Key Points
-
The relationship between sleep and immunity is important because in recent decades there has been a documented decrease in the mean duration of sleep and an increase in shift work. With ever-increasing pressures to work longer hours, sleep loss and sleep disruption have become occupational hazards. As well as those who are ill with infections, chronic disease, depression or sleep apnoea, sleep disturbance is also suffered by shift workers, military personnel, new parents and carers.
-
There is good evidence, mainly from animal studies, that various infections are associated with increased sleep. This has been shown for both infection with whole microorganisms or exposure to their component virulence factors, such as lipopolysaccharide.
-
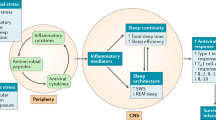
The host immune response (specifically, the production of cytokines) is responsible for the increased sleep associated with infection. Certain pro-inflammatory cytokines, in particular tumour-necrosis factor (TNF) and interleukin-1β (IL-1β), usually increase sleep, and anti-inflammatory cytokines inhibit sleep.
-
Certain cytokines are regulated endogenously during sleep. The fact that some cytokines show circadian rhythm indicates that they have a role in physiological sleep. This is supported by experiments using knockout mice, showing that both TNF- and type 1 IL-1 receptor-deficient mice have either decreased baseline sleep or do not show enhanced sleep after administration of TNF or IL-1β, respectively.
-
Sleep-deprivation experiments in both animals and humans provide the best evidence for a crucial role of sleep in the immune response. The results of these experiments show some inconsistencies, thereby underlining the complexities of measuring the interaction between different durations of sleep deprivation and different immune components. However, in humans, general patterns emerge, indicating that sleep deprivation has detrimental effects on immune-cell number, function and cytokine production.
-
There is also emerging evidence that chronic partial sleep loss might be more detrimental to immune function than short-term total sleep loss. This is important because it is chronic partial sleep loss that burdens the current population, through shift work, pressured lifestyles, and other stresses and changes in society.
-
The final piece of evidence for a reciprocal relationship between sleep and immunity is provided by clinical situations (for example, depression or narcolepsy) in which sleep disorders are associated with changes in the immune system.
-
The effects of sleep deprivation on the immune response might have important implications for protecting the population against infection and malignancy.
Abstract
It is a common belief that we are more susceptible to infections when deprived of sleep. Consistent with this, there is increasing evidence that sleep deprivation has detrimental effects on the immune response, indicating that sleep should be considered a vital part of the immune system and that there is a reciprocal relationship between sleep and immunity. This relationship is important because, over recent decades, there has been a documented decrease in the mean duration and quality of sleep in the population. The concept that lack of sleep might be compromising immunity in the population has far-reaching public-health implications for both individuals and society.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Girardin, J. L., Kripke, D. F., Ancoli-Israel, S., Klauber, M. R. & Sepulveda, R. S. Sleep duration, illumination, and activity patterns in a population sample: effects of gender and ethnicity. Biol. Psychiatry 47, 921–927 (2000).
Leger, D. The cost of sleep-related accidents: a report for the National Commission on Sleep Disorders Research. Sleep 17, 84–93 (1994).
Mandai, O., Guerrien, A., Sockeel, P., Dujardin, K. & Leconte, P. REM sleep modifications following a Morse code learning session in humans. Physiol. Behav. 46, 639–642 (1989).
Edinger, J. D., Glenn, D. M., Bastian, L. A. & Marsh, G. R. Slow-wave sleep and waking cognitive performance II: findings among middle-aged adults with and without insomnia complaints. Physiol. Behav. 70, 127–134 (2000).
Toth, L. A. & Krueger, J. M. Alteration of sleep in rabbits by Staphylococcus aureus infection. Infect. Immun. 56, 1785–1791 (1988). The first paper in a large body of work by this author, investigating the effects of infection with various microorganisms on sleep in animals.
Toth, L. A., Rehg, J. E. & Webster, R. G. Strain differences in sleep and other pathophysiological sequelae of influenza virus infection in naive and immunized mice. J. Neuroimmunol. 58, 89–99 (1995).
Toth, L. A., Gardiner, T. W. & Krueger, J. M. Modulation of sleep by cortisone in normal and bacterially infected rabbits. Am. J. Physiol. 263, R1339–R1346 (1992).
