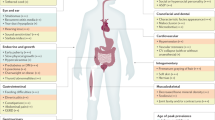
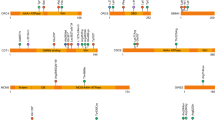
Key Points
-
The completion of the high-quality nucleotide sequence of chromosome 21 now provides the basis for the molecular analysis of trisomy 21.
-
Trisomy 21 is the prototype of all syndromes with triplicated genomic material, and provides ample opportunities to study gene dosage imbalance.
-
Comparative and functional genomics will uncover most of the functional genomic regions of the human genome, which in turn provides the basis for understanding the molecular pathophysiology of trisomies.
-
Genomic variability might contribute to the phenotypic variability of trisomy 21.
-
Mouse models with partial trisomy are now providing fundamental knowledge for trisomy 21 and will be useful for developing potential treatments.
-
There are numerous (non-mutually exclusive) molecular mechanisms for phenotypic consequences of protein dosage imbalance. Therefore, a considerable number of genes could contribute directly or indirectly to the Down syndrome phenotypes.
-
Functional genomic elements other than protein coding genes might be involved in the molecular pathogenesis of the phenotypic features of trisomy 21.
Abstract
The sequence of chromosome 21 was a turning point for the understanding of Down syndrome. Comparative genomics is beginning to identify the functional components of the chromosome and that in turn will set the stage for the functional characterization of the sequences. Animal models combined with genome-wide analytical methods have proved indispensable for unravelling the mysteries of gene dosage imbalance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hsu, L. Y. in Genetic Disorders and the Fetus (ed. Milunsky, A.) 179–248 (Johns Hopkins Univ. Press, Baltimore, 1998).
Bailey, J. A. et al. Recent segmental duplications in the human genome. Science 297, 1003–1007 (2002).
Pentao, L., Wise, C. A., Chinault, A. C., Patel, P. I. & Lupski, J. R. Charcot–Marie–Tooth type 1A duplication appears to arise from recombination at repeat sequences flanking the 1.5 Mb monomer unit. Nature Genet. 2, 292–300 (1992).
de Mollerat, X. J. et al. A genomic rearrangement resulting in a tandem duplication is associated with split hand-split foot malformation 3 (SHFM3) at 10q24. Hum. Mol. Genet. 12, 1959–1971 (2003).
Snijders, A. M. et al. Assembly of microarrays for genome-wide measurement of DNA copy number. Nature Genet. 29, 263–264 (2001).
Ellis, D. & Malcolm, S. Proteolipid protein gene dosage effect in Pelizaeus–Merzbacher disease. Nature Genet. 6, 333–4 (1994).
Inoue, K. & Lupski, J. R. Molecular mechanisms for genomic disorders. Annu. Rev. Genomics Hum. Genet. 3, 199–242 (2002).
Readhead, C., Schneider, A., Griffiths, I. & Nave, K. A. Premature arrest of myelin formation in transgenic mice with increased proteolipid protein gene dosage. Neuron 12, 583–595 (1994).
Magyar, J. P. et al. Impaired differentiation of Schwann cells in transgenic mice with increased PMP22 gene dosage. J. Neurosci. 16, 5351–5360 (1996).
Lettice, L. A. et al. A long-range Shh enhancer regulates expression in the developing limb and fin and is associated with preaxial polydactyly. Hum. Mol. Genet. 12, 1725–1735 (2003).
Albertson, D. G., Collins, C., McCormick, F. & Gray, J. W. Chromosome aberrations in solid tumors. Nature Genet. 34, 369–376 (2003).
Hassold, T. & Hunt, P. To err (meiotically) is human: the genesis of human aneuploidy. Nature Rev. Genet. 2, 280–291 (2001).
Antonarakis, S. E. 10 years of genomics, chromosome 21, and Down syndrome. Genomics 51, 1–16 (1998).
Bandyopadhyay, R. et al. Parental origin and timing of de novo Robertsonian translocation formation. Am. J. Hum. Genet. 71, 1456–1462 (2002).
Epstein, C. J. in The Metabolic and Molecular Bases of Inherited Diseases (eds Scriver, C. R., Beaudet, A. L., Sly, W. S. & Valle, D.) 1223–1256 (McGraw-Hill, New York, 2001).
