Key Points
-
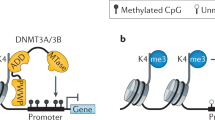
The basic pattern of genomic DNA methylation is established at the time of embryo implantation through a wave of de novo methylation, but CpG islands are protected through a mechanism that involves the recognition of histone H3 lysine 4 methylation.
-
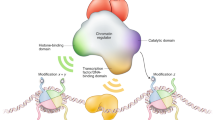
DNA methylation and histone modification influence each other during development. Histone methylation can help to direct DNA methylation patterns, and DNA methylation seems to serve as a template for rebuilding histone modification patterns following DNA replication.
-
Targeted de novo methylation during development involves SET domain-containing proteins that are capable of specifically methylating histones as well as recruiting DNA methyltransferases.
-
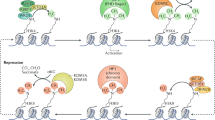
Targeted gene silencing can be mediated by repressor complexes, heterochromatinization and DNA methylation. DNA methylation may be a secondary event that provides long-term stability.
-
During reprogramming of somatic cells, pluripotency genes become reactivated in a process that involves changes in histone modification followed by demethylation of the DNA.
-
De novo methylation in cancer is probably targeted to some genes marked with histone H3 lysine 27 methylation.
Abstract
Both DNA methylation and histone modification are involved in establishing patterns of gene repression during development. Certain forms of histone methylation cause local formation of heterochromatin, which is readily reversible, whereas DNA methylation leads to stable long-term repression. It has recently become apparent that DNA methylation and histone modification pathways can be dependent on one another, and that this crosstalk can be mediated by biochemical interactions between SET domain histone methyltransferases and DNA methyltransferases. Relationships between DNA methylation and histone modification have implications for understanding normal development as well as somatic cell reprogramming and tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kafri, T. et al. Developmental pattern of gene-specific DNA methylation in the mouse embryo and germline. Genes Dev. 6, 705–714 (1992).
Monk, M., Boubelik, M. & Lehnert, S. Temporal and regional changes in DNA methylation in the embryonic, extraembryonic and germ cell lineages during mouse embryo development. Development 99, 371–382 (1987).
Okano, M., Bell, D. W., Haber, D. A. & Li, E. DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99, 247–257 (1999).
Brandeis, M. et al. Sp1 elements protect a CpG island from de novo methylation. Nature 371, 435–438 (1994).
Siegfried, Z. et al. DNA methylation represses transcription in vivo. Nature Genet. 22, 203–206 (1999).
Macleod, D., Charlton, J., Mullins, J. & Bird, A. P. Sp1 sites in the mouse aprt gene promoter are required to prevent methylation of the CpG island. Genes Dev. 8, 2282–2292 (1994).
Frank, D. et al. Demethylation of CpG islands in embryonic cells. Nature 351, 239–241 (1991).
Ooi, S. K. et al. DNMT3L connects unmethylated lysine 4 of histone H3 to de novo methylation of DNA. Nature 448, 714–717 (2007). References 8 and 10 show that DNMT3L interacts with unmethylated H3K4 through its N terminus and with DNMT3A through its C terminus, thus linking the DNA methylation machinery to the modification state of histone tails.
Guenther, M. G., Levine, S. S., Boyer, L. A., Jaenisch, R. & Young, R. A. A chromatin landmark and transcription initiation at most promoters in human cells. Cell 130, 77–88 (2007).
Jia, D., Jurkowska, R. Z., Zhang, X., Jeltsch, A. & Cheng, X. Structure of Dnmt3a bound to Dnmt3L suggests a model for de novo DNA methylation. Nature 449, 248–251 (2007).
Bourc'his, D., Xu, G. L., Lin, C. S., Bollman, B. & Bestor, T. H. Dnmt3L and the establishment of maternal genomic imprints. Science 294, 2536–2539 (2001).
Weber, M. et al. Distribution, silencing potential and evolutionary impact of promoter DNA methylation in the human genome. Nature Genet. 39, 457–466 (2007).
Mohn, F. et al. Lineage-specific polycomb targets and de novo DNA methylation define restriction and potential of neuronal progenitors. Mol. Cell 30, 755–766 (2008). References 13 and 14 present genome-wide DNA methylation maps of pluripotent and differentiated stem cells. They show a link between DNA methylation patterns and histone methylation patterns.
