Key Points
-
Both in vitro study and transplantation therapy in human patients require large numbers of cells of a desired type. Often, such cells cannot be readily obtained from primary sources (such as patient biopsies or cadavers). As a result, the development of methods to convert plentiful, readily available cell types into the types needed for study and therapy is highly desirable.
-
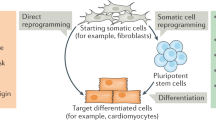
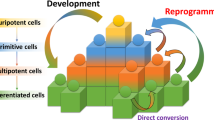
One strategy for producing cells of a desired type is termed 'directed differentiation'. In this process, a pluripotent stem cell, such as an embryonic stem cell or induced pluripotent stem cell, is pushed through a series of cell-fate decisions in order to achieve the desired fate.
-
Cell-fate decisions can be directed in vitro using a variety of methods, including the application of growth factors or small molecules, and through the use of co-culture systems.
-
Another strategy for producing desired cells is to begin with fully differentiated cells of a readily available type, such as fibroblasts, and convert them directly into the desired cell type through a process called 'reprogramming'.
-
A reprogramming strategy usually relies on the overexpression of key transcription factors that can activate the transcriptional programme that leads to the desired cellular phenotype.
-
Cells produced by either reprogramming or directed differentiation must be carefully evaluated and compared with their endogenous counterparts to determine the extent to which the cells produced in vitro are the functional equivalent of those produced in vivo.
-
Both directed-differentiation and reprogramming methods are currently extremely inefficient, and typically generate cells with an immature or embryonic phenotype. These challenges must be overcome in order for cells produced by these methods to achieve their full potential.
-
In spite of these limitations, two Phase I clinical trials on the safety of transplantable cells generated in vitro are currently under way, providing evidence of the promise of the methods described in this Review for research and medicine.
Abstract
Regenerative medicine offers the hope that cells for disease research and therapy might be created from readily available sources. To fulfil this promise, the cells available need to be converted into the desired cell types. We review two main approaches to accomplishing this goal: in vitro directed differentiation, which is used to push pluripotent stem cells, including embryonic stem cells or induced pluripotent stem cells, through steps similar to those that occur during embryonic development; and reprogramming (also known as transdifferentiation), in which a differentiated cell is converted directly into the cell of interest without proceeding through a pluripotent intermediate. We analyse the status of progress made using these strategies and highlight challenges that must be overcome to achieve the goal of cell-replacement therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lengner, C. J. iPS cell technology in regenerative medicine. Ann. N. Y. Acad. Sci. 1192, 38–44 (2010).
Laustriat, D., Gide, J. & Peschanski, M. Human pluripotent stem cells in drug discovery and predictive toxicology. Biochem. Soc. Trans. 38, 1051–1057 (2010).
Park, I. H. et al. Disease-specific induced pluripotent stem cells. Cell 134, 877–886 (2008).
Kiskinis, E. & Eggan, K. Progress toward the clinical application of patient-specific pluripotent stem cells. J. Clin. Invest. 120, 51–59 (2010).
Zhou, Q. & Melton, D. A. Extreme makeover: converting one cell into another. Cell Stem Cell 3, 382–388 (2008).
Vierbuchen, T. et al. Direct conversion of fibroblasts to functional neurons by defined factors. Nature 463, 1035–1041 (2010). This paper was the first to demonstrate the feasibility of producing neurons, a cell type that is highly desirable for both study and therapy, using a reprogramming strategy.
Ieda, M. et al. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell 142, 375–386 (2010). This work demonstrates that fibroblasts, which are a derivative of the ectoderm, can be reprogrammed into a mesodermal derivative, cardiomyocytes.
Szabo, E. et al. Direct conversion of human fibroblasts to multilineage blood progenitors. Nature 468, 521–526 (2010).
Manohar, R. & Lagasse, E. Transdetermination: a new trend in cellular reprogramming. Mol. Ther. 17, 936–938 (2009).