Roizen, N. J. & Patterson, D. Down's syndrome. Lancet 361, 1281–1289 (2003).
Pennington, B. F., Moon, J., Edgin, J., Stedron, J. & Nadel, L. The neuropsychology of Down syndrome: evidence for hippocampal dysfunction. Child Dev. 74, 75–93 (2003).
Hattori, M. et al. The DNA sequence of human chromosome 21. Nature 405, 311–319 (2000). Landmark study that reports the high-quality nearly-complete nucleotide sequence of HSA21. Important for molecular studies on HSA21.
Lander, E. S. et al. Initial sequencing and analysis of the human genome. Nature 409, 860–921 (2001).
Venter, J. C. et al. The sequence of the human genome. Science 291, 1304–1351 (2001).
Reymond, A. et al. From PREDs and open reading frames to cDNA isolation: revisiting the human chromosome 21 transcription map. Genomics 78, 46–54 (2001).
Reymond, A. et al. Nineteen additional unpredicted transcripts from human chromosome 21. Genomics 79, 824–832 (2002).
Davisson, M. et al. Evolutionary breakpoints on human chromosome 21. 78, 99–106 (2001).
Gardiner, K. & Davisson, M. The sequence of human chromosome 21 and implications for research into Down syndrome. Genome Biol. 1, 1–9 (2000).
Gardiner, K., Fortna, A., Bechtel, L. & Davisson, M. T. Mouse models of Down syndrome: how useful can they be? Comparison of the gene content of human chromosome 21 with orthologous mouse genomic regions. Gene 318, 137–147 (2003).
Pletcher, M. T., Wiltshire, T., Cabin, D. E., Villanueva, M. & Reeves, R. H. Use of comparative physical and sequence mapping to annotate mouse chromosome 16 and human chromosome 21. Genomics 74, 45–54 (2001).
Rogers, M. A. et al. Hair keratin associated proteins: characterization of a second high sulfur KAP gene domain on human chromosome 21. J. Invest. Dermatol. 122, 147–158 (2004).
Shibuya, K. et al. A cluster of 21 keratin-associated protein genes within introns of another gene on human chromosome 21q22. 3. Genomics 83, 679–693 (2004).
Mural, R. J. et al. A comparison of whole-genome shotgun-derived mouse chromosome 16 and the human genome. Science 296, 1661–1671 (2002).
Watanabe, H. et al. DNA sequence and comparative analysis of chimpanzee chromosome 22. Nature 429, 382–388 (2004).
Kapranov, P. et al. Large-scale transcriptional activity in chromosomes 21 and 22. Science 296, 916–919 (2002). DNA chip technology used to study the transcriptional potential of the genome.
Kampa, D. et al. Novel RNAs identified from an in-depth analysis of the transcriptome of human chromosomes 21 and 22. Genome Res. 14, 331–342 (2004).
Cawley, S. et al. Unbiased mapping of transcription factor binding sites along human chromosomes 21 and 22 points to widespread regulation of noncoding RNAs. Cell 116, 499–509 (2004). DNA chip technology used to identify functional elements of the genome.
Waterston, R. H. et al. Initial sequencing and comparative analysis of the mouse genome. Nature 420, 520–562 (2002). The sequence of the mouse genome provided the opportunity for comparative analysis to identify human functional genomic elements.
Dermitzakis, E. T. et al. Numerous potentially functional but non-genic conserved sequences on human chromosome 21. Nature 420, 578–582 (2002).
Dermitzakis, E. T. et al. Comparison of human chromosome 21 conserved nongenic sequences (CNGs) with the mouse and dog genomes shows that their selective constraint is independent of their genic environment. Genome Res. 14, 852–859 (2004).
Dermitzakis, E. T. et al. Evolutionary discrimination of mammalian conserved non-genic sequences (CNGs). Science 302, 1033–1035 (2003).
Kirkness, E. F. et al. The dog genome: survey sequencing and comparative analysis. Science 301, 1898–1903 (2003).
Frazer, K. A. et al. Noncoding sequences conserved in a limited number of mammals in the SIM2 interval are frequently functional. Genome Res. 14, 367–372 (2004).