Meissner, A. et al. Genome-scale DNA methylation maps of pluripotent and differentiated cells. Nature 454, 766–770 (2008).
Okitsu, C. Y. & Hsieh, C. L. DNA methylation dictates histone H3K4 methylation. Mol. Cell. Biol. 27, 2746–2757 (2007).
Weber, M. et al. Chromosome-wide and promoter-specific analyses identify sites of differential DNA methylation in normal and transformed human cells. Nature Genet. 37, 853–862 (2005).
Gidekel, S. & Bergman, Y. A unique developmental pattern of Oct-3/4 DNA methylation is controlled by a cis-demodification element. J. Biol. Chem. 277, 34521–34530 (2002).
Sylvester, I. & Scholer, H. R. Regulation of the Oct-4 gene by nuclear receptors. Nucleic Acids Res. 22, 901–911 (1994).
Ben-Shushan, E., Sharir, H., Pikarsky, E. & Bergman, Y. A dynamic balance between ARP-1/COUP-TFII, EAR-3/COUP-TFI, and retinoic acid receptor:retinoid X receptor heterodimers regulates Oct-3/4 expression in embryonal carcinoma cells. Mol. Cell. Biol. 15, 1034–1048 (1995).
Fuhrmann, G. et al. Mouse germline restriction of Oct4 expression by germ cell nuclear factor. Dev. Cell 1, 377–387 (2001).
Feldman, N. et al. G9a-mediated irreversible epigenetic inactivation of Oct-3/4 during early embryogenesis. Nature Cell Biol. 8, 188–194 (2006). References 21 and 22 show that G9a inactivates early embryonic genes. Histone methylation is shown to block target gene reactivation in the absence of repressors, whereas DNA methylation prevents reprogramming.
Epsztejn-Litman, S. et al. De novo DNA methylation promoted by G9a prevents reprogramming of embryonically silenced genes. Nature Struct. Mol. Biol. 15, 1176–1183 (2008).
Lehnertz, B. et al. Suv39h-mediated histone H3 lysine 9 methylation directs DNA methylation to major satellite repeats at pericentric heterochromatin. Curr. Biol. 13, 1192–1200 (2003).
Fuks, F., Hurd, P. J., Deplus, R. & Kouzarides, T. The DNA methyltransferases associate with HP1 and the SUV39H1 histone methyltransferase. Nucleic Acids Res. 31, 2305–2312 (2003).
Sugiyama, T., Cam, H., Verdel, A., Moazed, D. & Grewal, S. I. RNA-dependent RNA polymerase is an essential component of a self-enforcing loop coupling heterochromatin assembly to siRNA production. Proc. Natl. Acad. Sci. USA 102, 152–157 (2005).
Kanellopoulou, C. et al. Dicer-deficient mouse embryonic stem cells are defective in differentiation and centromeric silencing. Genes Dev. 19, 489–501 (2005).
Fukagawa, T. et al. Dicer is essential for formation of the heterochromatin structure in vertebrate cells. Nature Cell Biol. 6, 784–791 (2004).
Malinina, L. Possible involvement of the RNAi pathway in trinucleotide repeat expansion diseases. J. Biomol. Struct. Dyn. 23, 233–235 (2005).
Pandey, R. R. et al. Kcnq1ot1 antisense noncoding RNA mediates lineage-specific transcriptional silencing through chromatin-level regulation. Mol. Cell 32, 232–246 (2008).
Zhao, J., Sun, B. K., Erwin, J. A., Song, J. J. & Lee, J. T. Polycomb proteins targeted by a short repeat RNA to the mouse X chromosome. Science 322, 750–756 (2008).
Nagano, T. et al. The Air noncoding RNA epigenetically silences transcription by targeting G9a to chromatin. Science 322, 1717–1720 (2008).
Tamaru, H. & Selker, E. U. A histone H3 methyltransferase controls DNA methylation in Neurospora crassa. Nature 414, 277–283 (2001).
Jackson, J. P., Lindroth, A. M., Cao, X. & Jacobsen, S. E. Control of CpNpG DNA methylation by the KRYPTONITE histone H3 methyltransferase. Nature 416, 556–560 (2002). References 32 and 33 were the first studies to report crosstalk between histone methylation and DNA methylation in N. crassa and A. thaliana .
Osipovich, O. et al. Targeted inhibition of V(D)J recombination by a histone methyltransferase. Nature Immunol. 5, 309–316 (2004).
