Key Points
-
Regulatory T (TReg) cells are essential for maintaining peripheral tolerance, preventing autoimmunity and limiting chronic inflammatory diseases. However, they also limit beneficial responses by suppressing sterilizing immunity and limiting anti-tumour immunity.
-
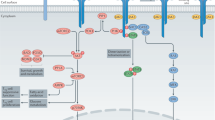
TReg cells have multiple mechanisms at their disposal to mediate their suppressive effects. These can be grouped into four basic 'modes of action': suppression by inhibitory cytokines, suppression by cytolysis, suppression by metabolic disruption and suppression by modulation of dendritic-cell (DC) maturation or function.
-
Suppression by inhibitory cytokines: interleukin-10 (IL-10), transforming growth factor-β (TGFβ) and the newly identified IL-35 are key mediators of TReg-cell function. Although they are all inhibitory, the extent to which they are used in distinct pathogenic or homeostatic settings differs, suggesting a non-overlapping function.
-
Suppression by cytolysis: both mouse and human TReg cells have been shown to mediate cytolysis via granzyme A and/or granzyme B and perforin in vitro and in vivo.
-
Suppression by metabolic disruption: a collection of intriguing mechanisms have recently been shown to either suppress or kill the target cell. Cytokine-deprivation-mediated apoptosis is mediated by the rapid consumption of IL-2 by CD25+ TReg cells, whereas the pericellular generation of adenosine and the intracellular transfer of cyclic AMP through membrane gap junctions expose the target cell to two potently inhibitory molecules.
-
Suppression by modulation of DC maturation or function: two mechanisms have been proposed. First, cytotoxic T-lymphocyte antigen 4 (CTLA4)–CD80/CD86 interactions induce the release of indoleamine 2,3-dioxygenase (IDO), a potent regulatory molecule, which induces the catabolism of tryptophan into pro-apoptotic metabolites. Second, lymphocyte-activation gene 3 (LAG3) binding to MHC class II molecules inhibits DC maturation and function.
-
Several complicating issues should be considered when evaluating the importance of these varied mechanisms. First, TReg-cell function is considered contact-dependent yet it is not clear how some mechanisms might mediate their function in this manner (for example, cytokines). Second, it is not clear for many of these mechanisms whether the primary target cell is the effector T cells and/or DCs or other antigen-presenting cells.
-
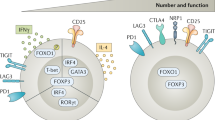
An important question is how many mechanisms do TReg cells need. There could be a single primary mechanism, multiple redundant mechanisms or multiple non-redundant mechanisms. Current data favour the latter but this remains to be fully defined and may vary depending on type of TReg cell involved and the context in which it is mediating its regulatory function.
-
We present the hypothesis that effector T cells may not be 'innocent' parties in this suppressive process and might in fact potentiate TReg-cell function.
Abstract
Regulatory T (TReg) cells are essential for maintaining peripheral tolerance, preventing autoimmune diseases and limiting chronic inflammatory diseases. However, they also limit beneficial responses by suppressing sterilizing immunity and limiting antitumour immunity. Given that TReg cells can have both beneficial and deleterious effects, there is considerable interest in determining their mechanisms of action. In this Review, we describe the basic mechanisms used by TReg cells to mediate suppression and discuss whether one or many of these mechanisms are likely to be crucial for TReg-cell function. In addition, we propose the hypothesis that effector T cells may not be 'innocent' parties in this suppressive process and might in fact potentiate TReg-cell function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Sakaguchi, S. et al. Immunologic tolerance maintained by CD25+ CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol. Rev. 182, 18–32 (2001).
Shevach, E. M. et al. The lifestyle of naturally occurring CD4+ CD25+ Foxp3+ regulatory T cells. Immunol. Rev. 212, 60–73 (2006).
Xystrakis, E., Boswell, S. E. & Hawrylowicz, C. M. T regulatory cells and the control of allergic disease. Expert. Opin. Biol. Ther. 6, 121–133 (2006).
Coombes, J. L., Robinson, N. J., Maloy, K. J., Uhlig, H. H. & Powrie, F. Regulatory T cells and intestinal homeostasis. Immunol. Rev. 204, 184–194 (2005).
