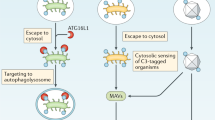
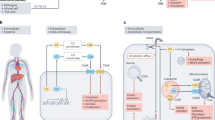
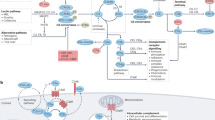
Abstract
Complement is traditionally known to be a system of serum proteins that provide protection against pathogens through direct cell lysis and the mobilization of innate and adaptive immunity. However, recent work indicates that the complement system has additional physiological roles beyond those in host defence. In this Opinion article, we describe the new modes and locations of complement activation that enable it to interact with other cell effector systems, such as growth factor receptors, inflammasomes and metabolic pathways. We propose that the location of complement activation dictates its function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Walport, M. Complement. First of two parts. N. Engl. J. Med. 344, 1058–1066 (2001).
Walport, M. Complement. Second of two parts. N. Engl. J. Med. 344, 1140–1144 (2001).
Morgan, B. P. The complement system: an overview. Methods Mol. Biol. 150, 1–13 (2000).
Ricklin, D., Hajishengallis, G., Yang, K. & Lambris, J. D. Complement: a key system for immune surveillance and homeostasis. Nature Immunol. 11, 785–797 (2010).
Sarma, J. V. & Ward, P. A. The complement system. Cell Tissue Res. 343, 227–235 (2011).
Köhl, J. The role of complement in danger sensing and transmission. Immunol. Res. 34, 157–176 (2006).
Carroll, M. The complement system in regulation of adaptive immunity. Nature Immunol. 5, 981–986 (2004).
Carroll, M. C. & Isenman, D. E. Regulation of humoral immunity by complement. Immunity 37, 199–207 (2012).
Strainic, M. G. et al. Locally produced complement fragments C5a and C3a provide both costimulatory and survival signals to naive CD4+ T cells. Immunity 28, 425–435 (2008).
Kemper, C. & Köhl, J. Novel roles for complement receptors in T cell regulation and beyond. Mol. Immunol. 56, 181–190 (2013).
Clarke, E. V. & Tenner, A. J. Complement modulation of T cell immune responses during homeostasis and disease. J. Leukoc. Biol. http://dx.doi.org/10.1189/jlb.3MR0214-109R (2014).
Mayilyan, K. R. Complement genetics, deficiencies, and disease associations. Protein Cell 3, 487–496 (2012).
Lewis, M. J. & Botto, M. Complement deficiencies in humans and animals: links to autoimmunity. Autoimmunity 39, 367–378 (2006).
Ghebrehiwet, B. & Peerschke, E. I. Role of C1q and C1q receptors in the pathogenesis of systemic lupus erythematosus. Curr. Dir. Autoimmun. 7, 87–97 (2004).
Chen, M., Daha, M. R. & Kallenberg, C. G. The complement system in systemic autoimmune disease. J. Autoimmun. 34, J276–J286 (2010).
Liszewski, M. K. et al. Intracellular complement activation sustains T cell homeostasis and mediates effector differentiation. Immunity 39, 1143–1157 (2013).
Morgan, B. & Gasque, P. Extrahepatic complement biosynthesis: where, when and why? Clin. Exp. Immunol. 107, 1–7 (1997).
Barnum, S. Complement biosynthesis in the central nervous system. Crit. Rev. Oral Biol. Med. 6, 132–146 (1995).
Naughton, M. A. et al. Extrahepatic secreted complement C3 contributes to circulating C3 levels in humans. J. Immunol. 156, 3051–3056 (1996).
Gerritsma, J. S., van Kooten, C., Gerritsen, A. F., van Es, L. A. & Daha, M. R. Transforming growth factor-β1 regulates chemokine and complement production by human proximal tubular epithelial cells. Kidney Int. 53, 609–616 (1998).
