Key Points
-
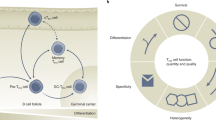
Naive CD4+ T cells have the potential to differentiate into specialized effector cell populations that have distinct functions during infection; T follicular helper (TFH) cells are the effector CD4+ T cell population responsible for mediating the activation and differentiation of B cells to generate protective humoral (antibody-based) immunity.
-
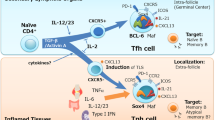
TFH cells are defined as CD4+ T cells that migrate to follicles and interact with antigen-specific B cells to support their differentiation into memory or plasma cells. They can be identified from other CD4+ T cells on the basis of their unique surface phenotype, as they express the highest levels of CXC-chemokine receptor 5 (CXCR5), together with the surface receptors inducible T cell co-stimulator (ICOS) and programmed cell death protein 1 (PD1), the transcriptional repressor B cell lymphoma 6 (BCL-6) and the cytokine interleukin-21 (IL-21).They also lack expression of CC-chemokine receptor 7 (CCR7) and IL-7 receptor-α (IL-7Rα), and are hence defined as CD4+CXCR5hiPD1hiICOShiBCl-6+IL-21+CCR7−IL-7Rα− cells.
-
TFH cell differentiation is dependent on interactions with antigen-presenting dendritic cells and B cells that are mediated through specific receptor–ligand pairs, including cytokines and their receptors. Collectively, these signals induce the expression of a range of transcription factors that induce the differentiation of the activated CD4+ T cell to a TFH cell fate. Many molecules have been found to regulate TFH cell formation, including: CD28, ICOS, CD40 ligand (CD40L) and SLAM-associated protein (SAP)-associated receptors; signal transducer and activator of transcription 3 (STAT3)-activating cytokines IL-6 and IL-21 (particularly in combination); and BCL-6, MAF, basic leucine zipper transcriptional factor ATF-like (BATF) and interferon-regulatory factor 4.
-
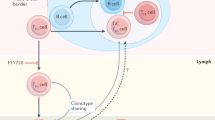
Although originally identified as cells that are important for controlling responses to conventional T cell-dependent antigens, additional subsets of TFH cells have now been characterized, such as natural killer TFH cells and γδ TFH cells, which presumably contribute to immune responses against lipid, glycolipid and phosphopeptide antigens. Moreover, a distinct subset of regulatory T (TReg) cells — follicular TReg cells — seem to co-evolve with TFH cells and to restrain TFH cell function.
-
TFH cells are associated with numerous immunopathologies that are characterized by aberrant humoral immune responses. These include primary and acquired immunodeficiencies, systemic and organ-specific autoimmune diseases and T cell malignancies. Thus, targeting the pathways that are important for TFH cell formation in an attempt to either attenuate or enhance their function represents an attractive novel therapeutic strategy to treat these conditions.
Abstract
Antibody production is an important feature of the vertebrate immune system. Antibodies neutralize and clear pathogens, thereby protecting against infectious diseases. Such humoral immunity has great longevity, often persisting for the host's lifetime. Long-lived humoral immunity depends on help provided by CD4+ T cells, namely T follicular helper (TFH) cells, which support the differentiation of antigen-specific B cells into memory and plasma cells. TFH cells are stringently regulated, as aberrant TFH cell activity is involved in immunopathologies such as autoimmunity, immunodeficiencies and lymphomas. The elucidation of the mechanisms that regulate TFH cell differentiation, function and fate should highlight targets for novel therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
O'Shea, J. J. & Paul, W. E. Mechanisms underlying lineage commitment and plasticity of helper CD4+ T cells. Science 327, 1098–1102 (2010).
Al-Herz, W. et al. Primary immunodeficiency diseases: an update on the classification from the International Union of Immunological Societies Expert Committee for Primary Immunodeficiency. Front. Immunol. 2, 54 (2011).
Miller, J. F., De Burgh, P. M. & Grant, G. A. Thymus and the production of antibody-plaque-forming cells. Nature 208, 1332–1334 (1965).
Breitfeld, D. et al. Follicular B helper T cells express CXC chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J. Exp. Med. 192, 1545–1552 (2000).
Schaerli, P. et al. CXC chemokine receptor 5 expression defines follicular homing T cells with B cell helper function. J. Exp. Med. 192, 1553–1562 (2000).
Kim, C. H. et al. Subspecialization of CXCR5+ T cells: B helper activity is focused in a germinal center — localized subset of CXCR5+ T cells. J. Exp. Med. 193, 1373–1381 (2001).