Belkaid, Y. Regulatory T cells and infection: a dangerous necessity. Nature Rev. Immunol. 7, 875–888 (2007).
Rouse, B. T., Sarangi, P. P. & Suvas, S. Regulatory T cells in virus infections. Immunol. Rev. 212, 272–286 (2006).
Kretschmer, K., Apostolou, I., Jaeckel, E., Khazaie, K. & von Boehmer, H. Making regulatory T cells with defined antigen specificity: role in autoimmunity and cancer. Immunol. Rev. 212, 163–169 (2006).
Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
Fontenot, J. D., Gavin, M. A. & Rudensky, A. Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nature Immunol. 4, 330–336 (2003). References 8 and 9 provided the first direct evidence that FOXP3 is required for T Reg -cell development and is sufficient to confer regulatory activity on naive T cells.
Bennett, C. L. et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nature Genet. 27, 20–21 (2001).
Wildin, R. S. et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nature Genet. 27, 18–20 (2001).
Brunkow, M. E. et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nature Genet. 27, 68–73 (2001). References 10–12 were the first to identify FOXP3 as the defective gene in patients with IPEX and in scurfy mice.
Rudensky, A. Foxp3 and dominant tolerance. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 360, 1645–1646 (2005).
Ramsdell, F. Foxp3 and natural regulatory T cells: key to a cell lineage? Immunity 19, 165–168 (2003).
Hill, J. A. et al. Foxp3 transcription-factor-dependent and -independent regulation of the regulatory T cell transcriptional signature. Immunity 27, 786–800 (2007).
Allan, S. E. et al. Activation-induced FOXP3 in human T effector cells does not suppress proliferation or cytokine production. Int. Immunol. 19, 345–354 (2007).
Morgan, M. E. et al. Expression of FOXP3 mRNA is not confined to CD4+CD25+ T regulatory cells in humans. Hum. Immunol. 66, 13–20 (2005).
Wang, J., Ioan-Facsinay, A., van der Voort, E. I., Huizinga, T. W. & Toes, R. E. Transient expression of FOXP3 in human activated nonregulatory CD4+ T cells. Eur. J. Immunol. 37, 129–138 (2007).
Gavin, M. A. et al. Single-cell analysis of normal and FOXP3-mutant human T cells: FOXP3 expression without regulatory T cell development. Proc. Natl Acad. Sci. USA 103, 6659–6664 (2006).
Tran, D. Q., Ramsey, H. & Shevach, E. M. Induction of FOXP3 expression in naive human CD4+FOXP3 T cells by T-cell receptor stimulation is transforming growth factor-β-dependent but does not confer a regulatory phenotype. Blood 110, 2983–2990 (2007).
Vignali, D. How many mechanisms do regulatory T cells need? Eur. J. Immunol. 38, 908–911 (2008).
Tang, Q. & Bluestone, J. A. The Foxp3+ regulatory T cell: a jack of all trades, master of regulation. Nature Immunol. 9, 239–244 (2008).
Sakaguchi, S., Sakaguchi, N., Asano, M., Itoh, M. & Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor α-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 155, 1151–1164 (1995). This seminal paper re-ignited interest in 'suppressor' cells by demonstrating that a small CD4+CD25+ T-cell population had regulatory activity.
Shevach, E. M. From vanilla to 28 flavors: multiple varieties of T regulatory cells. Immunity 25, 195–201 (2006).
Takahashi, T. et al. Immunologic self-tolerance maintained by CD25+CD4+ naturally anergic and suppressive T cells: induction of autoimmune disease by breaking their anergic/suppressive state. Int. Immunol. 10, 1969–1980 (1998).
Thornton, A. M. & Shevach, E. M. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J. Exp. Med. 188, 287–296 (1998).
Dieckmann, D., Plottner, H., Berchtold, S., Berger, T. & Schuler, G. Ex vivo isolation and characterization of CD4+CD25+ T cells with regulatory properties from human blood. J. Exp. Med. 193, 1303–1310 (2001).
Jonuleit, H. et al. Identification and functional characterization of human CD4+CD25+ T cells with regulatory properties isolated from peripheral blood. J. Exp. Med. 193, 1285–1294 (2001).
Hawrylowicz, C. M. & O'Garra, A. Potential role of interleukin-10-secreting regulatory T cells in allergy and asthma. Nature Rev. Immunol. 5, 271–283 (2005).
