Key Points
-
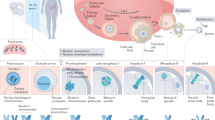
Aneuploidy is extraordinarily common in humans, occurring in an estimated 20–40% of all conceptions. It is the most common cause of miscarriages and congenital defects in our species and is a leading impediment to the treatment of infertility.
-
Most aneuploidy results from maternal meiotic nondisjunctional errors. However, there is remarkable variation among chromosomes in the way in which these errors originate, indicating that there are multiple mechanisms by which human aneuploidy occurs.
-
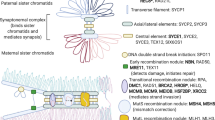
Studies of human fetal oocytes indicate a high level of recombination errors, indicating that some oocytes are predisposed to nondisjoin because of events that occurred before birth.
-
Cell cycle control checkpoints that operate in meiotic prophase and at the metaphase–anaphase transition are less stringent in females than in males. Consequently, abnormal cells that are eliminated in spermatogenesis may escape detection in the female, ultimately leading to aneuploid eggs.
-
Studies from mice suggest that loss of cohesin proteins over the reproductive life of the female contribute to the maternal age effect on human trisomy.
-
Exposure to endocrine disruptors (for example, bisphenol A) disrupts oogenesis at multiple stages and predisposes the oocyte to aneuploidy.
Abstract
Trisomic and monosomic (aneuploid) embryos account for at least 10% of human pregnancies and, for women nearing the end of their reproductive lifespan, the incidence may exceed 50%. The errors that lead to aneuploidy almost always occur in the oocyte but, despite intensive investigation, the underlying molecular basis has remained elusive. Recent studies of humans and model organisms have shed new light on the complexity of meiotic defects, providing evidence that the age-related increase in errors in the human female is not attributable to a single factor but to an interplay between unique features of oogenesis and a host of endogenous and exogenous factors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lejeune, J., Turpin, R. & Gautier, M. Mongolism; a chromosomal disease (trisomy). Bull. Acad. Natl Med. 143, 256–265 (1959).
Jacobs, P. A., Baikie, A. G., Court Brown, W. M. & Strong, J. A. The somatic chromosomes in mongolism. Lancet 1, 710 (1959).
Hassold, T. & Hunt, P. To err (meiotically) is human: the genesis of human aneuploidy. Nature Rev. Genet. 2, 280–291 (2001).
Gaulden, M. E. Maternal age effect: the enigma of Down syndrome and other trisomic conditions. Mutat. Res. 296, 69–88 (1992).
Jacobs, P. A. The chromosome complement of human gametes. Oxf. Rev. Reprod. Biol. 14, 47–72 (1992).
Jamieson, M. E., Coutts, J. R. & Connor, J. M. The chromosome constitution of human preimplantation embryos fertilized in vitro. Hum. Reprod. 9, 709–715 (1994).
Pellestor, F., Andreo, B., Anahory, T. & Hamamah, S. The occurrence of aneuploidy in human: lessons from the cytogenetic studies of human oocytes. Eur. J. Med. Genet. 49, 103–116 (2006).
Pacchierotti, F., Adler, I. D., Eichenlaub-Ritter, U. & Mailhes, J. B. Gender effects on the incidence of aneuploidy in mammalian germ cells. Environ. Res. 104, 46–69 (2007).
Fragouli, E. et al. Comprehensive molecular cytogenetic analysis of the human blastocyst stage. Hum. Reprod. 23, 2596–2608 (2008).
Fragouli, E. et al. The cytogenetics of polar bodies: insights into female meiosis and the diagnosis of aneuploidy. Mol. Hum. Reprod. 17, 286–295 (2011).
Gabriel, A. S. et al. Array comparative genomic hybridisation on first polar bodies suggests that non-disjunction is not the predominant mechanism leading to aneuploidy in humans. J. Med. Genet. 48, 433–437 (2011).
Gutierrez-Mateo, C. et al. Validation of microarray comparative genomic hybridization for comprehensive chromosome analysis of embryos. Fertil. Steril. 95, 953–958 (2011).