Toth, L. A. Immune-modulatory drugs alter Candida albicans-induced sleep patterns in rabbits. Pharmacol. Biochem. Behav. 51, 877–884 (1995).
Toth, L. A., Tolley, E. A., Broady, R., Blakely, B. & Krueger, J. M. Sleep during experimental trypanosomiasis in rabbits. Proc. Soc. Exp. Biol. Med. 205, 174–181 (1994).
Shoham, S., Ahokas, R. A., Blatteis, C. M. & Krueger, J. M. Effects of muramyl dipeptide on sleep, body temperature and plasma copper after intracerebral ventricular administration. Brain Res. 419, 223–228 (1987).
Lancel, M., Cronlein, J., Muller-Preuss, P. & Holsboer, F. Lipopolysaccharide increases EEG delta activity within non-REM sleep and disrupts sleep continuity in rats. Am. J. Physiol. 268, R1310–R1318 (1995).
Schiffelholz, T. & Lancel, M. Sleep changes induced by lipopolysaccharide in the rat are influenced by age. Am. J. Physiol. Regul. Integr. Comp. Physiol. 280, R398–R403 (2001).
Smith, A. in Sleep, Arousal and Performance (eds Broughton, R. J. & Ogilvie, R. D.) 233–242 (Birkhauser, Boston, 1992).
Drake, C. L. et al. Effects of an experimentally induced rhinovirus cold on sleep, performance, and daytime alertness. Physiol. Behav. 71, 75–81 (2000).
Mullington, J. et al. Dose-dependent effects of endotoxin on human sleep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 278, R947–R955 (2000). Shows that LPS has differential dose effects in humans and demonstrates the dissociation of its effects on sleep from its effects on other aspects of the acute-phase response.
Haack, M., Schuld, A., Kraus, T. & Pollmacher, T. Effects of sleep on endotoxin-induced host responses in healthy men. Psychosom. Med. 63, 568–578 (2001).
Gourmelon, P., Briet, D., Clarencon, D., Court, L. & Tsiang, H. Sleep alterations in experimental street rabies virus infection occur in the absence of major EEG abnormalities. Brain Res. 554, 159–165 (1991).
Hemachudha, T., Laothamatas, J. & Rupprecht, C. E. Human rabies: a disease of complex neuropathogenetic mechanisms and diagnostic challenges. Lancet Neurol. 1, 101–109 (2002).
Buguet, A. et al. Sleep-wake cycle in human African trypanosomiasis. J. Clin. Neurophysiol. 10, 190–196 (1993).
Buguet, A., Tapie, P. & Bert, J. Reversal of the sleep/wake cycle disorder of sleeping sickness after trypanosomicide treatment. J. Sleep Res. 8, 225–235 (1999).
White, J. L. et al. Early central nervous system response to HIV infection: sleep distortion and cognitive-motor decrements. AIDS 9, 1043–1050 (1995).
Darko, D. F., Mitler, M. M. & White, J. L. Sleep disturbance in early HIV infection. Focus 10, 5–6 (1995).
Gould, J. B., Lee, A. F., Cook, P. & Morelock, S. Apnea and sleep state in infants with nasopharyngitis. Pediatrics 65, 713–717 (1980).
Toth, L. A., Tolley, E. A. & Krueger, J. M. Sleep as a prognostic indicator during infectious disease in rabbits. Proc. Soc. Exp. Biol. Med. 203, 179–192 (1993).
Kripke, D. F., Simons, R. N., Garfinkel, L. & Hammond, E. C. Short and long sleep and sleeping pills. Is increased mortality associated? Arch. Gen. Psychiatry 36, 103–116 (1979).
Kripke, D. F., Garfinkel, L., Wingard, D. L., Klauber, M. R. & Marler, M. R. Mortality associated with sleep duration and insomnia. Arch. Gen. Psychiatry 59, 131–136 (2002). A prospective longitudinal study involving more than one million participants, which demonstrates that increased mortality is associated with sleep duration of less than or greater than 7 hours.
Dew, M. A. et al. Healthy older adults' sleep predicts all-cause mortality at 4 to 19 years of follow-up. Psychosom. Med. 65, 63–73 (2003).