Wagers, A. J. & Weissman, I. L. Plasticity of adult stem cells. Cell 116, 639–648 (2004).
Moustakas, A. & Heldin, C. H. The regulation of TGFβ signal transduction. Development 136, 3699–3714 (2009).
Rao, T. P. & Kuhl, M. An updated overview on Wnt signaling pathways: a prelude for more. Circ. Res. 106, 1798–1806 (2010).
Turner, N. & Grose, R. Fibroblast growth factor signalling: from development to cancer. Nature Rev. Cancer 10, 116–129 (2010).
Borowiak, M. et al. Small molecules efficiently direct endodermal differentiation of mouse and human embryonic stem cells. Cell Stem Cell 4, 348–358 (2009).
Kroon, E. et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nature Biotech. 26, 443–452 (2008).
Oldershaw, R. A. et al. Directed differentiation of human embryonic stem cells toward chondrocytes. Nature Biotech. 28, 1187–1194 (2010).
Yang, L. et al. Human cardiovascular progenitor cells develop from a KDR+ embryonic-stem-cell-derived population. Nature 453, 524–528 (2008).
Lamba, D. A., Karl, M. O., Ware, C. B. & Reh, T. A. Efficient generation of retinal progenitor cells from human embryonic stem cells. Proc. Natl Acad. Sci. USA 103, 12769–12774 (2006).
Oshima, K. et al. Mechanosensitive hair cell-like cells from embryonic and induced pluripotent stem cells. Cell 141, 704–716 (2010). This elegant study used various methods, including growth factors, small molecules and co-culture, to produce an extremely scarce cell type, the hair cell of the ear, by directed differentiation.
D'Amour, K. A. et al. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nature Biotech. 24, 1392–1401 (2006).
Ding, S. & Schultz, P. G. A role for chemistry in stem cell biology. Nature Biotech. 22, 833–840 (2004).
Rubin, L. L. Stem cells and drug discovery: the beginning of a new era? Cell 132, 549–552 (2008).
Frank-Kamenetsky, M. et al. Small-molecule modulators of Hedgehog signaling: identification and characterization of Smoothened agonists and antagonists. J. Biol. 1, 10 (2002). This work identified small-molecule modulators of the Hedgehog pathway, a developmental signalling network that is important to the specification of many tissues. This demonstrates the ability of small molecules to significantly influence cell-fate decisions through a known mechanism.
Wichterle, H., Lieberam, I., Porter, J. A. & Jessell, T. M. Directed differentiation of embryonic stem cells into motor neurons. Cell 110, 385–397 (2002).
Rubin, L. L. & de Sauvage, F. J. Targeting the Hedgehog pathway in cancer. Nature Rev. Drug Discov. 5, 1026–1033 (2006).
Laping, N. J. et al. Inhibition of transforming growth factor (TGF)-β1-induced extracellular matrix with a novel inhibitor of the TGF-β type I receptor kinase activity: SB-431542. Mol. Pharmacol. 62, 58–64 (2002).
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nature Biotech. 27, 275–280 (2009).
Smith, J. R. et al. Inhibition of Activin/Nodal signaling promotes specification of human embryonic stem cells into neuroectoderm. Dev. Biol. 313, 107–117 (2008).
Touboul, T. et al. Generation of functional hepatocytes from human embryonic stem cells under chemically defined conditions that recapitulate liver development. Hepatology 51, 1754–1765 (2010).
Jinnin, M., Ihn, H. & Tamaki, K. Characterization of SIS3, a novel specific inhibitor of Smad3, and its effect on transforming growth factor-β1-induced extracellular matrix expression. Mol. Pharmacol. 69, 597–607 (2006).
Chen, S. et al. A small molecule that directs differentiation of human ESCs into the pancreatic lineage. Nature Chem. Biol. 5, 258–265 (2009). This study demonstrates the efficacy of chemical screening for the identification of compounds that can affect cell fate — in this case, the conversion of endoderm cells into pancreatic progenitors.
