Key Points
-
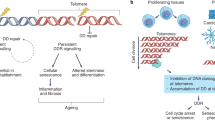
Mutations in telomerase and telomere gene components manifest as diverse clinical syndromes that vary in severity but share a single common molecular defect of shortened telomeres.
-
Telomere length determines disease severity and type in the monogenic telomere syndromes. This is most evidently seen in the pattern of genetic anticipation in families with autosomal-dominant inheritance because mutant telomerase genes cause haploinsufficiency and progressive telomere shortening across generations.
-
Telomere length is a heritable genetic trait even when the telomerase genes are wild-type. Because it is polymorphic, it may influence disease risk across populations.
-
Short telomere length limits the replicative potential of stem cells in high-turnover tissues, such as the bone marrow. This is seen clinically in a failure of haematopoiesis known as aplastic anaemia, which is a common complication of telomere syndromes.
-
Even in tissues of slow turnover, short telomere length causes degenerative disease, as seen in the high prevalence of pulmonary disease in telomerase mutation carriers. In these tissues, short telomere length lowers the threshold to acquired injuries, such as cigarette smoke in the lung.
Abstract
There has been mounting evidence of a causal role for telomere dysfunction in a number of degenerative disorders. Their manifestations encompass common disease states such as idiopathic pulmonary fibrosis and bone marrow failure. Although these disorders seem to be clinically diverse, collectively they comprise a single syndrome spectrum defined by the short telomere defect. Here we review the manifestations and unique genetics of telomere syndromes. We also discuss their underlying molecular mechanisms and significance for understanding common age-related disease processes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
12 February 2013
In the schematic chromosome diagrams of figure 3 of this article, the position of the mutant TERT or TR gene and the junction between the telomere and the rest of the chromosome, was inconsistent across the figure. The article has been corrected online. The authors and editors apologize for these errors.
References
Blackburn, E. H. Telomeres and telomerase: the means to the end (Nobel lecture). Angew. Chem. Int. Edn Engl. 49, 7405–7421 (2010).
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
Allsopp, R. C. et al. Telomere length predicts replicative capacity of human fibroblasts. Proc. Natl Acad. Sci. USA 89, 10114–10118 (1992). References 2 and 3 showed that telomere length shortens in human cells with age and limits their in vitro replicative potential.
Greider, C. W. & Blackburn, E. H. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 43, 405–413 (1985).
Greider, C. W. & Blackburn, E. H. The telomere terminal transferase of Tetrahymena is a ribonucleoprotein enzyme with two kinds of primer specificity. Cell 51, 887–898 (1987).
Blackburn, E. H., Greider, C. W. & Szostak, J. W. Telomeres and telomerase: the path from maize, Tetrahymena and yeast to human cancer and aging. Nature Med. 12, 1133–1138 (2006).
Kim, N. W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011–2015 (1994). In this study, telomerase activity was found to be enriched in cancer and germ cells, suggesting that telomerase has a role in extending replicative potential.
Buseman, C. M., Wright, W. E. & Shay, J. W. Is telomerase a viable target in cancer? Mutat. Res. 730, 90–97 (2012).
Heiss, N. S. et al. X-linked dyskeratosis congenita is caused by mutations in a highly conserved gene with putative nucleolar functions. Nature Genet. 19, 32–38 (1998). This study identified mutations in the X-linked DKC1 gene as a first cause of dyskeratosis congenita.
Mitchell, J. R., Cheng, J. & Collins, K. A box H/ACA small nucleolar RNA-like domain at the human telomerase RNA 3′ end. Mol. Cell. Biol. 19, 567–576 (1999).
Mitchell, J. R., Wood, E. & Collins, K. A telomerase component is defective in the human disease dyskeratosis congenita. Nature 402, 551–555 (1999). This study showed that dyskerin is essential for telomerase RNA stability and that dyskeratosis congenita cell lines have a short telomere length, linking this disease with defects in telomere maintenance.
Armanios, M. Telomerase and idiopathic pulmonary fibrosis. Mutat. Res. 730, 52–58 (2012).
Armanios, M. Syndromes of telomere shortening. Annu. Rev. Genom. Hum. Genet. 10, 45–61 (2009).