Opp, M. R. & Toth, L. A. Neural-immune interactions in the regulation of sleep. Front. Biosci. 8, d768–d779 (2003).
Vitkovic, L., Bockaert, J. & Jacque, C. 'Inflammatory' cytokines: neuromodulators in normal brain? J. Neurochem. 74, 457–471 (2000).
Shoham, S., Davenne, D., Cady, A. B., Dinarello, C. A. & Krueger, J. M. Recombinant tumor necrosis factor and interleukin 1 enhance slow-wave sleep. Am. J. Physiol. 253, R142–R149 (1987).
Dickstein, J. B., Moldofsky, H., Lue, F. A. & Hay, J. B. Intracerebroventricular injection of TNF-α promotes sleep and is recovered in cervical lymph. Am. J. Physiol. 276, R1018–R1022 (1999).
Opp, M. R. & Imeri, L. Rat strains that differ in corticotropin-releasing hormone production exhibit different sleep-wake responses to interleukin 1. Neuroendocrinology 73, 272–284 (2001).
Takahashi, S., Kapas, L., Fang, J. & Krueger, J. M. An anti-tumor necrosis factor antibody suppresses sleep in rats and rabbits. Brain Res. 690, 241–244 (1995).
Opp, M. R., Postlethwaite, A. E., Seyer, J. M. & Krueger, J. M. Interleukin 1 receptor antagonist blocks somnogenic and pyrogenic responses to an interleukin 1 fragment. Proc. Natl Acad. Sci. USA 89, 3726–3730 (1992).
Opp, M. R., Smith, E. M. & Hughes, T. K. Jr. Interleukin-10 (cytokine synthesis inhibitory factor) acts in the central nervous system of rats to reduce sleep. J. Neuroimmunol. 60, 165–168 (1995).
Walter, J. S., Meyers, P. & Krueger, J. M. Microinjection of interleukin-1 into brain: separation of sleep and fever responses. Physiol. Behav. 45, 169–176 (1989).
Opp, M. R. & Krueger, J. M. Anti-interleukin-1β reduces sleep and sleep rebound after sleep deprivation in rats. Am. J. Physiol. 266, R688–R695 (1994).
Takahashi, S., Kapas, L., Fang, J. & Krueger, J. M. Somnogenic relationships between tumor necrosis factor and interleukin-1. Am. J. Physiol. 276, R1132–R1140 (1999).
Fang, J., Wang, Y. & Krueger, J. M. Effects of interleukin-1β on sleep are mediated by the type I receptor. Am. J. Physiol. 274, R655–R660 (1998). Shows the importance of IL-1β to physiological sleep and, together with reference 40, the reciprocal relationship between TNF and IL-1β in sleep.
Fang, J., Wang, Y. & Krueger, J. M. Mice lacking the TNF 55 kDa receptor fail to sleep more after TNF-α treatment. J. Neurosci. 17, 5949–5955 (1997).
Kubota, T., Kushikata, T., Fang, J. & Krueger, J. M. Nuclear factor-κB inhibitor peptide inhibits spontaneous and interleukin-1β-induced sleep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279, R404–R413 (2000).
Kubota, T., Brown, R. A., Fang, J. & Krueger, J. M. Interleukin-15 and interleukin-2 enhance non-REM sleep in rabbits. Am. J. Physiol. Regul. Integr. Comp. Physiol. 281, R1004–R1012 (2001).
Kubota, T., Fang, J., Brown, R. A. & Krueger, J. M. Interleukin-18 promotes sleep in rabbits and rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 281, R828–R838 (2001).
Kubota, T., Majde, J. A., Brown, R. A. & Krueger, J. M. Tumor necrosis factor receptor fragment attenuates interferon-γ-induced non-REM sleep in rabbits. J. Neuroimmunol. 119, 192–198 (2001).
Krueger, J. M. et al. Interferon-α2 enhances slow-wave sleep in rabbits. Int. J. Immunopharmacol. 9, 23–30 (1987).
Kushikata, T., Fang, J. & Krueger, J. M. Interleukin-10 inhibits spontaneous sleep in rabbits. J. Interferon Cytokine Res. 19, 1025–1030 (1999).
Kubota, T., Fang, J., Kushikata, T. & Krueger, J. M. Interleukin-13 and transforming growth factor-β1 inhibit spontaneous sleep in rabbits. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279, R786–R792 (2000).