Zhu, S. et al. A small molecule primes embryonic stem cells for differentiation. Cell Stem Cell 4, 416–426 (2009).
McLean, A. B. et al. Activin A efficiently specifies definitive endoderm from human embryonic stem cells only when phosphatidylinositol 3-kinase signaling is suppressed. Stem Cells 25, 29–38 (2007).
Takahashi, T. et al. Ascorbic acid enhances differentiation of embryonic stem cells into cardiac myocytes. Circulation 107, 1912–1916 (2003).
Osakada, F. et al. In vitro differentiation of retinal cells from human pluripotent stem cells by small-molecule induction. J. Cell Sci. 122, 3169–3179 (2009).
Dhara, S. K. & Stice, S. L. Neural differentiation of human embryonic stem cells. J. Cell. Biochem. 105, 633–640 (2008).
Ng, E. S., Davis, R. P., Azzola, L., Stanley, E. G. & Elefanty, A. G. Forced aggregation of defined numbers of human embryonic stem cells into embryoid bodies fosters robust, reproducible hematopoietic differentiation. Blood 106, 1601–1603 (2005).
Ying, Q. L., Stavridis, M., Griffiths, D., Li, M. & Smith, A. Conversion of embryonic stem cells into neuroectodermal precursors in adherent monoculture. Nature Biotech. 21, 183–186 (2003).
Di Giorgio, F. P., Boulting, G. L., Bobrowicz, S. & Eggan, K. C. Human embryonic stem cell-derived motor neurons are sensitive to the toxic effect of glial cells carrying an ALS-causing mutation. Cell Stem Cell 3, 637–648 (2008).
Kehat, I. et al. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J. Clin. Invest. 108, 407–414 (2001).
Ikeda, H. et al. Generation of Rx+/Pax6+ neural retinal precursors from embryonic stem cells. Proc. Natl Acad. Sci. USA 102, 11331–11336 (2005).
Arai, A., Yamamoto, K. & Toyama, J. Murine cardiac progenitor cells require visceral embryonic endoderm and primitive streak for terminal differentiation. Dev. Dyn. 210, 344–353 (1997).
Mummery, C. L. et al. Visceral-endoderm-like cell lines induce differentiation of murine P19 embryonal carcinoma cells. Differentiation 46, 51–60 (1991).
Mummery, C. et al. Differentiation of human embryonic stem cells to cardiomyocytes: role of coculture with visceral endoderm-like cells. Circulation 107, 2733–2740 (2003).
Kodama, H. et al. MC3T3-G2/PA6 preadipocytes support in vitro proliferation of hemopoietic stem cells through a mechanism different from that of interleukin 3. J. Cell. Physiol. 129, 20–26 (1986).
Kawasaki, H. et al. Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron 28, 31–40 (2000).
Sun, N., Longaker, M. T. & Wu, J. C. Human iPS cell-based therapy: considerations before clinical applications. Cell Cycle 9, 880–885 (2010).
Ishii, S. et al. Stromal cell-secreted factors promote the survival of embryonic stem cell-derived early neural stem/progenitor cells via the activation of MAPK and PI3K-Akt pathways. J. Neurosci. Res. 88, 722–734 (2010).
Swistowska, A. M. et al. Stage-specific role for shh in dopaminergic differentiation of human embryonic stem cells induced by stromal cells. Stem Cells Dev. 19, 71–82 (2010).
Vazin, T. et al. A novel combination of factors, termed SPIE, which promotes dopaminergic neuron differentiation from human embryonic stem cells. PLoS ONE 4, e6606 (2009).
Kim, D. S. et al. Robust enhancement of neural differentiation from human ES and iPS cells regardless of their innate difference in differentiation propensity. Stem Cell Rev. 6, 270–281 (2010).
Osafune, K. et al. Marked differences in differentiation propensity among human embryonic stem cell lines. Nature Biotech. 26, 313–315 (2008).