Kim, J. R., Lim, H. W., Kang, S. G., Hillsamer, P. & Kim, C. H. Human CD57+ germinal center-T cells are the major helpers for GC-B cells and induce class switch recombination. BMC Immunol. 6, 3 (2005).
Chtanova, T. et al. T follicular helper cells express a distinctive transcriptional profile, reflecting their role as non-Th1/Th2 effector cells that provide help for B cells. J. Immunol. 173, 68–78 (2004).
Rasheed, A. U., Rahn, H. P., Sallusto, F., Lipp, M. & Muller, G. Follicular B helper T cell activity is confined to CXCR5hiICOShi CD4 T cells and is independent of CD57 expression. Eur. J. Immunol. 36, 1892–1903 (2006).
Vinuesa, C. G. et al. A RING-type ubiquitin ligase family member required to repress follicular helper T cells and autoimmunity. Nature 435, 452–458 (2005). References 8–10 determine the molecular profile of human and mouse T FH cells, and identify many of the defining characteristics of these cells.
Goodnow, C. C., Vinuesa, C. G., Randall, K. L., Mackay, F. & Brink, R. Control systems and decision making for antibody production. Nature Immunol. 11, 681–688 (2010).
Victora, G. D. & Nussenzweig, M. C. Germinal centers. Annu. Rev. Immunol. 30, 429–457 (2012).
Chan, T. D. & Brink, R. Affinity-based selection and the germinal center response. Immunol. Rev. 247, 11–23 (2012).
Haynes, N. M. et al. Role of CXCR5 and CCR7 in follicular Th cell positioning and appearance of a programmed cell death gene-1high germinal center-associated subpopulation. J. Immunol. 179, 5099–5108 (2007).
Kroenke, M. A. et al. Bcl6 and maf cooperate to instruct human follicular helper CD4 T cell differentiation. J. Immunol. 188, 3734–3744 (2012).
Ma, C. S. et al. Early commitment of naive human CD4+ T cells to the T follicular helper (TFH) cell lineage is induced by IL-12. Immunol. Cell Biol. 87, 590–600 (2009). This study determines that IL-12 is important for the differentiation of human naive CD4+ T cells to T FH cells.
Deenick, E. K. et al. Follicular helper T cell differentiation requires continuous antigen presentation that is independent of unique B cell signaling. Immunity 33, 241–253 (2010).
Yusuf, I. et al. Germinal center T follicular helper cell IL-4 production is dependent on signaling lymphocytic activation molecule receptor (CD150). J. Immunol. 185, 190–202 (2010).
Bentebibel, S. E., Schmitt, N., Banchereau, J. & Ueno, H. Human tonsil B-cell lymphoma 6 (BCL6)-expressing CD4+ T-cell subset specialized for B-cell help outside germinal centers. Proc. Natl Acad. Sci. USA 108, E488–E497 (2011).
Johnston, R. J. et al. Bcl6 and Blimp-1 are reciprocal and antagonistic regulators of T follicular helper cell differentiation. Science 325, 1006–1010 (2009).
Nurieva, R. I. et al. Bcl6 mediates the development of T follicular helper cells. Science 325, 1001–1005 (2009).
Yu, D. et al. The transcriptional repressor Bcl-6 directs T follicular helper cell lineage commitment. Immunity 31, 457–468 (2009). References 20–22 identify that BCL-6 is crucial for the generation of T FH cells, and in reference 20 the authorselegantly demonstrate the opposing functions of BCL-6 and BLIMP1 in this process.
Crotty, S. Follicular helper CD4 T cells (TFH). Annu. Rev. Immunol. 29, 621–663 (2011).
Ma, C. S., Deenick, E. K., Batten, M. & Tangye, S. G. The origins, function, and regulation of T follicular helper cells. J. Exp. Med. 209, 1241–1253 (2012).
Nutt, S. L. & Tarlinton, D. M. Germinal center B and follicular helper T cells: siblings, cousins or just good friends? Nature Immunol. 12, 472–477 (2011).
Craft, J. E. Follicular helper T cells in immunity and systemic autoimmunity. Nature Rev. Rheumatol. 8, 337–347 (2012).
Hu, H. et al. Noncanonical NF-κB regulates inducible costimulator (ICOS) ligand expression and T follicular helper cell development. Proc. Natl Acad. Sci. USA 108, 12827–12832 (2011).
Ou, X., Xu, S. & Lam, K. P. Deficiency in TNFRSF13B (TACI) expands T-follicular helper and germinal center B cells via increased ICOS-ligand expression but impairs plasma cell survival. Proc. Natl Acad. Sci. USA 109, 15401–15406 (2012).