Moyzis, R. K. et al. A highly conserved repetitive DNA sequence, (TTAGGG)n, present at the telomeres of human chromosomes. Proc. Natl Acad. Sci. USA 85, 6622–6626 (1988).
Allshire, R. C. et al. Telomeric repeat from T. thermophila cross hybridizes with human telomeres. Nature 332, 656–659 (1988).
Vaziri, H. et al. Loss of telomeric DNA during aging of normal and trisomy 21 human lymphocytes. Am. J. Hum. Genet. 52, 661–667 (1993).
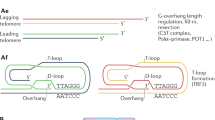
de Lange, T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 19, 2100–2110 (2005).
Palm, W. & de Lange, T. How shelterin protects mammalian telomeres. Annu. Rev. Genet. 42, 301–334 (2008).
Surovtseva, Y. V. et al. Conserved telomere maintenance component 1 interacts with STN1 and maintains chromosome ends in higher eukaryotes. Mol. Cell 36, 207–218 (2009).
Miyake, Y. et al. RPA-like mammalian Ctc1-Stn1-Ten1 complex binds to single-stranded DNA and protects telomeres independently of the Pot1 pathway. Mol. Cell 36, 193–206 (2009).
Price, C. M. et al. Evolution of CST function in telomere maintenance. Cell Cycle 9, 3157–3165 (2010).
Gu, P. et al. CTC1 deletion results in defective telomere replication, leading to catastrophic telomere loss and stem cell exhaustion. EMBO J 31, 2309–2321 (2012).
Greider, C. W. & Blackburn, E. H. A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature 337, 331–337 (1989).
Lingner, J. et al. Reverse transcriptase motifs in the catalytic subunit of telomerase. Science 276, 561–567 (1997).
Feng, J. et al. The RNA component of human telomerase. Science 269, 1236–1241 (1995).
Podlevsky, J. D. & Chen, J. J. It all comes together at the ends: telomerase structure, function, and biogenesis. Mutat. Res. 730, 3–11 (2012).
Blackburn, E. H. & Collins, K. Telomerase: an RNP enzyme synthesizes DNA. Cold Spring Harb. Perspect. Biol. 3, a003558 (2011).
Meier, U. T. The many facets of H/ACA ribonucleoproteins. Chromosoma 114, 1–14 (2005).
Chen, J. L., Blasco, M. A. & Greider, C. W. Secondary structure of vertebrate telomerase RNA. Cell 100, 503–514 (2000).
Venteicher, A. S. et al. A human telomerase holoenzyme protein required for Cajal body localization and telomere synthesis. Science 323, 644–648 (2009).
Tycowski, K. T., Shu, M. D., Kukoyi, A. & Steitz, J. A. A conserved WD40 protein binds the Cajal body localization signal of scaRNP particles. Mol. Cell 34, 47–57 (2009).
Diede, S. J. & Gottschling, D. E. Telomerase-mediated telomere addition in vivo requires DNA primase and DNA polymerases α and δ. Cell 99, 723–733 (1999).
Marcand, S., Brevet, V., Mann, C. & Gilson, E. Cell cycle restriction of telomere elongation. Curr. Biol. 10, 487–490 (2000).
Teixeira, M. T., Arneric, M., Sperisen, P. & Lingner, J. Telomere length homeostasis is achieved via a switch between telomerase- extendible and -nonextendible states. Cell 117, 323–335 (2004).
Erdmann, N., Liu, Y. & Harrington, L. Distinct dosage requirements for the maintenance of long and short telomeres in mTert heterozygous mice. Proc. Natl Acad. Sci. USA 101, 6080–6085 (2004).
Hao, L. Y. et al. Short telomeres, even in the presence of telomerase, limit tissue renewal capacity. Cell 123, 1121–1131 (2005). In reference 36, using CAST/EiJ mice that have telomere length similar to humans, this paper established that telomerase haploinsufficiency is sufficient to cause dyskeratosis-congenita-like phenotypes. It also showed that telomere length is inherited and can cause degenerative phenotypes even when telomerase is wild-type.
Mozdy, A. D. & Cech, T. R. Low abundance of telomerase in yeast: implications for telomerase haploinsufficiency. RNA 12, 1721–1737 (2006).