Kushikata, T., Fang, J., Wang, Y. & Krueger, J. M. Interleukin-4 inhibits spontaneous sleep in rabbits. Am. J. Physiol. 275, R1185–R1191 (1998).
Hogan, D., Morrow, J. D., Smith, E. M. & Opp, M. R. Interleukin-6 alters sleep of rats. J. Neuroimmunol. 137, 59–66 (2003).
Krueger, J. M. & Majde, J. A. Microbial products and cytokines in sleep and fever regulation. Crit. Rev. Immunol. 14, 355–379 (1994).
Spath-Schwalbe, E. et al. Acute effects of recombinant human interleukin-6 on endocrine and central nervous sleep functions in healthy men. J. Clin. Endocrinol. Metab. 83, 1573–1579 (1998).
Borbely, A. A., Achermann, P., Trachsel, L. & Tobler, I. Sleep initiation and initial sleep intensity: interactions of homeostatic and circadian mechanisms. J. Biol. Rhythms 4, 149–160 (1989).
Krueger, J. M. & Majde, J. A. Humoral links between sleep and the immune system: research issues. Ann. NY Acad. Sci. 992, 9–20 (2003). A recent review of how the endocrine system might be involved in mediating sleep and immunity.
Armstrong, M. D. & Klein, J. R. Immune-endocrine interactions of the hypothalamus–pituitary–thyroid axis: integration, communication and homeostasis. Arch. Immunol. Ther. Exp. (Warsz.) 49, 231–237 (2001).
Obal, F. Jr, Bodosi, B., Szilagyi, A., Kacsoh, B. & Krueger, J. M. Antiserum to growth hormone decreases sleep in the rat. Neuroendocrinology 66, 9–16 (1997).
Obal, F. Jr, Fang, J., Payne, L. C. & Krueger, J. M. Growth-hormone-releasing hormone mediates the sleep-promoting activity of interleukin-1 in rats. Neuroendocrinology 61, 559–565 (1995).
Redwine, L., Hauger, R. L., Gillin, J. C. & Irwin, M. Effects of sleep and sleep deprivation on interleukin-6, growth hormone, cortisol, and melatonin levels in humans. J. Clin. Endocrinol. Metab. 85, 3597–3603 (2000).
Cupps, T. R. & Fauci, A. S. Corticosteroid-mediated immunoregulation in man. Immunol. Rev. 65, 133–155 (1982).
Gala, R. R. Prolactin and growth hormone in the regulation of the immune system. Proc. Soc. Exp. Biol. Med. 198, 513–527 (1991).
Maestroni, G. J., Conti, A. & Pierpaoli, W. Role of the pineal gland in immunity. Circadian synthesis and release of melatonin modulates the antibody response and antagonizes the immunosuppressive effect of corticosterone. J. Neuroimmunol. 13, 19–30 (1986).
Born, J., Lange, T., Hansen, K., Molle, M. & Fehm, H. L. Effects of sleep and circadian rhythm on human circulating immune cells. J. Immunol. 158, 4454–4464 (1997). An impressive number of measurements of immune cells, plasma cytokines and in vitro responses to LPS and PHA, measured during both normal sleep and sleep deprivation. The results show a correlation between cytokine responses and the number of circulating immune cells.
Heiser, P. et al. White blood cells and cortisol after sleep deprivation and recovery sleep in humans. Eur. Arch. Psychiatry Clin. Neurosci. 250, 16–23 (2000).
Born, J. et al. Cytokine production and lymphocyte subpopulations in aged humans. An assessment during nocturnal sleep. Mech. Ageing Dev. 84, 113–126 (1995).
Moldofsky, H., Lue, F. A., Shahal, B., Jiang, C. G. & Gorczynski, R. M. Diurnal sleep/wake-related immune functions during the menstrual cycle of healthy young women. J. Sleep Res. 4, 150–159 (1995).
Entzian, P., Linnemann, K., Schlaak, M. & Zabel, P. Obstructive sleep apnea syndrome and circadian rhythms of hormones and cytokines. Am. J. Respir. Crit. Care Med. 153, 1080–1086 (1996).