Bialas, A. R. & Stevens, B. TGF-β signaling regulates neuronal C1q expression and developmental synaptic refinement. Nature Neurosci. 16, 1773–1782 (2013).
Shavva, V. S. et al. Hepatic nuclear factor 4α positively regulates complement C3 expression and does not interfere with TNFα-mediated stimulation of C3 expression in HepG2 cells. Gene 524, 187–192 (2013).
Kolev, M., Le Friec, G. & Kemper, C. The role of complement in CD4+ T cell homeostasis and effector functions. Semin. Immunol. 25, 12–19 (2013).
Jani, P. K. et al. MASP-1 induces a unique cytokine pattern in endothelial cells: a novel link between complement system and neutrophil granulocytes. PLoS ONE 9, e87104 (2014).
Asgari, E. et al. C3a modulates IL-1β secretion in human monocytes by regulating ATP efflux and subsequent NLRP3 inflammasome activation. Blood 122, 3473–3481 (2013).
Grailer, J. J., Bosmann, M. & Ward, P. A. Regulatory effects of C5a on IL-17A, IL-17F, and IL-23. Front. Immunol. 3, 387 (2012).
Gadjeva, M. et al. Macrophage-derived complement component C4 can restore humoral immunity in C4-deficient mice. J. Immunol. 169, 5489–5495 (2002).
Pratt, J. R., Basheer, S. A. & Sacks, S. H. Local synthesis of complement component C3 regulates acute renal transplant rejection. Nature Med. 8, 582–587 (2002).
Lalli, P. N. et al. Locally produced C5a binds to T cell-expressed C5aR to enhance effector T-cell expansion by limiting antigen-induced apoptosis. Blood 112, 1759–1766 (2008).
Liu, J. et al. The complement inhibitory protein DAF (CD55) suppresses T cell immunity in vivo. J. Exp. Med. 201, 567–577 (2005).
Le Friec, G. et al. The CD46–Jagged1 interaction is critical for human TH1 immunity. Nature Immunol. 13, 1213–1221 (2012).
Ghannam, A., Fauquert, J. L., Thomas, C., Kemper, C. & Drouet, C. Human complement C3 deficiency: Th1 induction requires T cell-derived complement C3a and CD46 activation. Mol. Immunol. 58, 98–107 (2014).
Cardone, J. et al. Complement regulator CD46 temporally regulates cytokine production by conventional and unconventional T cells. Nature Immunol. 11, 862–871 (2010).
Lachmann, P. J. & Smith, R. A. Taking complement to the clinic — has the time finally come? Scand. J. Immunol. 69, 471–478 (2009).
Yamamoto, H., Fara, A. F., Dasgupta, P. & Kemper, C. CD46: the 'multitasker' of complement proteins. Int. J. Biochem. Cell Biol. 45, 2808–2820 (2013).
Pavlov, V. et al. Donor deficiency of decay-accelerating factor accelerates murine T cell-mediated cardiac allograft rejection. J. Immunol. 181, 4580–4589 (2008).
Heeger, P. et al. Decay-accelerating factor modulates induction of T cell immunity. J. Exp. Med. 201, 1523–1530 (2005).
Strainic, M. G., Shevach, E. M., An, F., Lin, F. & Medof, M. E. Absence of signaling into CD4+ cells via C3aR and C5aR enables autoinductive TGF-β1 signaling and induction of Foxp3+ regulatory T cells. Nature Immunol. 14, 162–171 (2013).
Le Friec, G., Köhl, J. & Kemper, C. A complement a day keeps the Fox(p3) away. Nature Immunol. 14, 110–112 (2013).
Kwan, W. H., van der Touw, W., Paz-Artal, E., Li, M. O. & Heeger, P. S. Signaling through C5a receptor and C3a receptor diminishes function of murine natural regulatory T cells. J. Exp. Med. 210, 257–268 (2013).