Bossaller, L. et al. ICOS deficiency is associated with a severe reduction of CXCR5+CD4 germinal center Th cells. J. Immunol. 177, 4927–4932 (2006).
Nurieva, R. I. et al. Generation of T follicular helper cells is mediated by interleukin-21 but independent of T helper 1, 2, or 17 cell lineages. Immunity 29, 138–149 (2008).
Ma, C. S., Nichols, K. E. & Tangye, S. G. Regulation of cellular and humoral immune responses by the SLAM and SAP families of molecules. Annu. Rev. Immunol. 25, 337–379 (2007).
Qi, H., Cannons, J. L., Klauschen, F., Schwartzberg, P. L. & Germain, R. N. SAP-controlled T–B cell interactions underlie germinal centre formation. Nature 455, 764–769 (2008). In this study, the use of multiphoton in vivo imaging reveals the importance of SAP in sustaining conjugate formation between T FH cells and B cells for the generation of GC and antibody responses.
Cannons, J. L. et al. Optimal germinal center responses require a multistage T cell:B cell adhesion process involving integrins, SLAM-associated protein, and CD84. Immunity 32, 253–265 (2010).
Linterman, M. A. et al. Follicular helper T cells are required for systemic autoimmunity. J. Exp. Med. 206, 561–576 (2009).
Kageyama, R. et al. The receptor Ly108 functions as a SAP adaptor-dependent on-off switch for T cell help to B cells and NKT cell development. Immunity 36, 986–1002 (2012).
Good-Jacobson, K. L. et al. PD-1 regulates germinal center B cell survival and the formation and affinity of long-lived plasma cells. Nature Immunol. 11, 535–542 (2010).
Hamel, K. M. et al. B7-H1 expression on non-B and non-T cells promotes distinct effects on T- and B-cell responses in autoimmune arthritis. Eur. J. Immunol. 40, 3117–3127 (2010).
Hams, E. et al. Blockade of B7-H1 (programmed death ligand 1) enhances humoral immunity by positively regulating the generation of T follicular helper cells. J. Immunol. 186, 5648–5655 (2011).
Butler, N. S. et al. Therapeutic blockade of PD-L1 and LAG-3 rapidly clears established blood-stage Plasmodium infection. Nature Immunol. 13, 188–195 (2012).
Cubas, R. A. et al. Inadequate T follicular cell help impairs B cell immunity during HIV infection. Nature Med. 19, 494–499 (2013).
Sage, P. T., Francisco, L. M., Carman, C. V. & Sharpe, A. H. The receptor PD-1 controls follicular regulatory T cells in the lymph nodes and blood. Nature Immunol. 14, 152–161 (2012).
Eto, D. et al. IL-21 and IL-6 are critical for different aspects of B cell immunity and redundantly induce optimal follicular helper CD4 T cell (Tfh) differentiation. PLoS ONE 6, e17739 (2011).
Harker, J. A., Lewis, G. M., Mack, L. & Zuniga, E. I. Late interleukin-6 escalates T follicular helper cell responses and controls a chronic viral infection. Science 334, 825–829 (2011).
Karnowski, A. et al. B and T cells collaborate in antiviral responses via IL-6, IL-21, and transcriptional activator and coactivator, Oct2 and OBF-1. J. Exp. Med. 209, 2049–2064 (2012).
Choi, Y. S., Eto, D., Yang, J. A., Lao, C. & Crotty, S. Cutting edge: STAT1 is required for IL-6-mediated Bcl6 induction for early follicular helper cell differentiation. J. Immunol. 190, 3049–3053 (2013).
Ma, C. S. et al. Functional STAT3 deficiency compromises the generation of human T follicular helper cells. Blood 119, 3997–4008 (2012). This study establishes the importance of STAT3 and its related cytokines in generating human T FH cells.
Kim, S. J., Zou, Y. R., Goldstein, J., Reizis, B. & Diamond, B. Tolerogenic function of Blimp-1 in dendritic cells. J. Exp. Med. 208, 2193–2199 (2011).
Cai, G. et al. A regulatory role for IL-10 receptor signaling in development and B cell help of T follicular helper cells in mice. J. Immunol. 189, 1294–1302 (2012).
Johnston, R. J., Choi, Y. S., Diamond, J. A., Yang, J. A. & Crotty, S. STAT5 is a potent negative regulator of TFH cell differentiation. J. Exp. Med. 209, 243–250 (2012).