Moldofsky, H., Lue, F. A., Eisen, J., Keystone, E. & Gorczynski, R. M. The relationship of interleukin-1 and immune functions to sleep in humans. Psychosom. Med. 48, 309–318 (1986).
Gudewill, S. et al. Nocturnal plasma levels of cytokines in healthy men. Eur. Arch. Psychiatry Clin. Neurosci. 242, 53–56 (1992).
Lissoni, P., Rovelli, F., Brivio, F., Brivio, O. & Fumagalli, L. Circadian secretions of IL-2, IL-12, IL-6 and IL-10 in relation to the light/dark rhythm of the pineal hormone melatonin in healthy humans. Nat. Immun. 16, 1–5 (1998).
Moldofsky, H. et al. in Sleep '88 (ed. Horne, J.) 1 (Gustav Fischer, New York, 1989).
Redwine, L., Dang, J., Hall, M. & Irwin, M. Disordered sleep, nocturnal cytokines, and immunity in alcoholics. Psychosom. Med. 65, 75–85 (2003).
Lancel, M., Mathias, S., Faulhaber, J. & Schiffelholz, T. Effect of interleukin-1β on EEG power density during sleep depends on circadian phase. Am. J. Physiol. 270, R830–R837 (1996).
Mohren, D. C. et al. Prevalence of common infections among employees in different work schedules. J. Occup. Environ. Med. 44, 1003–1011 (2002).
Dinges, D. F. et al. Leukocytosis and natural killer cell function parallel neurobehavioral fatigue induced by 64 hours of sleep deprivation. J. Clin. Invest. 93, 1930–1939 (1994). A well-conducted sleep-deprivation study measuring changes in immune-cell number and function in humans. The first study to find alterations in immune parameters with different durations of sleep deprivation.
Ozturk, L. et al. Effects of 48 hours sleep deprivation on human immune profile. Sleep Res. Online 2, 107–111 (1999).
Boyum, A. et al. The effect of strenuous exercise, calorie deficiency and sleep deprivation on white blood cells, plasma immunoglobulins and cytokines. Scand. J. Immunol. 43, 228–235 (1996).
Palmblad, J., Petrini, B., Wasserman, J. & Akerstedt, T. Lymphocyte and granulocyte reactions during sleep deprivation. Psychosom. Med. 41, 273–278 (1979).
Irwin, M. et al. Partial night sleep deprivation reduces natural killer and cellular immune responses in humans. FASEB J. 10, 643–653 (1996).
Moldofsky, H., Lue, F. A., Davidson, J. R. & Gorczynski, R. Effects of sleep deprivation on human immune functions. FASEB J. 3, 1972–1977 (1989).
Irwin, M. et al. Partial sleep deprivation reduces natural killer cell activity in humans. Psychosom. Med. 56, 493–498 (1994).
Palmblad, J. et al. Stressor exposure and immunological response in man: interferon-producing capacity and phagocytosis. J. Psychosom. Res. 20, 193–199 (1976).
Bergmann, B. M., Rechtschaffen, A., Gilliland, M. A. & Quintans, J. Effect of extended sleep deprivation on tumor growth in rats. Am. J. Physiol. 271, R1460–R1464 (1996).
Brown, R., Price, R. J., King, M. G. & Husband, A. J. Interleukin-1β and muramyl dipeptide can prevent decreased antibody response associated with sleep deprivation. Brain Behav. Immun. 3, 320–330 (1989).
Renegar, K. B., Floyd, R. & Krueger, J. M. Effect of sleep deprivation on serum influenza-specific IgG. Sleep 21, 19–24 (1998).
Uthgenannt, D., Schoolmann, D., Pietrowsky, R., Fehm, H. L. & Born, J. Effects of sleep on the production of cytokines in humans. Psychosom. Med. 57, 97–104 (1995).
Shearer, W. T. et al. Soluble TNF-α receptor 1 and IL-6 plasma levels in humans subjected to the sleep deprivation model of spaceflight. J. Allergy Clin. Immunol. 107, 165–170 (2001).
Mackiewicz, M., Sollars, P. J., Ogilvie, M. D. & Pack, A. I. Modulation of IL-1β gene expression in the rat CNS during sleep deprivation. Neuroreport 7, 529–533 (1996).