Thomas, J. W. et al. Comparative analyses of multi-species sequences from targeted genomic regions. Nature 424, 788–793 (2003).
Patil, N. et al. Blocks of limited haplotype diversity revealed by high-resolution scanning of human chromosome 21. Science 294, 1719–1723 (2001). A whole chromosome linkage disequilibrium mapping that provides tools for association studies of human complex phenotypes.
Davisson, M. T. et al. Segmental trisomy as a mouse model for Down syndrome. Prog. Clin. Biol. Res. 384, 117–133 (1993).
Kahlem, P. et al. Transcript level alterations reflect gene dosage effects across multiple tissues in a mouse model of down syndrome. Genome Res. 14, 1258–1267 (2004).
Reeves, R. H. et al. A mouse model for Down syndrome exhibits learning and behavioural deficits. Nature Genet. 11, 177–184 (1995). First phenotypic analysis of the most-used mouse model with partial trisomy 16.
Baxter, L. L., Moran, T. H., Richtsmeier, J. T., Troncoso, J. & Reeves, R. H. Discovery and genetic localization of Down syndrome cerebellar phenotypes using the Ts65Dn mouse. Hum. Mol. Genet. 9, 195–202 (2000).
Costa, A. C., Walsh, K. & Davisson, M. T. Motor dysfunction in a mouse model for Down syndrome. Physiol. Behav. 68, 211–220 (1999).
Escorihuela, R. M. et al. Impaired short- and long-term memory in Ts65Dn mice, a model for Down syndrome. Neurosci. Lett. 247, 171–174 (1998).
Dierssen, M. et al. Murine models for Down syndrome. Physiol. Behav. 73, 859–871 (2001).
Richtsmeier, J. T., Baxter, L. L. & Reeves, R. H. Parallels of craniofacial maldevelopment in Down syndrome and Ts65Dn mice. Dev. Dyn. 217, 137–145 (2000).
Delcroix, J. D. et al. Trafficking the NGF signal: implications for normal and degenerating neurons. Prog. Brain Res. 146, 3–23 (2004).
Cooper, J. D. et al. Failed retrograde transport of NGF in a mouse model of Down's syndrome: reversal of cholinergic neurodegenerative phenotypes following NGF infusion. Proc. Natl Acad. Sci. USA 98, 10439–10444 (2001).
Sago, H. et al. Ts1Cje, a partial trisomy 16 mouse model for Down syndrome, exhibits learning and behavioral abnormalities. Proc. Natl Acad. Sci. USA 95, 6256–6261 (1998).
Sago, H. et al. Genetic dissection of region associated with behavioral abnormalities in mouse models for Down syndrome. Pediatr. Res. 48, 606–613 (2000).
Shinohara, T. et al. Mice containing a human chromosome 21 model behavioral impairment and cardiac anomalies of Down's syndrome. Hum. Mol. Genet. 10, 1163–1175 (2001).
Maroun, L. E., Heffernan, T. N. & Hallam, D. M. Partial IFN-α/β and IFN-γ receptor knockout trisomy 16 mouse fetuses show improved growth and cultured neuron viability. J. Interferon Cytokine Res. 20, 197–203 ( 2000).
Epstein, C. J. et al. Transgenic mice with increased Cu/Zn-superoxide dismutase activity: animal model of dosage effects in Down syndrome. Proc. Natl Acad. Sci. USA 84, 8044–8048 (1987). First description of single-gene transgenic mouse model to study the overexpression consequences of a HSA21 gene.
Chrast, R. et al. Mice trisomic for a bacterial artificial chromosome with the single-minded 2 gene (Sim2) show phenotypes similar to some of those present in the partial trisomy 16 mouse models of Down syndrome. Hum. Mol. Genet. 9, 1853–1864 (2000).
Sumarsono, S. H. et al. Down's syndrome-like skeletal abnormalities in Ets2 transgenic mice. Nature 379, 534–537 (1996).
Lejeune, J., Gautier, M. & Turpin, R. Études des chromosomes somatiques de neuf enfants mongoliens. C. R. Acad. Sci. 248, 1721–1722 (1959). A classic paper. The first description of trisomy 21.
