Key Points
-
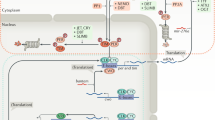
Daily rhythms in biochemical, cellular and behavioural activities are controlled by the biological clock, which consists of one or more endogenous oscillators.
-
The clock exerts its effects on a wide variety of processes, ranging from development in fungi, cell division in the marine protist Gonyaulax polyedra, photosynthesis in plants, sleep in animals, to cognitive functions in humans. Although circadian rhythms are present in different organisms, several aspects of the clock mechanism, and its complexity, are not conserved among these organisms.
-
In prokaryotic and eukaryotic microorganisms, the circadian clock system seems to consist of several oscillators. These oscillators might respond to different environmental signals and direct rhythms in specific genes and behaviours. The coupling of oscillators is thought to provide stability and precision to the timing mechanism.
-
In eukaryotes with differentiated tissues, a network of cell autonomous oscillators is found not within a single cell, but among cells in different tissue types. The clock system in eukaryotes also regulates rhythms in diverse biological processes, but these rhythms can be specific to different tissue types.
-
In mammals and birds, a circadian pacemaker in the brain responds to input from the environment and coordinates overt rhythmicity throughout the peripheral tissues.
-
In mammals, lesions and metabolic and electrophysiological studies have provided incontrovertible evidence that the SCN of the hypothalamus serves as the master circadian pacemaker. This pacemaker can coordinate rhythmicity in downstream cells and tissues.
-
In non-mammalian vertebrates, the circadian clock system seems to be more complex. In birds, the circadian system consists of at least three anatomically distinct circadian pacemakers; the retina, the pineal gland and an avian homologue of the mammalian SCN.
-
In Drosophila melanogaster, the hierarchal model of a central pacemaker setting the time of peripheral oscillators does not hold; virtually all tissues harbour circadian oscillators that can be entrained directly by light.
-
So, the need for a centralized pacemaker to entrain peripheral oscillators in organisms seems to be dependent on the ability of peripheral tissues to be directly entrained.
Abstract
The organization of biological activities into daily cycles is universal in organisms as diverse as cyanobacteria, fungi, algae, plants, flies, birds and man. Comparisons of circadian clocks in unicellular and multicellular organisms using molecular genetics and genomics have provided new insights into the mechanisms and complexity of clock systems. Whereas unicellular organisms require stand-alone clocks that can generate 24-hour rhythms for diverse processes, organisms with differentiated tissues can partition clock function to generate and coordinate different rhythms. In both cases, the temporal coordination of a multi-oscillator system is essential for producing robust circadian rhythms of gene expression and biological activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Reppert, S. M. & Weaver, D. R. Coordination of circadian timing in mammals. Nature 418, 935–941 (2002).
Young, M. W. & Kay, S. A. Time zones: a comparative genetics of circadian clocks. Nature Rev. Genet. 2, 702–715 (2001).
Hardin, P. E. Transcription regulation within the circadian clock: the E-box and beyond. J. Biol. Rhythms 19, 348–360 (2004).
Hastings, M. H. & Herzog, E. D. Clock genes, oscillators, and cellular networks in the suprachiasmatic nuclei. J. Biol. Rhythms 19, 400–413 (2004).
Dunlap, J. C. & Loros, J. J. The Neurospora circadian system. J. Biol. Rhythms 19, 414–424 (2004).
Panda, S., Hogenesch, J. B. & Kay, S. A. Circadian rhythms from flies to human. Nature 417, 329–335 (2002).
Harms, E., Kivimae, S., Young, M. W. & Saez, L. Posttranscriptional and posttranslational regulation of clock genes. J. Biol. Rhythms 19, 361–373 (2004).
Salome, P. A. & McClung, C. R. The Arabidopsis thaliana clock. J. Biol. Rhythms 19, 425–435 (2004).
Iwasaki, H. & Kondo, T. Circadian timing mechanism in the prokaryotic clock system of cyanobacteria. J. Biol. Rhythms 19, 436–444 (2004).
Lakin-Thomas, P. L. & Brody, S. Circadian rhythms in microorganisms: new complexities. Annu. Rev. Microbiol. 58, 489–519 (2004).
Michel, S., Geusz, M. E., Zaritsky, J. J. & Block, G. D. Circadian rhythm in membrane conductance expressed in isolated neurons. Science 259, 239–241 (1993).
