Key Points
-
Reprogramming of the epigenome in mammals occurs in both the germline and the early developing embryo. Some regions of the epigenome appear to escape this process.
-
Parentally imprinted genes are not reprogrammed during early development but are reprogrammed in the germline.
-
A small number of genes escape reprogramming at both stages. This includes some transgenes (that are made up of head-to-tail repeats) and some single-copy genes that are driven off repeated elements, such as retrotransposons.
-
The molecular nature of the molecules involved in this transgenerational epigenetic inheritance via the gametes is under investigation. The prevailing view has been that DNA methylation is the most likely candidate.
-
Here we suggest that RNA that is carried from the mature gametes to the zygote may be the molecule that carries this memory of transcriptional state across generations.
Abstract
It is known that information that is not contained in the DNA sequence — epigenetic information — can be inherited from the parent to the offspring. However, many questions remain unanswered regarding the extent and mechanisms of such inheritance. In this Review, we consider the evidence for transgenerational epigenetic inheritance via the gametes, including cases of environmentally induced epigenetic changes. The molecular basis of this inheritance remains unclear, but recent evidence points towards diffusible factors, in particular RNA, rather than DNA methylation or chromatin. Interestingly, many cases of epigenetic inheritance seem to involve repeat sequences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Peaston, A. E. & Whitelaw, E. Epigenetics and phenotypic variation in mammals. Mamm. Genome 17, 365–374 (2006).
Monk, M., Boubelik, M. & Lehnert, S. Temporal and regional changes in DNA methylation in the embryonic, extraembryonic and germ cell lineages during mouse embryo development. Development 99, 371–382 (1987).
DeChiara, T. M., Robertson, E. J. & Efstratiadis, A. Parental imprinting of the mouse insulin-like growth factor II gene. Cell 64, 849–859 (1991).
Bartolomei, M. S., Webber, A. L., Brunkow, M. E. & Tilghman, S. M. Epigenetic mechanisms underlying the imprinting of the mouse H19 gene. Genes Dev. 7, 1663–1673 (1993).
Bartolomei, M. S., Zemel, S. & Tilghman, S. M. Parental imprinting of the mouse H19 gene. Nature 351, 153–155 (1991).
Gregg, C. et al. High-resolution analysis of parent-of-origin allelic expression in the mouse brain. Science 329, 643–648 (2010).
Gregg, C., Zhang, J., Butler, J. E., Haig, D. & Dulac, C. Sex-specific parent-of-origin allelic expression in the mouse brain. Science 329, 682–685 (2010).
Hadchouel, M., Farza, H., Simon, D., Tiollais, P. & Pourcel, C. Maternal inhibition of hepatitis B surface antigen gene expression in transgenic mice correlates with de novo methylation. Nature 329, 454–456 (1987).
Kearns, M., Preis, J., McDonald, M., Morris, C. & Whitelaw, E. Complex patterns of inheritance of an imprinted murine transgene suggest incomplete germline erasure. Nucleic Acids Res. 28, 3301–3309 (2000).
Allen, N. D., Norris, M. L. & Surani, M. A. Epigenetic control of transgene expression and imprinting by genotype-specific modifiers. Cell 61, 853–861 (1990).
Swain, J. L., Stewart, T. A. & Leder, P. Parental legacy determines methylation and expression of an autosomal transgene: a molecular mechanism for parental imprinting. Cell 50, 719–727 (1987).
Sutherland, H. G. et al. Reactivation of heritably silenced gene expression in mice. Mamm. Genome 11, 347–355 (2000).
Sapienza, C., Peterson, A. C., Rossant, J. & Balling, R. Degree of methylation of transgenes is dependent on gamete of origin. Nature 328, 251–254 (1987).
McGowan, R., Campbell, R., Peterson, A. & Sapienza, C. Cellular mosaicism in the methylation and expression of hemizygous loci in the mouse. Genes Dev. 3, 1669–1676 (1989).
Garrick, D., Fiering, S., Martin, D. I. & Whitelaw, E. Repeat-induced gene silencing in mammals. Nature Genet. 18, 56–59 (1998).
Morgan, H. D., Sutherland, H. G., Martin, D. I. & Whitelaw, E. Epigenetic inheritance at the agouti locus in the mouse. Nature Genet. 23, 314–318 (1999).