Lyle, R., Gehrig, C., Neergaard-Henrichsen, C., Deutsch, S. & Antonarakis, S. E. Gene expression from the aneuploid chromosome in a trisomy mouse model of Down syndrome. Genome Res. 14, 1268–1274 (2004).
Reymond, A. et al. Human chromosome 21 gene expression atlas in the mouse. Nature 420, 582–586 (2002). HSA21 gene expression atlas in the mouse.
Gitton, Y. et al. A gene expression map of human chromosome 21 orthologues in the mouse. Nature 420, 586–590 (2002). A gene expression map of HSA21 orthologues in the mouse.
FitzPatrick, D. R. et al. Transcriptome analysis of human autosomal trisomy. Hum. Mol. Genet. 11, 3249–3256 (2002).
Mao, R., Zielke, C. L., Zielke, H. R. & Pevsner, J. Global up-regulation of chromosome 21 gene expression in the developing Down syndrome brain. Genomics 81, 457–467 (2003).
Saran, N. G., Pletcher, M. T., Natale, J. E., Cheng, Y. & Reeves, R. H. Global disruption of the cerebellar transcriptome in a Down syndrome mouse model. Hum. Mol. Genet. 12, 2013–2019 (2003).
Chrast, R. et al. The mouse brain transcriptome by SAGE: differences in gene expression between P30 brains of the partial trisomy 16 mouse model of Down syndrome (Ts65Dn) and normals. Genome Res. 10, 2006–2021 (2000).
Barlow, G. M. et al. Down syndrome congenital heart disease: a narrowed region and a candidate gene. Genet. Med. 3, 91–101 (2001).
Korenberg, J. R. et al. Down syndrome phenotypes: the consequences of chromosomal imbalance. Proc. Natl Acad. Sci. USA 91, 4997–5001 (1994).
Delabar, J. M. et al. Molecular mapping of twenty-four features of Down syndrome on chromosome 21. Eur. J. Hum. Genet. 1, 114–124 (1993). Systematic attempt to define chromosomal trisomic regions resulting in Down syndrome phenotypes.
Kuo, W. L. et al. Detection of aneuploidy involving chromosomes 13, 18, or 21, by fluorescence in situ hybridization (FISH) to interphase and metaphase amniocytes. Am. J. Hum. Genet. 49, 112–119 (1991).
Armour, J. A., Sismani, C., Patsalis, P. C. & Cross, G. Measurement of locus copy number by hybridisation with amplifiable probes. Nucleic Acids Res. 28, 605–609 (2000).
Slater, H. R. et al. Rapid, high throughput prenatal detection of aneuploidy using a novel quantitative method (MLPA). J. Med. Genet. 40, 907–912 (2003).
Deutsch, S., Choudhury, U. & Antonarakis, S. E. Detection of trisomy 21 and other aneuploidies by paralogous gene quantification. J. Med. Genet. (in the press).
Ishkanian, A. S. et al. A tiling resolution DNA microarray with complete coverage of the human genome. Nature Genet. 36, 299–303 (2004).
Epstein, C. J., Epstein, L. B., Weil, J. & Cox, D. R. Trisomy 21: mechanisms and models. Ann. NY Acad. Sci. 396, 107–118 (1982).
Epstein, C. J. Mechanisms of the effects of aneuploidy in mammals. Annu. Rev. Genet. 22, 51–75 (1988).
Epstein, C. J. Consequences of Chromosome Imbalance: Principles, Mechanisms, and Models, (Cambridge Univ. Press, New York, 1986).
Meeks-Wagner, D. & Hartwell, L. H. Normal stoichiometry of histone dimer sets is necessary for high fidelity of mitotic chromosome transmission. Cell 44, 43–52 (1986).
Zwart, R. & Vijverberg, H. P. Four pharmacologically distinct subtypes of α4β2 nicotinic acetylcholine receptor expressed in Xenopus laevis oocytes. Mol. Pharmacol. 54, 1124–1131 (1998).
Nelson, M. E., Kuryatov, A., Choi, C. H., Zhou, Y. & Lindstrom, J. Alternate stoichiometries of α4β2 nicotinic acetylcholine receptors. Mol. Pharmacol. 63, 332–341 (2003).