Welsh, D. K., Logothetis, D. E., Meister, M. & Reppert, S. M. Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 14, 697–706 (1995).
Plautz, J. D., Kaneko, M., Hall, J. C. & Kay, S. A. Independent photoreceptive circadian clocks throughout Drosophila. Science 278, 1632–1635 (1997).
Giebultowicz, J. M., Stanewsky, R., Hall, J. C. & Hege, D. M. Transplanted Drosophila excretory tubules maintain circadian clock cycling out of phase with the host. Curr. Biol. 10, 107–110 (2000).
Balsalobre, A., Damiola, F. & Schibler, U. A serum shock induces circadian gene expression in mammalian tissue culture cells. Cell 93, 929–937 (1998).
Welsh, D. K., Yoo, S. H., Liu, A. C., Takahashi, J. S. & Kay, S. A. Bioluminescence imaging of individual fibroblasts reveals persistent, independently phased circadian rhythms of clock gene expression. Curr. Biol. 14, 2289–2295 (2004).
Herzog, E. D., Takahashi, J. S. & Block, G. D. Clock controls circadian period in isolated suprachiasmatic nucleus neurons. Nature Neurosci. 1, 708–713 (1998).
Quintero, J. E., Kuhlman, S. J. & McMahon, D. G. The biological clock nucleus: a multiphasic oscillator network regulated by light. J. Neurosci. 23, 8070–8076 (2003).
Yoo, S. H. et al. PERIOD2::LUCIFERASE real-time reporting of circadian dynamics reveals persistent circadian oscillations in mouse peripheral tissues. Proc. Natl Acad. Sci. USA 101, 5339–5346 (2004). The authors demonstrate that peripheral tissues maintain circadian rhythms of gene expression in SCN-lesioned mice, but lose synchrony of phase among the tissues of individual animals and between animals.
Yamazaki, S. et al. Resetting central and peripheral circadian oscillators in transgenic rats. Science 288, 682–685 (2000). This study led to the hypothesis that a self-sustained circadian pacemaker in the SCN entrains circadian oscillators in the periphery to maintain adaptive phase control.
Abe, M. et al. Circadian rhythms in isolated brain regions. J. Neurosci. 22, 350–356 (2002).
Tosini, G. & Menaker, M. Circadian rhythms in cultured mammalian retina. Science 272, 419–421 (1996).
Besharse, J. C. & Iuvone, P. M. Circadian clock in Xenopus eye controlling retinal serotonin N-acetyltransferase. Nature 305, 133–135 (1983).
Deguchi, T. A circadian oscillator in cultured cells of chicken pineal gland. Nature 282, 94–96 (1979).
Green, C. B., Cahill, G. M. & Besharse, J. C. Regulation of tryptophan hydroxylase expression by a retinal circadian oscillator in vitro. Brain Res. 677, 283–290 (1995).
Underwood, H. & Groos, G. Vertebrate circadian rhythms: retinal and extraretinal photoreception. Experientia 38, 1013–1021 (1982).
Greve, P. et al. Serotonin N-acetyltransferase mRNA levels in photoreceptor-enriched chicken retinal cell cultures: elevation by cyclic AMP. J. Neurochem. 73, 1894–1900 (1999).
Binkley, S. A., Riebman, J. B. & Reilly, K. B. The pineal gland: a biological clock in vitro. Science 202, 1198–1120 (1978).
Kasal, C. A., Menaker, M. & Perez-Polo, J. R. Circadian clock in culture: N-acetyltransferase activity of chick pineal glands oscillates in vitro. Science 203, 656–658 (1979).
Whitmore, D., Foulkes, N. S., Strahle, U. & Sassone-Corsi, P. Zebrafish Clock rhythmic expression reveals independent peripheral circadian oscillators. Nature Neurosci. 1, 701–707 (1998).
Cahill, G. M. Circadian regulation of melatonin production in cultured zebrafish pineal and retina. Brain Res. 708, 177–181 (1996).
Zimmerman, N. H. & Menaker, M. The pineal gland: a pacemaker within the circadian system of the house sparrow. Proc. Natl Acad. Sci. USA 76, 999–1003 (1979).