Perrier, A. L. et al. Derivation of midbrain dopamine neurons from human embryonic stem cells. Proc. Natl Acad. Sci. USA 101, 12543–12548 (2004).
Chambers, I. et al. Nanog safeguards pluripotency and mediates germline development. Nature 450, 1230–1234 (2007).
Fischer, Y. et al. NANOG reporter cell lines generated by gene targeting in human embryonic stem cells. PLoS ONE 5, e12533 (2010).
Rashid, S. T. et al. Modeling inherited metabolic disorders of the liver using human induced pluripotent stem cells. J. Clin. Invest. 120, 3127–3136 (2010).
Davis, R. L., Weintraub, H. & Lassar, A. B. Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell 51, 987–1000 (1987).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Kondo, M. et al. Cell-fate conversion of lymphoid-committed progenitors by instructive actions of cytokines. Nature 407, 383–386 (2000).
Cobaleda, C., Jochum, W. & Busslinger, M. Conversion of mature B cells into T cells by dedifferentiation to uncommitted progenitors. Nature 449, 473–477 (2007).
Bussmann, L. H. et al. A robust and highly efficient immune cell reprogramming system. Cell Stem Cell 5, 554–566 (2009).
Schafer, B. W., Blakely, B. T., Darlington, G. J. & Blau, H. M. Effect of cell history on response to helix-loop-helix family of myogenic regulators. Nature 344, 454–458 (1990).
Xie, H., Ye, M., Feng, R. & Graf, T. Stepwise reprogramming of B cells into macrophages. Cell 117, 663–676 (2004).
Zhou, Q., Brown, J., Kanarek, A., Rajagopal, J. & Melton, D. A. In vivo reprogramming of adult pancreatic exocrine cells to β-cells. Nature 455, 627–632 (2008). The authors of this paper succeeded in reprogramming pancreatic exocrine cells into insulin-producing β-cells in vivo through the expression of three reprogramming factors.
Collombat, P. et al. Embryonic endocrine pancreas and mature β cells acquire α and PP cell phenotypes upon Arx misexpression. J. Clin. Invest. 117, 961–970 (2007).
Collombat, P. et al. The ectopic expression of Pax4 in the mouse pancreas converts progenitor cells into α and subsequently β cells. Cell 138, 449–462 (2009).
Park, I. H. et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature 451, 141–146 (2008).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Nishimura, K. et al. Development of defective and persistent sendai virus vector: a unique gene delivery/expression system ideal for cell reprogramming. J. Biol. Chem. 286, 4760–4771 (2010).
Stadtfeld, M., Nagaya, M., Utikal, J., Weir, G. & Hochedlinger, K. Induced pluripotent stem cells generated without viral integration. Science 322, 945–949 (2008).
Zhou, W. & Freed, C. R. Adenoviral gene delivery can reprogram human fibroblasts to induced pluripotent stem cells. Stem Cells 27, 2667–2674 (2009).
Kim, D. et al. Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell 4, 472–476 (2009).
Woltjen, K. et al. piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature 458, 766–770 (2009).
Okita, K., Nakagawa, M., Hyenjong, H., Ichisaka, T. & Yamanaka, S. Generation of mouse induced pluripotent stem cells without viral vectors. Science 322, 949–953 (2008).
Si-Tayeb, K. et al. Generation of human induced pluripotent stem cells by simple transient transfection of plasmid DNA encoding reprogramming factors. BMC Dev. Biol. 10, 81 (2010).
Yu, J. et al. Human induced pluripotent stem cells free of vector and transgene sequences. Science 324, 797–801 (2009).
Warren, L. et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7, 618–630 (2010). This paper describes the transfection of modified mRNAs as a means of expressing reprogramming factors in a target cell without permanent genetic modification of the cell. The high efficiency of this method, combined with the potential to make mRNA encoding any reprogramming factor of interest, suggest that this method may gain widespread use for various reprogramming applications.
Huangfu, D. et al. Induction of pluripotent stem cells by defined factors is greatly improved by small-molecule compounds. Nature Biotech. 26, 795–797 (2008).
