Key Points
-
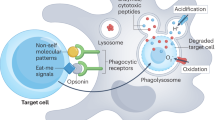
In addition to mediating the killing of microorganisms and the clearance of debris, the process of phagocytosis itself is an integral part of how macrophages and dendritic cells acquire information about a threat and elicit inflammatory responses that are appropriate to that threat.
-
Interactions of target particles with the cell surface can yield information about particle size, shape and composition. This information is interpreted by immune cells and incorporated into decisions such as whether to internalize the targets and what type of pro-inflammatory cytokines and chemokines to produce.
-
The process of forming a phagosome and pinching it off from the plasma membrane can influence the duration of signalling from membrane receptors, the concentration of such receptors into the newly formed phagosomes, and the way in which signalling molecules inside the cell interact with those receptors.
-
Signalling from within a phagosome can influence the processing and presentation of antigens derived specifically from the cargo in that phagosome.
-
The degradation of microorganisms within phagosomes releases cryptic ligands, which can subsequently be detected by receptors such as Toll-like receptor 2 (TLR2) and TLR9 to further regulate inflammatory responses.
-
Inflammasomes — which are caspase 1-containing protein complexes that regulate the release of interleukin-1β (IL-1β) and IL-18 — have been shown to be regulated by phagocytosis in various settings.
Abstract
Phagocytosis — the process by which macrophages, dendritic cells and other myeloid phagocytes internalize diverse particulate targets — is a key mechanism of innate immunity. The molecular and cellular events that underlie the binding of targets to a phagocyte and their engulfment into phagosomes have been extensively studied. More recent data suggest that the process of phagocytosis itself provides information to myeloid phagocytes about the nature of the targets they are engulfing and that this helps to tailor inflammatory responses. In this Review, we discuss how such information is acquired during phagocytosis and how it is processed to coordinate an immune response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jaumouille, V. & Grinstein, S. Receptor mobility, the cytoskeleton, and particle binding during phagocytosis. Curr. Opin. Cell Biol. 23, 22–29 (2011).
Kerrigan, A. M. & Brown, G. D. C-type lectins and phagocytosis. Immunobiology 214, 562–575 (2009).
Swanson, J. A. Shaping cups into phagosomes and macropinosomes. Nature Rev. Mol. Cell Biol. 9, 639–649 (2008).
Kawai, T. & Akira, S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 34, 637–650 (2011).
Moresco, E. M., LaVine, D. & Beutler, B. Toll-like receptors. Curr. Biol. 21, R488–R493 (2011).
Brown, G. D. & Gordon, S. Immune recognition. A new receptor for β-glucans. Nature 413, 36–37 (2001).
Indik, Z., Kelly, C., Chien, P., Levinson, A. I. & Schreiber, A. D. Human FcγRII, in the absence of other Fcγ receptors, mediates a phagocytic signal. J. Clin. Invest. 88, 1766–1771 (1991).
Henson, P. M., Bratton, D. L. & Fadok, V. A. The phosphatidylserine receptor: a crucial molecular switch? Nature Rev. Mol. Cell Biol. 2, 627–633 (2001).
Hoffmann, P. R. et al. Phosphatidylserine (PS) induces PS receptor-mediated macropinocytosis and promotes clearance of apoptotic cells. J. Cell Biol. 155, 649–659 (2001).
Toda, S., Hanayama, R. & Nagata, S. Two-step engulfment of apoptotic cells. Mol. Cell. Biol. 32, 118–125 (2012).
N'Diaye, E. N. et al. TREM-2 (triggering receptor expressed on myeloid cells 2) is a phagocytic receptor for bacteria. J. Cell Biol. 184, 215–223 (2009).
Ito, H. & Hamerman, J. A. TREM-2, triggering receptor expressed on myeloid cell-2, negatively regulates TLR responses in dendritic cells. Eur. J. Immunol. 42, 176–185 (2012).
Hamerman, J. A. et al. Cutting edge: inhibition of TLR and FcR responses in macrophages by triggering receptor expressed on myeloid cells (TREM)-2 and DAP12. J. Immunol. 177, 2051–2055 (2006).
Alpuche-Aranda, C. M., Racoosin, E. L., Swanson, J. A. & Miller, S. I. Salmonella stimulate macrophage macropinocytosis and persist within spacious phagosomes. J. Exp. Med. 179, 601–608 (1994).
Goodridge, H. S., Simmons, R. M. & Underhill, D. M. Dectin-1 stimulation by Candida albicans yeast or zymosan triggers NFAT activation in macrophages and dendritic cells. J. Immunol. 178, 3107–3115 (2007).