Annacker, O., Asseman, C., Read, S. & Powrie, F. Interleukin-10 in the regulation of T cell-induced colitis. J. Autoimmun. 20, 277–279 (2003).
Joetham, A. et al. Naturally occurring lung CD4+CD25+ T cell regulation of airway allergic responses depends on IL-10 induction of TGF-β. J. Immunol. 178, 1433–1442 (2007).
Kearley, J., Barker, J. E., Robinson, D. S. & Lloyd, C. M. Resolution of airway inflammation and hyperreactivity after in vivo transfer of CD4+CD25+ regulatory T cells is interleukin 10 dependent. J. Exp. Med. 202, 1539–1547 (2005). This paper revealed the interesting distinction that IL-10 is required for the T Reg -cell-mediated control of airway hyper-reactivity but is derived from the suppressed effector T cells rather than the T Reg cells.
Rubtsov, Y. P. et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity 28, 546–558 (2008).
Stoop, J. N. et al. Tumor necrosis factor α inhibits the suppressive effect of regulatory T cells on the hepatitis B virus-specific immune response. Hepatology 46, 699–705 (2007).
Molitor-Dart, M. L. et al. Developmental exposure to noninherited maternal antigens induces CD4+ T regulatory cells: relevance to mechanism of heart allograft tolerance. J. Immunol. 179, 6749–6761 (2007).
Kursar, M. et al. Cutting Edge: regulatory T cells prevent efficient clearance of Mycobacterium tuberculosis. J. Immunol. 178, 2661–2665 (2007).
Jankovic, D. et al. Conventional T-bet+Foxp3− Th1 cells are the major source of host-protective regulatory IL-10 during intracellular protozoan infection. J. Exp. Med. 204, 273–283 (2007).
Anderson, C. F., Oukka, M., Kuchroo, V. J. & Sacks, D. CD4+CD25−Foxp3−Th1 cells are the source of IL-10-mediated immune suppression in chronic cutaneous leishmaniasis. J. Exp. Med. 204, 285–297 (2007).
Beiting, D. P. et al. Coordinated control of immunity to muscle stage Trichinella spiralis by IL-10, regulatory T cells, and TGF-β. J. Immunol. 178, 1039–1047 (2007).
Asseman, C., Mauze, S., Leach, M. W., Coffman, R. L. & Powrie, F. An essential role for interleukin 10 in the function of regulatory T cells that inhibit intestinal inflammation. J. Exp. Med. 190, 995–1004 (1999). This paper demonstrated that T Reg cells require IL-10 for their maximal regulatory activity.
Bergmann, C., Strauss, L., Zeidler, R., Lang, S. & Whiteside, T. L. Expansion and characteristics of human T regulatory type 1 cells in co-cultures simulating tumor microenvironment. Cancer Immunol. Immunother. 56, 1429–1442 (2007).
Loser, K. et al. IL-10 controls ultraviolet-induced carcinogenesis in mice. J. Immunol. 179, 365–371 (2007).
Erhardt, A., Biburger, M., Papadopoulos, T. & Tiegs, G. IL-10, regulatory T cells, and Kupffer cells mediate tolerance in concanavalin A-induced liver injury in mice. Hepatology 45, 475–485 (2007).
Ivars, F. T cell subset-specific expression of antigen receptor β chains in α chain-transgenic mice. Eur. J. Immunol. 22, 635–639 (1992).
Schumacher, A. et al. Mechanisms of action of regulatory T cells specific for paternal antigens during pregnancy. Obstet. Gynecol. 110, 1137–1145 (2007).
Mann, M. K., Maresz, K., Shriver, L. P., Tan, Y. & Dittel, B. N. B cell regulation of CD4+CD25+ T regulatory cells and IL-10 via B7 is essential for recovery from experimental autoimmune encephalomyelitis. J. Immunol. 178, 3447–3456 (2007).
Piccirillo, C. A. et al. CD4+CD25+ regulatory T cells can mediate suppressor function in the absence of transforming growth factor β1 production and responsiveness. J. Exp. Med. 196, 237–246 (2002).