Treff, N. R. et al. Single nucleotide polymorphism microarray-based concurrent screening of 24-chromosome aneuploidy and unbalanced translocations in preimplantation human embryos. Fertil. Steril. 95, 1606–1612.e2 (2011).
Treff, N. R. et al. SNP microarray-based 24 chromosome aneuploidy screening is significantly more consistent than FISH. Mol. Hum. Reprod. 16, 583–589 (2010).
Geraedts, J. et al. Polar body array CGH for prediction of the status of the corresponding oocyte. Part I: clinical results. Hum. Reprod. 26, 3173–3180 (2011).
Lamb, N. E. et al. Susceptible chiasmate configurations of chromosome 21 predispose to non-disjunction in both maternal meiosis I and meiosis II. Nature Genet. 14, 400–405 (1996).
Lynn, A. et al. Covariation of synaptonemal complex length and mammalian meiotic exchange rates. Science 296, 2222–2225 (2002).
Cheng, E. Y. et al. Meiotic recombination in human oocytes. PLoS Genet. 5, e1000661 (2009). This is a study of human fetal oocytes that provides evidence that recombination errors occurring during fetal development set the stage for nondisjunction in the adult.
Baudat, F. et al. PRDM9 is a major determinant of meiotic recombination hotspots in humans and mice. Science 327, 836–840 (2010).
Kong, A. et al. Sequence variants in the RNF212 gene associate with genome-wide recombination rate. Science 319, 1398–1401 (2008).
Lynn, A., Ashley, T. & Hassold, T. Variation in human meiotic recombination. Annu. Rev. Genom. Hum. Genet. 5, 317–349 (2004).
O'Connell, M. J., Walworth, N. C. & Carr, A. M. The G2-phase DNA-damage checkpoint. Trends Cell Biol. 10, 296–303 (2000).
Hochwagen, A. & Amon, A. Checking your breaks: surveillance mechanisms of meiotic recombination. Curr. Biol. 16, R217–R228 (2006).
Li, X. C. & Schimenti, J. C. Mouse pachytene checkpoint 2 (trip13) is required for completing meiotic recombination but not synapsis. PLoS Genet. 3, e130 (2007).
Hunt, P. A. & Hassold, T. J. Sex matters in meiosis. Science 296, 2181–2183 (2002).
Celeste, A. et al. Genomic instability in mice lacking histone H2AX. Science 296, 922–927 (2002).
Bannister, L. A. et al. A dominant, recombination-defective allele of Dmc1 causing male-specific sterility. PLoS Biol. 5, e105 (2007).
Kuznetsov, S. et al. RAD51C deficiency in mice results in early prophase I arrest in males and sister chromatid separation at metaphase II in females. J. Cell Biol. 176, 581–592 (2007).
Herran, Y. et al. The cohesin subunit RAD21L functions in meiotic synapsis and exhibits sexual dimorphism in fertility. EMBO J. 30, 3091–3105 (2011).
McKee, B. D. & Handel, M. A. Sex chromosomes, recombination, and chromatin conformation. Chromosoma 102, 71–80 (1993).
Turner, J. M. Meiotic sex chromosome inactivation. Development 134, 1823–1831 (2007).
Burgoyne, P. S., Mahadevaiah, S. K. & Turner, J. M. The consequences of asynapsis for mammalian meiosis. Nature Rev. Genet. 10, 207–216 (2009). This is an informative Review of the meiotic consequences of synaptic defects that emphasizes the mechanisms and consequences of transcriptional silencing of unsynapsed regions.
Cloutier, J. M. & Turner, J. M. Meiotic sex chromosome inactivation. Curr. Biol. 20, R962–963 (2010).
Baarends, W. M. et al. Silencing of unpaired chromatin and histone H2A ubiquitination in mammalian meiosis. Mol. Cell. Biol. 25, 1041–1053 (2005).
Turner, J. M. et al. Silencing of unsynapsed meiotic chromosomes in the mouse. Nature Genet. 37, 41–47 (2005).