Pepper, M., Pagan, A. J., Igyarto, B. Z., Taylor, J. J. & Jenkins, M. K. Opposing signals from the Bcl6 transcription factor and the interleukin-2 receptor generate T helper 1 central and effector memory cells. Immunity 35, 583–595 (2011).
Hale, J. S. et al. Distinct memory CD4+ T cells with commitment to T follicular helper- and T helper 1-cell lineages are generated after acute viral infection. Immunity 38, 805–817 (2013).
Choi, Y. S. et al. Bcl6 expressing follicular helper CD4 T cells are fate committed early and have the capacity to form memory. J. Immunol. 190, 4014–4026 (2013).
Liu, X. et al. Bcl6 expression specifies the T follicular helper cell program in vivo. J. Exp. Med. 209, 1841–1852 (2012).
Luthje, K. et al. The development and fate of follicular helper T cells defined by an IL-21 reporter mouse. Nature Immunol. 13, 491–498 (2012).
Weber, J. P., Fuhrmann, F. & Hutloff, A. T-follicular helper cells survive as long-term memory cells. Eur. J. Immunol. 42, 1981–1988 (2012). References 50–55 examine the 'memory' capability of T FH cells in murine models of disease. T FH cells that are generated in response to protein antigen or pathogen infection can contribute to the long-term pool of memory CD4+ T cells; however, these cells do not necessarily retain a T FH cell phenotype, which demonstrates the plasticity that exists in this T cell subsetin terms of their capacity to differentiate into alternative effector subsets.
Morita, R. et al. Human blood CXCR5+CD4+ T cells are counterparts of T follicular cells and contain specific subsets that differentially support antibody secretion. Immunity 34, 108–121 (2011). This study is a comprehensive analysis of circulating CD4+CXCR5+ T cells in humans, establishing that they exhibit hallmarks of tissue T FH cells (they secrete IL-21, IL-10 and CXCL13, express ICOS and provide B cell help) and that they can be further divided into T H 1-, T H 2- and T H 17-type T FH cells. Importantly, the T H 2 and T H 17 subset, but not the T H 1 subset, are over-represented in human autoimmunity.
Bentebibel, S. E. et al. Induction of ICOS+CXCR3+CXCR5+ TH cells correlates with antibody responses to influenza vaccination. Sci. Transl. Med. 5, 176ra132 (2013).
Galli, G. et al. Invariant NKT cells sustain specific B cell responses and memory. Proc. Natl Acad. Sci. USA 104, 3984–3989 (2007).
Barral, P. et al. B cell receptor-mediated uptake of CD1d-restricted antigen augments antibody responses by recruiting invariant NKT cell help in vivo. Proc. Natl Acad. Sci. USA 105, 8345–8350 (2008).
Leadbetter, E. A. et al. NK T cells provide lipid antigen-specific cognate help for B cells. Proc. Natl Acad. Sci. USA 105, 8339–8344 (2008).
Tonti, E. et al. NKT-cell help to B lymphocytes can occur independently of cognate interaction. Blood 113, 370–376 (2009).
Chang, P. P. et al. Identification of Bcl-6-dependent follicular helper NKT cells that provide cognate help for B cell responses. Nature Immunol. 13, 35–43 (2012).
Tonti, E. et al. Follicular helper NKT cells induce limited B cell responses and germinal center formation in the absence of CD4+ T cell help. J. Immunol. 188, 3217–3222 (2012).
King, I. L. et al. Invariant natural killer T cells direct B cell responses to cognate lipid antigen in an IL-21- dependent manner. Nature Immunol. 13, 44–50 (2012). References 62–64 characterize NKT FH cells and their contribution to responses to αGalCer antigen.
Vantourout, P. & Hayday, A. Six-of-the-best: unique contributions of γδ T cells to immunology. Nature Rev. Immunol. 13, 88–100 (2013).
Brandes, M. et al. Flexible migration program regulates γδ T-cell involvement in humoral immunity. Blood 102, 3693–3701 (2003).
Caccamo, N. et al. CXCR5 identifies a subset of Vγ9Vδ2 T cells which secrete IL-4 and IL-10 and help B cells for antibody production. J. Immunol. 177, 5290–5295 (2006).
Caccamo, N. et al. IL-21 regulates the differentiation of a human γδ T cell subset equipped with B cell helper activity. PLoS ONE 7, e41940 (2012).
Vermijlen, D. et al. Distinct cytokine-driven responses of activated blood γδ T cells: insights into unconventional T cell pleiotropy. J. Immunol. 178, 4304–4314 (2007).
Chan, T. D. et al. Elimination of germinal-center-derived self-reactive B cells is governed by the location and concentration of self-antigen. Immunity 37, 893–904 (2012).