Nakamura, K., Kitani, A. & Strober, W. Cell contact-dependent immunosuppression by CD4+CD25+ regulatory T cells is mediated by cell surface-bound transforming growth factor β. J. Exp. Med. 194, 629–644 (2001). This paper demonstrated that T Reg cells require cell-surface-bound TGFβ for their maximal regulatory activity.
Green, E. A., Gorelik, L., McGregor, C. M., Tran, E. H. & Flavell, R. A. CD4+CD25+ T regulatory cells control anti-islet CD8+ T cells through TGF-β–TGF-β receptor interactions in type 1 diabetes. Proc. Natl Acad. Sci. USA 100, 10878–10883 (2003).
Fahlen, L. et al. T cells that cannot respond to TGF-β escape control by CD4+CD25+ regulatory T cells. J. Exp. Med. 201, 737–746 (2005).
Li, M. O., Wan, Y. Y. & Flavell, R. A. T cell-produced transforming growth factor-b1 controls T cell tolerance and regulates Th1- and Th17-cell differentiation. Immunity 26, 579–591 (2007).
Strauss, L. et al. A unique subset of CD4+CD25highFoxp3+ T cells secreting interleukin-10 and transforming growth factor-β1 mediates suppression in the tumor microenvironment. Clin. Cancer Res. 13, 4345–4354 (2007).
Hilchey, S. P., De, A., Rimsza, L. M., Bankert, R. B. & Bernstein, S. H. Follicular lymphoma intratumoral CD4+CD25+GITR+ regulatory T cells potently suppress CD3/CD28-costimulated autologous and allogeneic CD8+. J. Immunol. 178, 4051–4061 (2007).
Li, H. et al. CD4+CD25+ regulatory T cells decreased the antitumor activity of cytokine-induced killer (CIK) cells of lung cancer patients. J. Clin. Immunol. 27, 317–326 (2007).
Clayton, A., Mitchell, J. P., Court, J., Mason, M. D. & Tabi, Z. Human tumor-derived exosomes selectively impair lymphocyte responses to interleukin-2. Cancer Res. 67, 7458–7466 (2007).
Xia, Z. W. et al. Heme oxygenase-1 attenuates ovalbumin-induced airway inflammation by up-regulation of Foxp3 T-regulatory cells, interleukin-10, and membrane-bound transforming growth factor-β1. Am. J. Pathol. 171, 1904–1914 (2007).
Ostroukhova, M. et al. Treg-mediated immunosuppression involves activation of the Notch-HES1 axis by membrane-bound TGF-β. J. Clin. Invest. 116, 996–1004 (2006).
Collison, L. W. et al. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature 450, 566–569 (2007). This paper was the first to describe the inhibitory cytokine IL-35 and its requirement for maximal T Reg -cell maximal regulatory activity.
Gavin, M. A. et al. Foxp3-dependent programme of regulatory T-cell differentiation. Nature 445, 771–775 (2007).
Lieberman, J. The ABCs of granule-mediated cytotoxicity: new weapons in the arsenal. Nature Rev. Immunol. 3, 361–370 (2003).
Grossman, W. J. et al. Differential expression of granzymes A and B in human cytotoxic lymphocyte subsets and T regulatory cells. Blood 104, 2840–2848 (2004).
McHugh, R. S. et al. CD4+CD25+ immunoregulatory T cells: gene expression analysis reveals a functional role for the glucocorticoid-induced TNF receptor. Immunity 16, 311–323 (2002).
Herman, A. E., Freeman, G. J., Mathis, D. & Benoist, C. CD4+CD25+ T regulatory cells dependent on ICOS promote regulation of effector cells in the prediabetic lesion. J. Exp. Med. 199, 1479–1489 (2004).
Gondek, D. C., Lu, L. F., Quezada, S. A., Sakaguchi, S. & Noelle, R. J. Cutting edge: contact-mediated suppression by CD4+CD25+ regulatory cells involves a granzyme B-dependent, perforin-independent mechanism. J. Immunol. 174, 1783–1786 (2005). This paper was the first to demonstrate that T Reg cells have cytolytic capacity and regulate in a granzyme-B-dependent manner. Reference 66 subsequently showed that the granzyme-dependent lytic activity of T Reg cells was required for their regulatory activity in vivo.