Huangfu, D. et al. Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nature Biotech. 26, 1269–1275 (2008).
Lyssiotis, C. A. et al. Reprogramming of murine fibroblasts to induced pluripotent stem cells with chemical complementation of Klf4. Proc. Natl Acad. Sci. USA 106, 8912–8917 (2009).
Shi, Y. et al. Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell 3, 568–574 (2008).
Fomina-Yadlin, D. et al. Small-molecule inducers of insulin expression in pancreatic α-cells. Proc. Natl Acad. Sci. USA 107, 15099–15104 (2010).
Idelson, M. et al. Directed differentiation of human embryonic stem cells into functional retinal pigment epithelium cells. Cell Stem Cell 5, 396–408 (2009).
Lamba, D. A., Gust, J. & Reh, T. A. Transplantation of human embryonic stem cell-derived photoreceptors restores some visual function in Crx-deficient mice. Cell Stem Cell 4, 73–79 (2009).
Peljto, M., Dasen, J. S., Mazzoni, E. O., Jessell, T. M. & Wichterle, H. Functional diversity of ESC-derived motor neuron subtypes revealed through intraspinal transplantation. Cell Stem Cell 7, 355–366 (2010).
D'Amour, K. A. et al. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nature Biotech. 23, 1534–1541 (2005).
Acknowledgements
We apologize to authors whose studies could not be cited owing to space limitations. Work in the laboratory of D.M. is funded by the US National Institutes of Health, The Leona M. and Harry B. Helmsley Charitable Trust, the Juvenile Diabetes Research Foundation and the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Directed differentiation
-
The process by which pluripotent stem cells are induced to assume a particular cell fate, through the application of specific culture conditions designed to produce cell-fate changes similar to those observed in the formation of the target cell type in vivo.
- Reprogramming
-
(Also referred to as transdifferentiation.) The direct interconversion of one fully differentiated cell type to another without a pluripotent or multipotent intermediate, often achieved through transcription-factor overexpression.
- Transdetermination
-
A switch in commitment from one lineage to another, closely related lineage that occurs in a multipotent stem or progenitor cell.
- Embryonic stem cells
-
(ESCs). Pluripotent stem cells derived from the inner cell mass of a mammalian embryo.
- Induced pluripotent stem cells
-
(iPSCs). Pluripotent stem cells derived from somatic cells by reprogramming.
- Ectoderm
-
One of the three germ layers formed in early embryonic development; this layer gives rise to tissues including the skin and the nervous system.
- Mesoderm
-
One of the three embryonic germ layers; the mesoderm gives rise to connective tissue, the heart and blood, among other tissue types.
- Endoderm
-
One of the three embryonic germ layers; this layer produces tissues such as the gut, liver, pancreas and lungs.
- Spontaneous differentiation
-
The process by which pluripotent stem cells take on a mixture of cell fates in vitro on transfer from media containing factors that maintain pluripotency to media lacking such factors.
- Embryoid bodies
-
Clusters of pluripotent stem cells, usually grown in suspension culture, that are undergoing spontaneous differentiation.
- Multiplicity of infection
-
The ratio of viral particles present in a transduction experiment divided by the number of target cells present.
Rights and permissions
About this article
Cite this article
Cohen, D., Melton, D. Turning straw into gold: directing cell fate for regenerative medicine. Nat Rev Genet 12, 243–252 (2011). https://doi.org/10.1038/nrg2938
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg2938
This article is cited by
-
Development of a quantitative prediction algorithm for target organ-specific similarity of human pluripotent stem cell-derived organoids and cells
Nature Communications (2021)
-
Stem cells: past, present, and future
Stem Cell Research & Therapy (2019)
-
Cell identity reprogrammed
Nature (2019)
-
Regeneration of a bioengineered 3D integumentary organ system from iPS cells
Nature Protocols (2019)
-
Generation of orthotopically functional salivary gland from embryonic stem cells
Nature Communications (2018)