Mahadevaiah, S. K. et al. Extensive meiotic asynapsis in mice antagonizes meiotic silencing of unsynapsed chromatin and consequently disrupts meiotic sex chromosome inactivation. J. Cell Biol. 182, 263–276 (2008).
Royo, H. et al. Evidence that meiotic sex chromosome inactivation is essential for male fertility. Curr. Biol. 20, 2117–2123 (2010).
Homolka, D., Jansa, P. & Forejt, J. Genetically enhanced asynapsis of autosomal chromatin promotes transcriptional dysregulation and meiotic failure. Chromosoma 121, 91–104 (2011).
Burgoyne, P. S. & Baker, T. G. Perinatal oocyte loss in XO mice and its implications for the aetiology of gonadal dysgenesis in XO women. J. Reprod. Fertil. 75, 633–645 (1985).
de Boer, P. & de Jong, J. H. in Fertility and Chromosome Pairing: Recent Studies in Plants and Animals (ed. Gilles, C. B.) 77–107 (CRC Press, 1989).
Baudat, F., Manova, K., Yuen, J. P., Jasin, M. & Keeney, S. Chromosome synapsis defects and sexually dimorphic meiotic progression in mice lacking Spo11. Mol. Cell 6, 989–998 (2000).
Hassold, T. J. & Jacobs, P. A. Trisomy in man. Annu. Rev. Genet. 18, 69–97 (1984).
Hunt, P. & Hassold, T. Female meiosis: coming unglued with age. Curr. Biol. 20, R699–702 (2010).
Henderson, S. A. & Edwards, R. G. Chiasma frequency and maternal age in mammals. Nature 218, 22–28 (1968).
Koehler, K. E. et al. Spontaneous X chromosome MI and MII nondisjunction events in Drosophila melanogaster oocytes have different recombinational histories. Nature Genet. 14, 406–414 (1996).
Ross, L. O., Maxfield, R. & Dawson, D. Exchanges are not equally able to enhance meiotic chromosome segregation in yeast. Proc. Natl Acad. Sci. USA 93, 4979–4983 (1996).
Angell, R. R. Predivision in human oocytes at meiosis I: a mechanism for trisomy formation in man. Hum. Genet. 86, 383–387 (1991).
Jeffreys, C. A., Burrage, P. S. & Bickel, S. E. A model system for increased meiotic nondisjunction in older oocytes. Curr. Biol. 13, 498–503 (2003).
Hodges, C. A., Revenkova, E., Jessberger, R., Hassold, T. J. & Hunt, P. A. SMC1β-deficient female mice provide evidence that cohesins are a missing link in age-related nondisjunction. Nature Genet. 37, 1351–1355 (2005).
Liu, L. & Keefe, D. L. Defective cohesin is associated with age-dependent misaligned chromosomes in oocytes. Reprod. Biomed. Online 16, 103–112 (2008).
Chiang, T., Duncan, F. E., Schindler, K., Schultz, R. M. & Lampson, M. A. Evidence that weakened centromere cohesion is a leading cause of age-related aneuploidy in oocytes. Curr. Biol. 20, 1522–1528 (2010).
Lister, L. M. et al. Age-related meiotic segregation errors in Mammalian oocytes are preceded by depletion of cohesin and Sgo2. Curr. Biol. 20, 1511–1521 (2010).
Chiang, T., Schultz, R. M. & Lampson, M. Meiotic origins of maternal age-related aneuploidy. Biol. Reprod. 86, 1–7 (2011).
Revenkova, E., Herrmann, K., Adelfalk, C. & Jessberger, R. Oocyte cohesin expression restricted to predictyate stages provides full fertility and prevents aneuploidy. Curr. Biol. 20, 1529–1533 (2010).