Zhao, D. M., Thornton, A. M., DiPaolo, R. J. & Shevach, E. M. Activated CD4+CD25+ T cells selectively kill B lymphocytes. Blood 107, 3925–3932 (2006).
Cao, X. et al. Granzyme B and perforin are important for regulatory T cell-mediated suppression of tumor clearance. Immunity 27, 635–646 (2007).
Ren, X. et al. Involvement of cellular death in TRAIL/DR5-dependent suppression induced by CD4+CD25+ regulatory T cells. Cell Death. Differ. 14, 2076–2084 (2007).
Garin, M. I. et al. Galectin-1: a key effector of regulation meditated by CD4+CD25+ T cells. Blood 109, 2058–2065 (2007).
de la Rosa, M., Rutz, S., Dorninger, H. & Scheffold, A. Interleukin-2 is essential for CD4+CD25+ regulatory T cell function. Eur. J. Immunol. 34, 2480–2488 (2004).
Fontenot, J. D., Rasmussen, J. P., Gavin, M. A. & Rudensky, A. Y. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nature Immunol. 6, 1142–1151 (2005).
Duthoit, C. T., Mekala, D. J., Alli, R. S. & Geiger, T. L. Uncoupling of IL-2 signaling from cell cycle progression in naive CD4+ T cells by regulatory CD4+CD25+ T lymphocytes. J. Immunol. 174, 155–163 (2005).
Pandiyan, P., Zheng, L., Ishihara, S., Reed, J. & Lenardo, M. J. CD4+CD25+Foxp3+ regulatory T cells induce cytokine deprivation-mediated apoptosis of effector CD4+ T cells. Nature Immunol. 8, 1353–1362 (2007).
Oberle, N., Eberhardt, N., Falk, C. S., Krammer, P. H. & Suri-Payer, E. Rapid suppression of cytokine transcription in human CD4+CD25− T cells by CD4+Foxp3+ regulatory T cells: independence of IL-2 consumption, TGF-β, and various inhibitors of TCR signaling. J. Immunol. 179, 3578–3587 (2007).
Deaglio, S. et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J. Exp. Med. 204, 1257–1265 (2007).
Borsellino, G. et al. Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood 110, 1225–1232 (2007).
Kobie, J. J. et al. T regulatory and primed uncommitted CD4 T cells express CD73, which suppresses effector CD4 T cells by converting 5′-adenosine monophosphate to adenosine. J. Immunol. 177, 6780–6786 (2006). References 74–76 collectively revealed the ability of T Reg cells to generate the inhibitory molecule adenosine by selective expression of CD39 and CD73. Reference 79 showed that another inhibitory adenosine nucleoside, cAMP, is directly transferred into effector T cells via gap junctions.
Zarek, P. E. et al. A2A receptor signaling promotes peripheral tolerance by inducing T-cell anergy and the generation of adaptive regulatory T cells. Blood 111, 251–259 (2008).
Oukka, M. Interplay between pathogenic Th17 and regulatory T cells. Ann. Rheum. Dis. 66 (Suppl 3), iii87–90 (2007).
Bopp, T. et al. Cyclic adenosine monophosphate is a key component of regulatory T cell-mediated suppression. J. Exp. Med. 204, 1303–1310 (2007).
Bluestone, J. A. & Tang, Q. How do CD4+CD25+ regulatory T cells control autoimmunity? Curr. Opin. Immunol. 17, 638–642 (2005).
Tang, Q. et al. Visualizing regulatory T cell control of autoimmune responses in nonobese diabetic mice. Nature Immunol. 7, 83–92 (2006).
Tadokoro, C. E. et al. Regulatory T cells inhibit stable contacts between CD4+ T cells and dendritic cells in vivo. J. Exp. Med. 203, 505–511 (2006). References 81 and 82 revealed the importance of T Reg -cell–DC interactions as a mechanism for blocking effector-T-cell activation.
Read, S., Malmstrom, V. & Powrie, F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+ CD4+ regulatory cells that control intestinal inflammation. J. Exp. Med. 192, 295–302 (2000). This paper demonstrated that T Reg cells require CTLA4 for their maximal regulatory activity in vivo.
Oderup, C., Cederbom, L., Makowska, A., Cilio, C. M. & Ivars, F. Cytotoxic T lymphocyte antigen-4-dependent down-modulation of costimulatory molecules on dendritic cells in CD4+ CD25+ regulatory T-cell-mediated suppression. Immunology 118, 240–249 (2006).
