).
Li, H. B., Jin, C., Chen, Y. & Flavell, R. A. Inflammasome activation and metabolic disease progression. Cytokine Growth Factor Rev. 25, 699–706 (2014).
Razani, B. et al. Autophagy links inflammasomes to atherosclerotic progression. Cell. Metab. 15, 534–544 (2012).
Nakahira, K. et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nature Immunol. 12, 222–230 (2011).
Shi, C. S. et al. Activation of autophagy by inflammatory signals limits IL-1β production by targeting ubiquitinated inflammasomes for destruction. Nature Immunol. 13, 255–263 (2012).
Ouimet, M. et al. Autophagy regulates cholesterol efflux from macrophage foam cells via lysosomal acid lipase. Cell. Metab. 13, 655–667 (2011).
Sheedy, F. J. & Moore, K. J. IL-1 signaling in atherosclerosis: sibling rivalry. Nature Immunol. 14, 1030–1032 (2013).
Blankenberg, S. et al. Interleukin-18 is a strong predictor of cardiovascular death in stable and unstable angina. Circulation 106, 24–30 (2002).
Whitman, S. C., Ravisankar, P. & Daugherty, A. Interleukin-18 enhances atherosclerosis in apolipoprotein E−/− mice through release of interferon-γ. Circ. Res. 90, e34–e38 (2002).
Mallat, Z. et al. Interleukin-18/interleukin-18 binding protein signaling modulates atherosclerotic lesion development and stability. Circ. Res. 89, e41–e45 (2001).
Menu, P. et al. Atherosclerosis in ApoE-deficient mice progresses independently of the NLRP3 inflammasome. Cell Death Dis. 2, e137 (2011).
Usui, F. et al. Critical role of caspase-1 in vascular inflammation and development of atherosclerosis in Western diet-fed apolipoprotein E-deficient mice. Biochem. Biophys. Res. Commun. 425, 162–168 (2012).
Gage, J., Hasu, M., Thabet, M. & Whitman, S. C. Caspase-1 deficiency decreases atherosclerosis in apolipoprotein E-null mice. Can. J. Cardiol 28, 222–229 (2012).
Zheng, F., Xing, S., Gong, Z., Mu, W. & Xing, Q. Silence of NLRP3 suppresses atherosclerosis and stabilizes plaques in apolipoprotein E-deficient mice. Mediators Inflamm. 2014, 507208 (2014).
Small, D. M. & Shipley, G. G. Physical-chemical basis of lipid deposition in atherosclerosis. Science 185, 222–229 (1974).
Small, D. M. George Lyman Duff memorial lecture. Progression and regression of atherosclerotic lesions. Insights from lipid physical biochemistry. Arteriosclerosis 8, 103–129 (1988).
Kellner-Weibel, G. et al. Crystallization of free cholesterol in model macrophage foam cells. Arterioscler Thromb. Vasc. Biol. 19, 1891–1898 (1999).
Tangirala, R. K. et al. Formation of cholesterol monohydrate crystals in macrophage-derived foam cells. J. Lipid Res. 35, 93–104 (1994).
McDonald, J. G. & Russell, D. W. Editorial: 25-Hydroxycholesterol: a new life in immunology. J. Leukoc. Biol. 88, 1071–1072 (2010).
Diczfalusy, U. et al. Marked upregulation of cholesterol 25-hydroxylase expression by lipopolysaccharide. J. Lipid Res. 50, 2258–2264 (2009).
Park, K. & Scott, A. L. Cholesterol 25-hydroxylase production by dendritic cells and macrophages is regulated by type I interferons. J. Leukoc. Biol. 88, 1081–1087 (2010).
Liu, S. Y. et al. Interferon-inducible cholesterol-25-hydroxylase broadly inhibits viral entry by production of 25-hydroxycholesterol. Immunity 38, 92–105 (2013).
Gold, E. S. et al. 25-Hydroxycholesterol acts as an amplifier of inflammatory signaling. Proc. Natl Acad. Sci. USA 111, 10666–10671 (2014).
Reboldi, A. et al. Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon. Science 345, 679–684 (2014). This study shows that the synthesis of 25-OH cholesterol that occurs during the acute phase response leads to suppression of inflammasome activity in vivo , probably as a result of the ability of 25-OH cholesterol to inhibit the synthesis of cholesterol or other sterols.
Gurcel, L., Abrami, L., Girardin, S., Tschopp, J. & van der Goot, F. G. Caspase-1 activation of lipid metabolic pathways in response to bacterial pore-forming toxins promotes cell survival. Cell 126, 1135–1145 (2006).