Carlson, L. L., Page, A. W. & Bestor, T. H. Properties and localization of DNA methyltransferase in preimplantation mouse embryos: implications for genomic imprinting. Genes Dev. 6, 2536–2541 (1992).
Leonhardt, H., Page, A. W., Weier, H. U. & Bestor, T. H. A targeting sequence directs DNA methyltransferase to sites of DNA replication in mammalian nuclei. Cell 71, 865–873 (1992).
Bostick, M. et al. UHRF1 plays a role in maintaining DNA methylation in mammalian cells. Science 317, 1760–1764 (2007). References 37–39 show that UHRF1 contains an SRA domain that binds to hemimethylated CG sites and forms a complex with DNMT1, thus mediating epigenetic inheritance of DNA methylation.
Sharif, J. et al. The SRA protein Np95 mediates epigenetic inheritance by recruiting Dnmt1 to methylated DNA. Nature 450, 908–912 (2007).
Achour, M. et al. The interaction of the SRA domain of ICBP90 with a novel domain of DNMT1 is involved in the regulation of VEGF gene expression. Oncogene 27, 2187–2197 (2008).
Suzuki, M. M. & Bird, A. DNA methylation landscapes: provocative insights from epigenomics. Nature Rev. Genet. 9, 465–476 (2008).
Weber, M. & Schubeler, D. Genomic patterns of DNA methylation: targets and function of an epigenetic mark. Curr. Opin. Cell Biol. 19, 273–280 (2007).
Eden, S., Hashimshony, T., Keshet, I., Thorne, A. W. & Cedar, H. DNA methylation models histone acetylation. Nature 394, 842–843 (1998).
Hashimshony, T., Zhang, J., Keshet, I., Bustin, M. & Cedar, H. The role of DNA methylation in setting up chromatin structure during development. Nature Genet. 34, 187–192 (2003).
Nan, X. et al. Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393, 386–389 (1998).
Jones, P. L. et al. Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nature Genet. 19, 187–191 (1998).
Esteve, P. O. et al. Direct interaction between DNMT1 and G9a coordinates DNA and histone methylation during replication. Genes Dev. 20, 3089–3103 (2006).
Lande-Diner, L. et al. Role of DNA methylation in stable gene repression. J. Biol. Chem. 282, 12194–12200 (2007).
Zilberman, D., Coleman-Derr, D., Ballinger, T. & Henikoff, S. Histone H2A.Z and DNA methylation are mutually antagonistic chromatin marks. Nature 456, 125–129 (2008).
Tachibana, M., Matsumura, Y., Fukuda, M., Kimura, H. & Shinkai, Y. G9a/GLP complexes independently mediate H3K9 and DNA methylation to silence transcription. EMBO J. 27, 2681–2690 (2008). References 49 and 50 show that G9a promotes DNA methylation of retrotransposons and a number of genes in embryonic stem cells independently of its catalytic activity.
Dong, K. B. et al. DNA methylation in ES cells requires the lysine methyltransferase G9a but not its catalytic activity. EMBO J. 27, 2691–2701 (2008).
Vire, E. et al. The Polycomb group protein EZH2 directly controls DNA methylation. Nature 439, 871–874 (2006).
Li, H. et al. The histone methyltransferase SETDB1 and the DNA methyltransferase DNMT3A interact directly and localize to promoters silenced in cancer cells. J. Biol. Chem. 281, 19489–19500 (2006).
Smallwood, A., Esteve, P. O., Pradhan, S. & Carey, M. Functional cooperation between HP1 and DNMT1 mediates gene silencing. Genes Dev. 21, 1169–1178 (2007).
Freitag, M., Hickey, P. C., Khlafallah, T. K., Read, N. D. & Selker, E. U. HP1 is essential for DNA methylation in Neurospora. Mol. Cell 13, 427–434 (2004).
Lande-Diner, L. & Cedar, H. Silence of the genes — mechanisms of long-term repression. Nature Rev. Genet. 6, 648–654 (2005).
Schoenherr, C. J. & Anderson, D. J. The neuron-restrictive silencer factor (NRSF): a coordinate repressor of multiple neuron-specific genes. Science 267, 1360–1363 (1995).
Franke, A. et al. Polycomb and polyhomeotic are constituents of a multimeric protein complex in chromatin of Drosophila melanogaster. EMBO J. 11, 2941–2950 (1992).
Boyer, L. A. et al. Polycomb complexes repress developmental regulators in murine embryonic stem cells. Nature 441, 349–353 (2006).