Wolff, G. L. Influence of maternal phenotype on metabolic differentiation of agouti locus mutants in the mouse. Genetics 88, 529–539 (1978).
Duhl, D. M., Vrieling, H., Miller, K. A., Wolff, G. L. & Barsh, G. S. Neomorphic agouti mutations in obese yellow mice. Nature Genet. 8, 59–65 (1994).
Dolinoy, D. C., Weinhouse, C., Jones, T. R., Rozek, L. S. & Jirtle, R. L. Variable histone modifications at the Avy metastable epiallele. Epigenetics 5, 637–644 (2010).
Rakyan, V. K. et al. Transgenerational inheritance of epigenetic states at the murine AxinFu allele occurs after maternal and paternal transmission. Proc. Natl Acad. Sci. USA 100, 2538–2543 (2003).
Druker, R., Bruxner, T. J., Lehrbach, N. J. & Whitelaw, E. Complex patterns of transcription at the insertion site of a retrotransposon in the mouse. Nucleic Acids Res. 32, 5800–5808 (2004).
Weinhouse, C. et al. An expression microarray approach for the identification of metastable epialleles in the mouse genome. Epigenetics 6, 1105–1113 (2011).
Rakyan, V. K., Blewitt, M. E., Druker, R., Preis, J. I. & Whitelaw, E. Metastable epialleles in mammals. Trends Genet. 18, 348–351 (2002).
Waterland, R. A. et al. Season of conception in rural Gambia affects DNA methylation at putative human metastable epialleles. PLoS Genet. 6, e1001252 (2010). This paper was the first to identify what are thought to be metastable epialleles in humans.
Roemer, I., Reik, W., Dean, W. & Klose, J. Epigenetic inheritance in the mouse. Curr. Biol. 7, 277–280 (1997).
Reik, W. et al. Adult phenotype in the mouse can be affected by epigenetic events in the early embryo. Development 119, 933–942 (1993).
Wolff, G. L., Kodell, R. L., Moore, S. R. & Cooney, C. A. Maternal epigenetics and methyl supplements affect agouti gene expression in Avy/a mice. FASEB J. 12, 949–957 (1998).
Blewitt, M. E., Vickaryous, N. K., Paldi, A., Koseki, H. & Whitelaw, E. Dynamic reprogramming of DNA methylation at an epigenetically sensitive allele in mice. PLoS Genet. 2, e49 (2006).
Kaminen-Ahola, N. et al. Maternal ethanol consumption alters the epigenotype and the phenotype of offspring in a mouse model. PLoS Genet. 6, e1000811 (2010).
Waterland, R. A. & Jirtle, R. L. Transposable elements: targets for early nutritional effects on epigenetic gene regulation. Mol. Cell. Biol. 23, 5293–5300 (2003).
Cooney, C. A., Dave, A. A. & Wolff, G. L. Maternal methyl supplements in mice affect epigenetic variation and DNA methylation of offspring. J. Nutr. 132, 2393S–2400S (2002).
Dolinoy, D. C., Weidman, J. R., Waterland, R. A. & Jirtle, R. L. Maternal genistein alters coat colour and protects Avy mouse offspring from obesity by modifying the fetal epigenome. Environ. Health Perspect. 114, 567–572 (2006).
Rakyan, V. K., Down, T. A., Balding, D. J. & Beck, S. Epigenome-wide association studies for common human diseases. Nature Rev. Genet. 12, 529–541 (2011).
Fraga, M. F. et al. Epigenetic differences arise during the lifetime of monozygotic twins. Proc. Natl Acad. Sci. USA 102, 10604–10609 (2005).
Oates, N. A. et al. Increased DNA methylation at the AXIN1 gene in a monozygotic twin from a pair discordant for a caudal duplication anomaly. Am. J. Hum. Genet. 79, 155–162 (2006).
Mill, J. et al. Evidence for monozygotic twin (MZ) discordance in methylation level at two CpG sites in the promoter region of the catechol-O-methyltransferase (COMT) gene. Am. J. Med. Genet. B 141, 421–425 (2006).
Baranzini, S. E. et al. Genome, epigenome and RNA sequences of monozygotic twins discordant for multiple sclerosis. Nature 464, 1351–1356 (2010). This is currently the most comprehensive study of the epigenome in monozygotic twins.
Kaminsky, Z. A. et al. DNA methylation profiles in monozygotic and dizygotic twins. Nature Genet. 41, 240–245 (2009).