Xiao, H. et al. Sterol regulatory element binding protein 2 activation of NLRP3 inflammasome in endothelium mediates hemodynamic-induced atherosclerosis susceptibility. Circulation 128, 632–642 (2013). This study suggests that the NLRP3 inflammasome is activated in endothelial cells in response to disturbed blood flow.
Choi, J. H. et al. Flt3 signaling-dependent dendritic cells protect against atherosclerosis. Immunity 35, 819–831 (2011).
Randolph, G. J. Mechanisms that regulate macrophage burden in atherosclerosis. Circ. Res. 114, 1757–1771 (2014).
Li, P. et al. NCoR repression of LXRs restricts macrophage biosynthesis of insulin-sensitizing omega 3 fatty acids. Cell 155, 200–214 (2013).
Serhan, C. N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 510, 92–101 (2014).
Rong, X. et al. LXRs regulate ER stress and inflammation through dynamic modulation of membrane phospholipid composition. Cell. Metab. 18, 685–697 (2013).
Ghisletti, S. et al. Parallel SUMOylation-dependent pathways mediate gene- and signal-specific transrepression by LXRs and PPARγ. Mol. Cell 25, 57–70 (2007).
N., A. G. et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity 31, 245–258 (2009).
Gerbod-Giannone, M. C. et al. TNFα induces ABCA1 through NF-κB in macrophages and in phagocytes ingesting apoptotic cells. Proc. Natl Acad. Sci. USA 103, 3112–3117 (2006).
Murphy, A. J. et al. ApoE regulates hematopoietic stem cell proliferation, monocytosis, and monocyte accumulation in atherosclerotic lesions in mice. J. Clin. Invest. 121, 4138–4149 (2011). This study shows that APOE is expressed in HSCs, in which it seems to be bound to cell-surface proteoglycans, promoting cholesterol efflux via ABCA1 and ABCG1 and thus suppressing the proliferation of HSCs and myelopoiesis with anti-atherogenic consequences.
Coller, B. S. Leukocytosis and ischemic vascular disease morbidity and mortality: is it time to intervene? Arterioscler. Thromb. Vasc. Biol. 25, 658–670 (2005).
Swirski, F. K. & Nahrendorf, M. Leukocyte behavior in atherosclerosis, myocardial infarction, and heart failure. Science 339, 161–166 (2013).
Yvan-Charvet, L. et al. ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation. Science 328, 1689–1693 (2010). This is the first study to show that cholesterol efflux pathways suppress the proliferation of HSCs, myelopoiesis and atherogenesis.
Gao, M. et al. Regulation of high-density lipoprotein on hematopoietic stem/progenitor cells in atherosclerosis requires scavenger receptor type BI expression. Arterioscler. Thromb. Vasc. Biol. 34, 1900–1909 (2014).
Tolani, S. et al. Hypercholesterolemia and reduced HDL-C promote hematopoietic stem cell proliferation and monocytosis: studies in mice and FH children. Atherosclerosis 229, 79–85 (2013).
Feng, Y. et al. Hematopoietic stem/progenitor cell proliferation and differentiation is differentially regulated by high-density and low-density lipoproteins in mice. PLoS ONE 7, e47286 (2012).
Wang, M. et al. Interleukin-3/granulocyte macrophage colony-stimulating factor receptor promotes stem cell expansion, monocytosis, and atheroma macrophage burden in mice with hematopoietic ApoE deficiency. Arterioscler. Thromb. Vasc. Biol. 34, 976–984 (2014).
van Kampen, E., Jaminon, A., van Berkel, T. J. & Van Eck, M. Diet-induced (epigenetic) changes in bone marrow augment atherosclerosis. J. Leukoc. Biol. 96, 833–841 (2014).
Westerterp, M. et al. Regulation of hematopoietic stem and progenitor cell mobilization by cholesterol efflux pathways. Cell Stem Cell 11, 195–206 (2012). This paper shows that cholesterol efflux pathways suppress the mobilization of HSCs to the spleen and thus extramedullary haematopoiesis, which is relevant to both atherosclerosis and leukaemia.
Hong, C. et al. Coordinate regulation of neutrophil homeostasis by liver X receptors in mice. J. Clin. Invest. 122, 337–347 (2012).
Robbins, C. S. et al. Extramedullary hematopoiesis generates Ly-6Chigh monocytes that infiltrate atherosclerotic lesions. Circulation 125, 364–374 (2012).
Rauch, P. J. et al. Innate response activator B cells protect against microbial sepsis. Science 335, 597–601 (2012).
Hilgendorf, I. et al. Innate response activator B cells aggravate atherosclerosis by stimulating T helper-1 adaptive immunity. Circulation 129, 1677–1687 (2014).
