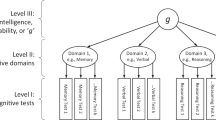
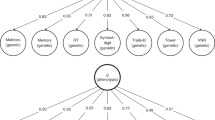
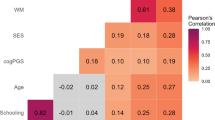
Key Points
-
Until 2017, genome-wide polygenic scores derived from genome-wide association studies (GWAS) of intelligence were able to predict only 1% of the variance in intelligence in independent samples.
-
Polygenic scores derived from GWAS of intelligence can now predict 4% of the variance in intelligence.
-
More than 10% of the variance in intelligence can be predicted by multipolygenic scores derived from GWAS of both intelligence and years of education. This accounts for more than 20% of the 50% heritability of intelligence.
-
Polygenic scores are unique predictors in two ways. First, they predict psychological and behavioural outcomes just as well from birth as later in life. Second, polygenic scores are causal predictors in the sense that nothing in our brains, behaviour or environment can change the differences in DNA sequence that we inherited from our parents.
-
Polygenic scores for intelligence can bring the powerful construct of intelligence to any research in the life sciences without having to assess intelligence through the use of tests.
Abstract
Intelligence — the ability to learn, reason and solve problems — is at the forefront of behavioural genetic research. Intelligence is highly heritable and predicts important educational, occupational and health outcomes better than any other trait. Recent genome-wide association studies have successfully identified inherited genome sequence differences that account for 20% of the 50% heritability of intelligence. These findings open new avenues for research into the causes and consequences of intelligence using genome-wide polygenic scores that aggregate the effects of thousands of genetic variants.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Gottfredson, L. S. Why g matters: The complexity of everyday life. Intelligence 24, 79–132 (1997).
Deary, I. J. et al. Genetic contributions to stability and change in intelligence from childhood to old age. Nature 482, 212–214 (2012).
Deary, I. J., Strand, S., Smith, P. & Fernandes, C. Intelligence and educational achievement. Intelligence 35, 13–21 (2007).
Schmidt, F. L. & Hunter, J. General mental ability in the world of work: occupational attainment and job performance. J. Pers. Soc. Psychol. 86, 162–173 (2004).
Strenze, T. Intelligence and socioeconomic success: a meta-analytic review of longitudinal research. Intelligence 35, 401–426 (2007).
Calvin, C. M. et al. Childhood intelligence in relation to major causes of death in 68 year follow-up: prospective population study. Brit. Med. J. 357, 2708 (2017).
Deary, I. J., Pattie, A. & Starr, J. M. The stability of intelligence from age 11 to age 90 years: the Lothian birth cohort of 1921. Psychol. Sci. 24, 2361–2368 (2013).
[No authors listed] Intelligence research should not be held back by its past. Nature 545, 385–386 (2017). This editorial is a landmark in the acceptance of genetic influence on intelligence, concluding, “it's well established and uncontroversial among geneticists that together, differences in genetics underwrite significant variation in intelligence between people.”
Pinker, S. The Blank Slate: The Modern Denial of Human Nature (Penguin, 2003).
Block, N. J. & Dworkin, G. E. The IQ Controversy: Critical Readings (Pantheon, 1976).
Gould, S. J. The Mismeasure of Man (W.W. Norton, 1982).
Kamin, L. J. The Science and Politics of IQ (Routledge, 1974).
Bouchard, T. J. & McGue, M. Familial studies of intelligence: a review. Science 212, 1055–1059 (1981).
Knopik, V. S., Neiderheiser, J., DeFries, J. C. & Plomin, R. Behavioral Genetics. 7th edn (Worth, 2017).
Haier, R. J. The Neuroscience of Intelligence (Cambridge Univ. Press, 2016).
Hare, B. Survival of the friendliest: Homo sapiens evolved via selection for prosociality. Annu. Rev. Psychol. 68, 155–186 (2017).
Sternberg, R. J. & Kaufman, J. C. The Evolution of Intelligence (Psychology Press, 2013).
Chabris, C. F. et al. Most reported genetic associations with general intelligence are probably false positives. Psychol. Sci. 23, 1314–1323 (2012).