Dunkelberger, J., Zhou, L., Miwa, T. & Song, W. C. C5aR expression in a novel GFP reporter gene knockin mouse: implications for the mechanism of action of C5aR signaling in T cell immunity. J. Immunol. 188, 4032–4042 (2012).
Wetsel, R. A. Structure, function and cellular expression of complement anaphylatoxin receptors. Curr. Opin. Immunol. 7, 48–53 (1995).
Soruri, A., Kim, S., Kiafard, Z. & Zwirner, J. Characterization of C5aR expression on murine myeloid and lymphoid cells by the use of a novel monoclonal antibody. Immunol. Lett. 88, 47–52 (2003).
Zhou, W., Peng, Q., Li, K. & Sacks, S. H. Role of dendritic cell synthesis of complement in the allospecific T cell response. Mol. Immunol. 44, 57–63 (2007).
Ghannam, A. et al. Human C3 deficiency associated with impairments in dendritic cell differentiation, memory B cells, and regulatory T cells. J. Immunol. 181, 5158–5166 (2008).
Weaver, D. J. et al. C5a receptor-deficient dendritic cells promote induction of Treg and Th17 cells. Eur. J. Immunol. 40, 710–721 (2010).
Irannejad, R. et al. Conformational biosensors reveal GPCR signalling from endosomes. Nature 495, 534–538 (2013).
Baudino, L. et al. C3 opsonization regulates endocytic handling of apoptotic cells resulting in enhanced T-cell responses to cargo-derived antigens. Proc. Natl Acad. Sci. USA 111, 1503–1508 (2014).
Tam, J. C., Bidgood, S. R., McEwan, W. A. & James, L. C. Intracellular sensing of complement C3 activates cell autonomous immunity. Science 345, 1256070 (2014).
Krus, U. et al. The complement inhibitor CD59 regulates insulin secretion by modulating exocytotic events. Cell. Metab. 19, 883–890 (2014).
Krarup, A., Wallis, R., Presanis, J. S., Gál, P. & Sim, R. B. Simultaneous activation of complement and coagulation by MBL-associated serine protease 2. PLoS ONE 2, e623 (2007).
Amara, U. et al. Interaction between the coagulation and complement system. Adv. Exp. Med. Biol. 632, 71–79 (2008).
Song, W. C. Crosstalk between complement and Toll-like receptors. Toxicol. Pathol. 40, 174–182 (2012).
Liao, W., Lin, J. X. & Leonard, W. J. IL-2 family cytokines: new insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Curr. Opin. Immunol. 23, 598–604 (2011).
Gregory, S. G. et al. Interleukin 7 receptor α-chain (IL7R) shows allelic and functional association with multiple sclerosis. Nature Genet. 39, 1083–1091 (2007).
Astier, A. L., Meiffren, G., Freeman, S. & Hafler, D. A. Alterations in CD46-mediated Tr1 regulatory T cells in patients with multiple sclerosis. J. Clin. Invest. 116, 3252–3257 (2006).
Liao, W., Lin, J. X. & Leonard, W. J. Interleukin-2 at the crossroads of effector responses, tolerance, and immunotherapy. Immunity 38, 13–25 (2013).
Samstad, E. O. et al. Cholesterol crystals induce complement-dependent inflammasome activation and cytokine release. J. Immunol. 192, 2837–2845 (2014).
Laudisi, F. et al. Cutting edge: the NLRP3 inflammasome links complement-mediated inflammation and IL-1β release. J. Immunol. 191, 1006–1010 (2013).
Triantafilou, K., Hughes, T. R., Triantafilou, M. & Morgan, B. P. The complement membrane attack complex triggers intracellular Ca2+ fluxes leading to NLRP3 inflammasome activation. J. Cell Sci. 126, 2903–2913 (2013).
Doyle, S. L. et al. NLRP3 has a protective role in age-related macular degeneration through the induction of IL-18 by drusen components. Nature Med. 18, 791–798 (2012).