Tassi, I. et al. Requirement of phospholipase C-γ2 (PLCγ2) for Dectin-1-induced antigen presentation and induction of TH1/TH17 polarization. Eur. J. Immunol. 39, 1369–1378 (2009).
Xu, S., Huo, J., Lee, K. G., Kurosaki, T. & Lam, K. P. Phospholipase Cγ2 is critical for Dectin-1-mediated Ca2+ flux and cytokine production in dendritic cells. J. Biol. Chem. 284, 7038–7046 (2009).
Brown, G. D. et al. Dectin-1 mediates the biological effects of β-glucans. J. Exp. Med. 197, 1119–1124 (2003).
Gantner, B. N., Simmons, R. M., Canavera, S. J., Akira, S. & Underhill, D. M. Collaborative induction of inflammatory responses by dectin-1 and Toll-like receptor 2. J. Exp. Med. 197, 1107–1117 (2003).
Brewer, J. M., Pollock, K. G., Tetley, L. & Russell, D. G. Vesicle size influences the trafficking, processing, and presentation of antigens in lipid vesicles. J. Immunol. 173, 6143–6150 (2004).
Rettig, L. et al. Particle size and activation threshold: a new dimension of danger signaling. Blood 115, 4533–4541 (2010).
Doshi, N. & Mitragotri, S. Macrophages recognize size and shape of their targets. PLoS ONE 5, e10051 (2010).
Champion, J. A. & Mitragotri, S. Role of target geometry in phagocytosis. Proc. Natl Acad. Sci. USA 103, 4930–4934 (2006).
Rosas, M. et al. The induction of inflammation by dectin-1 in vivo is dependent on myeloid cell programming and the progression of phagocytosis. J. Immunol. 181, 3549–3557 (2008).
Beningo, K. A. & Wang, Y. L. Fc-receptor-mediated phagocytosis is regulated by mechanical properties of the target. J. Cell Sci. 115, 849–856 (2002).
Goodridge, H. S. et al. Activation of the innate immune receptor Dectin-1 upon formation of a 'phagocytic synapse'. Nature 472, 471–475 (2011). This study provides a mechanism to explain how dectin 1 is able to discriminate between polymeric soluble ligands and particulate ligands.
Dustin, M. L. & Depoil, D. New insights into the T cell synapse from single molecule techniques. Nature Rev. Immunol. 11, 672–684 (2011).
Flach, T. L. et al. Alum interaction with dendritic cell membrane lipids is essential for its adjuvanticity. Nature Med. 17, 479–487 (2011).
Ng, G. et al. Receptor-independent, direct membrane binding leads to cell-surface lipid sorting and Syk kinase activation in dendritic cells. Immunity 29, 807–818 (2008).
Ozinsky, A. et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between Toll-like receptors. Proc. Natl Acad. Sci. USA 97, 13766–13771 (2000).
Kagan, J. C. et al. TRAM couples endocytosis of Toll-like receptor 4 to the induction of interferon-β. Nature Immunol. 9, 361–368 (2008).
Husebye, H. et al. The Rab11a GTPase controls Toll-like receptor 4-induced activation of interferon regulatory factor-3 on phagosomes. Immunity 33, 583–596 (2010).
Zanoni, I. et al. CD14 controls the LPS-induced endocytosis of Toll-like receptor 4. Cell 147, 868–880 (2011). Together, references 32 and 33 report that TLR4 recruitment to phagosomes for TRIF signalling requires RAB11A but not CD14.
Torchinsky, M. B., Garaude, J. & Blander, J. M. Infection and apoptosis as a combined inflammatory trigger. Curr. Opin. Immunol. 22, 55–62 (2010).
Sauter, B. et al. Consequences of cell death: exposure to necrotic tumor cells, but not primary tissue cells or apoptotic cells, induces the maturation of immunostimulatory dendritic cells. J. Exp. Med. 191, 423–434 (2000).
Erwig, L. P. et al. Differential regulation of phagosome maturation in macrophages and dendritic cells mediated by Rho GTPases and ezrin-radixin-moesin (ERM) proteins. Proc. Natl Acad. Sci. USA 103, 12825–12830 (2006).
Blander, J. M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006).
Torchinsky, M. B., Garaude, J., Martin, A. P. & Blander, J. M. Innate immune recognition of infected apoptotic cells directs TH17 cell differentiation. Nature 458, 78–82 (2009).
West, A. P. et al. TLR signalling augments macrophage bactericidal activity through mitochondrial ROS. Nature 472, 476–480 (2011).
Huang, J. et al. Activation of antibacterial autophagy by NADPH oxidases. Proc. Natl Acad. Sci. USA 106, 6226–6231 (2009).
Sanjuan, M. A. et al. Toll-like receptor signalling in macrophages links the autophagy pathway to phagocytosis. Nature 450, 1253–1257 (2007).