Serra, P. et al. CD40 ligation releases immature dendritic cells from the control of regulatory CD4+CD25+ T cells. Immunity 19, 877–889 (2003).
Fallarino, F. et al. Modulation of tryptophan catabolism by regulatory T cells. Nature Immunol. 4, 1206–1212 (2003). This paper shows that T Reg cells initiate the IDO-mediated catabolism of tryptophan in a CTLA4-dependent manner.
Mellor, A. L. & Munn, D. H. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nature Rev. Immunol. 4, 762–774 (2004).
Cederbom, L., Hall, H. & Ivars, F. CD4+CD25+ regulatory T cells down-regulate co-stimulatory molecules on antigen-presenting cells. Eur. J. Immunol. 30, 1538–1543 (2000).
Kryczek, I. et al. Cutting edge: induction of B7-H4 on APCs through IL-10: novel suppressive mode for regulatory T cells. J. Immunol. 177, 40–44 (2006).
Lewkowich, I. P. et al. CD4+CD25+ T cells protect against experimentally induced asthma and alter pulmonary dendritic cell phenotype and function. J. Exp. Med. 202, 1549–1561 (2005).
Houot, R., Perrot, I., Garcia, E., Durand, I. & Lebecque, S. Human CD4+CD25high regulatory T cells modulate myeloid but not plasmacytoid dendritic cells activation. J. Immunol. 176, 5293–5298 (2006).
Misra, N., Bayry, J., Lacroix-Desmazes, S., Kazatchkine, M. D. & Kaveri, S. V. Cutting edge: human CD4+CD25+ T cells restrain the maturation and antigen-presenting function of dendritic cells. J. Immunol. 172, 4676–4680 (2004).
Taams, L. S. et al. Modulation of monocyte/macrophage function by human CD4+CD25+ regulatory T cells. Hum. Immunol. 66, 222–230 (2005).
Tiemessen, M. M. et al. CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages. Proc. Natl Acad. Sci. USA 104, 19446–19451 (2007).
Workman, C. J. & Vignali, D. A. A. Negative regulation of T cell homeostasis by LAG-3 (CD223). J. Immunol. 174, 688–695 (2004).
Huang, C. T. et al. Role of LAG-3 in regulatory T cells. Immunity 21, 503–513 (2004).
Liang, B. et al. Regulatory T cells inhibit dendritic cells by LAG-3 engagement of MHC class II. J. Immunol. 180, 5916–5926 (2008).
Baecher-Allan, C., Wolf, E. & Hafler, D. A. MHC class II expression identifies functionally distinct human regulatory T cells. J. Immunol. 176, 4622–4631 (2006).
Sarris, M., Andersen, K. G., Randow, F., Mayr, L. & Betz, A. G. Neuropilin-1 expression on regulatory T cells enhances their interactions with dendritic cells during antigen recognition. Immunity 28, 402–413 (2008).
Lu, L. F. et al. Mast cells are essential intermediaries in regulatory T-cell tolerance. Nature 442, 997–1002 (2006).
Kaplan, D. Autocrine secretion and the physiological concentration of cytokines. Immunol. Today 17, 303–304 (1996).
Kleinewietfeld, M. et al. CCR6 expression defines regulatory effector/memory-like cells within the CD25+CD4+ T-cell subset. Blood 105, 2877–2886 (2005).
Baecher-Allan, C., Wolf, E. & Hafler, D. A. MHC class II expression identifies functionally distinct human regulatory T cells. J. Immunol. 176, 4622–4631 (2006).
Thornton, A. M., Donovan, E. E., Piccirillo, C. A. & Shevach, E. M. Cutting edge: IL-2 is critically required for the in vitro activation of CD4+CD25+ T cell suppressor function. J. Immunol. 172, 6519–6523 (2004).
Kim, J. M., Rasmussen, J. P. & Rudensky, A. Y. Regulatory T cells prevent catastrophic autoimmunity throughout the lifespan of mice. Nature Immunol. 8, 191–197 (2007).
Fontenot, J. D. et al. Regulatory T cell lineage specification by the forkhead transcription factor foxp3. Immunity 22, 329–341 (2005).