Robertson, L. M. & Takahashi, J. S. Circadian clock in cell culture: I. Oscillation of melatonin release from dissociated chick pineal cells in flow-through microcarrier culture. J. Neurosci. 8, 12–21 (1988).
Tosini, G. & Menaker, M. Multioscillatory circadian organization in a vertebrate, Iguana iguana. J. Neurosci. 18, 1105–1114 (1998).
Dunlap, J. C., Loros, J. J., Liu, Y. & Crosthwaite, S. K. Eukaryotic circadian systems: cycles in common. Genes Cells 4, 1–10 (1999).
Hall, J. C. Genetics of biological rhythms in Drosophila. Adv. Genet. 38, 135–184 (1998).
Harmer, S. L., Panda, S. & Kay, S. A. Molecular bases of circadian rhythms. Annu. Rev. Cell Dev. Biol. 17, 215–253 (2001).
King, D. P. & Takahashi, J. S. Molecular genetics of circadian rhythms in mammals. Annu. Rev. Neurosci. 23, 713–742 (2000).
Johnson, C. H. Endogenous timekeepers in photosynthetic organisms. Annu. Rev. Physiol. 63, 695–728 (2001).
Ditty, J. L., Williams, S. B. & Golden, S. S. A cyanobacterial circadian timing mechanism. Annu. Rev. Genet. 37, 513–543 (2003).
Tomita, J., Nakajima, M., Kondo, T. & Iwasaki, H. No transcription-translation feedback in circadian rhythm of KaiC phosphorylation. Science 307, 251–254 (2004).
Duffield, G. E. DNA microarray analyses of circadian timing: the genomic basis of biological time. J. Neuroendocrinol. 15, 991–1002 (2003).
Lowrey, P. L. & Takahashi, J. S. Mammalian circadian biology: elucidating genome-wide levels of temporal organization. Annu. Rev. Genomics Hum. Genet. 5, 407–441 (2004).
Hastings, M. H., Reddy, A. B. & Maywood, E. S. A clockwork web: circadian timing in brain and periphery, in health and disease. Nature Rev. Neurosci. 4, 649–661 (2003).
Panda, S. & Hogenesch, J. B. It's all in the timing: many clocks, many outputs. J. Biol. Rhythms 19, 374–387 (2004).
Ueda, H. R. et al. Genome-wide transcriptional orchestration of circadian rhythms in Drosophila. J. Biol. Chem. 277, 14048–14052 (2002).
Correa, A. et al. Multiple oscillators regulate circadian gene expression in Neurospora. Proc. Natl Acad. Sci. USA 100, 13597–13602 (2003). The authors identify CCGs that maintain circadian rhythmicity in the absence of the FRQ/WC oscillator, confirming the existence of a second oscillator in N. crassa cells that is involved in regulating rhythmic gene expression.
McDonald, M. J. & Rosbash, M. Microarray analysis and organization of circadian gene expression in Drosophila. Cell 107, 567–578 (2001).
Claridge-Chang, A. et al. Circadian regulation of gene expression systems in the Drosophila head. Neuron 32, 657–671 (2001).
Shimomura, K. et al. Genome-wide epistatic interaction analysis reveals complex genetic determinants of circadian behavior in mice. Genome Res. 11, 959–980 (2001).
Morse, D., Hastings, J. W. & Roenneberg, T. Different phase responses of the two circadian oscillators in Gonyaulax. J. Biol. Rhythms 9, 263–274 (1994).
Pittendrigh, C. S. Temporal organization: reflections of a Darwinian clock-watcher. Annu. Rev. Physiol. 55, 16–54 (1993). A must-read for any student of circadian biology.
Golden, S. S. & Canales, S. R. Cyanobacterial circadian clocks — timing is everything. Nature Rev. Microbiol. 1, 191–199 (2003).
Johnson, C. H. Precise circadian clocks in prokaryotic cyanobacteria. Curr. Issues Mol. Biol. 6, 103–110 (2004).
Kondo, T. et al. Circadian rhythms in rapidly dividing cyanobacteria. Science 275, 224–227 (1997).
Ishiura, M. et al. Expression of a gene cluster kaiABC as a circadian feedback process in cyanobacteria. Science 281, 1519–1523 (1998).