Francis, D. D. & Meaney, M. J. Maternal care and the development of stress responses. Curr. Opin. Neurobiol. 9, 128–134 (1999).
Weaver, I. C. et al. Epigenetic programming by maternal behaviour. Nature Neurosci. 7, 847–854 (2004).
Youngson, N. A. & Whitelaw, E. Transgenerational epigenetic effects. Annu. Rev. Genomics Hum. Genet. 9, 233–257 (2008).
Anway, M. D., Cupp, A. S., Uzumcu, M. & Skinner, M. K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 308, 1466–1469 (2005).
Guerrero-Bosagna, C., Settles, M., Lucker, B. & Skinner, M. K. Epigenetic transgenerational actions of vinclozolin on promoter regions of the sperm epigenome. PLoS ONE 5, e13100 (2010).
Waterland, R. A., Travisano, M. & Tahiliani, K. G. Diet-induced hypermethylation at agouti viable yellow is not inherited transgenerationally through the female. FASEB J. 21, 3380–3385 (2007).
Cropley, J. E., Suter, C. M., Beckman, K. B. & Martin, D. I. Germ-line epigenetic modification of the murine Avy allele by nutritional supplementation. Proc. Natl Acad. Sci. USA 103, 17308–17312 (2006).
Cropley, J. E., Suter, C. M. & Martin, D. I. Methyl donors change the germline epigenetic state of the Avy allele. FASEB J. 21, 3021; author reply 3021–3022 (2007).
Hales, C. N. & Barker, D. J. Type 2 (non-insulin-dependent) diabetes mellitus: the thrifty phenotype hypothesis. Diabetologia 35, 595–601 (1992).
McMillen, I. C. & Robinson, J. S. Developmental origins of the metabolic syndrome: prediction, plasticity, and programming. Physiol. Rev. 85, 571–633 (2005).
Gluckman, P. D., Hanson, M. A. & Beedle, A. S. Non-genomic transgenerational inheritance of disease risk. Bioessays 29, 145–154 (2007).
Kaati, G., Bygren, L. O. & Edvinsson, S. Cardiovascular and diabetes mortality determined by nutrition during parents' and grandparents' slow growth period. Eur. J. Hum. Genet. 10, 682–688 (2002).
Lumey, L. H., Stein, A. D., Kahn, H. S. & Romijn, J. A. Lipid profiles in middle-aged men and women after famine exposure during gestation: the Dutch Hunger Winter Families Study. Am. J. Clin. Nutr. 89, 1737–1743 (2009).
Pembrey, M. E. et al. Sex-specific, male-line transgenerational responses in humans. Eur. J. Hum. Genet. 14, 159–166 (2006).
Heijmans, B. T. et al. Persistent epigenetic differences associated with prenatal exposure to famine in humans. Proc. Natl Acad. Sci. USA 105, 17046–17049 (2008).
Jimenez-Chillaron, J. C. et al. Intergenerational transmission of glucose intolerance and obesity by in utero undernutrition in mice. Diabetes 58, 460–468 (2009).
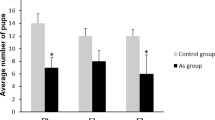
Ng, S. F. et al. Chronic high-fat diet in fathers programs β-cell dysfunction in female rat offspring. Nature 467, 963–966 (2010). This is a carefully executed study revealing transgenerational epigenetic inheritance of phenotype in rats. This is a clear case of inheritance of non-genetic information via the ejaculate.
Carone, B. R. et al. Paternally induced transgenerational environmental reprogramming of metabolic gene expression in mammals. Cell 143, 1084–1096 (2010). This is another study that reveals the inheritance of epigenetic information (in this case, DNA methylation) from sire to offspring.
Lillycrop, K. A. et al. Feeding pregnant rats a protein-restricted diet persistently alters the methylation of specific cytosines in the hepatic PPARα promoter of the offspring. Br. J. Nutr. 100, 278–282 (2008).
Seong, K. H., Li, D., Shimizu, H., Nakamura, R. & Ishii, S. Inheritance of stress-induced, ATF-2-dependent epigenetic change. Cell 145, 1049–1061 (2011). This elegant study uses genetics in D. melanogaster to show that transgenerational memory of an environmental event (heat stress) can occur in trans.
Santos, F., Hendrich, B., Reik, W. & Dean, W. Dynamic reprogramming of DNA methylation in the early mouse embryo. Dev. Biol. 241, 172–182 (2002).