Laffitte, B. A. et al. LXRs control lipid-inducible expression of the apolipoprotein E gene in macrophages and adipocytes. Proc. Natl Acad. Sci. USA 98, 507–512 (2001).
Werb, Z. & Chin, J. R. Endotoxin suppresses expression of apoprotein E by mouse macrophages in vivo and in culture. A biochemical and genetic study. J. Biol. Chem. 258, 10642–10648 (1983).
Claudel, T. et al. Reduction of atherosclerosis in apolipoprotein E knockout mice by activation of the retinoid X receptor. Proc. Natl Acad. Sci. USA 98, 2610–2615 (2001).
Levin, N. et al. Macrophage liver X receptor is required for antiatherogenic activity of LXR agonists. Arterioscler. Thromb. Vasc. Biol. 25, 135–142 (2005).
Kappus, M. S. et al. Activation of liver X receptor decreases atherosclerosis in Ldlr−/− mice in the absence of ATP-binding cassette transporters A1 and G1 in myeloid cells. Arterioscler. Thromb. Vasc. Biol. 34, 279–284 (2013).
Hong, C. & Tontonoz, P. Liver X receptors in lipid metabolism: opportunities for drug discovery. Nature Rev. Drug Discov. 13, 433–444 (2014).
Schmuth, M., Moosbrugger-Martinz, V., Blunder, S. & Dubrac, S. Role of PPAR, LXR, and PXR in epidermal homeostasis and inflammation. Biochim. Biophys. Acta 1841, 463–473 (2014).
Yi, T. et al. Oxysterol gradient generation by lymphoid stromal cells guides activated B cell movement during humoral responses. Immunity 37, 535–548 (2012).
Hannedouche, S. et al. Oxysterols direct immune cell migration via EBI2. Nature 475, 524–527 (2011).
Plump, A. S., Scott, C. J. & Breslow, J. L. Human apolipoprotein A-I gene expression increases high density lipoprotein and suppresses atherosclerosis in the apolipoprotein E-deficient mouse. Proc. Natl Acad. Sci. USA 91, 9607–9611 (1994).
Badimon, J. J., Badimon, L. & Fuster, V. Regression of atherosclerotic lesions by high density lipoprotein plasma fraction in the cholesterol-fed rabbit. J. Clin. Invest. 85, 1234–1241 (1990).
Rubin, E. M., Krauss, R. M., Spangler, E. A., Verstuyft, J. G. & Clift, S. M. Inhibition of early atherogenesis in transgenic mice by human apolipoprotein AI. Nature 353, 265–267 (1991).
Nissen, S. E. et al. Effect of recombinant ApoA-I Milano on coronary atherosclerosis in patients with acute coronary syndromes: a randomized controlled trial. JAMA 290, 2292–2300 (2003).
Tardif, J. C. et al. Effects of reconstituted high-density lipoprotein infusions on coronary atherosclerosis: a randomized controlled trial. JAMA 297, 1675–1682 (2007).
Khera, A. V. et al. Cholesterol efflux capacity, high-density lipoprotein function, and atherosclerosis. N. Engl. J. Med. 364, 127–135 (2011).
Cockerill, G. W., Rye, K. A., Gamble, J. R., Vadas, M. A. & Barter, P. J. High-density lipoproteins inhibit cytokine-induced expression of endothelial cell adhesion molecules. Arterioscler. Thromb. Vasc. Biol. 15, 1987–1994 (1995).
Yvan-Charvet, L. et al. Cholesterol efflux potential and antiinflammatory properties of high-density lipoprotein after treatment with niacin or anacetrapib. Arterioscler. Thromb. Vasc. Biol. 30, 1430–1438 (2010).
Suzuki, M. et al. High-density lipoprotein suppresses the type I interferon response, a family of potent antiviral immunoregulators, in macrophages challenged with lipopolysaccharide. Circulation 122, 1919–1927 (2010).
De Nardo, D. et al. High-density lipoprotein mediates anti-inflammatory reprogramming of macrophages via the transcriptional regulator ATF3. Nature Immunol. 15, 152–160 (2014).
Gold, E. S. et al. ATF3 protects against atherosclerosis by suppressing 25-hydroxycholesterol-induced lipid body formation. J. Exp. Med. 209, 807–817 (2012).
Tardif, J. C. et al. Effects of the high-density lipoprotein mimetic agent CER-001 on coronary atherosclerosis in patients with acute coronary syndromes: a randomized trial. Eur. Heart J. http://dx.doi.org/10.1093/eurheartj/ehu171 (2014).