Nakajima, M. et al. Reconstitution of circadian oscillation of cyanobacterial KaiC phosphorylation in vitro. Science 308, 414–415 (2005). The authors show that a mixture of KaiA, KaiB, KaiC and ATP can reconstitute a temperature-compensated circadian rhythm of KaiC phosphorylation in vitro.
Mori, T. & Johnson, C. H. Circadian programming in cyanobacteria. Semin. Cell Dev. Biol. 12, 271–278 (2001).
Liu, Y. et al. Circadian orchestration of gene expression in cyanobacteria. Genes Dev. 9, 1469–1478 (1995).
Ivleva, N. B., Bramlett, M. R., Lindahl, P. A. & Golden, S. S. LdpA: a component of the circadian clock senses redox state of the cell. EMBO J. (2005).
Nakahira, Y. et al. Global gene repression by KaiC as a master process of prokaryotic circadian system. Proc. Natl Acad. Sci. USA 101, 881–885 (2004).
Nair, U., Ditty, J. L., Min, H. & Golden, S. S. Roles for sigma factors in global circadian regulation of the cyanobacterial genome. J. Bacteriol. 184, 3530–3538 (2002). This paper shows that separate timing circuits with different periods coexist in cyanobacterial cells.
Mihalcescu, I., Hsing, W. & Leibler, S. Resilient circadian oscillator revealed in individual cyanobacteria. Nature 430, 81–85 (2004).
Aronson, B. D., Johnson, K. A., Loros, J. J. & Dunlap, J. C. Negative feedback defining a circadian clock: autoregulation of the clock gene frequency. Science 263, 1578–1584 (1994).
Crosthwaite, S. K., Loros, J. J. & Dunlap, J. C. Light-induced resetting of a circadian clock is mediated by a rapid increase in frequency transcript. Cell 81, 1003–1012 (1995).
Liu, Y., Merrow, M., Loros, J. J. & Dunlap, J. C. How temperature changes reset a circadian oscillator. Science 281, 825–829 (1998).
Loros, J. J. & Dunlap, J. C. Genetic and molecular analysis of circadian rhythms in Neurospora. Annu. Rev. Physiol. 63, 757–794 (2001).
Loros, J. J. & Feldman, J. F. Loss of temperature compensation of circadian period length in the frq-9 mutant of Neurospora crassa. J. Biol. Rhythms 1, 187–198 (1986).
Aronson, B. D., Johnson, K. A. & Dunlap, J. C. Circadian clock locus frequency: protein encoded by a single open reading frame defines period length and temperature compensation. Proc. Natl Acad. Sci. USA 91, 7683–7687 (1994).
Granshaw, T., Tsukamoto, M. & Brody, S. Circadian rhythms in Neurospora crassa: farnesol or geraniol allow expression of rhythmicity in the otherwise arrhythmic strains frq10, wc-1, and wc-2. J. Biol. Rhythms 18, 287–296 (2003).
Merrow, M., Brunner, M. & Roenneberg, T. Assignment of circadian function for the Neurospora clock gene frequency. Nature 399, 584–586 (1999).
Pregueiro, A. M. et al. Assignment of an essential role for the Neurospora frequency gene in circadian entrainment to temperature cycles. Proc. Natl Acad. Sci. USA 102, 2210–2215 (2005).
Christensen, M. K. et al. A nitrate-induced frq-less oscillator in Neurospora crassa. J. Biol. Rhythms 19, 280–286 (2004).
Ramsdale, M. & Lakin-Thomas, P. L. SN-1,2-diacylglycerol levels in the fungus Neurospora crassa display circadian rhythmicity. J. Biol. Chem. 275, 27541–27550 (2000).
Nowrousian, M., Duffield, G. E., Loros, J. J. & Dunlap, J. C. The frequency gene is required for temperature-dependent regulation of many clock-controlled genes in Neurospora crassa. Genetics 164, 923–933 (2003).
Turek, F. W. Circadian neural rhythms in mammals. Annu. Rev. Physiol. 47, 49–64 (1985).
Ralph, M. R., Foster, R. G., Davis, F. C. & Menaker, M. Transplanted suprachiasmatic nucleus determines circadian period. Science 247, 975–978 (1990). The authors show that the period of behavioural rhythms in rats is determined by cells of the SCN.
Klein, D., Moore, R. & Reppert, S. Suprachiasmatic Nucleus: the Mind's Clock (Oxford Univ. Press, New York, 1991).