Lane, N. et al. Resistance of IAPs to methylation reprogramming may provide a mechanism for epigenetic inheritance in the mouse. Genesis 35, 88–93 (2003).
Hajkova, P. et al. Epigenetic reprogramming in mouse primordial germ cells. Mech. Dev. 117, 15–23 (2002).
Hajkova, P. et al. Chromatin dynamics during epigenetic reprogramming in the mouse germ line. Nature 452, 877–881 (2008).
Popp, C. et al. Genome-wide erasure of DNA methylation in mouse primordial germ cells is affected by AID deficiency. Nature 463, 1101–1105 (2010).
Borgel, J. et al. Targets and dynamics of promoter DNA methylation during early mouse development. Nature Genet. 42, 1093–1100 (2010).
Chan, T. L. et al. Heritable germline epimutation of MSH2 in a family with hereditary nonpolyposis colorectal cancer. Nature Genet. 38, 1178–1183 (2006).
Ligtenberg, M. J. et al. Heritable somatic methylation and inactivation of MSH2 in families with Lynch syndrome due to deletion of the 3′ exons of TACSTD1. Nature Genet. 41, 112–117 (2009).
Suter, C. M., Martin, D. I. & Ward, R. L. Germline epimutation of MLH1 in individuals with multiple cancers. Nature Genet. 36, 497–501 (2004).
Hitchins, M. P. et al. Inheritance of a cancer-associated MLH1 germ-line epimutation. N. Engl. J. Med. 356, 697–705 (2007).
Chong, S., Youngson, N. A. & Whitelaw, E. Heritable germline epimutation is not the same as transgenerational epigenetic inheritance. Nature Genet. 39, 574–575; author reply 575–576 (2007).
Suter, C. M. & Martin, D. I. Inherited epimutation or a haplotypic basis for the propensity to silence? Nature Genet. 39, 573; author reply 576 (2007).
Hitchins, M. P. & Ward, R. L. Erasure of MLH1 methylation in spermatozoa—implications for epigenetic inheritance. Nature Genet. 39, 1289 (2007).
Goel, A. et al. De novo constitutional MLH1 epimutations confer early-onset colorectal cancer in two new sporadic Lynch syndrome cases, with derivation of the epimutation on the paternal allele in one. Int. J. Cancer 128, 869–878 (2011).
Lister, R. et al. Human DNA methylomes at base resolution show widespread epigenomic differences. Nature 462, 315–322 (2009).
Kriaucionis, S. & Heintz, N. The nuclear DNA base 5-hydroxymethylcytosine is present in Purkinje neurons and the brain. Science 324, 929–930 (2009).
Tahiliani, M. et al. Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 324, 930–935 (2009).
Ito, S. et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333, 1300–1303 (2011).
He, Y. F. et al. Tet-mediated formation of 5-carboxylcytosine and its excision by TDG in mammalian DNA. Science 333, 1303–1307 (2011).
Hammoud, S. S. et al. Distinctive chromatin in human sperm packages genes for embryo development. Nature 460, 473–478 (2009). This paper uses genome-wide methods to show that nucleosomes and histone marks retained in human sperm are enriched at loci important for development.
Brykczynska, U. et al. Repressive and active histone methylation mark distinct promoters in human and mouse spermatozoa. Nature Struct. Mol. Biol. 17, 679–687 (2010). This study shows that promoters with distinct gene function and developmental expression patterns are selectively marked by active and repressive histone methylation in human and mouse sperm. They propose a model in which sperm-inherited nucleosomes are retained during zygotic reprogramming.
Puschendorf, M. et al. PRC1 and Suv39h specify parental asymmetry at constitutive heterochromatin in early mouse embryos. Nature Genet. 40, 411–420 (2008).
Festenstein, R. et al. Modulation of heterochromatin protein 1 dynamics in primary Mammalian cells. Science 299, 719–721 (2003).
Deal, R. B., Henikoff, J. G. & Henikoff, S. Genome-wide kinetics of nucleosome turnover determined by metabolic labelling of histones. Science 328, 1161–1164 (2010).
Henikoff, S. & Shilatifard, A. Histone modification: cause or cog? Trends Genet. 27, 389–396 (2011).
Chandler, V. L. Paramutation's properties and puzzles. Science 330, 628–629 (2010).