Tachibana-Konwalski, K. et al. Rec8-containing cohesin maintains bivalents without turnover during the growing phase of mouse oocytes. Genes Dev. 24, 2505–2516 (2010). This series of recent papers focuses on meiotic cohesins during oogenesis in the mouse. References 50–53 link loss of cohesin proteins with maternal age-dependent aneuploidy. References 54 and 55 provide evidence that cohesin proteins loaded during fetal development are necessary and sufficient to orchestrate meiotic chromosome segregation in the adult.
Angell, R. R., Xian, J., Keith, J., Ledger, W. & Baird, D. T. First meiotic division abnormalities in human oocytes: mechanism of trisomy formation. Cytogenet. Cell Genet. 65, 194–202 (1994).
Pellestor, F., Andreo, B., Arnal, F., Humeau, C. & Demaille, J. Maternal aging and chromosomal abnormalities: new data drawn from in vitro unfertilized human oocytes. Hum. Genet. 112, 195–203 (2003).
Fisher, J. M., Harvey, J. F., Morton, N. E. & Jacobs, P. A. Trisomy 18: studies of the parent and cell division of origin and the effect of aberrant recombination on nondisjunction. Am. J. Hum. Genet. 56, 669–675 (1995).
Bugge, M. et al. Non-disjunction of chromosome 18. Hum. Mol. Genet. 7, 661–669 (1998).
Hassold, T., Merrill, M., Adkins, K., Freeman, S. & Sherman, S. Recombination and maternal age-dependent nondisjunction: molecular studies of trisomy 16. Am. J. Hum. Genet. 57, 867–874 (1995).
Oliver, T. R. et al. New insights into human nondisjunction of chromosome 21 in oocytes. PLoS Genet. 4, e1000033 (2008).
Bond, D. J. & Chandley, A. C. in Aneuploidy 83–90 (Oxford Univ. Press, 1983).
Garcia-Cruz, R. et al. Dynamics of cohesin proteins REC8, STAG3, SMC1 β and SMC3 are consistent with a role in sister chromatid cohesion during meiosis in human oocytes. Hum. Reprod. 25, 2316–2327 (2010).
Musacchio, A. & Salmon, E. D. The spindle-assembly checkpoint in space and time. Nature Rev. Mol. Cell Biol. 8, 379–393 (2007).
Kot, M. C. & Handel, M. A. Spermatogenesis in XO,Sxr mice: role of the Y chromosome. J. Exp. Zool. 256, 92–105 (1990).
Sutcliffe, M. J., Darling, S. M. & Burgoyne, P. S. Spermatogenesis in XY, XYSxra and XOSxra mice: a quantitative analysis of spermatogenesis throughout puberty. Mol. Reprod. Dev. 30, 81–89 (1991).
LeMaire-Adkins, R., Radke, K. & Hunt, P. A. Lack of checkpoint control at the metaphase/anaphase transition: a mechanism of meiotic nondisjunction in mammalian females. J. Cell Biol. 139, 1611–1619 (1997).
Kouznetsova, A., Lister, L., Nordenskjold, M., Herbert, M. & Hoog, C. Bi-orientation of achiasmatic chromosomes in meiosis I oocytes contributes to aneuploidy in mice. Nature Genet. 39, 966–968 (2007).
LeMaire-Adkins, R. & Hunt, P. A. Nonrandom segregation of the mouse univalent X chromosome: evidence of spindle-mediated meiotic drive. Genetics 156, 775–783 (2000).
Nagaoka, S. I., Hodges, C. A., Albertini, D. F. & Hunt, P. A. Oocyte-specific differences in cell-cycle control create an innate susceptibility to meiotic errors. Curr. Biol. 21, 651–657 (2011).
Gui, L. & Homer, H. Spindle assembly checkpoint signalling is uncoupled from chromosomal position in mouse oocytes. Development 139, 1941–1946 (2012).
Kolano, A., Brunet, S., Silk, A. D., Cleveland, D. W. & Verlhac, M. H. Error prone mammalian female meiosis from silencing the spindle assembly checkpoint without normal interkinetochore tension. Proc. Natl Acad. Sci. USA 2 May 2012 (doi:10.1073/pnas.1204686109).