Johnson, R. F., Moore, R. Y. & Morin, L. P. Loss of entrainment and anatomical plasticity after lesions of the hamster retinohypothalamic tract. Brain Res. 460, 297–313 (1988).
Sato, T. K. et al. A functional genomics strategy reveals RORa as a component of the mammalian circadian clock. Neuron 43, 527–537 (2004).
Preitner, N. et al. The orphan nuclear receptor REV-ERBα controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell 110, 251–260 (2002).
Shearman, L. P., Zylka, M. J., Weaver, D. R., Kolakowski, L. F., Jr & Reppert, S. M. Two period homologs: circadian expression and photic regulation in the suprachiasmatic nuclei. Neuron 19, 1261–1269 (1997).
Zylka, M. J., Shearman, L. P., Weaver, D. R. & Reppert, S. M. Three period homologs in mammals: differential light responses in the suprachiasmatic circadian clock and oscillating transcripts outside of brain. Neuron 20, 1103–1110 (1998). These are the first experiments to show that circadian oscillators in mammals exist in tissues outside the brain and retina.
Balsalobre, A. et al. Resetting of circadian time in peripheral tissues by glucocorticoid signaling. Science 289, 2344–2347 (2000).
Brandstaetter, R. Circadian lessons from peripheral clocks: is the time of the mammalian pacemaker up? Proc. Natl Acad. Sci. USA 101, 5699–5700 (2004).
Sujino, M. et al. Suprachiasmatic nucleus grafts restore circadian behavioural rhythms of genetically arrhythmic mice. Curr. Biol. 13, 664–668 (2003).
Ralph, M. R. & Lehman, M. N. Transplantation: a new tool in the analysis of the mammalian hypothalamic circadian pacemaker. Trends Neurosci. 14, 362–366 (1991).
Earnest, D. J. et al. Establishment and characterization of adenoviral E1A immortalized cell lines derived from the rat suprachiasmatic nucleus. J. Neurobiol. 39, 1–13 (1999).
Earnest, D. J., Liang, F. Q., Ratcliff, M. & Cassone, V. M. Immortal time: circadian clock properties of rat suprachiasmatic cell lines. Science 283, 693–695 (1999).
Allen, G., Rappe, J., Earnest, D. J. & Cassone, V. M. Oscillating on borrowed time: diffusible signals from immortalized suprachiasmatic nucleus cells regulate circadian rhythmicity in cultured fibroblasts. J. Neurosci. 21, 7937–7943 (2001). The capacity to generate circadian rhythms endogenously and to confer this rhythmicity to other cells was compared in immortalized cells derived from the SCN and a fibroblast line. This allowed differentiation between SCN pacemaker properties and the oscillatory behaviour of non-clock tissues.
Jin, X. et al. A molecular mechanism regulating rhythmic output from the suprachiasmatic circadian clock. Cell 96, 57–68 (1999).
Damiola, F. et al. Restricted feeding uncouples circadian oscillators in peripheral tissues from the central pacemaker in the suprachiasmatic nucleus. Genes Dev. 14, 2950–2961 (2000).
Stokkan, K. A., Yamazaki, S., Tei, H., Sakaki, Y. & Menaker, M. Entrainment of the circadian clock in the liver by feeding. Science 291, 490–493 (2001).
Schibler, U., Ripperger, J. & Brown, S. A. Peripheral circadian oscillators in mammals: time and food. J. Biol. Rhythms 18, 250–260 (2003).
Cheng, M. Y. et al. Prokineticin 2 transmits the behavioural circadian rhythm of the suprachiasmatic nucleus. Nature 417, 405–410 (2002).
Kramer, A. et al. Regulation of daily locomotor activity and sleep by hypothalamic EGF receptor signaling. Science 294, 2511–2515 (2001).
Bailey, M. J. et al. Transcriptional profiling of the chick pineal gland, a photoreceptive circadian oscillator and pacemaker. Mol. Endocrinol. 17, 2084–2095 (2003).
Bailey, M. J. et al. Transcriptional profiling of circadian patterns of mRNA expression in the chick retina. J. Biol. Chem. 279, 52247–52254 (2004).
Sato, T. K., Panda, S., Kay, S. A. & Hogenesch, J. B. DNA arrays: applications and implications for circadian biology. J. Biol. Rhythms 18, 96–105 (2003).