Arteaga-Vazquez, M. A. & Chandler, V. L. Paramutation in maize: RNA mediated trans-generational gene silencing. Curr. Opin. Genet. Dev. 20, 156–163 (2010).
Patterson, G. I., Thorpe, C. J. & Chandler, V. L. Paramutation, an allelic interaction, is associated with a stable and heritable reduction of transcription of the maize b regulatory gene. Genetics 135, 881–894 (1993).
Cavalli, G. Chromosome kissing. Curr. Opin. Genet. Dev. 17, 443–450 (2007).
Alleman, M. et al. An RNA-dependent RNA polymerase is required for paramutation in maize. Nature 442, 295–298 (2006).
Sidorenko, L. et al. A dominant mutation in mediator of paramutation2, one of three second-largest subunits of a plant-specific RNA polymerase, disrupts multiple siRNA silencing processes. PLoS Genet. 5, e1000725 (2009).
Stam, M. & Mittelsten Scheid, O. Paramutation: an encounter leaving a lasting impression. Trends Plant Sci. 10, 283–290 (2005).
Chong, S. et al. Modifiers of epigenetic reprogramming show paternal effects in the mouse. Nature Genet. 39, 614–622 (2007).
Yazbek, S. N., Spiezio, S. H., Nadeau, J. H. & Buchner, D. A. Ancestral paternal genotype controls body weight and food intake for multiple generations. Hum. Mol. Genet. 19, 4134–4144 (2010).
Krawetz, S. A. Paternal contribution: new insights and future challenges. Nature Rev. Genet. 6, 633–642 (2005).
Zhao, Y. et al. Characterization and quantification of mRNA transcripts in ejaculated spermatozoa of fertile men by serial analysis of gene expression. Hum. Reprod. 21, 1583–1590 (2006).
Watanabe, T. et al. Identification and characterization of two novel classes of small RNAs in the mouse germline: retrotransposon-derived siRNAs in oocytes and germline small RNAs in testes. Genes Dev. 20, 1732–1743 (2006).
Watanabe, T. et al. Endogenous siRNAs from naturally formed dsRNAs regulate transcripts in mouse oocytes. Nature 453, 539–543 (2008).
Tam, O. H. et al. Pseudogene-derived small interfering RNAs regulate gene expression in mouse oocytes. Nature 453, 534–538 (2008).
Melnyk, C. W., Molnar, A. & Baulcombe, D. C. Intercellular and systemic movement of RNA silencing signals. EMBO J. 30, 3553–3563 (2011).
Slotkin, R. K. et al. Epigenetic reprogramming and small RNA silencing of transposable elements in pollen. Cell 136, 461–472 (2009).
Murchison, E. P. et al. Critical roles for Dicer in the female germline. Genes Dev. 21, 682–693 (2007).
Saito, K. & Siomi, M. C. Small RNA-mediated quiescence of transposable elements in animals. Dev. Cell 19, 687–697 (2010).
Pal-Bhadra, M. et al. Heterochromatic silencing and HP1 localization in Drosophila are dependent on the RNAi machinery. Science 303, 669–672 (2004).
Gan, H. et al. piRNA profiling during specific stages of mouse spermatogenesis. RNA 17, 1191–1203 (2011).
Watanabe, T. et al. Role for piRNAs and noncoding RNA in de novo DNA methylation of the imprinted mouse Rasgrf1 locus. Science 332, 848–852 (2011). This study reveals a role for piRNAs in directing DNA methylation at the imprinted Rasgrf1 locus in the mouse.
Bucheton, A., Paro, R., Sang, H. M., Pelisson, A. & Finnegan, D. J. The molecular basis of I-R. hybrid dysgenesis in Drosophila melanogaster: identification, cloning, and properties of the I factor. Cell 38, 153–163 (1984).
Bregliano, J. C. et al. Hybrid dysgenesis in Drosophila melanogaster. Science 207, 606–611 (1980).
Brennecke, J. et al. An epigenetic role for maternally inherited piRNAs in transposon silencing. Science 322, 1387–1392 (2008).
Hwang, H. W., Wentzel, E. A. & Mendell, J. T. A hexanucleotide element directs microRNA nuclear import. Science 315, 97–100 (2007).
Taft, R. J. et al. Nuclear-localized tiny RNAs are associated with transcription initiation and splice sites in metazoans. Nature Struct. Mol. Biol. 17, 1030–1034 (2010).