Lim, H. W., Hillsamer, P. & Kim, C. H. Regulatory T cells can migrate to follicles upon T cell activation and suppress GC-Th cells and GC-Th cell-driven B cell responses. J. Clin. Invest. 114, 1640–1649 (2004).
Chung, Y. et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nature Med. 17, 983–988 (2011).
Linterman, M. A. et al. Foxp3+ follicular regulatory T cells control the germinal center response. Nature Med. 17, 975–982 (2011).
Wollenberg, I. et al. Regulation of the germinal center reaction by Foxp3+ follicular regulatory T cells. J. Immunol. 187, 4553–4560 (2011). References 72–74 characterize follicular T Reg cells in human and mouse lymphoid tissues.
Josefowicz, S. Z., Lu, L. F. & Rudensky, A. Y. Regulatory T cells: mechanisms of differentiation and function. Annu. Rev. Immunol. 30, 531–564 (2012).
Wu, H. Y., Quintana, F. J. & Weiner, H. L. Nasal anti-CD3 antibody ameliorates lupus by inducing an IL-10-secreting CD4+ CD25− LAP+ regulatory T cell and is associated with down-regulation of IL-17+ CD4+ ICOS+ CXCR5+ follicular helper T cells. J. Immunol. 181, 6038–6050 (2008).
Simpson, N. et al. Expansion of circulating T cells resembling follicular helper T cells is a fixed phenotype that identifies a subset of severe systemic lupus erythematosus. Arthritis Rheum. 62, 234–244 (2010). This study demonstrates that numbers of circulating CXCR5+CD4+ T cells in human blood are increased in the autoimmune diseases SLE and Sjögren's syndrome.
Chevalier, N. et al. CXCR5 expressing human central memory CD4 T cells and their relevance for humoral immune responses. J. Immunol. 186, 5556–5568 (2011).
Kitano, M. et al. Bcl6 protein expression shapes pre-germinal center B cell dynamics and follicular helper T cell heterogeneity. Immunity 34, 961–972 (2011).
Pallikkuth, S. et al. Upregulation of IL-21 receptor on B cells and IL-21 secretion distinguishes novel 2009 H1N1 vaccine responders from nonresponders among HIV-infected persons on combination antiretroviral therapy. J. Immunol. 186, 6173–6181 (2011).
Pallikkuth, S. et al. Impaired peripheral blood T-follicular helper cell function in HIV-infected nonresponders to the 2009 H1N1/09 vaccine. Blood 120, 985–993 (2012). References 57, 80 and 81 establish the benefits of analysing circulating CD4+CXCR5+ T cells as a biomarker of successful vaccination.
Banchereau, J. & Rousset, F. Human B lymphocytes: phenotype, proliferation, and differentiation. Adv. Immunol. 52, 125–262 (1992).
Bryant, V. L. et al. Cytokine-mediated regulation of human B cell differentiation into Ig-secreting cells: predominant role of IL-21 produced by CXCR5+ T follicular helper cells. J. Immunol. 179, 8180–8190 (2007).
Salomonsson, S. et al. Expression of the B cell-attracting chemokine CXCL13 in the target organ and autoantibody production in ectopic lymphoid tissue in the chronic inflammatory disease Sjögren's syndrome. Scand. J. Immunol. 55, 336–342 (2002).
Hutloff, A. et al. Involvement of inducible costimulator in the exaggerated memory B cell and plasma cell generation in systemic lupus erythematosus. Arthritis Rheum. 50, 3211–3220 (2004).
Saito, R. et al. Altered expression of chemokine receptor CXCR5 on T cells of myasthenia gravis patients. J. Neuroimmunol. 170, 172–178 (2005).
Tackenberg, B. et al. Clonal expansions of CD4+ B helper T cells in autoimmune myasthenia gravis. Eur. J. Immunol. 37, 849–863 (2007).
Feng, X. et al. Inhibition of aberrant circulating Tfh cell proportions by corticosteroids in patients with systemic lupus erythematosus. PLoS ONE 7, e51982 (2012).
Terrier, B. et al. Interleukin 21 correlates with T cell and B cell subset alterations in systemic lupus erythematosus. J. Rheumatol 39, 1819–1828 (2012).
Ma, J. et al. Increased frequency of circulating follicular helper T cells in patients with rheumatoid arthritis. Clin. Dev. Immunol. 2012, 827–480 (2012).
Zhu, C. et al. Increased frequency of follicular helper T cells in patients with autoimmune thyroid disease. J. Clin. Endocrinol. Metab. 97, 943–950 (2012).