Yi, H., Zhen, Y., Jiang, L., Zheng, J. & Zhao, Y. The phenotypic characterization of naturally occurring regulatory CD4+CD25+ T cells. Cell. Mol. Immunol. 3, 189–195 (2006).
Seddiki, N. et al. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 203, 1693–1700 (2006).
Liu, W. et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ TReg cells. J. Exp. Med. 203, 1701–1711 (2006).
Yamaguchi, T. et al. Control of immune responses by antigen-specific regulatory T cells expressing the folate receptor. Immunity 27, 145–159 (2007).
Fontenot, J. D., Dooley, J. L., Farr, A. G. & Rudensky, A. Y. Developmental regulation of Foxp3 expression during ontogeny. J. Exp. Med. 202, 901–906 (2005).
Hsieh, C. S. et al. Recognition of the peripheral self by naturally arising CD25+CD4+ T cell receptors. Immunity 21, 267–277 (2004).
Izcue, A., Coombes, J. L. & Powrie, F. Regulatory T cells suppress systemic and mucosal immune activation to control intestinal inflammation. Immunol. Rev. 212, 256–271 (2006).
Roncarolo, M. G. et al. Interleukin-10-secreting type 1 regulatory T cells in rodents and humans. Immunol. Rev. 212, 28–50 (2006).
Chen, W. et al. Conversion of peripheral CD4+CD25−T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Mucida, D. et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260 (2007).
Coombes, J. L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β and retinoic acid-dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Sun, C. M. et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 204, 1775–1785 (2007).
Benson, M. J., Pino-Lagos, K., Rosemblatt, M. & Noelle, R. J. All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J. Exp. Med. 204, 1765–1774 (2007).
Schambach, F., Schupp, M., Lazar, M. A. & Reiner, S. L. Activation of retinoic acid receptor-α favours regulatory T cell induction at the expense of IL-17-secreting T helper cell differentiation. Eur. J. Immunol. 37, 2396–2399 (2007).
Kang, S. G., Lim, H. W., Andrisani, O. M., Broxmeyer, H. E. & Kim, C. H. Vitamin A metabolites induce gut-homing FoxP3+ regulatory T cells. J. Immunol. 179, 3724–3733 (2007).
Travis, M. A. et al. Loss of integrin αvβ8 on dendritic cells causes autoimmunity and colitis in mice. Nature 449, 361–365 (2007).
Matsumura, Y. et al. Selective expansion of Foxp3-positive regulatory T cells and immunosuppression by suppressors of cytokine signaling 3-deficient dendritic cells. J. Immunol. 179, 2170–2179 (2007).
Pyzik, M. & Piccirillo, C. A. TGF-β1 modulates Foxp3 expression and regulatory activity in distinct CD4+ T cell subsets. J. Leukoc. Biol. 82, 335–346 (2007).
Wei, J. et al. Antagonistic nature of T helper 1/2 developmental programs in opposing peripheral induction of Foxp3+ regulatory T cells. Proc. Natl Acad. Sci. USA 104, 18169–18174 (2007).
Grossman, W. J. et al. Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity 21, 589–601 (2004).
Bluestone, J. A. & Abbas, A. K. Natural versus adaptive regulatory T cells. Nature Rev. Immunol. 3, 253–257 (2003).
Liu, V. C. et al. Tumor evasion of the immune system by converting CD4+CD25− T cells into CD4+CD25+ T regulatory cells: role of tumor-derived TGF-β. J. Immunol. 178, 2883–2892 (2007).
Acknowledgements
We thank Randolph Noelle and Peter Ernst for granting permission to cite their unpublished observations. This work is supported by the US National Institutes of Health (NIH), the Juvenile Diabetes Research Foundation (JDRF), a Cancer Center Support CORE grant and the American Lebanese Syrian Associated Charities (ALSAC). We apologize to those authors whose work we could not cite due to space limitations.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Peripheral tolerance
-
The lack of self-responsiveness of mature lymphocytes in the periphery to specific antigens. These mechanisms control potentially self-reactive lymphocytes that have escaped central-tolerance mechanisms. Peripheral tolerance is associated with suppression of the production of self-reactive antibodies by B cells and inhibition of self-reactive effector T cells, such as cytotoxic T lymphocytes. The actions of regulatory T cells constitute one mechanism of peripheral tolerance.