Lee, T. I. et al. Control of developmental regulators by polycomb in human embryonic stem cells. Cell 125, 301–313 (2006).
Hansen, K. H. et al. A model for transmission of the H3K27me3 epigenetic mark. Nature Cell Biol. 10, 1291–1300 (2008). The authors suggest a mechanism by which H3K27me3 is propagated during the cell division cycle. Once H3K27me3 is established it recruits the PRC2 complex, leading to methylation of histone H3 on the newly synthesized DNA.
Hajkova, P. et al. Chromatin dynamics during epigenetic reprogramming in the mouse germ line. Nature 452, 877–881 (2008). This study examines the erasure of parental imprints in mouse primordial germ cells during embryogenesis. The data suggest that DNA demethylation occurs prior to histone replacement, thus supporting a repair model for demethylation.
Schwartz, Y. B. & Pirrotta, V. Polycomb complexes and epigenetic states. Curr. Opin. Cell Biol. 20, 266–273 (2008).
Pietersen, A. M. & van Lohuizen, M. Stem cell regulation by polycomb repressors: postponing commitment. Curr. Opin. Cell Biol. 20, 201–207 (2008).
Bernstein, B. E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006).
Mikkelsen, T. S. et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448, 553–560 (2007).
Pan, G. et al. Whole-genome analysis of histone H3 lysine 4 and lysine 27 methylation in human embryonic stem cells. Cell Stem Cell 1, 299–312 (2007).
Zhao, X. D. et al. Whole-genome mapping of histone H3 Lys4 and 27 trimethylations reveals distinct genomic compartments in human embryonic stem cells. Cell Stem Cell 1, 286–298 (2007).
Barski, A. et al. High-resolution profiling of histone methylations in the human genome. Cell 129, 823–837 (2007).
Hershko, A. Y., Kafri, T., Fainsod, A. & Razin, A. Methylation of HoxA5 and HoxB5 and its relevance to expression during mouse development. Gene 302, 65–72 (2003).
Payer, B. & Lee, J. T. X chromosome dosage compensation: how mammals keep the balance. Annu. Rev. Genet. 42, 733–772 (2008).
Lock, L. F., Takagi, N. & Martin, G. R. Methylation of the Hprt gene on the inactive X occurs after chromosome inactivation. Cell 48, 39–46 (1987).
Samollow, P. B., Robinson, E. S., Ford, A. L. & Vandeberg, J. L. Developmental progression of Gpd expression from the inactive X chromosome of the virginia opossum. Dev. Genet. 16, 367–378 (1995).
Migeon, B. R., Jan de Beur, S. & Axelman, J. Frequent derepression of G6PD and HPRT on the marsupial inactive X chromosome associated with cell proliferation in vitro. Exp. Cell Res. 182, 597–609 (1989).
Gautsch, J. W. & Wilson, M. C. Delayed de novo methylation in teratocarcinoma suggests additional tissue-specific mechanisms for controlling gene expression. Nature 301, 32–37 (1983).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006). This is the first report showing the generation of iPS cells by introduction of four transcription factor genes into somatic cells.
Maherali, N. et al. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell 1, 55–70 (2007).
Wernig, M. et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448, 318–324 (2007).
Welstead, G. G., Schorderet, P. & Boyer, L. A. The reprogramming language of pluripotency. Curr. Opin. Genet. Dev. 18, 123–129 (2008).
Mikkelsen, T. S. et al. Dissecting direct reprogramming through integrative genomic analysis. Nature 454, 49–55 (2008). This paper shows that partially reprogrammed cell lines have DNA hypermethylation at pluripotency-related loci. This suggests that DNA demethylation is an inefficient step accomplished late in the transition to pluripotency.
Shi, Y. et al. A combined chemical and genetic approach for the generation of induced pluripotent stem cells. Cell Stem Cell 2, 525–528 (2008).
Shi, Y. et al. Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell 3, 568–574 (2008).
Ma, D. K., Chiang, C. H., Ponnusamy, K., Ming, G. L. & Song, H. G9a and Jhdm2a regulate embryonic stem cell fusion-induced reprogramming of adult neural stem cells. Stem Cells 26, 2131–2141 (2008).
Jones, P. A. & Baylin, S. B. The epigenomics of cancer. Cell 128, 683–692 (2007).
De Marzo, A. M. et al. Abnormal regulation of DNA methyltransferase expression during colorectal carcinogenesis. Cancer Res. 59, 3855–3860 (1999).