Ogawa, M. et al. Autophagy targeting of Listeria monocytogenes and the bacterial countermeasure. Autophagy 7, 310–314 (2011).
Anand, P. K. et al. TLR2 and RIP2 pathways mediate autophagy of Listeria monocytogenes via extracellular signal-regulated kinase (ERK) activation. J. Biol. Chem. 286, 42981–42991 (2011).
Birmingham, C. L. et al. Listeria monocytogenes evades killing by autophagy during colonization of host cells. Autophagy 3, 442–451 (2007).
Meyer-Morse, N. et al. Listeriolysin O is necessary and sufficient to induce autophagy during Listeria monocytogenes infection. PLoS ONE 5, e8610 (2010).
Cooney, R. et al. NOD2 stimulation induces autophagy in dendritic cells influencing bacterial handling and antigen presentation. Nature Med. 16, 90–97 (2010). This study implicates the cytosolic innate immune sensor NOD2 and autophagy components in regulating the acquisition and processing of antigens derived from phagocytosed bacteria.
Travassos, L. H. et al. Nod1 and Nod2 direct autophagy by recruiting ATG16L1 to the plasma membrane at the site of bacterial entry. Nature Immunol. 11, 55–62 (2010).
Mostowy, S. et al. Entrapment of intracytosolic bacteria by septin cage-like structures. Cell Host Microbe 8, 433–444 (2010).
Ip, W. K. et al. Phagocytosis and phagosome acidification are required for pathogen processing and MyD88-dependent responses to Staphylococcus aureus. J. Immunol. 184, 7071–7081 (2010).
Ip, W. K., Takahashi, K., Moore, K. J., Stuart, L. M. & Ezekowitz, R. A. Mannose-binding lectin enhances Toll-like receptors 2 and 6 signaling from the phagosome. J. Exp. Med. 205, 169–181 (2008).
Underhill, D. M. et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 401, 811–815 (1999).
Wolf, A. J. et al. Phagosomal degradation increases TLR access to bacterial ligands and enhances macrophage sensitivity to bacteria. J. Immunol. 187, 6002–6010 (2011).
Shimada, T. et al. Staphylococcus aureus evades lysozyme-based peptidoglycan digestion that links phagocytosis, inflammasome activation, and IL-1β secretion. Cell Host Microbe 7, 38–49 (2010). Together, references 50, 52 and 53 explore the role of phagosomal degradation of S. aureus in the inflammatory response.
Rae, C. S., Geissler, A., Adamson, P. C. & Portnoy, D. A. Mutations of the Listeria monocytogenes peptidoglycan N-deacetylase and O-acetylase result in enhanced lysozyme sensitivity, bacteriolysis, and hyperinduction of innate immune pathways. Infect. Immun. 79, 3596–3606 (2011).
Hornung, V. et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nature Immunol. 9, 847–856 (2008). This was the first study to implicate the rupture of intracellular compartments as a key mechanism for triggering inflammasome activation.
Halle, A. et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-β. Nature Immunol. 9, 857–865 (2008).
Davis, M. J. & Swanson, J. A. Technical advance: caspase-1 activation and IL-1β release correlate with the degree of lysosome damage, as illustrated by a novel imaging method to quantify phagolysosome damage. J. Leukoc. Biol. 88, 813–822 (2010).
Fernandes-Alnemri, T. et al. The AIM2 inflammasome is critical for innate immunity to Francisella tularensis. Nature Immunol. 11, 385–393 (2010).
Sander, L. E. et al. Detection of prokaryotic mRNA signifies microbial viability and promotes immunity. Nature 474, 385–389 (2011). This study provides a mechanism by which macrophages can distinguish between the phagocytosis of live and dead E. coli.
Gross, O. et al. Syk kinase signalling couples to the Nlrp3 inflammasome for anti-fungal host defence. Nature 459, 433–436 (2009).
Joly, S. et al. Cutting edge: Candida albicans hyphae formation triggers activation of the Nlrp3 inflammasome. J. Immunol. 183, 3578–3581 (2009).
Sandvig, K., Pust, S., Skotland, T. & van Deurs, B. Clathrin-independent endocytosis: mechanisms and function. Curr. Opin. Cell Biol. 23, 413–420 (2011).
Kerr, M. C. & Teasdale, R. D. Defining macropinocytosis. Traffic 10, 364–371 (2009).
Overholtzer, M. & Brugge, J. S. The cell biology of cell-in-cell structures. Nature Rev. Mol. Cell Biol. 9, 796–809 (2008).
Lamkanfi, M. & Dixit, V. M. Modulation of inflammasome pathways by bacterial and viral pathogens. J. Immunol. 187, 597–602 (2011).