Benoit, M. E., Clarke, E. V., Morgado, P., Fraser, D. A. & Tenner, A. J. Complement protein C1q directs macrophage polarization and limits inflammasome activity during the uptake of apoptotic cells. J. Immunol. 188, 5682–5693 (2012).
Strowig, T., Henao-Mejia, J., Elinav, E. & Flavell, R. Inflammasomes in health and disease. Nature 481, 278–286 (2012).
Liu, H. et al. Mannan binding lectin attenuates double-stranded RNA-mediated TLR3 activation and innate immunity. FEBS Lett. 588, 866–872 (2014).
Tang, D., Kang, R., Coyne, C. B., Zeh, H. J. & Lotze, M. T. PAMPs and DAMPs: signal 0s that spur autophagy and immunity. Immunol. Rev. 249, 158–175 (2012).
Xu, L., Xiao, N., Liu, F., Ren, H. & Gu, J. Inhibition of RIG-I and MDA5-dependent antiviral response by gC1qR at mitochondria. Proc. Natl Acad. Sci. USA 106, 1530–1535 (2009).
Wang, Y., Tong, X., Zhang, J. & Ye, X. The complement C1qA enhances retinoic acid-inducible gene-I- mediated immune signalling. Immunology 136, 78–85 (2012).
Karsten, C. M. & Köhl, J. The immunoglobulin, IgG Fc receptor and complement triangle in autoimmune diseases. Immunobiology 217, 1067–1079 (2012).
Karsten, C. M. et al. Anti-inflammatory activity of IgG1 mediated by Fc galactosylation and association of FcγRIIB and dectin-1. Nature Med. 18, 1401–1406 (2012).
Carmona-Fontaine, C. et al. Complement fragment C3a controls mutual cell attraction during collective cell migration. Dev. Cell 21, 1026–1037 (2011).
Rooryck, C. et al. Mutations in lectin complement pathway genes COLEC11 and MASP1 cause 3MC syndrome. Nature Genet. 43, 197–203 (2011).
Tan, D. W. et al. Single-cell gene expression profiling reveals functional heterogeneity of undifferentiated human epidermal cells. Development 140, 1433–1444 (2013).
Hawksworth, O. A., Coulthard, L. G., Taylor, S. M., Wolvetang, E. J. & Woodruff, T. M. Brief report: complement C5a promotes human embryonic stem cell pluripotency in the absence of FGF2. Stem Cells http://dx.doi.org/10.1002/stem.1801 (2014).
Borkowska, S., Suszynska, M., Wysoczynski, M. & Ratajczak, M. Z. Mobilization studies in C3-deficient mice unravel the involvement of a novel crosstalk between the coagulation and complement cascades in mobilization of hematopoietic stem/progenitor cells. Leukemia 27, 1928–1930 (2013).
Lara-Astiaso, D. et al. Complement anaphylatoxins C3a and C5a induce a failing regenerative program in cardiac resident cells. Evidence of a role for cardiac resident stem cells other than cardiomyocyte renewal. Springerplus 1, 63 (2012).
Anaraki, P. K. et al. Urokinase receptor mediates osteogenic differentiation of mesenchymal stem cells and vascular calcification via the complement C5a receptor. Stem Cells Dev. 23, 352–362 (2014).
Matsuoka, K., Park, K. A., Ito, M., Ikeda, K. & Takeshita, S. Osteoclast-derived complement component 3a stimulates osteoblast differentiation. J. Bone Miner. Res. 29, 1522–1530 (2014).
Schafer, D. P. et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74, 691–705 (2012).
Benoit, M. E. et al. C1q-induced LRP1B and GPR6 proteins expressed early in Alzheimer disease mouse models, are essential for the C1q-mediated protection against amyloid-β neurotoxicity. J. Biol. Chem. 288, 654–665 (2013).
Stephan, A. H. et al. A dramatic increase of C1q protein in the CNS during normal aging. J. Neurosci. 33, 13460–13474 (2013).