Antoch, M. P. et al. Functional identification of the mouse circadian Clock gene by transgenic BAC rescue. Cell 89, 655–667 (1997).
Alkema, M. J., van der Lugt, N. M., Bobeldijk, R. C., Berns, A. & van Lohuizen, M. Transformation of axial skeleton due to overexpression of bmi-1 in transgenic mice. Nature 374, 724–727 (1995).
Heitzler, P. & Simpson, P. The choice of cell fate in the epidermis of Drosophila. Cell 64, 1083–1092 (1991).
Semenza, G. L., Koury, S. T., Nejfelt, M. K., Gearhart, J. D. & Antonarakis, S. E. Cell-type-specific and hypoxia-inducible expression of the human erythropoietin gene in transgenic mice. Proc. Natl Acad. Sci. USA 88, 8725–8729 (1991).
Singaraja, R. R. et al. Human ABCA1 BAC transgenic mice show increased high density lipoprotein cholesterol and ApoAI-dependent efflux stimulated by an internal promoter containing liver X receptor response elements in intron 1. J. Biol. Chem. 276, 33969–33979 (2001).
Hoffman, S. & Edelman, G. M. Kinetics of homophilic binding by embryonic and adult forms of the neural cell adhesion molecule. Proc. Natl Acad. Sci. USA 80, 5762–5766 (1983).
Struhl, G., Struhl, K. & Macdonald, P. M. The gradient morphogen bicoid is a concentration-dependent transcriptional activator. Cell 57, 1259–1273 (1989).
MacKay, V. L. et al. Gene expression analyzed by high-resolution state array analysis and quantitative proteomics: response of yeast to mating pheromone. Mol. Cell. Proteomics 3, 478–489 (2004).
Baptista, M. J. et al. Heterotrisomy, a significant contributing factor to ventricular septal defect associated with Down syndrome? Hum. Genet. 107, 476–482 (2000).
Ward, O. C., John Langdon Down (Royal Society of Medicine, London, 1998).
Wang, S. Y. et al. A high-resolution physical map of human chromosome 21p using yeast artificial chromosomes. Genome Res. 9, 1059–1073 (1999).
Kong, A. et al. A high-resolution recombination map of the human genome. Nature Genet. 31, 241–247 ( 2002).
Lynn, A. et al. Patterns of meiotic recombination on the long arm of human chromosome 21. Genome Res. 10, 1319–1332 (2000).
Harrison, P. M. et al. Molecular fossils in the human genome: identification and analysis of the pseudogenes in chromosomes 21 and 22. Genome Res. 12, 272–280 (2002).
Worton, R. G. et al. Human ribosomal RNA genes: orientation of the tandem array and conservation of the 5′ end. Science 239, 64–68 (1988).
Yamada, Y. et al. A comprehensive analysis of allelic methylation status of CpG islands on human chromosome 21q. Genome Res. 14, 247–266 (2004).
Antonarakis, S. E., Lyle, R., Deutsch, S. & Reymond, A. Chromosome 21: a small land of fascinating disorders with unknown pathophysiology. Int. J. Dev. Biol. 46, 89–96 (2002).
Olson, L. et al. Down syndrome mouse models Ts65Dn, Ts1Cje, and Ms1Cje/Ts65Dn exhibit variable severity of cerebellar phenotypes. Dev. Dyn. 230, 581–589 (2004).
Acknowledgements
We thank H. Attar, E. Hafen, U. Schibler, K. Basler, A. Estreicher, M. Friedli, K. Casada, L. Curtis, D. Sutter, C. Borel, and C. Attanasio for their contributions in the preparation of the manuscript; S. Dahoun and D. Marelli for figures 4a,b, and F. Bena for figure 4c. We also thank R. Reeves and three anonymous reviewers for critical reading of the manuscript and numerous insightful suggestions; all members (past and present) of the Antonarakis laboratory for discussions, debates and experimental data; the Swiss National Science Foundation, NCCR 'Frontiers in Genetics', European Union/Swiss OFES, Lejeune and 'Childcare' Foundations for support. We also thank the patients and their families for the donation of their samples and the continuous inspiration for research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1: The origin of supernumerary human chromosome 21 (HSA21) in free trisomy 21
Supplementary Table 2: Frequency of Down syndrome (DS) phenotypes (PDF 41 kb)
Related links
Related links
DATABASES
Entrez Gene
OMIM
FURTHER INFORMATION
Glossary
- ROBERTSONIAN TRANSLOCATIONS
-
Occurs when the long arms of two acrocentric chromosomes fuse at the centromere and the two short arms are lost. A non-robertsonian rearrangement is a chromosomal rearrangement other than a Robertsonian translocation.