Liu, R. et al. A regulatory effect of IL-21 on T follicular helper-like cell and B cell in rheumatoid arthritis. Arthritis Res. Ther. 14, R255 (2012).
Li, X. Y. et al. Role of the frequency of blood CD4+ CXCR5+ CCR6+ T cells in autoimmunity in patients with Sjögren's syndrome. Biochem. Biophys. Res. Commun. 422, 238–244 (2012).
Wong, C. K. et al. Elevated production of B cell chemokine CXCL13 is correlated with systemic lupus erythematosus disease activity. J. Clin. Immunol. 30, 45–52 (2010).
Chakera, A. et al. The phenotype of circulating follicular-helper T cells in patients with rheumatoid arthritis defines CD200 as a potential therapeutic target. Clin. Dev. Immunol. 2012, 948–218 (2012).
Ma, C. S. et al. Impaired humoral immunity in X-linked lymphoproliferative disease is associated with defective IL-10 production by CD4+ T cells. J. Clin. Invest. 115, 1049–1059 (2005). This study determines that compromised humoral immunity in XLP is the result of a defect in CD4+ T cell-mediated B cell help; this was the first study to implicate a T FH cell defect as a cause of impaired antibody responses in primary immunodeficiencies.
Schmitt, N. et al. IL-12 receptor β1 deficiency alters in vivo T follicular helper cell response in humans. Blood 121, 3375–3385 (2013).
Moir, S. & Fauci, A. S. B cells in HIV infection and disease. Nature Rev. Immunol. 9, 235–245 (2009).
Lindqvist, M. et al. Expansion of HIV-specific T follicular helper cells in chronic HIV infection. J. Clin. Invest. 122, 3271–3280 (2012).
Petrovas, C. et al. CD4 T follicular helper cell dynamics during SIV infection. J. Clin. Invest. 122, 3281–3294 (2012).
Perreau, M. et al. Follicular helper T cells serve as the major CD4 T cell compartment for HIV-1 infection, replication, and production. J. Exp. Med. 210, 143–156 (2012).
Xu, Y. et al. Simian immunodeficiency virus infects follicular helper CD4 T cells in lymphoid tissues during pathogenic infection of pigtail macaques. J. Virol. 87, 3760–3773 (2013).
de Leval, L., Gisselbrecht, C. & Gaulard, P. Advances in the understanding and management of angioimmunoblastic T-cell lymphoma. Br. J. Haematol. 148, 673–689 (2010).
Hu, S., Young, K. H., Konoplev, S. N. & Medeiros, L. J. Follicular T-cell lymphoma: a member of an emerging family of follicular helper T-cell derived T-cell lymphomas. Hum. Pathol. 43, 1789–1798 (2012).
Krenacs, L., Schaerli, P., Kis, G. & Bagdi, E. Phenotype of neoplastic cells in angioimmunoblastic T-cell lymphoma is consistent with activated follicular B helper T cells. Blood 108, 1110–1111 (2006).
Roncador, G. et al. Expression of two markers of germinal center T cells (SAP and PD-1) in angioimmunoblastic T-cell lymphoma. Haematologica 92, 1059–1066 (2007).
Marafioti, T. et al. The inducible T-cell co-stimulator molecule is expressed on subsets of T cells and is a new marker of lymphomas of T follicular helper cell-derivation. Haematologica 95, 432–439 (2010).
de Leval, L. et al. The gene expression profile of nodal peripheral T-cell lymphoma demonstrates a molecular link between angioimmunoblastic T-cell lymphoma (AITL) and follicular helper T (TFH) cells. Blood 109, 4952–4963 (2007). This study uses comprehensive gene array analysis to show that T FH cells are the malignant cell type in AITL.
Iqbal, J. et al. Molecular signatures to improve diagnosis in peripheral T-cell lymphoma and prognostication in angioimmunoblastic T-cell lymphoma. Blood 115, 1026–1036 (2010).
Rodriguez-Pinilla, S. M. et al. Peripheral T-cell lymphoma with follicular T-cell markers. Am. J. Surg. Pathol. 32, 1787–1799 (2008).
Miyoshi, H. et al. Clinicopathologic analysis of peripheral T-cell lymphoma, follicular variant, and comparison with angioimmunoblastic T-cell lymphoma: Bcl-6 expression might affect progression between these disorders. Am. J. Clin. Pathol. 137, 879–889 (2012).
Quivoron, C. et al. TET2 inactivation results in pleiotropic hematopoietic abnormalities in mouse and is a recurrent event during human lymphomagenesis. Cancer Cell 20, 25–38 (2011).