Hastings, M. H. et al. Expression of clock gene products in the suprachiasmatic nucleus in relation to circadian behaviour. Novartis Found. Symp. 253, 203–217 (2003).
Etter, P. D. & Ramaswami, M. The ups and downs of daily life: profiling circadian gene expression in Drosophila. Bioessays 24, 494–498 (2002).
Lin, Y. et al. Influence of the period-dependent circadian clock on diurnal, circadian, and aperiodic gene expression in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 99, 9562–9567 (2002).
Menger, G. J., Lu, K., Thomas, T., Cassone, V. M. & Earnest, D. J. Circadian profiling of the transcriptome in immortalized rat SCN cells. Physiol. Genomics (in the press).
Storch, K. F. et al. Extensive and divergent circadian gene expression in liver and heart. Nature 417, 78–83 (2002).
Cassone, V. M. & Moore, R. Y. Retinohypothalamic projection and suprachiasmatic nucleus of the house sparrow, Passer domesticus. J. Comp. Neurol. 266, 171–182 (1987).
Brandstatter, R. & Abraham, U. Hypothalamic circadian organization in birds. I. Anatomy, functional morphology, and terminology of the suprachiasmatic region. Chronobiol. Int. 20, 637–655 (2003).
Gwinner, E. & Brandstatter, R. Complex bird clocks. Philos. Trans. R. Soc. Lond. B 356, 1801–1810 (2001).
Cassone, V. M. & Menaker, M. Is the avian circadian system a neuroendocrine loop? J. Exp. Zool. 232, 539–549 (1984). This paper provides evidence for the neuroendocrine loop model in birds.
Gwinner, E. Melatonin in the Circadian System of Birds: Model of Internal Resonance (eds Hiroshige, T. & Homma, K.) (Hokkaido Univ. Press, Sapporo, Japan, 1989).
Silver, R., LeSauter, J., Tresco, P. A. & Lehman, M. N. A diffusible coupling signal from the transplanted suprachiasmatic nucleus controlling circadian locomotor rhythms. Nature 382, 810–813 (1996).
Cassone, V. M., Forsyth, A. M. & Woodlee, G. L. Hypothalamic regulation of circadian noradrenergic input to the chick pineal gland. J. Comp. Physiol. A 167, 187–192 (1990).
Cassone, V. M., Takahashi, J. S., Blaha, C. D., Lane, R. F. & Menaker, M. Dynamics of noradrenergic circadian input to the chicken pineal gland. Brain Res. 384, 334–341 (1986).
Chong, N. W., Chaurasia, S. S., Haque, R., Klein, D. C. & Iuvone, P. M. Temporal-spatial characterization of chicken clock genes: circadian expression in retina, pineal gland, and peripheral tissues. J. Neurochem. 85, 851–860 (2003).
Fu, Z., Inaba, M., Noguchi, T. & Kato, H. Molecular cloning and circadian regulation of cryptochrome genes in Japanese quail (Coturnix coturnix japonica). J. Biol. Rhythms 17, 14–27 (2002).
Yoshimura, T. et al. Molecular analysis of avian circadian clock genes. Brain Res. Mol. Brain Res. 78, 207–215 (2000).
Bailey, M. J., Chong, N. W., Xiong, J. & Cassone, V. M. Chickens' Cry2: molecular analysis of an avian cryptochrome in retinal and pineal photoreceptors. FEBS Lett. 513, 169–174 (2002).
Cassone, V. M. Melatonin's role in vertebrate circadian rhythms. Chronobiol. Int. 15, 457–473 (1998).
Abraham, U., Albrecht, U. & Brandstatter, R. Hypothalamic circadian organization in birds. II. Clock gene expression. Chronobiol. Int. 20, 657–669 (2003).
Yasuo, S. et al. Effect of melatonin administration on qPer2, qPer3, and qClock gene expression in the suprachiasmatic nucleus of Japanese quail. Eur. J. Neurosci. 16, 1541–1546 (2002).
Abraham, U., Albrecht, U., Gwinner, E. & Brandstatter, R. Spatial and temporal variation of Passer Per2 gene expression in two distinct cell groups of the suprachiasmatic hypothalamus in the house sparrow (Passer domesticus). Eur. J. Neurosci. 16, 429–436 (2002).