Lane, S. I. R., Yun, Y. & Jones, K. T. Timing of anaphase promoting complex activation in mouse oocytes is predicted by microtubule–kinetochore attachment, but not by bivalent alignment or tension. Development 139, 1947–1955 (2012). The studies described in references 70–73 provide evidence that the spindle assembly checkpoint mechanism differs in the oocyte and that metaphase alignment of all chromosomes is not a prerequisite for anaphase onset. This difference provides a mechanism whereby various different factors can all lead to aneuploidy.
Steuerwald, N., Cohen, J., Herrera, R. J., Sandalinas, M. & Brenner, C. A. Association between spindle assembly checkpoint expression and maternal age in human oocytes. Mol. Hum. Reprod. 7, 49–55 (2001).
Brunet, S., Pahlavan, G., Taylor, S. & Maro, B. Functionality of the spindle checkpoint during the first meiotic division of mammalian oocytes. Reproduction 126, 443–450 (2003).
Wassmann, K., Niault, T. & Maro, B. Metaphase I arrest upon activation of the Mad2-dependent spindle checkpoint in mouse oocytes. Curr. Biol. 13, 1596–1608 (2003).
Homer, H. A., McDougall, A., Levasseur, M., Murdoch, A. P. & Herbert, M. Mad2 is required for inhibiting securin and cyclin B degradation following spindle depolymerisation in meiosis I mouse oocytes. Reproduction 130, 829–843 (2005).
McGuinness, B. E. et al. Regulation of APC/C activity in oocytes by a Bub1-dependent spindle assembly checkpoint. Curr. Biol. 19, 369–380 (2009).
Woods, L. M. et al. Chromosomal influence on meiotic spindle assembly: abnormal meiosis I in female Mlh1 mutant mice. J. Cell Biol. 145, 1395–1406 (1999).
Yin, H., Cukurcam, S., Betzendahl, I., Adler, I. D. & Eichenlaub-Ritter, U. Trichlorfon exposure, spindle aberrations and nondisjunction in mammalian oocytes. Chromosoma 107, 514–522 (1998).
Hodges, C. A. et al. Experimental evidence that changes in oocyte growth influence meiotic chromosome segregation. Hum. Reprod. 17, 1171–1180 (2002).
Hunt, P. A. et al. Bisphenol a exposure causes meiotic aneuploidy in the female mouse. Curr. Biol. 13, 546–553 (2003).
Jin, F. et al. Cdc20 is critical for meiosis I and fertility of female mice. PLoS Genet 6, e1001147 (2010).
Selesniemi, K., Lee, H. J., Muhlhauser, A. & Tilly, J. L. Prevention of maternal aging-associated oocyte aneuploidy and meiotic spindle defects in mice by dietary and genetic strategies. Proc. Natl Acad. Sci. USA 108, 12319–12324 (2011). This provocative report links caloric restriction with decreased levels of aneuploidy in the ageing female mouse.
Battaglia, D. E., Goodwin, P., Klein, N. A. & Soules, M. R. Influence of maternal age on meiotic spindle assembly in oocytes from naturally cycling women. Hum. Reprod. 11, 2217–2222 (1996).
Volarcik, K. et al. The meiotic competence of in-vitro matured human oocytes is influenced by donor age: evidence that folliculogenesis is compromised in the reproductively aged ovary. Hum. Reprod. 13, 154–160 (1998).
Reis, A. et al. Prometaphase APCcdh1 activity prevents non-disjunction in mammalian oocytes. Nature Cell Biol. 9, 1192–1198 (2007).
Homer, H., Gui, L. & Carroll, J. A spindle assembly checkpoint protein functions in prophase I arrest and prometaphase progression. Science 326, 991–994 (2009).
Can, A., Semiz, O. & Cinar, O. Bisphenol-A induces cell cycle delay and alters centrosome and spindle microtubular organization in oocytes during meiosis. Mol. Hum. Reprod. 11, 389–396 (2005).