Cairns, R. A. et al. IDH2 mutations are frequent in angioimmunoblastic T-cell lymphoma. Blood 119, 1901–1903 (2012).
Lemonnier, F. et al. Recurrent TET2 mutations in peripheral T-cell lymphomas correlate with TFH-like features and adverse clinical parameters. Blood 120, 1466–1469 (2012).
Ellyard, J. I. et al. Heterozygosity for Roquinsan leads to angioimmunoblastic T-cell lymphoma-like tumors in mice. Blood 120, 812–821 (2012).
Parrish-Novak, J. et al. Interleukin 21 and its receptor are involved in NK cell expansion and regulation of lymphocyte function. Nature 408, 57–63 (2000).
Ettinger, R. et al. IL-21 induces differentiation of human naive and memory B cells into antibody-secreting plasma cells. J. Immunol. 175, 7867–7879 (2005).
Avery, D. T. et al. B cell-intrinsic signaling through IL-21 receptor and STAT3 is required for establishing long-lived antibody responses in humans. J. Exp. Med. 207, 155–171 (2010).
Pene, J. et al. Cutting edge: IL-21 is a switch factor for the production of IgG1 and IgG3 by human B cells. J. Immunol. 172, 5154–5157 (2004).
Avery, D. T., Bryant, V. L., Ma, C. S., de Waal Malefyt, R. & Tangye, S. G. IL-21-induced isotype switching to IgG and IgA by human naive B cells is differentially regulated by IL-4. J. Immunol. 181, 1767–1779 (2008).
Steinman, L. A brief history of TH17, the first major revision in the TH1/TH2 hypothesis of T cell-mediated tissue damage. Nature Med. 13, 139–145 (2007).
Annunziato, F., Cosmi, L., Liotta, F., Maggi, E. & Romagnani, S. Type 17 T helper cells — origins, features and possible roles in rheumatic disease. Nature Rev. Rheumatol. 5, 325–331 (2009).
Afshar, R., Medoff, B. D. & Luster, A. D. Allergic asthma: a tale of many T cells. Clin. Exp. Allergy 38, 1847–1857 (2008).
Korn, T., Bettelli, E., Oukka, M. & Kuchroo, V. K. IL-17 and Th17 Cells. Annu. Rev. Immunol. 27, 485–517 (2009).
Jabeen, R. & Kaplan, M. H. The symphony of the ninth: the development and function of Th9 cells. Curr. Opin. Immunol. 24, 303–307 (2012).
Witte, E., Witte, K., Warszawska, K., Sabat, R. & Wolk, K. Interleukin-22: a cytokine produced by T, NK and NKT cell subsets, with importance in the innate immune defense and tissue protection. Cytokine Growth Factor Rev. 21, 365–379 (2010).
Acknowledgements
Studies in the authors' laboratories into T follicular helper cells are supported by grants and fellowships that have been awarded by the National Health and Medical Research Council of Australia. The authors thank S. Crotty and H. Ueno for providing papers before publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
FURTHER INFORMATION
Supplementary information
Supplementary information S1 (table)
Effects of gene deficiency on TFH cell formation (PDF 280 kb)
Glossary
- Germinal centres
-
The structures that are formed by the expansion of antigen-activated B cell blasts that have migrated into the follicles of lymph nodes. The B cells in these structures proliferate and the immunoglobulin genes undergo somatic hypermutation before the cells leave as plasma cells or memory cells.
- B cell lymphoma 6
-
(BCL-6). A transcriptional repressor identified as being crucial for the formation of T follicular helper (TFH) cells. Several mechanisms have been proposed for the role of BCL-6 in TFH cell commitment, including suppression of the expression of transcription factors that are required for the generation of alternative TH fates, suppression of microRNAs and cooperation with other transcriptional regulators to induce the expression of important TFH cell-related genes.
- X-linked lymphoproliferative disease
-
A rare, often fatal, primary immunodeficiency disease that is characterized by an inability to mount an effective immune response against Epstein–Barr virus, as well as a susceptibility to developing lymphoma and/or hypogammaglobulinaemia.
- SH2 domain-containing protein tyrosine phosphatase 1
-
(SHP1). A protein tyrosine phosphatase that is involved in suppressing intracellular signals delivered via numerous activating receptors, including T cell and B cell antigen receptors, as well as members of the signalling lymphocytic activation molecule (SLAM) family of surface receptors. One proposed mechanism of action is the direct or indirect dephosphorylation of components of the T cell receptor signalling pathway, such as CD3ζ, LCK, ζ-chain-associated protein kinase of 70 kDa (ZAP70) and phosphoinositide 3-kinase.