Menaker, M., Moreira, L. F. & Tosini, G. Evolution of circadian organization in vertebrates. Braz. J. Med. Biol. Res. 30, 305–313 (1997).
Cassone, V. M. Melatonin: time in a bottle. Oxf. Rev. Reprod. Biol. 12, 319–367 (1990).
Cassone, V. M. & Natesan, A. K. Time and time again: the phylogeny of melatonin as a transducer of biological time. J. Biol. Rhythms 12, 489–497 (1997).
Goldsmith, T. H. Optimization, constraint, and history in the evolution of eyes. Q. Rev. Biol. 65, 281–322 (1990).
Brocchieri, L. Phylogenetic inferences from molecular sequences: review and critique. Theor. Popul. Biol. 59, 27–40 (2001).
Glossop, N. R., Lyons, L. C. & Hardin, P. E. Interlocked feedback loops within the Drosophila circadian oscillator. Science 286, 766–768 (1999).
Glossop, N. R. et al. VRILLE feeds back to control circadian transcription of Clock in the Drosophila circadian oscillator. Neuron 37, 249–261 (2003).
Cyran, S. A. et al. vrille, Pdp1, and dClock form a second feedback loop in the Drosophila circadian clock. Cell 112, 329–341 (2003).
Darlington, T. K. et al. Closing the circadian loop: CLOCK-induced transcription of its own inhibitors per and tim. Science 280, 1599–1603 (1998).
Lee, C., Bae, K. & Edery, I. The Drosophila CLOCK protein undergoes daily rhythms in abundance, phosphorylation, and interactions with the PER–TIM complex. Neuron 21, 857–867 (1998).
Lee, C., Bae, K. & Edery, I. PER and TIM inhibit the DNA binding activity of a Drosophila CLOCK–CYC/dBMAL1 heterodimer without disrupting formation of the heterodimer: a basis for circadian transcription. Mol. Cell. Biol. 19, 5316–5325 (1999).
Hall, J. C. Genetics and molecular biology of rhythms in Drosophila and other insects. Adv. Genet. 48, 1–280 (2003).
Glossop, N. R. & Hardin, P. E. Central and peripheral circadian oscillator mechanisms in flies and mammals. J. Cell Sci. 115, 3369–3377 (2002).
Grima, B., Chelot, E., Xia, R. & Rouyer, F. Morning and evening peaks of activity rely on different clock neurons of the Drosophila brain. Nature 431, 869–873 (2004).
Frisch, B., Hardin, P. E., Hamblen-Coyle, M. J., Rosbash, M. & Hall, J. C. A promoterless period gene mediates behavioral rhythmicity and cyclical per expression in a restricted subset of the Drosophila nervous system. Neuron 12, 555–570 (1994).
Renn, S. C., Park, J. H., Rosbash, M., Hall, J. C. & Taghert, P. H. A pdf neuropeptide gene mutation and ablation of PDF neurons each cause severe abnormalities of behavioral circadian rhythms in Drosophila. Cell 99, 791–802 (1999).
Krishnan, B., Dryer, S. E. & Hardin, P. E. Circadian rhythms in olfactory responses of Drosophila melanogaster. Nature 400, 375–378 (1999).
Tanoue, S., Krishnan, P., Krishnan, B., Dryer, S. E. & Hardin, P. E. Circadian clocks in antennal neurons are necessary and sufficient for olfaction rhythms in Drosophila. Curr. Biol. 14, 638–649 (2004). This is a demonstration that antennal neurons are both necessary and sufficient for olfaction rhythms, showing for the first time that a peripheral tissue can function as an autonomous pacemaker in D. melanogaster.
Hege, D. M., Stanewsky, R., Hall, J. C. & Giebultowicz, J. M. Rhythmic expression of a PER-reporter in the Malpighian tubules of decapitated Drosophila: evidence for a brain-independent circadian clock. J. Biol. Rhythms 12, 300–308 (1997).
Stoleru, D., Peng, Y., Agosto, J. & Rosbash, M. Coupled oscillators control morning and evening locomotor behaviour of Drosophila. Nature 431, 862–868 (2004).
Peng, Y., Stoleru, D., Levine, J. D., Hall, J. C. & Rosbash, M. Drosophila free-running rhythms require intercellular communication. PLoS Biol. 1, e13 (2003).