Takahashi, S., Kapas, L., Seyer, J. M., Wang, Y. & Krueger, J. M. Inhibition of tumor necrosis factor attenuates physiological sleep in rabbits. Neuroreport 7, 642–646 (1996).
Everson, C. A. Sustained sleep deprivation impairs host defense. Am. J. Physiol. 265, R1148–R1154 (1993).
Rechtschaffen, A., Gilliland, M. A., Bergmann, B. M. & Winter, J. B. Physiological correlates of prolonged sleep deprivation in rats. Science 221, 182–184 (1983).
Landis, C. A. & Whitney, J. D. Effects of 72 hours sleep deprivation on wound healing in the rat. Res. Nurs. Health 20, 259–267 (1997).
Horohov, D. W., Pourciau, S. S., Mistric, L., Chapman, A. & Ryan, D. H. Increased dietary fat prevents sleep deprivation-induced immune suppression in rats. Comp. Med. 51, 230–233 (2001).
Brown, R., Pang, G., Husband, A. J. & King, M. G. Suppression of immunity to influenza virus infection in the respiratory tract following sleep disturbance. Reg. Immunol. 2, 321–325 (1989).
Renegar, K. B., Crouse, D., Floyd, R. A. & Krueger, J. Progression of influenza viral infection through the murine respiratory tract: the protective role of sleep deprivation. Sleep 23, 859–863 (2000).
Renegar, K. B., Floyd, R. A. & Krueger, J. M. Effects of short-term sleep deprivation on murine immunity to influenza virus in young adult and senescent mice. Sleep 21, 241–248 (1998).
Toth, L. A. & Rehg, J. E. Effects of sleep deprivation and other stressors on the immune and inflammatory responses of influenza-infected mice. Life Sci. 63, 701–709 (1998).
Spiegel, K., Sheridan, J. F. & Van Cauter, E. Effect of sleep deprivation on response to immunization. JAMA 288, 1471–1472 (2002). The first demonstration of the detrimental functional effects of sleep deprivation on immunization in humans.
Lange, T., Perras, B., Fehm, H. L. & Born, J. Sleep enhances the human antibody response to hepatitis A vaccination. Psychosom. Med. 65, 831–835 (2003).
Gabor, J. Y., Cooper, A. B. & Hanly, P. J. Sleep disruption in the intensive care unit. Curr. Opin. Crit. Care 7, 21–27 (2001).
Moldofsky, H. Fibromyalgia, sleep disorder and chronic fatigue syndrome. Ciba Found. Symp. 173, 262–271 (1993).
Mullington, J. M., Hinze-Selch, D. & Pollmacher, T. Mediators of inflammation and their interaction with sleep: relevance for chronic fatigue syndrome and related conditions. Ann. NY Acad. Sci. 933, 201–210 (2001).
Borish, L. et al. Chronic fatigue syndrome: identification of distinct subgroups on the basis of allergy and psychologic variables. J. Allergy Clin. Immunol. 102, 222–230 (1998).
Gupta, S., Aggarwal, S., See, D. & Starr, A. Cytokine production by adherent and non-adherent mononuclear cells in chronic fatigue syndrome. J. Psychiatr. Res. 31, 149–156 (1997).
Krouse, H. J., Davis, J. E. & Krouse, J. H. Immune mediators in allergic rhinitis and sleep. Otolaryngol. Head Neck Surg. 126, 607–613 (2002).
Song, C. et al. The inflammatory response system and the availability of plasma tryptophan in patients with primary sleep disorders and major depression. J. Affect. Disord. 49, 211–219 (1998).
Savard, J. et al. Association between subjective sleep quality and depression on immunocompetence in low-income women at risk for cervical cancer. Psychosom. Med. 61, 496–507 (1999).
Hall, M. et al. Sleep as a mediator of the stress–immune relationship. Psychosom. Med. 60, 48–51 (1998).
Cover, H. & Irwin, M. Immunity and depression: insomnia, retardation, and reduction of natural killer cell activity. J. Behav. Med. 17, 217–223 (1994).
Bauer, J. et al. Induction of cytokine synthesis and fever suppresses REM sleep and improves mood in patients with major depression. Biol. Psychiatry 38, 611–621 (1995). Shows the effects on sleep and lifting-of-mood in depression following the induction of pro-inflammatory cytokine expression (TNF, IL-1β and IL-6) mediated by the administration of LPS.