Rassoulzadegan, M. et al. RNA-mediated non-Mendelian inheritance of an epigenetic change in the mouse. Nature 441, 469–474 (2006).
Lewis, B. P., Shih, I. H., Jones-Rhoades, M. W., Bartel, D. P. & Burge, C. B. Prediction of mammalian microRNA targets. Cell 115, 787–798 (2003).
Wagner, K. D. et al. RNA induction and inheritance of epigenetic cardiac hypertrophy in the mouse. Dev. Cell 14, 962–969 (2008).
Grandjean, V. et al. The miR-124–Sox9 paramutation: RNA-mediated epigenetic control of embryonic and adult growth. Development 136, 3647–3655 (2009).
Katz, D. J., Edwards, T. M., Reinke, V. & Kelly, W. G. A C. elegans LSD1 demethylase contributes to germline immortality by reprogramming epigenetic memory. Cell 137, 308–320 (2009).
Metivier, R. et al. Cyclical DNA methylation of a transcriptionally active promoter. Nature 452, 45–50 (2008).
Kangaspeska, S. et al. Transient cyclical methylation of promoter DNA. Nature 452, 112–115 (2008).
Scharf, A. N., Barth, T. K. & Imhof, A. Establishment of histone modifications after chromatin assembly. Nucleic Acids Res. 37, 5032–5040 (2009).
Yisraeli, J., Frank, D., Razin, A. & Cedar, H. Effect of in vitro DNA methylation on beta-globin gene expression. Proc. Natl Acad. Sci. USA 85, 4638–4642 (1988).
Montero, L. M. et al. The distribution of 5-methylcytosine in the nuclear genome of plants. Nucleic Acids Res. 20, 3207–3210 (1992).
Ehrlich, M. et al. Amount and distribution of 5-methylcytosine in human DNA from different types of tissues of cells. Nucleic Acids Res. 10, 2709–2721 (1982).
Antequera, F., Tamame, M., Villanueva, J. R. & Santos, T. DNA methylation in the fungi. J. Biol. Chem. 259, 8033–8036 (1984).
Lyko, F. et al. The honey bee epigenomes: differential methylation of brain DNA in queens and workers. PLoS Biol. 8, e1000506 (2010).
Lyko, F., Ramsahoye, B. H. & Jaenisch, R. DNA methylation in Drosophila melanogaster. Nature 408, 538–540 (2000).
Bird, A. P., Taggart, M. H., Nicholls, R. D. & Higgs, D. R. Non-methylated CpG-rich islands at the human alpha-globin locus: implications for evolution of the α-globin pseudogene. EMBO J. 6, 999–1004 (1987).
Smallwood, S. A. et al. Dynamic CpG island methylation landscape in oocytes and preimplantation embryos. Nature Genet. 43, 811–814 (2011).
Kouzminova, E. & Selker, E. U. dim-2 encodes a DNA methyltransferase responsible for all known cytosine methylation in Neurospora. EMBO J. 20, 4309–4323 (2001).
Tamaru, H. & Selker, E. U. A histone H3 methyltransferase controls DNA methylation in Neurospora crassa. Nature 414, 277–283 (2001).
Weber, M. et al. Chromosome-wide and promoter-specific analyses identify sites of differential DNA methylation in normal and transformed human cells. Nature Genet. 37, 853–862 (2005).
Henikoff, S. Position-effect variegation after 60 years. Trends Genet. 6, 422–426 (1990).
Ebert, A., Lein, S., Schotta, G. & Reuter, G. Histone modification and the control of heterochromatic gene silencing in Drosophila. Chromosome Res. 14, 377–392 (2006).
Acknowledgements
E.W. is a National Health and Medical Research Council (NHMRC) Australia Fellow. We thank N. Youngson for helpful discussions and M. Blewitt for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Heterochromatin
-
The portion of the genome that stays highly condensed throughout the cell cycle. It contains a high proportion of repetitive sequences, is gene-poor overall and is enriched for histone marks, such as histone H3 lysine 9 trimethylation (H3K9me3) and H4K20me3, as well as DNA methylation. Heterochromatin is generally associated with gene silencing.
- Histone modifications
-
Covalent alterations of histone tail residues that can alter chromatin structure. Modifications include phosphorylation, methylation, acetylation, sumoylation and ubiquitylation.