Pacchierotti, F., Ranaldi, R., Eichenlaub-Ritter, U., Attia, S. & Adler, I. D. Evaluation of aneugenic effects of bisphenol A in somatic and germ cells of the mouse. Mutat. Res. 651, 64–70 (2008).
Eichenlaub-Ritter, U. et al. Exposure of mouse oocytes to bisphenol A causes meiotic arrest but not aneuploidy. Mutat. Res. 651, 82–92 (2008).
Lenie, S., Cortvrindt, R., Eichenlaub-Ritter, U. & Smitz, J. Continuous exposure to bisphenol A during in vitro follicular development induces meiotic abnormalities. Mutat. Res. 651, 71–81 (2008).
Peretz, J., Gupta, R. K., Singh, J., Hernandez-Ochoa, I. & Flaws, J. A. Bisphenol A impairs follicle growth, inhibits steroidogenesis, and downregulates rate-limiting enzymes in the estradiol biosynthesis pathway. Toxicol. Sci. 119, 209–217 (2011).
Bloom, M. S. et al. Bisphenol A exposure reduces the estradiol response to gonadotropin stimulation during in vitro fertilization. Fertil. Steril. 96, 672–677.e2 (2011).
Fujimoto, V. Y. et al. Serum unconjugated bisphenol A concentrations in women may adversely influence oocyte quality during in vitro fertilization. Fertil. Steril. 95, 1816–1819 (2011).
Susiarjo, M., Hassold, T. J., Freeman, E. & Hunt, P. A. Bisphenol A exposure in utero disrupts early oogenesis in the mouse. PLoS Genet. 3, e5 (2007).
Allard, P. & Colaiacovo, M. P. Bisphenol A impairs the double-strand break repair machinery in the germline and causes chromosome abnormalities. Proc. Natl Acad. Sci. USA 107, 20405–20410 (2010). These studies provide evidence that exposure to the bisphenol A (BPA) disrupts the prophase events of meiosis in the ovaries of mice and worms, setting the stage for nondisjunctional events during the meiotic divisions.
Brieno-Enriquez, M. A. et al. Human meiotic progression and recombination are affected by bisphenol A exposure during in vitro human oocyte development. Hum. Reprod. 26, 2807–2818 (2011).
Doherty, A. S., Mann, M. R., Tremblay, K. D., Bartolomei, M. S. & Schultz, R. M. Differential effects of culture on imprinted H19 expression in the preimplantation mouse embryo. Biol. Reprod. 62, 1526–1535 (2000).
Khosla, S., Dean, W., Brown, D., Reik, W. & Feil, R. Culture of preimplantation mouse embryos affects fetal development and the expression of imprinted genes. Biol. Reprod. 64, 918–926 (2001).
Mann, M. R. et al. Selective loss of imprinting in the placenta following preimplantation development in culture. Development 131, 3727–3735 (2004).
Rivera, R. M. et al. Manipulations of mouse embryos prior to implantation result in aberrant expression of imprinted genes on day 9.5 of development. Hum. Mol. Genet. 17, 1–14 (2008).
Market-Velker, B. A., Zhang, L., Magri, L. S., Bonvissuto, A. C. & Mann, M. R. Dual effects of superovulation: loss of maternal and paternal imprinted methylation in a dose-dependent manner. Hum. Mol. Genet. 19, 36–51 (2010).
Denomme, M. M., Zhang, L. & Mann, M. R. Embryonic imprinting perturbations do not originate from superovulation-induced defects in DNA methylation acquisition. Fertil. Steril. 96, 734–738.e2 (2011).
Harlap, S. et al. Chromosome abnormalities in oral contraceptive breakthrough pregnancies. Lancet 1, 1342–1343 (1979).
Maudlin, I. & Fraser, L. R. The effect of PMSG dose on the incidence of chromosomal anomalies in mouse embryos fertilized in vitro. J. Reprod. Fertil. 50, 275–280 (1977).
Munne, S. et al. Treatment-related chromosome abnormalities in human embryos. Hum. Reprod. 12, 780–784 (1997).