Vulliamy, T. et al. The RNA component of telomerase is mutated in autosomal dominant dyskeratosis congenita. Nature 413, 432–435 (2001).
Armanios, M. et al. Haploinsufficiency of telomerase reverse transcriptase leads to anticipation in autosomal dominant dyskeratosis congenita. Proc. Natl Acad. Sci. USA 102, 15960–15964 (2005). References 38 and 39 identified mutations in the essential telomerase genes, TERT and TR , in dyskeratosis congenita.
Strong, M. A. et al. Phenotypes in mTERT+/− and mTERT+/− mice are due to short telomeres, not telomere-independent functions of telomerase reverse transcriptase. Mol. Cell. Biol. 31, 2369–2379 (2011).
Yamaguchi, H. et al. Mutations in TERT, the gene for telomerase reverse transcriptase, in aplastic anemia. N. Engl. J. Med. 352, 1413–1424 (2005). This study identified TERT mutations in patients with aplastic anaemia.
Chiu, C. P. et al. Differential expression of telomerase activity in hematopoietic progenitors from adult human bone marrow. Stem Cells 14, 239–248 (1996).
Vaziri, H. et al. Evidence for a mitotic clock in human hematopoietic stem cells: loss of telomeric DNA with age. Proc. Natl Acad. Sci. USA 91, 9857–9860 (1994).
Wright, W. E., Piatyszek, M. A., Rainey, W. E., Byrd, W. & Shay, J. W. Telomerase activity in human germline and embryonic tissues and cells. Dev. Genet. 18, 173–179 (1996).
Greider, C. W. Telomerase RNA levels limit the telomere length equilibrium. Cold Spring Harb. Symp. Quant. Biol. 71, 225–229 (2006).
Zhou, J., Monson, E. K., Teng, S. C., Schulz, V. P. & Zakian, V. A. Pif1p helicase, a catalytic inhibitor of telomerase in yeast. Science 289, 771–774 (2000).
Makovets, S. & Blackburn, E. H. DNA damage signalling prevents deleterious telomere addition at DNA breaks. Nature Cell Biol. 11, 1383–1386 (2009).
Hemann, M. T., Strong, M. A., Hao, L. Y. & Greider, C. W. The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell 107, 67–77 (2001). This study established that the shortest telomeres have a genetically dominant influence on cellular phenotypes.
McEachern, M. J. & Blackburn, E. H. Cap-prevented recombination between terminal telomeric repeat arrays (telomere CPR) maintains telomeres in Kluyveromyces lactis lacking telomerase. Genes Dev. 10, 1822–1834 (1996).
Kaul, Z., Cesare, A. J., Huschtscha, L. I., Neumann, A. A. & Reddel, R. R. Five dysfunctional telomeres predict onset of senescence in human cells. EMBO Rep. 13, 52–59 (2011).
Lin, J. et al. Analyses and comparisons of telomerase activity and telomere length in human T and B cells: insights for epidemiology of telomere maintenance. J. Immunol. Methods 352, 71–80 (2010).
Rufer, N. et al. Telomere fluorescence measurements in granulocytes and T lymphocyte subsets point to a high turnover of hematopoietic stem cells and memory T cells in early childhood. J. Exp. Med. 190, 157–167 (1999).
Aubert, G., Hills, M. & Lansdorp, P. M. Telomere length measurement-Caveats and a critical assessment of the available technologies and tools. Mutat. Res. 730, 59–67 (2012).
Alder, J. K. et al. Short telomeres are a risk factor for idiopathic pulmonary fibrosis. Proc. Natl Acad. Sci. USA 105, 13051–13056 (2008).
Alter, B. P. et al. Very short telomere length by flow fluorescence in situ hybridization identifies patients with dyskeratosis congenita. Blood 110, 1439–1447 (2007).
Armanios, M. Y. et al. Telomerase mutations in families with idiopathic pulmonary fibrosis. N. Engl. J. Med. 356, 1317–1326 (2007). References 54–56 showed that lymphocyte telomere length using flow cytometry and fluorescence in situ hybridization could be a useful diagnostic marker in identifying individuals with telomere-related disease.