- Intracisternal A particle
-
(IAP). A long terminal repeat (LTR)-containing retrotransposon of mice that resembles a retrovirus but has a defective env gene.
- Genistein
-
An isoflavone found in plants that acts as an antioxidant and binds to the oestrogen receptor, hence its classification as a phytoestrogen.
- Hypothalamic–pituitary–adrenal axis
-
(HPA axis). A set of interactions between the hypothalamus, the pituitary gland and the adrenal glands that control reactions to stress.
- Glucocorticoid receptor
-
The receptor to which cortisol and other glucocorticoids bind. It is expressed throughout the body and controls transcription of many genes involved in development, metabolism and the immune response.
- Hippocampus
-
A neurogenic region of the forebrain that has important functions in learning and memory.
- Vinclozolin
-
A fungicide used on vines, fruits and vegetables. It is associated with the development of testicular tumours. There is some evidence that it is carcinogenic and can act as an endrocrine disruptor.
- Peroxisome proliferator-activated receptor alpha
-
(PPARα). A nuclear receptor and transcription factor involved in lipid metabolism.
- Bisulphite sequencing
-
Treatment of DNA with sulphite ions increases the relative resistance of the conversion of methylcytosine to uracil compared with cytosine. PCR amplification and sequencing of the DNA following conversion shows a thymine where a cytosine was located, whereas persistence of a cytosine reflects its methylation in the starting DNA sample.
- Epimutations
-
Mitotically heritable changes in epigenetic state but not gene sequence. Epimutation usually takes place by an abnormal increase or decrease in the methylation status of a gene. The heritability of epimutations across generations is currently under debate.
- MLH1
-
MutL homologue 1, colon cancer nonpolyposis type 2 (Escherichia coli) is a human gene coding for a protein that has an important role in DNA repair.
- MSH2
-
MutS homologue 2, colon cancer nonpolyposis type 2 (Escherichia coli) is another human gene coding for a protein involved in DNA repair.
- Polycomb repressive complex 1
-
(PRC1). Silencing of the homeotic genes in development requires the Polycomb group proteins (PcGs). PcGs form two distinct multiprotein complexes, PRC1 and PRC2.
- MicroRNAs
-
(miRNAs). Evolutionarily conserved small non-coding RNAs (~22-nucleotides long) that silence gene expression by degrading or inhibiting translation of mRNA transcripts in a sequence-specific manner.
- Endogenous small interfering RNAs
-
(endo-siRNAs). Small RNAs that originate, in a Dicer-dependent manner, from long double-stranded (sense–antisense or hairpin) precursors. Initially mainly thought of as a mechanism of host defence against exogenous double-stranded RNA, endo-siRNAs are now known to also regulate endogenous mRNAs in mouse oocytes and Caenorhabditis elegans.
- PIWI-interacting RNAs
-
(piRNAs). Small (24–31 bp) RNAs that are associated with PIWI-clade proteins of the Argonaute family. They ensure genome stability in the germline of flies, mice and zebrafish by silencing transposable and repetitive elements.
- Position effect variegation
-
(PEV). This term describes a type of phenotypic variegation among cells of the same type that is the result of mosaic silencing of a particular gene. The variegation in these cases is due to the position of the gene adjacent to a heterochromatic region of the chromosome.
Rights and permissions
About this article
Cite this article
Daxinger, L., Whitelaw, E. Understanding transgenerational epigenetic inheritance via the gametes in mammals. Nat Rev Genet 13, 153–162 (2012). https://doi.org/10.1038/nrg3188
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg3188
This article is cited by
-
Transferability of Nikita and Sukkula retrotransposons in domestic goose (Anser anser domesticus) genome
Journal of Genetics (2024)
-
Sperm DNA methylation is predominantly stable in mice offspring born after transplantation of long-term cultured spermatogonial stem cells
Clinical Epigenetics (2023)
-
Epigenetic programming for obesity and noncommunicable disease: From womb to tomb
Reviews in Endocrine and Metabolic Disorders (2023)
-
Elevated RIF1 participates in the epigenetic abnormalities of zygotes by regulating histone modifications on MuERV-L in obese mice
Molecular Medicine (2022)
-
The miR-98-3p/JAG1/Notch1 axis mediates the multigenerational inheritance of osteopenia caused by maternal dexamethasone exposure in female rat offspring
Experimental & Molecular Medicine (2022)