Ringel, B. L. & Szuba, M. P. Potential mechanisms of the sleep therapies for depression. Depress. Anxiety 14, 29–36 (2001).
Irwin, M., Clark, C., Kennedy, B., Gillin, J. C. & Ziegler, M. Nocturnal catecholamines and immune function in insomniacs, depressed patients, and control subjects. Brain Behav. Immun. 17, 365–372 (2003).
Savard, J., Laroche, L., Simard, S., Ivers, H. & Morin, C. M. Chronic insomnia and immune functioning. Psychosom. Med. 65, 211–221 (2003).
Sakami, S. et al. Coemergence of insomnia and a shift in the TH1/TH2 balance toward TH2 dominance. Neuroimmunomodulation 10, 337–343 (2002).
Alberti, A. et al. Plasma cytokine levels in patients with obstructive sleep apnea syndrome: a preliminary study. J. Sleep Res. 12, 305–311 (2003). Describes cytokine changes in patients with obstructive sleep apnoea. Patients show differences in T H 2-cell-type cytokine patterns, compared with controls at baseline, and these become more pronounced after an obstructive episode.
Mignot, E. Perspectives in narcolepsy research and therapy. Curr. Opin. Pulm. Med. 2, 482–487 (1996).
Lin, L., Hungs, M. & Mignot, E. Narcolepsy and the HLA region. J. Neuroimmunol. 117, 9–20 (2001).
Nishino, S., Ripley, B., Overeem, S., Lammers, G. J. & Mignot, E. Hypocretin (orexin) deficiency in human narcolepsy. Lancet 355, 39–40 (2000).
Waleh, N. S., Apte-Deshpande, A., Terao, A., Ding, J. & Kilduff, T. S. Modulation of the promoter region of prepro-hypocretin by α-interferon. Gene 262, 123–128 (2001).
Tafti, M., Chollet, D., Valatx, J. L. & Franken, P. Quantitative trait loci approach to the genetics of sleep in recombinant inbred mice. J. Sleep Res. 8 (Suppl. 1), 37–43 (1999). Defines genes that might greatly influence sleep amount and the immune system.
Dement, W. C. & Gelb, M. Somnolence: its importance in society. Neurophysiol. Clin. 23, 5–14 (1993).
Weatherstone, K. B., Franck, L. S. & Klein, N. J. Are there opportunities to decrease nosocomial infection by choice of analgesic regimen? Evidence for immunity and pain interactions. Arch. Pediatr. Adolesc. Med. 157, 1108–1114 (2003).
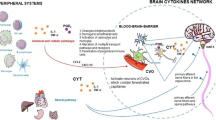
Plata-Salaman, C. R. Brain cytokines and disease. Acta Neuropsychiatr. 14, 262–278 (2002). A thorough review of cytokines in the brain, their relationship to peripheral cytokines and how this might lead to disease.
Krueger, J. M., Obal, F. Jr. & Fang, J. Humoral regulation of physiological sleep: cytokines and GHRH. J. Sleep Res. 8 (Suppl 1), 53–59 (1999).
Quan, N., Whiteside, M. & Herkenham, M. Time course and localization patterns of interleukin-1β messenger RNA expression in brain and pituitary after peripheral administration of lipopolysaccharide. Neuroscience 83, 281–293 (1998).
Cartmell, T., Luheshi, G. N. & Rothwell, N. J. Brain sites of action of endogenous interleukin-1 in the febrile response to localized inflammation in the rat. J. Physiol. 518, 585–594 (1999).
Laye, S. et al. Endogenous brain IL-1 mediates LPS-induced anorexia and hypothalamic cytokine expression. Am. J. Physiol. Regul. Integr. Comp. Physiol. 279, R93–R98 (2000).
Schoning, B., Elepfandt, P., Lanksch, W. R., Volk, H. D. & Woiciechowsky, C. Continuous infusion of proinflammatory cytokines into the brain to study brain cytokine induced local and systemic immune effects. Brain Res. Brain Res. Protoc. 4, 217–222 (1999).
Woiciechowsky, C. et al. Sympathetic activation triggers systemic interleukin-10 release in immunodepression induced by brain injury. Nature Med. 4, 808–813 (1998).