Baerlocher, G. M., Vulto, I., de Jong, G. & Lansdorp, P. M. Flow cytometry and FISH to measure the average length of telomeres (flow FISH). Nature Protoc. 1, 2365–2376 (2006).
de la Fuente, J. & Dokal, I. Dyskeratosis congenita: advances in the understanding of the telomerase defect and the role of stem cell transplantation. Pediatr. Transplant 11, 584–594 (2007).
Savage, S. A. & Alter, B. P. Dyskeratosis congenita. Hematol. Oncol. Clin. North Am. 23, 215–231 (2009).
Vulliamy, T. et al. Mutations in the telomerase component NHP2 cause the premature ageing syndrome dyskeratosis congenita. Proc. Natl Acad. Sci. USA 105, 8073–8078 (2008).
Walne, A. J. et al. Genetic heterogeneity in autosomal recessive dyskeratosis congenita with one subtype due to mutations in the telomerase-associated protein NOP10. Hum. Mol. Genet. 16, 1619–1629 (2007).
Savage, S. A. et al. TINF2, a component of the shelterin telomere protection complex, is mutated in dyskeratosis congenita. Am. J. Hum. Genet. 82, 501–509 (2008). This paper was the first to show that mutations in a shelterin component are a cause of dyskeratosis congenita.
Zhong, F. et al. Disruption of telomerase trafficking by TCAB1 mutation causes dyskeratosis congenita. Genes Dev. 25, 11–16.
Keller, R. B. et al. CTC1 mutations in a patient with dyskeratosis congenita. Pediatr. Blood Cancer 59, 311–314 (2012).
Jyonouchi, S., Forbes, L., Ruchelli, E. & Sullivan, K. E. Dyskeratosis congenita: a combined immunodeficiency with broad clinical spectrum—a single-center pediatric experience. Pediatr. Allergy Immunol. 22, 313–319 (2011).
Anderson, B. H. et al. Mutations in CTC1, encoding conserved telomere maintenance component 1, cause Coats plus. Nature Genet. 44, 338–342 (2012).
Polvi, A. et al. Mutations in CTC1, encoding the CTS telomere maintenance complex component 1, cause cerebroretinal microangiopathy with calcifications and cysts. Am. J. Hum. Genet. 90, 540–549 (2012). References 66 and 67 identified CTC1 mutations in Coats plus syndrome. Reference 66 identified short telomere length in these patients, suggesting a role for telomere dysfunction in the pathophysiology of this syndrome.
Tsakiri, K. D. et al. Adult-onset pulmonary fibrosis caused by mutations in telomerase. Proc. Natl Acad. Sci. USA 104, 7552–7557 (2007). References 56 and 68 identified mutations in the essential telomerase genes in familial pulmonary fibrosis.
Cronkhite, J.T. et al. Telomere shortening in familial and sporadic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 178, 729–737 (2008).
Diaz de Leon, A. et al. Telomere lengths, pulmonary fibrosis and telomerase (TERT) mutations. PLoS ONE 5, e10680 (2010).
Parry, E. M., Alder, J. K., Qi, X., Chen, J. J. & Armanios, M. Syndrome complex of bone marrow failure and pulmonary fibrosis predicts germline defects in telomerase. Blood 117, 5607–5611 (2011).
Du, H. Y. et al. TERC and TERT gene mutations in patients with bone marrow failure and the significance of telomere length measurements. Blood 113, 309–316 (2008).
Yamaguchi, H. et al. Mutations of the human telomerase RNA gene (TERC) in aplastic anemia and myelodysplastic syndrome. Blood 102, 916–918 (2003). This study identified TR mutations in patients with bone marrow failure.
Calado, R. T. et al. A spectrum of severe familial liver disorders associate with telomerase mutations. PLoS ONE 4, e7926 (2009).
Alter, B. P., Giri, N., Savage, S. A. & Rosenberg, P. S. Cancer in dyskeratosis congenita. Blood 113, 6549–6557 (2009).
Alder, J. K. et al. Ancestral mutation in telomerase causes defects in repeat addition processivity and manifests as familial pulmonary fibrosis. PLoS Genet. 7, e1001352 (2011).
Kirwan, M. et al. Defining the pathogenic role of telomerase mutations in myelodysplastic syndrome and acute myeloid leukemia. Hum. Mutat. 30, 1567–1573 (2009).