Robertson, K. D. et al. The human DNA methyltransferases (DNMTs) 1, 3a and 3b: coordinate mRNA expression in normal tissues and overexpression in tumors. Nucleic Acids Res. 27, 2291–2298 (1999).
Keshet, I. et al. Evidence for an instructive mechanism of de novo methylation in cancer cells. Nature Genet. 38, 149–153 (2006).
Schlesinger, Y. et al. Polycomb mediated histone H3(K27) methylation pre-marks genes for de novo methylation in cancer. Nature Genet. 39, 232–236 (2007). References 87–89 show that in cancer cells a large number of CpG islands marked by H3K27me3 undergo de novo methylation, indicating that Polycomb-directed de novo methylation might play an important part in carcinogenesis.
Ohm, J. E. et al. A stem cell-like chromatin pattern may predispose tumor suppressor genes to DNA hypermethylation and heritable silencing. Nature Genet. 39, 237–242 (2007).
Widschwendter, M. et al. Epigenetic stem cell signature in cancer. Nature Genet. 39, 157–158 (2007).
Varambally, S. et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 419, 624–629 (2002).
Varambally, S. et al. Genomic loss of microRNA-101 leads to overexpression of histone methyltransferase EZH2 in cancer. Science 322, 1695–1699 (2008).
Benetti, R. et al. A mammalian microRNA cluster controls DNA methylation and telomere recombination via Rbl2-dependent regulation of DNA methyltransferases. Nature Struct. Mol. Biol. 15, 268–279 (2008).
Sinkkonen, L. et al. MicroRNAs control de novo DNA methylation through regulation of transcriptional repressors in mouse embryonic stem cells. Nature Struct. Mol. Biol. 15, 259–267 (2008).
Gal-Yam, E. N. et al. Frequent switching of Polycomb repressive marks and DNA hypermethylation in the PC3 prostate cancer cell line. Proc. Natl. Acad. Sci. USA 105, 12979–12984 (2008).
McGarvey, K. M., Greene, E., Fahrner, J. A., Jenuwein, T. & Baylin, S. B. DNA methylation and complete transcriptional silencing of cancer genes persist after depletion of EZH2. Cancer Res. 67, 5097–5102 (2007).
Lindroth, A. M. et al. Antagonism between DNA and H3K27 methylation at the imprinted Rasgrf1 locus. PLoS Genet. 4, e1000145 (2008).
Zhao, Q. et al. PRMT5-mediated methylation of histone H4R3 recruits DNMT3A, coupling histone and DNA methylation in gene silencing. Nature Struct. Mol. Biol. 16, 304–311 (2009).
Wang, J. et al. The lysine demethylase LSD1 (KDM1) is required for maintenance of global DNA methylation. Nature Genet. 41, 125–129 (2009).
Goldmit, M. et al. Epigenetic ontogeny of the κ locus during B cell development. Nature Immunol. 6, 198–203 (2005).
Loh, Y. H., Zhang, W., Chen, X., George, J. & Ng, H. H. Jmjd1a and Jmjd2c histone H3 Lys 9 demethylases regulate self-renewal in embryonic stem cells. Genes Dev. 21, 2545–2557 (2007).
Lan, F., Nottke, A. C. & Shi, Y. Mechanisms involved in the regulation of histone lysine demethylases. Curr. Opin. Cell Biol. 20, 316–325 (2008).
Agger, K., Christensen, J., Cloos, P. A. & Helin, K. The emerging functions of histone demethylases. Curr. Opin. Genet. Dev. 18, 159–168 (2008).
Paroush, Z., Keshet, I., Yisraeli, J. & Cedar, H. Dynamics of demethylation and activation of the α actin gene in myoblasts. Cell 63, 1229–1237 (1990).
Barreto, G. et al. Gadd45a promotes epigenetic gene activation by repair-mediated DNA demethylation. Nature 445, 671–675 (2007). References 104, 105, 107 and 108 show that active DNA demethylation might be accomplished through DNA repair promoted by GADD45.
Rai, K. et al. DNA Demethylation in zebrafish involves the coupling of a deaminase, a glycosylase, and Gadd45. Cell 135, 1201–1212 (2008).
Weiss, A., Keshet, I., Razin, A. & Cedar, H. DNA demethylation in vitro: involvement of RNA. Cell 86, 709–718 (1996).