Baart, E. B. et al. Milder ovarian stimulation for in-vitro fertilization reduces aneuploidy in the human preimplantation embryo: a randomized controlled trial. Hum. Reprod. 22, 980–988 (2007).
Rubio, C. et al. Prospective cohort study in high responder oocyte donors using two hormonal stimulation protocols: impact on embryo aneuploidy and development. Hum. Reprod. 25, 2290–2297 (2010).
Penrose, L. in The Early Conceptus, Normal and Abnormal 94–97 (Univ. St Andrews, 1964).
Bond, D. J. & Chandley, A. C. in Aneuploidy 67–75 (Oxford Univ. Press, 1983).
Hubner, K. et al. Derivation of oocytes from mouse embryonic stem cells. Science 300, 1251–1256 (2003).
Toyooka, Y., Tsunekawa, N., Akasu, R. & Noce, T. Embryonic stem cells can form germ cells in vitro. Proc. Natl Acad. Sci. USA 100, 11457–11462 (2003).
Geijsen, N. et al. Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature 427, 148–154 (2004).
Nayernia, K. et al. In vitro-differentiated embryonic stem cells give rise to male gametes that can generate offspring in mice. Dev. Cell 11, 125–132 (2006).
Aflatoonian, B. et al. In vitro post-meiotic germ cell development from human embryonic stem cells. Hum. Reprod. 24, 3150–3159 (2009).
Kee, K., Angeles, V. T., Flores, M., Nguyen, H. N. & Reijo Pera, R. A. Human DAZL, DAZ and BOULE genes modulate primordial germ-cell and haploid gamete formation. Nature 462, 222–225 (2009).
Nicholas, C. R., Haston, K. M., Grewall, A. K., Longacre, T. A. & Reijo Pera, R. A. Transplantation directs oocyte maturation from embryonic stem cells and provides a therapeutic strategy for female infertility. Hum. Mol. Genet. 18, 4376–4389 (2009).
White, Y. A. et al. Oocyte formation by mitotically active germ cells purified from ovaries of reproductive-age women. Nature Med. 18, 413–421 (2012).
The Practice Committee of the Society for Assisted Reproductive Technology, Practice Committee of the American Society for Reproductive Medicine. Preimplantation genetic testing: a Practice Committee opinion. Fertil. Steril. 90, S136–S143 (2008).
Committee on Genetics. ACOG Committee Opinion No. 430: preimplantation genetic screening for aneuploidy. Obstet. Gynecol. 113, 766–767 (2009).
Munne, S., Wells, D. & Cohen, J. Technology requirements for preimplantation genetic diagnosis to improve assisted reproduction outcomes. Fertil. Steril. 94, 408–430 (2010).
Handyside, A. H. PGD and aneuploidy screening for 24 chromosomes by genome-wide SNP analysis: seeing the wood and the trees. Reprod. Biomed. Online 23, 686–691 (2011).
Lamb, N. E. et al. Characterization of susceptible chiasma configurations that increase the risk for maternal nondisjunction of chromosome 21. Hum. Mol. Genet. 6, 1391–1399 (1997).
Hassold, T. et al. Human aneuploidy: incidence, origin, and etiology. Environ. Mol. Mutagen. 28, 167–175 (1996).
Zenzes, M. T. & Casper, R. F. Cytogenetics of human oocytes, zygotes, and embryos after in vitro fertilization. Hum. Genet. 88, 367–375 (1992).
Magli, M. C., Gianaroli, L. & Ferraretti, A. P. Chromosomal abnormalities in embryos. Mol. Cell Endocrinol. 183, S29–S34 (2001).
Staessen, C. et al. Comparison of blastocyst transfer with or without preimplantation genetic diagnosis for aneuploidy screening in couples with advanced maternal age: a prospective randomized controlled trial. Hum. Reprod. 19, 2849–2858 (2004).
Munne, S. et al. Maternal age, morphology, development and chromosome abnormalities in over 6000 cleavage-stage embryos. Reprod. Biomed. Online 14, 628–634 (2007).