Calado, R. T. et al. Constitutional hypomorphic telomerase mutations in patients with acute myeloid leukemia. Proc. Natl Acad. Sci. USA 106, 1187–1192 (2009).
Artandi, S. E. & DePinho, R. A. Telomeres and telomerase in cancer. Carcinogenesis 31, 9–18 (2010).
Deng, Y., Chan, S. S. & Chang, S. Telomere dysfunction and tumour suppression: the senescence connection. Nature Rev. Cancer 8, 450–458 (2008).
Knudson, M., Kulkarni, S., Ballas, Z. K., Bessler, M. & Goldman, F. Association of immune abnormalities with telomere shortening in autosomal-dominant dyskeratosis congenita. Blood 105, 682–688 (2005).
Armanios, M. et al. Short telomeres are sufficient to cause the degenerative defects associated with aging. Am. J. Hum. Genet. 85, 823–832 (2009).
Vulliamy, T. et al. Disease anticipation is associated with progressive telomere shortening in families with dyskeratosis congenita due to mutations in TERC. Nature Genet. 36, 447–449 (2004). References 83 and 39 (above) showed that mutations in the essential telomerase genes cause genetic anticipation in autosomal-dominant dyskeratosis congenita.
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
Lee, H. W. et al. Essential role of mouse telomerase in highly proliferative organs. Nature 392, 569–574 (1998). References 84 and 85 are two landmark papers that established that telomerase itself is not essential and that short telomeres cause apoptosis and cell loss in high-turnover tissues.
Alter, B. P. et al. Telomere length is associated with disease severity and declines with age in dyskeratosis congenita. Haematologica 97, 353–359 (2012).
Makovets, S., Williams, T. L. & Blackburn, E. H. The telotype defines the telomere state in Saccharomyces cerevisiae and is inherited as a dominant non-Mendelian characteristic in cells lacking telomerase. Genetics 178, 245–257 (2008).
Hemann, M. T. & Greider, C. W. Wild-derived inbred mouse strains have short telomeres. Nucleic Acids Res. 28, 4474–4478 (2000).
Goldman, F. et al. The effect of TERC haploinsufficiency on the inheritance of telomere length. Proc. Natl Acad. Sci. USA 102, 17119–17124 (2005).
Aviv, A. Genetics of leukocyte telomere length and its role in atherosclerosis. Mutat. Res. 730, 68–74 (2012).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Enomoto, S., Glowczewski, L. & Berman, J. MEC3, MEC1, and DDC2 are essential components of a telomere checkpoint pathway required for cell cycle arrest during senescence in Saccharomyces cerevisiae. Mol. Biol. Cell 13, 2626–2638 (2002).
IJpma, A. & Greider, C. W. Short telomeres induce a DNA damage response in Saccharomyces cerevisiae. Mol. Biol. Cell 14, 987–1001 (2003).
Nautiyal, S., DeRisi, J. L. & Blackburn, E. H. The genome-wide expression response to telomerase deletion in Saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 99, 9316–9321 (2002).
Hemann, M. T. et al. Telomere dysfunction triggers developmentally regulated germ cell apoptosis. Mol. Biol. Cell 12, 2023–2030 (2001).
Chin, L. et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 97, 527–538 (1999).
Guo, N. et al. Short telomeres compromise β-cell signaling and survival. PLoS ONE 6, e17858 (2011).
Campisi, J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120, 513–522 (2005).
Alder, J. K. et al. Telomere length is a determinant of emphysema susceptibility. Am. J. Respir. Crit. Care Med. 183, 904–912 (2011).
Leri, A. et al. Ablation of telomerase and telomere loss leads to cardiac dilatation and heart failure associated with p53 upregulation. EMBO J. 22, 131–139 (2003).
Rudolph, K. L. et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 96, 701–712 (1999).
Rudolph, K. L., Chang, S., Millard, M., Schreiber-Agus, N. & DePinho, R. A. Inhibition of experimental liver cirrhosis in mice by telomerase gene delivery. Science 287, 1253–1258 (2000).
Sahin, E. et al. Telomere dysfunction induces metabolic and mitochondrial compromise. Nature 470, 359–365 (2011). References 97 and 103 implicate short telomeres as a cause of mitochondrial dysfunction, a pathogenic mechanism that highlights a role for telomere dysfunction in slow-turnover tissues.