- Type 1 diabetes
-
A chronic autoimmune disease that is characterized by the T-cell-mediated destruction of β-cells (which secrete insulin) in the pancreas. Individuals with type 1 diabetes develop hyperglycaemia and can develop diabetes-associated complications in multiple organ systems owing to a lack of insulin.
- Inflammatory bowel disease
-
(IBD). A T-cell-mediated inflammatory response that affects the gastrointestinal tract. There are two forms of IBD in humans; Crohn's disease, which can affect any part of the gastrointestinal tract but usually descends from the terminal ileum, and ulcerative colitis, which mainly affects the colon. In the mouse model of IBD, most of the inflammation is confined to the large intestine. The target antigen for the pathogenic T cells is unknown.
- Sterilizing immunity
-
An immune response that leads to the complete removal of the pathogen.
- Airway hyper-reactivity
-
Initiated by exposure to a defined stimulus that is usually tolerated by normal individuals and that causes broncho-constriction and airway infiltration of inflammatory cells in allergic individuals.
- Experimental autoimmune encephalomyelitis
-
(EAE). An animal model of the human autoimmune disease multiple sclerosis. EAE is experimentally induced in animals by immunization with myelin or with peptides derived from myelin. The animals develop a paralytic disease with inflammation and demyelination in the brain and spinal cord.
- Exosomes
-
Small, lipid-bilayer vesicles that are released from activated cells. They comprise either plasma membrane or membrane derived from intracellular vesicles.
- Notch
-
A transmembrane receptor involved in the pathway for direct cell–cell signalling that regulates cell-fate choice in the development of many cell lineages, and therefore is vital in the regulation of embryonic differentiation and development.
- Granzymes
-
A family of serine proteases that are found primarily in the cytoplasmic granules of cytotoxic T lymphocytes and natural killer cells. They enter target cells through perforin pores, and cleave and activate intracellular caspases, resulting in target-cell apoptosis.
- Perforin
-
A component of cytolytic granules that participates in the permeabilization of plasma membranes, allowing granzymes and other cytotoxic components to enter target cells.
- Adenosine nucleosides
-
Adenosine (C10H13N5O4) is a ribonucleoside (adenine linked to ribose) that is a structural component of nucleic acids. It is also the primary molecular component of cyclic AMP (an important intracellular second messenger), AMP, ADP and ATP (a key sourse of chemical energy for many enzymatic reactions).
- Ectoenzymes
-
Enzymes that are outside the cell membrane and therefore can cleave extracellular substrates. These are typically tethered to the outside of the cell by a transmembrane domain.
- TH17 cells
-
(T helper 17 cells). A subset of CD4+ T helper cells that produce interleukin-17 (IL-17) and that are thought to be important in inflammatory and autoimmune diseases. Their generation involves IL-6, IL-21 and IL-23, as well as the transcription factors RORγt (retinoic-acid-receptor-related orphan receptor-γt) and STAT3 (signal transducer and activator of transcription 3).
- Intravital microscopy
-
This is used for examination of biological processes, such as leukocyte–endothelial-cell interactions, in living tissue. In general, translucent tissues are used, such as the mesentery or cremaster muscle, which can be exposed and mounted for microscopic observation.
Rights and permissions
About this article
Cite this article
Vignali, D., Collison, L. & Workman, C. How regulatory T cells work. Nat Rev Immunol 8, 523–532 (2008). https://doi.org/10.1038/nri2343
Issue Date:
DOI: https://doi.org/10.1038/nri2343
This article is cited by
-
The regulation and differentiation of regulatory T cells and their dysfunction in autoimmune diseases
Nature Reviews Immunology (2024)
-
Pan-cancer analysis reveals potential immunological and prognostic roles of METTL7A in human cancers
Scientific Reports (2024)
-
Smith-specific regulatory T cells halt the progression of lupus nephritis
Nature Communications (2024)
-
Reduced numbers of regulatory T cells in chronic heart failure seems not to be restored by cardiac resynchronization therapy
BMC Cardiovascular Disorders (2023)
-
LFA-1 knockout inhibited the tumor growth and is correlated with treg cells
Cell Communication and Signaling (2023)