Ercelen, N. et al. Successful preimplantation genetic aneuploidy screening in Turkish patients. Genet. Mol. Res. 10, 4093–4103 (2011).
Obradors, A. et al. Whole-chromosome aneuploidy analysis in human oocytes: focus on comparative genomic hybridization. Cytogenet. Genome Res. 133, 119–126 (2011).
Martin, R. H. & Rademaker, A. The frequency of aneuploidy among individual chromosomes in 6,821 human sperm chromosome complements. Cytogenet. Cell Genet. 53, 103–107 (1990).
Martin, R. H., Ko, E. & Rademaker, A. Distribution of aneuploidy in human gametes: comparison between human sperm and oocytes. Am. J. Med. Genet. 39, 321–331 (1991).
Templado, C., Vidal, F. & Estop, A. Aneuploidy in human spermatozoa. Cytogenet. Genome Res. 133, 91–99 (2011).
Acknowledgements
Research conducted in the Hunt and Hassold laboratories and discussed in this Review was supported by US National Institutes of Health grants HD21341 (to T.J.H.) and ES013527 (to P.A.H.). In addition, the authors would gratefully like to acknowledge the three 'grand dames' of human aneuploidy research who sparked our interest and shaped our thinking: D. Warburton, M. Mikkelsen and, most of all, P. Jacobs.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Aneuploidy
-
A chromosome abnormality in which the chromosome number is not a multiple of the haploid number.
- Assisted reproductive technology
-
(ART). Clinical approaches that are used to help infertile couples achieve a normal pregnancy. These include ovarian stimulation protocols using exogenous hormones, in vitro fertilization, intracytoplasmic sperm injection and preimplantation genetic diagnosis.
- Nondisjunction
-
The failure of chromosomes to segregate normally during cell division. Nondisjunction at meiosis I results in products with additional or missing whole chromosomes; nondisjunction at meiosis II results in products with additional or missing sister chromatids.
- Pachytene
-
The stage of meiotic prophase characterized by complete synapsis of all homologues. Importantly, crossover sites can be visualized in pachytene stage cells using appropriate markers.
- Bivalent
-
Paired homologous chromosomes that are tethered by a crossover (or crossovers).
- Synapsis
-
The intimate pairing of homologous chromosomes that occurs during prophase of meiosis and is essential for meiotic recombination. Synapsis is facilitated by the formation of a meiosis-specific protein scaffold called the synaptonemal complex.
- Pseudoautosomal region
-
(PAR). The small region of homology at the distal ends of the X and Y chromosomes that allows for synapsis and recombination.
- Sister chromatid cohesion
-
Replicated chromosomes, or sister chromatids, are held together by cohesin, which is a protein complex that is loaded onto the chromosomes during S phase. In meiosis, sister chromatid cohesion is sequentially released from the chromosome arms at anaphase I and from sister centromeres at anaphase II, allowing for the orderly segregation of homologues and sister chromatids, respectively.
- Univalents
-
Homologous chromosomes that are not associated with one another (for example, owing to failure to recombine).
Rights and permissions
About this article
Cite this article
Nagaoka, S., Hassold, T. & Hunt, P. Human aneuploidy: mechanisms and new insights into an age-old problem. Nat Rev Genet 13, 493–504 (2012). https://doi.org/10.1038/nrg3245
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg3245
This article is cited by
-
Whole transcriptome screening for novel genes involved in meiosis and fertility in Drosophila melanogaster
Scientific Reports (2024)
-
Evaluating the developmental potential of 2.1PN-derived embryos and associated chromosomal analysis
Journal of Assisted Reproduction and Genetics (2024)
-
Association Between Anti-Müllerian Hormone and Early Spontaneous Abortion in Assisted Reproduction Treatment: A Case–Control Study Integrated with Biological Evidence
Reproductive Sciences (2024)
-
Meiotic and mitotic aneuploidies drive arrest of in vitro fertilized human preimplantation embryos
Genome Medicine (2023)
-
Population monitoring of trisomy 21: problems and approaches
Molecular Cytogenetics (2023)