- ACROCENTRIC CHROMOSOMES
-
A chromosome, the centromere of which is located near one end.
- ECTRODACTYLY
-
Limb malformations characterized by digit loss.
- BAC ARRAY CGH
-
A microarray that contains DNA from bacterial artificial chromosomes, which is used in comparative genomic hybridization to determine copy number differences of DNA sequences.
- PREAXIAL POLYDACTYLY
-
Polydactyly (additional fingers or toes) on the thumb side of the hand and the big-toe side of the foot.
- ATRIOVENTRICULAR CANAL
-
A complex congenital heart defect characterized by atrial septal defect, ventricular septal defect and abnormalities of the tricuspid and mitral valves.
- DUODENAL STENOSIS/ATRESIA
-
Narrowing (stenosis) or complete obliteration of the duodenal lumen.
- GIEMSA BANDS
-
Chromosome banding pattern produced by Giemsa staining.
- CPG ISLANDS
-
A genomic region of about one kilobase that contains close to the theoretical, expected frequency of the CpG dinucleotide.
- LINKAGE DISEQUILIBRIUM
-
Refers to the fact that particular alleles at nearby sites can co-occur on the same haplotype more often than is expected by chance.
- HAPLOTYPE BLOCKS
-
Long stretches (tens of megabases) along a chromosome that have low recombination rates (and relatively few haplotypes). Adjacent blocks are separated by recombination hot spots (short regions with high recombination rates).
- NOCICEPTION
-
The perception of pain.
- SAGE
-
Serial analysis of gene expression: a method for comprehensive analysis of gene expression patterns using a short sequence tag (10–14bp) for each RNA molecule.
- PYROSEQUENCING
-
A method for DNA sequencing, in which the inorganic pyrophosphate (PPi) that is released from a nucleoside triphosphate on DNA chain elongation is detected by a bioluminometric assay.
- FISH
-
Fluorescent in situ hybridization is a method that uses fluorescent molecular DNA probes to visualize specific regions in chromosomes that hybridize to the probe.
- CHROMOSOME PAINTING
-
Fluorescent in situ hybridization to chromosomes using a probe that represents a whole chromosome or a part of a chromosome.
- POLYCYTHAEMIA
-
The increase of the red blood cell count, haemoglobin and the total red blood cell volume, accompanied by an increase in total blood volume.
- HAEMATOCRIT
-
Percentage of red blood cells in blood.
Rights and permissions
About this article
Cite this article
Antonarakis, S., Lyle, R., Dermitzakis, E. et al. Chromosome 21 and Down syndrome: from genomics to pathophysiology. Nat Rev Genet 5, 725–738 (2004). https://doi.org/10.1038/nrg1448
Issue Date:
DOI: https://doi.org/10.1038/nrg1448
This article is cited by
-
Genome dosage alteration caused by chromosome pyramiding and shuffling effects on karyotypic heterogeneity, reproductive diversity, and phenotypic variation in Zea–Tripsacum allopolyploids
Theoretical and Applied Genetics (2024)
-
Transcriptomic analysis of stem cells from chorionic villi uncovers the impact of chromosomes 2, 6 and 22 in the clinical manifestations of Down syndrome
Stem Cell Research & Therapy (2023)
-
Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging
Archives of Toxicology (2023)
-
The aetiology of atypical bone health in individuals with Down syndrome
Archives of Osteoporosis (2023)
-
Non-invasive prenatal testing (NIPT) and pregnant women’s views on good motherhood: a qualitative study
European Journal of Human Genetics (2022)