Diditchenko, S. et al. Novel formulation of a reconstituted high-density lipoprotein (CSL112) dramatically enhances ABCA1-dependent cholesterol efflux. Arterioscler. Thromb. Vasc. Biol. 33, 2202–2211 (2013).
Murphy, A. J., Funt, S., Gorman, D., Tall, A. R. & Wang, N. Pegylation of high-density lipoprotein decreases plasma clearance and enhances antiatherogenic activity. Circ. Res. 113, e1–e9 (2013).
Nicholls, S. J. et al. ApoA-I induction as a potential cardioprotective strategy: rationale for the SUSTAIN and ASSURE studies. Cardiovasc. Drugs Ther. 26, 181–187 (2012).
Picaud, S. et al. RVX-208, an inhibitor of BET transcriptional regulators with selectivity for the second bromodomain. Proc. Natl Acad. Sci. USA 110, 19754–19759 (2013).
McLure, K. G. et al. RVX-208, an inducer of ApoA-I in humans, is a BET bromodomain antagonist. PLoS ONE 8, e83190 (2013).
Halley, P. et al. Regulation of the apolipoprotein gene cluster by a long noncoding RNA. Cell Rep. 6, 222–230 (2014). This paper shows that a long non-coding RNA functions at the APOA1 gene locus to suppress gene expression in rodents and primates, whereas antagonism increases plasma APOA1 and HDL levels.
Watson, C. E. et al. Treatment of patients with cardiovascular disease with L-4F, an apo-A1 mimetic, did not improve select biomarkers of HDL function. J. Lipid Res. 52, 361–373 (2011).
Bloedon, L. T. et al. Safety, pharmacokinetics, and pharmacodynamics of oral apoA-I mimetic peptide D-4F in high-risk cardiovascular patients. J. Lipid Res. 49, 1344–1352 (2008).
Moore, K. J., Sheedy, F. J. & Fisher, E. A. Macrophages in atherosclerosis: a dynamic balance. Nature Rev. Immunol. 13, 709–721 (2013).
Libby, P., Lichtman, A. H. & Hansson, G. K. Immune effector mechanisms implicated in atherosclerosis: from mice to humans. Immunity 38, 1092–1104 (2013).
Westerterp, M. et al. ATP-binding cassette transporters, atherosclerosis, and inflammation. Circ. Res. 114, 157–170 (2014).
Murphy, A. J. et al. Cholesterol efflux in megakaryocyte progenitors suppresses platelet production and thrombocytosis. Nature Med. 19, 586–594 (2013).
Swirski, F. K. et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325, 612–616 (2009).
Nahrendorf, M. et al. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J. Exp. Med. 204, 3037–3047 (2007).
Dutta, P. et al. Myocardial infarction accelerates atherosclerosis. Nature 487, 325–329 (2012).
Acknowledgements
This work was supported by the US National Institutes of Health (HL107653) and by the Leducq Foundation (to A.R.T.), and ATIP-AVENIR and ANR (to L.Y.-C).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
A.R.T. is a consultant to Amgen, Arisaph, Pfizer and CSL Behring. L.Y.-C. declares no competing financial interests.
Glossary
- Low-density lipoprotein
-
(LDL). A 20–25 nm low-density (1.016–1.063 g ml−1) lipoprotein with ~45% cholesterol, 20% phospholipids, 10% triglycerides and 25% protein (with apolipoprotein B (APOB) as the major apolipoprotein).
- High-density lipoprotein
-
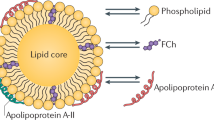
(HDL). An 8–11 nm high-density (1.063–1.210 g ml−1) lipoprotein with 40–55% protein (with apolipoprotein A1 (APOA1) as the major apolipoprotein), 25% phospholipids, 15% cholesterol and 5% triglycerides. HDL particles carry cholesterol from peripheral tissues to the liver.
- Liver X receptor
-
(LXR). LXRα and LXRβ are transcription factors that function as heterodimeric partners with retinoid X receptors (RXRs) on the promoters of many genes that are involved in cholesterol metabolism and lipogenesis. LXRs are activated by cholesterol biosynthetic intermediates, such as desmosterol, and by oxysterols derived from cholesterol. LXRs are key regulators of cellular cholesterol efflux and reverse cholesterol transport and also block the cellular uptake of low-density lipoprotein (LDL) cholesterol through the LDL receptor.
- ABC transporters
-
A family of membrane transport proteins that use the energy of ATP hydrolysis to transport various molecules, including cholesterol and other lipids, across the membrane.