Schmmitz, K. M. et al. TAF12 recruits Gadd45a and the nucleotide excision repair complex to the promoter of rRNA genes leading to active DNA demethylation. Mol. Cell 33, 344–353 (2009).
Ma, D. K. et al. Neuronal sctivity-induced Gadd45b promotes epigenetic DNA demethylation and adult neurogenesis. Science 323, 1074–1077 (2009).
Acknowledgements
This work was supported by grants from the Israel Academy of Science (Y.B. and H.C.), the National Institutes of Health (Y.B. and H.C.), the Israel Cancer Research Fund (Y.B. and H.C.) and Lew Sherman (H.C.).
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Histone
-
Protein component of chromatin that is involved in regulation of gene expression. Two of each of the core histones, H2A, H2B, H3 or H4, make up an octameric nucleosome, around which DNA winds. N-terminal tails of histones can be subject to covalent modification, including methylation and acetylation.
- CpG island
-
A sequence of at least 200 bp with a greater number of CpG sites than expected given the average GC content of the genome. These regions are typically undermethylated and are found upstream of many mammalian genes.
- Chromodomain
-
Initially identified in the Drosophila melanogaster heterochromatin protein 1 and Polycomb proteins, this is an ∼50 amino acid, highly conserved domain that binds to histone tails that are methylated at certain lysine residues. Different classes of chromodomains have been implicated in binding histones, RNA and DNA.
- Heterochromatin protein 1
-
(HP1). Conserved component of silent heterochromatic regions, which contains a chromodomain that binds nucleosomes containing histone H3 that is methylated on lysine 9.
- Heterochromatin
-
Highly compacted regions of chromatin, in which transcription is repressed. Constitutive heterochromatin is a common feature of highly repetitive DNA sequences.
- Satellite repeat
-
DNA that contains many tandem repeats of a short basic repeating unit. Both the major and minor satellite repeats are located at pericentromeric heterochromatin.
- SET domain
-
An evolutionarily conserved sequence motif that was initially identified in the Drosophila melanogaster position effect variegation suppressor Su(var)3–9, the Polycomb-group protein Enhancer of zeste, and Trithorax (a Trithorax group protein). It is present in many histone methyltransferases and is required for enzyme activity.
- Dicer
-
An RNA endonuclease that cleaves double-stranded RNA into small interfering RNAs of approximately 21 bp.
- RNA-induced silencing complex
-
(RISC). A complex made up of an Argonaute protein and small RNA, which inhibits translation of target RNAs through degradative or non-degradative mechanisms.
- Imprinted locus
-
A locus at which the expression of an allele is different depending on whether it is inherited from the mother or the father.
- X chromosome inactivation
-
The process that occurs in female mammals by which gene expression from one of the two X chromosomes is downregulated to match the levels of gene expression from the single X chromosome that is present in males. Inactivation involves changes in DNA methylation and histone modifications.
- Chromatin immunoprecipitation
-
(ChIP). A technique that is used to analyse the genomic location of DNA-associated proteins that involves crosslinking DNA–protein complexes then immunoprecipitation using an antibody against a protein of interest. This is followed by analysis of the recovered DNA sequences.
- Polycomb repressive complex
-
(PRC). A group of repressive chromatin proteins that maintain states of gene expression throughout development. Originally identified in Drosophila melanogaster as genes in which mutations caused homeotic transformations.
Rights and permissions
About this article
Cite this article
Cedar, H., Bergman, Y. Linking DNA methylation and histone modification: patterns and paradigms. Nat Rev Genet 10, 295–304 (2009). https://doi.org/10.1038/nrg2540
Issue Date:
DOI: https://doi.org/10.1038/nrg2540
This article is cited by
-
DNA demethylation and tri-methylation of H3K4 at the TACSTD2 promoter are complementary players for TROP2 regulation in colorectal cancer cells
Scientific Reports (2024)
-
DNA methylation of GDF-9 and GHR genes as epigenetic regulator of milk production in Egyptian Zaraibi goat
Genes & Genomics (2024)
-
Cognitive Dysfunction and Exercise: From Epigenetic to Genetic Molecular Mechanisms
Molecular Neurobiology (2024)
-
Transgenerational Epigenetic Inheritance of Cardiovascular Diseases: A Network Medicine Perspective
Maternal and Child Health Journal (2024)
-
Multilayered regulation of secondary metabolism in medicinal plants
Molecular Horticulture (2023)