Whitmore, D. et al. A clockwork organ. Biol. Chem. 381, 793–800 (2000).
Thain, S. C., Murtas, G., Lynn, J. R., McGrath, R. B. & Millar, A. J. The circadian clock that controls gene expression in Arabidopsis is tissue specific. Plant Physiol. 130, 102–110 (2002).
Acknowledgements
The authors are members of the Center for Research on Biological Clocks at Texas A&M University, Houston, USA. The work described in this review from the authors' laboratories was funded by the US National Institutes of Health, including an NINDS Program Project Grant and an NIEHS Center Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- CLOCK
-
A circadian clock is a 24-hour timing mechanism that is composed of molecular oscillators.
- CIRCADIAN RHYTHM
-
A biological rhythm with a ∼24-hour period that persists in constant conditions.
- OSCILLATOR
-
A system of components that interact to produce a rhythm with a definable period length. A circadian oscillator can drive a rhythmic output, but requires other oscillators (pacemakers) for its entrainment and/or function. A circadian oscillator can therefore be self-sustained, but cannot operate properly independently of other oscillators.
- ENTRAINMENT
-
The process by which an environmental rhythm, such as the light–dark cycle, regulates the period and phase relationship of a self-sustained oscillator.
- PACEMAKER
-
An oscillator that drives an output and/or entrains another oscillator. A circadian pacemaker is a specialized oscillator that operates independently of other oscillators to drive rhythmic outputs, either directly or through other oscillators, and is entrained by environmental cues.
- SUPRACHIASMATIC NUCLEUS
-
A small region of the brain that sits on top of the optic chiasm in the anteroventral region of the hypothalamus. Each of the bilaterally paired nuclei that comprise the SCN contains 8,000–10,000 cells packed together.
- PERIOD
-
The time after which a defined phase of an oscillation (such as a peak or trough) recurs.
- NUCLEOID
-
A structure in a prokaryotic cell that is composed of chromosomal DNA and its associated chromatin-like scaffolding proteins.
- PHASE
-
The instantaneous state of an oscillation relative to a reference point.
- SUBJECTIVE MIDDAY
-
The portion of a circadian day that is spent in constant darkness, which corresponds to the midday phase of a light–dark cycle.
- GROUP 2 SIGMA FACTORS
-
Members of a family of sigma factor proteins that are responsible for conferring promoter-specific contacts on the RNA polymerase enzyme of eubacteria, thereby allowing specific genes to be transcribed.
- CONIDIOSPORES
-
Asexually produced haploid fungal spores that are formed on a specialized aerial hypha — the conidiophore — that rises above the substratum.
- HUMORAL
-
Pertaining to elements that are dissolved in the blood or body fluid, typically serum.
- HYPOTHALAMUS
-
The part of the brain that lies below the thalamus, forming the main portion of the ventral region of the diencephalon and functioning to regulate bodily temperature, certain metabolic processes and other autonomic activities.
- SYMPATHETIC NERVOUS SYSTEM
-
Refers to a part of the autonomic nervous system that generally has excitatory function and regulates heart rate and blood pressure.
- OPSINS
-
A family of visual pigments.
- MALPHIGHIAN TUBULES
-
The part of an insect's gastrointestinal tract that excretes nitrogenous waste and maintains ionic balance.
Rights and permissions
About this article
Cite this article
Bell-Pedersen, D., Cassone, V., Earnest, D. et al. Circadian rhythms from multiple oscillators: lessons from diverse organisms. Nat Rev Genet 6, 544–556 (2005). https://doi.org/10.1038/nrg1633
Issue Date:
DOI: https://doi.org/10.1038/nrg1633
This article is cited by
-
Generation of CRISPR-Cas9-mediated knockin mutant models in mice and MEFs for studies of polymorphism in clock genes
Scientific Reports (2023)
-
A synthetic population-level oscillator in non-microfluidic environments
Communications Biology (2023)
-
Phenotype Design Space Provides a Mechanistic Framework Relating Molecular Parameters to Phenotype Diversity Available for Selection
Journal of Molecular Evolution (2023)
-
The current situations and limitations of genetic engineering in cyanobacteria: a mini review
Molecular Biology Reports (2023)
-
Identification of the Relationship Between DNA Methylation of Circadian Rhythm Genes and Obesity
Biochemical Genetics (2023)