- Apolipoprotein A1
-
(APOA1). The liver and the intestine secrete lipid-poor APOA1, the major protein component of high-density lipoprotein (HDL) particles. APOA1 functions as an acceptor for phospholipids and cholesterol on hepatocytes, enterocytes and macrophages. Thus, it may be involved in HDL formation as well as in the efflux of cholesterol from cells.
- Reverse cholesterol transport
-
(RCT). A multistep process that results in the net movement of cholesterol from peripheral tissues back to the liver via the blood. Cholesterol from peripheral tissues is transferred to apolipoprotein A1 (APOA1) and high-density lipoprotein (HDL) by the ATP-binding cassette transporters ABCA1 and ABCG1, respectively. The cholesteryl esters present within HDL can then be transferred, with the help of cholesteryl ester transfer protein in exchange for triglycerides, to APOB-rich lipoproteins (such as low-density lipoprotein and very low-density lipoprotein) or can be taken up in the liver by scavenger receptor B1 (SRB1). In the liver, cholesterol can be converted into bile acids for elimination.
- Acute phase response
-
The early immune response to infection, which results in the production of cytokines and other mediators, and in an increase in the number of peripheral leukocytes.
- Chylomicrons
-
50–200 nm diameter lowest density (< 1.006 g ml−1) lipoproteins that are composed of 85% triglycerides, 9% phospholipids, 4% cholesterol, and 2% protein (with apolipoprotein B48 (APOB48) as the major apolipoprotein).
- Very low-density lipoprotein
-
(VLDL). A 30–70 nm very low-density (0.95–1.006 g ml−1) lipoprotein, with ~50% triglycerides, 20% cholesterol, 20% phospholipids and 10% protein (with apolipoprotein B100 (APOB100) as the major apolipoprotein).
- MicroRNA
-
Small RNA molecules that regulate the expression of genes by binding to the 3′-untranslated regions of specific mRNAs.
- NLRP3 inflammasome
-
The NLRP3 (NOD-, LRR- and pyrin domain-containing 3) inflammasome consists of the NOD-like receptor NLRP3, caspase 1 and the adaptor protein ASC. It is activated by many signals, including microbial products, and stress- and injury-induced host factors, leading to caspase 1 activation, cleavage of pro-interleukin-1β (pro-IL-1β) and pro-IL-18, secretion of IL-1β and IL-18 and, in some cases, pyroptosis, which is a pro-inflammatory and lytic form of cell death.
- 25-hydroxycholesterol
-
(25-OH cholesterol). An oxysterol formed from cholesterol by the enzyme cholesterol 25-hydroxylase, which is present in the endoplasmic reticulum.
- Sterol regulatory element-binding protein 2
-
(SREBP2). A transcription factor that begins as a multi-transmembrane endoplasmic reticulum protein and is cleaved in the Golgi to release the basic helix–loop–helix leucine zipper transcription factor domain that binds to sterol regulatory elements in DNA.
- Sumoylation
-
The post-translational modification of proteins that involves the covalent attachment of a small ubiquitin-related modifier (SUMO) and that regulates the interactions of those proteins with other macromolecules.
- Innate response activator B cells
-
(IRA B cells). An effector B cell population and a transitional B1a-derived inflammatory subset that control IgM production and protect against microbial sepsis.
- Statins
-
A family of inhibitors of hydroxymethylglutaryl-coenzyme A reductase (HMG-CoA reductase), which is an enzyme that catalyses the conversion of HMG-CoA to L-mevalonate. These molecules are mainly used as cholesterol-lowering drugs but they also have immunoregulatory and anti-inflammatory properties. L-Mevalonate and its metabolites are implicated in cholesterol synthesis and other intracellular pathways.
Rights and permissions
About this article
Cite this article
Tall, A., Yvan-Charvet, L. Cholesterol, inflammation and innate immunity. Nat Rev Immunol 15, 104–116 (2015). https://doi.org/10.1038/nri3793
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3793
This article is cited by
-
Blood glucose and lipids are associated with sarcoidosis: findings from observational and mendelian randomization studies
Respiratory Research (2024)
-
LDLR is an entry receptor for Crimean-Congo hemorrhagic fever virus
Cell Research (2024)
-
Vitamin D is inversely associated with Monocyte to HDL-C ratio among medical staff in Chengdu, China
BMC Endocrine Disorders (2023)
-
Biomimetic nanoparticles to enhance the reverse cholesterol transport for selectively inhibiting development into foam cell in atherosclerosis
Journal of Nanobiotechnology (2023)
-
Prenylcysteine oxidase 1 like protein is required for neutrophil bactericidal activities
Nature Communications (2023)