- Follicular T regulatory cells
-
A subset of T regulatory (TReg) cells that co-opts the transcriptional machinery of T follicular helper (TFH) cells to facilitate their migration to germinal centres, where they can appropriately restrain humoral immune responses, thereby potentially preventing overzealous antibody responses. Follicular TReg cells can be identified by the expression of typical TFH cell surface markers (CXC-chemokine receptor 5 (CXCR5), inducible T cell co-stimulator (ICOS) and programmed cell death protein 1 (PD1)) along with the TReg transcription factor forkhead box P3. Their mechanism of action remains to be completely elucidated.
- Follicular dendritic cells
-
Specialized non-haematopoietic stromal cells that reside in lymphoid follicles and germinal centres. These cells possess long dendrites and carry intact antigen on their surface. They are crucial for the optimal selection of B cells that produce antigen-binding antibodies.
- γδ T cells
-
T cells that express the γδ T cell receptor. These T cells are present in the skin, vagina and intestinal epithelium as intraepithelial lymphocytes. Although the exact function of γδ T cells is unknown, it has been suggested that mucosal γδ T cells are involved in innate immune responses.
- Sjögren's syndrome
-
A systemic autoimmune disease in which autoantibodies target and destroy exocrine glands such as the tear ducts and the salivary glands.
- Systemic lupus erythematosus
-
(SLE). An autoimmune disease in which autoantibodies that are specific for DNA, RNA or proteins associated with nucleic acids form immune complexes. These complexes damage small blood vessels, especially in the kidneys. Patients with SLE generally have abnormal B and T cell function.
- Myasthenia gravis
-
A chronic autoimmune disease that involves the generation of T cell-dependent autoantibodies that are specific for the acetylcholine receptor. These antibodies interfere with the transmission of signals at neuromuscular junctions.
- Rheumatoid arthritis
-
An immunological disorder that is characterized by symmetrical polyarthritis, often progressing to crippling deformation after years of synovitis. It is associated with systemic immune activation and the presence of acute-phase reactants in the peripheral blood, as well as rheumatoid factor (immunoglobulins that are specific for IgG), which forms immune complexes that are deposited in many tissues.
- Graves' disease
-
A type of autoimmune disease, and the most common form of hyperthyroidism in humans. It results from activating antibodies that are specific for the thyroid stimulating hormone receptor (TSHR). In mouse models of Graves' thyroiditis, the disease is induced by immunization with the TSHR.
- Hashimoto's thyroiditis
-
An autoimmune disease in which self-reactive B cells and T cells target the thyroid, resulting in hypothyroidism.
- Juvenile dermatomyositis
-
A chronic, multisystem autoimmune and inflammatory disease involving muscle, skin, blood vessels, the gastrointestinal tract and other organs. Autoantibodies are often detected in these patients, but their specificities have not yet been completely defined.
- Isocitrate dehydrogenase 2
-
(IDH2). An enzyme that catalyses the oxidative decarboxylation of isocitrate to 2-oxoglutarate and is a component of the tricarboxylic acid cycle. Mutations in IDH1 and IDH3 have been detected in glioma, glioblastomas and acute myeloid leukaemia.
- TET2
-
The TET2 gene encodes an oxygenase that catalyses the oxidation of 5-methylcytosine to 5-hydroxymethylcytosine to alter the epigenetic status of DNA. It is frequently mutated in human lymphomas.
- Roquin
-
A RING-type ubiquitin ligase that represses the expression of inducible T cell co-stimulator (ICOS), thereby restraining the development and function of T follicular helper cells. A mutation in the Roquin (also known as Rc3h1) gene results in lupus-like disease in mice.
Rights and permissions
About this article
Cite this article
Tangye, S., Ma, C., Brink, R. et al. The good, the bad and the ugly — TFH cells in human health and disease. Nat Rev Immunol 13, 412–426 (2013). https://doi.org/10.1038/nri3447
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3447
This article is cited by
-
Ocrelizumab effect on humoral and cellular immunity in multiple sclerosis and its clinical correlates: a 3-year observational study
Journal of Neurology (2023)
-
Niclosamide suppresses the expansion of follicular helper T cells and alleviates disease severity in two murine models of lupus via STAT3
Journal of Translational Medicine (2021)
-
The concerted change in the distribution of cell cycle phases and zone composition in germinal centers is regulated by IL-21
Nature Communications (2021)
-
METTL3-dependent m6A modification programs T follicular helper cell differentiation
Nature Communications (2021)
-
The metabolic hormone leptin promotes the function of TFH cells and supports vaccine responses
Nature Communications (2021)