Ravetch, J. V. & Bolland, S. IgG Fc receptors. Annu. Rev. Immunol. 19, 275–290 (2001).
van Lookeren Campagne, M., Wiesmann, C. & Brown, E. J. Macrophage complement receptors and pathogen clearance. Cell. Microbiol. 9, 2095–2102 (2007).
Dupuy, A. G. & Caron, E. Integrin-dependent phagocytosis: spreading from microadhesion to new concepts. J. Cell Sci. 121, 1773–1783 (2008).
Blystone, S. D., Slater, S. E., Williams, M. P., Crow, M. T. & Brown, E. J. A molecular mechanism of integrin crosstalk: αvβ3 suppression of calcium/calmodulin-dependent protein kinase II regulates α5β1 function. J. Cell Biol. 145, 889–897 (1999).
Brown, G. D. Dectin-1: a signalling non-TLR pattern-recognition receptor. Nature Rev. Immunol. 6, 33–43 (2006).
Elomaa, O. et al. Cloning of a novel bacteria-binding receptor structurally related to scavenger receptors and expressed in a subset of macrophages. Cell 80, 603–609 (1995).
Bowdish, D. M. & Gordon, S. Conserved domains of the class A scavenger receptors: evolution and function. Immunol. Rev. 227, 19–31 (2009).
West, M. A. et al. Enhanced dendritic cell antigen capture via Toll-like receptor-induced actin remodeling. Science 305, 1153–1157 (2004).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Toll-like receptors
-
(TLRs). A type of pattern-recognition receptor that recognizes unique structures that are associated with infection or tissue damage. Signalling through TLRs promotes inflammatory immune responses, cytokine production and cell activation.
- Phagocyte NADPH oxidase
-
An enzyme system that consists of multiple cytosolic and membrane-bound subunits. The complex is assembled in activated myeloid cells, mainly on the phagolysosomal membrane. NADPH oxidase uses electrons from NADPH to reduce molecular oxygen to form superoxide anions. Superoxide anions are enzymatically converted to hydrogen peroxide, which is converted by myeloperoxidase to hypochlorous acid, a highly toxic and microbicidal agent.
- Pseudopodia
-
Transient cell membrane protrusions that extend around a target particle to engulf it. Pseudopodia also occur during cell migration.
- Frustrated phagocytosis
-
Occurs when a phagocyte is unable to engulf its target because the target is physically too large to encompass. This can lead to the release of potentially toxic pro-inflammatory mediators into the surrounding environment.
- Immunological synapse
-
A stable region of contact between a T cell and an antigen-presenting cell that forms through the interaction of adhesion molecules on the surface of both cells. The mature immunological synapse contains two distinct stable membrane domains: a central cluster of T cell receptors known as the central supramolecular activation cluster (cSMAC) and a surrounding ring of adhesion molecules known as the peripheral supramolecular activation cluster (pSMAC).
- Mechanotransducer
-
A system for converting mechanical energy into chemical signals.
- Pathogen-associated molecular patterns
-
(PAMPs). Molecular patterns that are found in microorganisms but not in mammalian cells. Examples include terminally mannosylated and polymannosylated compounds (which bind to the mannose receptor) and various microbial components, such as bacterial lipopolysaccharide, hypomethylated DNA, flagellin and double-stranded RNA (all of which bind to Toll-like receptors).
- Inflammasome
-
A large multiprotein complex that contains certain NOD-like receptors, RIG-I-like receptors and HIN200 proteins, together with the adaptor protein ASC and pro-caspase 1. Assembly of the inflammasome leads to the activation of caspase 1, which cleaves pro-interleukin-1β (pro-IL-1β) and pro-IL-18 to generate the active forms of these pro-inflammatory cytokines.
- Pyroptosis
-
An inflammatory form of cell death that is associated with antimicrobial responses and is dependent on inflammasome activation.
Rights and permissions
About this article
Cite this article
Underhill, D., Goodridge, H. Information processing during phagocytosis. Nat Rev Immunol 12, 492–502 (2012). https://doi.org/10.1038/nri3244
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3244
This article is cited by
-
Profiling phagosome proteins identifies PD-L1 as a fungal-binding receptor
Nature (2024)
-
Engineering nanomaterial physical characteristics for cancer immunotherapy
Nature Reviews Bioengineering (2023)
-
Significance of an altered lncRNA landscape in schizophrenia and cognition: clues from a case–control association study
European Archives of Psychiatry and Clinical Neuroscience (2023)
-
Efferocytosis requires periphagosomal Ca2+-signaling and TRPM7-mediated electrical activity
Nature Communications (2022)
-
Study of tribological properties of human buccal epithelium cell membranes using probe microscopy
Scientific Reports (2022)