Naito, A. T. et al. Complement C1q activates canonical Wnt signaling and promotes aging-related phenotypes. Cell 149, 1298–1313 (2012).
Cianflone, K., Rodriguez, M. A., Walsh, M., Vu, H. & Sniderman, A. D. The effect of a plasma protein fraction on lipid synthesis in cultured skin fibroblasts from normals and patients with hyperapobetalipoproteinemia. Clin. Invest. Med. 11, 99–107 (1988).
Kalant, D. et al. C5L2 is a functional receptor for acylation-stimulating protein. J. Biol. Chem. 280, 23936–23944 (2005).
Cui, W. et al. Acylation-stimulating protein/C5L2-neutralizing antibodies alter triglyceride metabolism in vitro and in vivo. Am. J. Physiol. Endocrinol. Metab. 293, E1482–E1491 (2007).
Phieler, J., Garcia-Martin, R., Lambris, J. D. & Chavakis, T. The role of the complement system in metabolic organs and metabolic diseases. Semin. Immunol. 25, 47–53 (2013).
Kim, E., Goraksha-Hicks, P., Li, L., Neufeld, T. P. & Guan, K. L. Regulation of TORC1 by Rag GTPases in nutrient response. Nature Cell Biol. 10, 935–945 (2008).
Delgoffe, G. M. et al. The kinase mTOR regulates the differentiation of helper T cells through the selective activation of signaling by mTORC1 and mTORC2. Nature Immunol. 12, 295–303 (2011).
Ramos de Carvalho, J. E. et al. Complement factor C3a alters proteasome function in human RPE cells and in an animal model of age-related RPE degeneration. Invest. Ophthalmol. Vis. Sci. 54, 6489–6501 (2013).
Berger, M., Wetzler, E. M., Welter, E., Turner, J. R. & Tartakoff, A. M. Intracellular sites for storage and recycling of C3b receptors in human neutrophils. Proc. Natl Acad. Sci. USA 88, 3019–3023 (1991).
Wirthmueller, U. et al. Properdin, a positive regulator of complement activation, is released from secondary granules of stimulated peripheral blood neutrophils. J. Immunol. 158, 4444–4451 (1997).
Martinon, F., Mayor, A. & Tschopp, J. The inflammasomes: guardians of the body. Annu. Rev. Immunol. 27, 229–265 (2009).
Berends, E. T., Kuipers, A., Ravesloot, M. M., Urbanus, R. T. & Rooijakkers, S. H. Bacteria under stress by complement and coagulation. FEMS Microbiol. Rev. http://dx.doi.org/10.1111/1574-6976.12080 (2014).
Frade, R. et al. Procathepsin-L, a proteinase that cleaves human C3 (the third component of complement), confers high tumorigenic and metastatic properties to human melanoma cells. Cancer Res. 58, 2733–2736 (1998).
Kanse, S. M. et al. Factor VII-activating protease is activated in multiple trauma patients and generates anaphylatoxin C5a. J. Immunol. 188, 2858–2865 (2012).
Huber-Lang, M. et al. Generation of C5a in the absence of C3: a new complement activation pathway. Nature Med. 12, 682–687 (2006).
Huber-Lang, M. et al. Cathepsin D is released after severe tissue trauma in vivo and is capable of generating C5a in vitro. Mol. Immunol. 50, 60–65 (2012).
Perl, M., Denk, S., Kalbitz, M. & Huber-Lang, M. Granzyme B: a new crossroad of complement and apoptosis. Adv. Exp. Med. Biol. 946, 135–146 (2012).
van den Berg, C. W. et al. Mechanism of neutrophil dysfunction: neutrophil serine proteases cleave and inactivate the C5a receptor. J. Immunol. 192, 1787–1795 (2014).
Klapper, Y. et al. Mediation of a non-proteolytic activation of complement component C3 by phospholipid vesicles. Biomaterials 35, 3688–3696 (2014).