Herrera, E., Martinez, A. C. & Blasco, M. A. Impaired germinal center reaction in mice with short telomeres. EMBO J. 19, 472–481 (2000).
Sacco, A. et al. Short telomeres and stem cell exhaustion model Duchenne muscular dystrophy in mdx/mTR mice. Cell 143, 1059–1071 (2010).
Pignolo, R. J. et al. Defects in telomere maintenance molecules impair osteoblast differentiation and promote osteoporosis. Aging Cell 7, 23–31 (2008).
Chang, S. et al. Essential role of limiting telomeres in the pathogenesis of Werner syndrome. Nature Genet. 36, 877–882 (2004).
Minamino, T. et al. A crucial role for adipose tissue p53 in the regulation of insulin resistance. Nature Med. 15, 1082–1087 (2009).
Lee, B. W., Yap, H. K., Quah, T. C., Chong, A. & Seah, C. C. T cell immunodeficiency in dyskeratosis congenita. Arch. Dis. Child 67, 524–526 (1992).
Sznajer, Y. et al. Further delineation of the congenital form of X-linked dyskeratosis congenita (Hoyeraal-Hreidarsson syndrome). Eur. J. Pediatr. 162, 863–867 (2003).
Vulliamy, T., Marrone, A., Dokal, I. & Mason, P. J. Association between aplastic anaemia and mutations in telomerase RNA. Lancet 359, 2168–2170 (2002).
Herrera, E. et al. Disease states associated with telomerase deficiency appear earlier in mice with short telomeres. EMBO J. 18, 2950–2960 (1999).
Metcalf, D. The Molecular Control of Blood Cells 1–22 (Harvard Univ. Press, 1988).
Allsopp, R. C., Morin, G. B., DePinho, R., Harley, C. B. & Weissman, I. L. Telomerase is required to slow telomere shortening and extend replicative lifespan of HSCs during serial transplantation. Blood 102, 517–520 (2003).
Goldman, F. D. et al. Characterization of primitive hematopoietic cells from patients with dyskeratosis congenita. Blood 111, 4523–4531 (2008).
Fogarty, P. F. et al. Late presentation of dyskeratosis congenita as apparently acquired aplastic anaemia due to mutations in telomerase RNA. Lancet 362, 1628–1630 (2003).
Rossi, D. J. et al. Deficiencies in DNA damage repair limit the function of haematopoietic stem cells with age. Nature 447, 725–729 (2007).
Choudhury, A. R. et al. Cdkn1a deletion improves stem cell function and lifespan of mice with dysfunctional telomeres without accelerating cancer formation. Nature Genet. 39, 99–105 (2007).
Wong, K. K. et al. Telomere dysfunction impairs DNA repair and enhances sensitivity to ionizing radiation. Nature Genet. 26, 85–88 (2000).
Coppe, J. P., Desprez, P. Y., Krtolica, A. & Campisi, J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu. Rev. Pathol. 5, 99–118 (2010).
Jiang, H. et al. Proteins induced by telomere dysfunction and DNA damage represent biomarkers of human aging and disease. Proc. Natl Acad. Sci. USA 105, 11299–11304 (2008).
Zhang, H., Pan, K. H. & Cohen, S. N. Senescence-specific gene expression fingerprints reveal cell-type-dependent physical clustering of up-regulated chromosomal loci. Proc. Natl Acad. Sci. USA 100, 3251–3256 (2003).
Prentki, M. & Nolan, C. J. Islet β cell failure in type 2 diabetes. J. Clin. Invest. 116, 1802–1812 (2006).
Knight, S. W. et al. Unexplained aplastic anaemia, immunodeficiency, and cerebellar hypoplasia (Hoyeraal-Hreidarsson syndrome) due to mutations in the dyskeratosis congenita gene, DKC1. Br. J. Haematol. 107, 335–339 (1999).
Yaghmai, R. et al. Overlap of dyskeratosis congenita with the Hoyeraal-Hreidarsson syndrome. J. Pediatr. 136, 390–393 (2000).