Thibeault, I., Laflamme, N. & Rivest, S. Regulation of the gene encoding the monocyte chemoattractant protein 1 (MCP-1) in the mouse and rat brain in response to circulating LPS and proinflammatory cytokines. J. Comp. Neurol. 434, 461–477 (2001).
Dantzer, R. et al. Cytokines and sickness behavior. Ann. NY Acad. Sci. 840, 586–590 (1998).
Elenkov, I. J., Wilder, R. L., Chrousos, G. P. & Vizi, E. S. The sympathetic nerve—an integrative interface between two supersystems: the brain and the immune system. Pharmacol. Rev. 52, 595–638 (2000).
Hattori, N. et al. GH, GH receptor, GH secretagogue receptor, and ghrelin expression in human T cells, B cells, and neutrophils. J. Clin. Endocrinol. Metab. 86, 4284–4291 (2001).
Acknowledgements
The authors thank R. Robins-Browne, J. McCluskey and D. Ventor for helpful comments on the manuscript. P.A.B. is the recipient of a European Society of Paediatric Infectious Diseases Fellowship Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- ELECTROENCEPHALOGRAM
-
Graphical representation of the electrical activity of the brain, recorded by attaching electrodes to the scalp. The shape, frequency and amplitude of the waveforms provide information about the stage and intensity of sleep.
- MURAMYL PEPTIDES
-
Fragments of peptidoglycans, from the cell walls of Gram-positive bacteria, that are thought to have a crucial role in the generation of the immune response to Gram-positive bacterial infection.
- LIPOPOLYSACCHARIDE
-
A constituent of the cell walls of Gram-negative bacteria that is thought to be important for eliciting the immune response to Gram-negative bacterial infection. Also known as endotoxin.
- PYROGENIC RESPONSE
-
The response to infection that leads to fever. Cytokines induced by microbial products — particularly tumour-necrosis factor and interleukins — function to increase the 'set point' for body temperature (through eliciting prostaglandin synthesis in the hypothalamus) and consequently produce fever.
- ACUTE-PHASE RESPONSE
-
The early immune response to infection, which results in the production of cytokines and other mediators and an increase in the number of peripheral leukocytes.
- CIRCADIAN RHYTHMICITY
-
Having an approximately 24-hour variation. This can be a property of biological or behavioural processes. It can be a direct consequence of an endogenous circadian mechanism or be secondary to other processes, such as the sleep–wake cycle. From the Latin words circa meaning 'about' and dies meaning 'day'.
- OBSTRUCTIVE SLEEP APNOEA
-
A medical condition in which the obstruction of upper airways causes episodes of breathing cessation during sleep, leading to recurrent arousals from sleep and other complications.
- CIRCADIAN OSCILLATOR
-
The biological clock responsible for organizing many of the circadian rhythms of the body. It is a function of the suprachiasmatic nucleus in the brain.
- CACHEXIA
-
Severe weight loss, muscle wasting and debility caused by prolonged disease. It is thought to be mediated through neuroimmunoendocrine interactions.
- CHRONIC FATIGUE AND FIBROMYALGIA
-
Clinical conditions characterized by debilitating fatigue, often following a viral illness. In the latter, fatigue occurs together with chronic pain and tenderness in muscles.
Rights and permissions
About this article
Cite this article
Bryant, P., Trinder, J. & Curtis, N. Sick and tired: does sleep have a vital role in the immune system?. Nat Rev Immunol 4, 457–467 (2004). https://doi.org/10.1038/nri1369
Issue Date:
DOI: https://doi.org/10.1038/nri1369
This article is cited by
-
Association between sleep duration and dental caries in a nationally representative U.S. population
BMC Oral Health (2023)
-
Do nocturnal asthma attacks influence sleep parameters and inflammatory markers? A cross-sectional population-based study
Sleep and Breathing (2023)
-
Prediction of the local and total thermal insulations of a bedding system based on the 3D virtual simulation technology
Building Simulation (2023)
-
Effect of sleep on development of early childhood caries: a systematic review
European Archives of Paediatric Dentistry (2023)
-
Circadian rhythms and cancers: the intrinsic links and therapeutic potentials
Journal of Hematology & Oncology (2022)