Nilsson, B. & Nilsson Ekdahl, K. The tick-over theory revisited: is C3 a contact-activated protein? Immunobiology 217, 1106–1110 (2012).
Ekdahl, K. N. & Nilsson, B. Alterations in C3 activation and binding caused by phosphorylation by a casein kinase released from activated human platelets. J. Immunol. 162, 7426–7433 (1999).
Pidde-Queiroz, G. et al. P-I snake venom metalloproteinase is able to activate the complement system by direct cleavage of central components of the cascade. PLoS Negl. Trop. Dis. 7, e2519 (2013).
Wiggins, R. C., Giclas, P. C. & Henson, P. M. Chemotactic activity generated from the fifth component of complement by plasma kallikrein of the rabbit. J. Exp. Med. 153, 1391–1404 (1981).
Claesson, R., Kanasi, E., Johansson, A. & Kalfas, S. A new cleavage site for elastase within the complement component 3. APMIS 118, 765–768 (2010).
Markiewski, M. M. et al. The regulation of liver cell survival by complement. J. Immunol. 182, 5412–5418 (2009).
Skoberne, M. et al. The apoptotic-cell receptor CR3, but not αvβ5, is a regulator of human dendritic-cell immunostimulatory function. Blood 108, 947–955 (2006).
Wang, R., Lu, B., Gerard, C. & Gerard, N. P. Disruption of the complement anaphylatoxin receptor C5L2 exacerbates inflammation in allergic contact dermatitis. J. Immunol. 191, 4001–4009 (2013).
Strey, C. W. et al. The proinflammatory mediators C3a and C5a are essential for liver regeneration. J. Exp. Med. 198, 913–923 (2003).
Markiewski, M. M. et al. C3a and C3b activation products of the third component of complement (C3) are critical for normal liver recovery after toxic injury. J. Immunol. 173, 747–754 (2004).
Mastellos, D., Papadimitriou, J. C., Franchini, S., Tsonis, P. A. & Lambris, J. D. A novel role of complement: mice deficient in the fifth component of complement (C5) exhibit impaired liver regeneration. J. Immunol. 166, 2479–2486 (2001).
Ehrnthaller, C. et al. Complement C3 and C5 deficiency affects fracture healing. PLoS ONE 8, e81341 (2013).
Haynes, T. et al. Complement anaphylatoxin C3a is a potent inducer of embryonic chick retina regeneration. Nature Commun. 4, 2312 (2013).
Zipfel, P. & Skerka, C. Complement regulators and inhibitory proteins. Nature Rev. Immunol. 9, 729–740 (2009).
Acknowledgements
The authors acknowledge their funding sources: the Medical Research Council, The European Union and The Wellcome Trust. Their research was also funded by the National Institute for Health Research (NIHR) Biomedical Research Centre at Guy's and St Thomas' NHS Foundation Trust and King's College London, UK. The views expressed are those of the authors and not necessarily those of the National Health Service, the NIHR or the Department of Health. The authors also acknowledge and thank P. Garred (University of Copenhagen, Denmark). The authors apologize to the groups whose work they were unable to cite owing to space constraints.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Kolev, M., Friec, G. & Kemper, C. Complement — tapping into new sites and effector systems. Nat Rev Immunol 14, 811–820 (2014). https://doi.org/10.1038/nri3761
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3761
This article is cited by
-
Complement System Deficiencies in Elite Athletes
Sports Medicine - Open (2024)
-
A guide to complement biology, pathology and therapeutic opportunity
Nature Reviews Immunology (2024)
-
Complosome — the intracellular complement system
Nature Reviews Nephrology (2023)
-
Endothelial-derived complement factor D contributes to endothelial dysfunction in malignant nephrosclerosis via local complement activation
Hypertension Research (2023)
-
Senescent fibro-adipogenic progenitors are potential drivers of pathology in inclusion body myositis
Acta Neuropathologica (2023)