Walne, A. J., Vulliamy, T., Beswick, R., Kirwan, M. & Dokal, I. TINF2 mutations result in very short telomeres: analysis of a large cohort of patients with dyskeratosis congenita and related bone marrow failure syndromes. Blood 112, 3594–3600 (2008).
Chiang, Y. J., Kim, S. H., Tessarollo, L., Campisi, J. & Hodes, R. J. Telomere-associated protein TIN2 is essential for early embryonic development through a telomerase-independent pathway. Mol. Cell. Biol. 24, 6631–6634 (2004).
Acknowledgements
Much of the basic biology that is relevant to disease and that is discussed in this Review has been studied in a number of model organisms, and we acknowledge that owing to space limitations we could not reference that important work comprehensively. We are grateful to several colleagues and laboratory members for helpful discussions and comments on the manuscript. M.A. acknowledges research support from the US National Institutes of Health Heart, Lung and Blood Institute (NHLBI), the US National Cancer Institute (NCI) and the Maryland Stem Cell and Flight Attendants Medical Research Foundations. E.H.B. acknowledges support from the US National Institute of General Medical Sciences (NIGMS) and the NCI.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Elizabeth H. Blackburn is a co-founder and shareholder of Telome Health, a company that specializes in telomere length measurement. Mary Armanios declares no competing financial interests.
Glossary
- Senescence
-
Classically defined as a permanent arrest in the cell cycle in G0.
- Pulmonary fibrosis
-
A scarring disorder of the lung in which alveolar structures are replaced with extracellular matrix components such as collagen.
- Cajal body
-
A small subnuclear organelle that contains the telomerase ribonucleoprotein complex, as well as other newly assembled ribonucleoproteins.
- Haploinsufficiency
-
A state in a diploid organism whereby one normal gene copy is insufficient for normal function.
- Oral leukoplakia
-
White patches in the mucosa of the mouth; this is often considered to be a precancerous state.
- Nail dystrophy
-
Abnormal or absent finger nails.
- Exudative retinopathy
-
A condition in which white–yellow spots are seen in the retina, indicating damage to retina blood vessels. When it is an isolated finding, it is often referred to as Coats disease.
- Coats plus syndrome
-
A syndrome defined by multiple congenital anomalies that are beyond the retinal abnormalities of Coats disease patients.
- Aplastic anaemia
-
A bone marrow failure state characterized by low blood counts and a paucity of haematopoietic cells in the bone marrow.
- Genetic anticipation
-
A pattern by which a certain phenotype manifests at an earlier age and with increasing severity with successive generations in autosomal-dominant disorders.
- Heritability
-
A quantification of the genetic component contributing to a specific trait.
- Missing heritability
-
The state in which the specific genotypes underlying the inheritance of a certain trait are not known.
- Allogeneic stem cell transplant
-
Transplant of stem cells, most frequently bone-marrow-derived, from an alternative donor to replace a failed organ.
- Cell-autonomous
-
An effect that is intrinsic to a specific cell type and not to an independent factor beyond that cell type.
- Crypt stem cells
-
Cells in the intestinal crypt that are responsible for the regenerative capacity of the epithelial protective barrier in the intestine.
- Tissue remodelling
-
The process by which tissue structures change, often in the setting of recovery from injury or healing.
- Senescence-associated secretory phenotype
-
(SASP). The phenomenon by which cultured senescent cells secrete growth factors, cytokines and proteases.
Rights and permissions
About this article
Cite this article
Armanios, M., Blackburn, E. The telomere syndromes. Nat Rev Genet 13, 693–704 (2012). https://doi.org/10.1038/nrg3246
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg3246
This article is cited by
-
Telomeres and aging: on and off the planet!
Biogerontology (2024)
-
Telomere Biology Disorder: A Focus on Gastrointestinal and Hepatic Manifestations
Current Hematologic Malignancy Reports (2024)
-
Emerging Treatments for Childhood Interstitial Lung Disease
Pediatric Drugs (2024)
-
Studying the pathogenicity of 26 variants characterized in the first molecular analyses of Egyptian aplastic anemia patients
Journal of Genetic Engineering and Biotechnology (2023)
-
Non-canonical telomere protection role of FOXO3a of human skeletal muscle cells regulated by the TRF2-redox axis
Communications Biology (2023)