Benyamin, B. et al. Childhood intelligence is heritable, highly polygenic and associated with FNBP1L. Mol. Psychiatry 19, 253–258 (2014).
Butcher, L. M., Davis, O. S., Craig, I. W. & Plomin, R. Genome-wide quantitative trait locus association scan of general cognitive ability using pooled DNA and 500K single nucleotide polymorphism microarrays. Genes Brain Behav. 7, 435–446 (2008).
Davies, G. et al. Genome-wide association studies establish that human intelligence is highly heritable and polygenic. Mol. Psychiatry 16, 996–1005 (2011).
Davies, G. et al. Genetic contributions to variation in general cognitive function: a meta-analysis of genome-wide association studies in the CHARGE consortium (N = 53 949). Mol. Psychiatry 20, 183–192 (2015).
Davies, G. et al. Genome-wide association study of cognitive functions and educational attainment in UK Biobank (N = 112 151). Mol. Psychiatry 21, 758–767 (2016).
Plomin, R. et al. A genome-wide scan of 1842 DNA markers for allelic associations with general cognitive ability: a five-stage design using DNA pooling and extreme selected groups. Behav. Genet. 31, 497–509 (2001).
Trampush, J. et al. GWAS meta-analysis reveals novel loci and genetic correlates for general cognitive function: a report from the COGENT consortium. Mol. Psychiatry 22, 336 (2017).
Cesarini, D. & Visscher, P. M. Genetics and educational attainment. Sci. Learn. 2, 1–7 (2017).
Rietveld, C. A. et al. Common genetic variants associated with cognitive performance identified using the proxy-phenotype method. Proc. Natl Acad. Sci. USA 111, 13790–13794 (2014). This study uses EA1 SNPs to predict intelligence, although less than 1% of the variance is predicted.
Rietveld, C. A. et al. GWAS of 126,559 individuals identifies genetic variants associated with educational attainment. Science 340, 1467–1471 (2013). This is the GWAS origin of EA1, which yields a GPS that predicts 1% of the variance in years of education.
Rietveld, C. A. et al. Replicability and robustness of genome-wide-association studies for behavioral traits. Psychol. Sci. 25, 1975–1986 (2014).
Okbay, A. et al. Genome-wide association study identifies 74 loci associated with educational attainment. Nature 533, 539–542 (2016). This is the GWAS origin of EA2 GPS, which increases the prediction of educational attainment from 1% to 3% of the variance.
Behavior Genetics Association 47th Annual Meeting Abstracts. Okbay, A. et al. GWAS of educational attainment – phase 3: main results [abstract]. Behav. Genet. 47, 699 (2017). This study refers to the largest GWAS of educational attainment ( n = 1,100,000), which increases the power of its GPS, EA3, to predict more than 10% of the variance in the targeted trait.
von Stumm, S. & Plomin, R. Socioeconomic status and the growth of intelligence from infancy through adolescence. Intelligence 48, 30–36 (2015).
Sniekers, S. et al. Genome-wide association meta-analysis of 78,308 individuals identifies new loci and genes influencing human intelligence. Nat. Genet. 49, 1107–1112 (2017). This is the GWAS origin of IQ2 GPS, which increases the prediction of intelligence from 1% to 3%.
Savage, J. E. et al. GWAS meta-analysis (N = 279,930) identifies new genes and functional links to intelligence. Preprint at https://doi.org/10.1101/184853 (2017). This paper describes the largest GWAS of intelligence to date, which yields a GPS (IQ3) that predicts 4% of the variance in intelligence.
Davies, G. et al. Ninety-nine independent genetic loci influencing general cognitive function include genes associated with brain health and structure (N = 280,360). Preprint at https://doi.org/10.1101/176511 (2017).
Krapohl, E. et al. Multi-polygenic score approach to trait prediction. Mol. Psychiatry https://doi.org/10.1038/mp.2017.163 (2017). This study employs a multiple-GPS approach and finds that 81 GPSs derived from well-powered GWAS predict 5% of the variance in intelligence.
Hill, W. D., Davies, G., McIntosh, A. M., Gale, C. R. & Deary, I. J. A combined analysis of genetically correlated traits identifies 107 loci associated with intelligence. Preprint at https://doi.org/10.1101/160291 (2017). This study employs multiple-trait analysis of GWAS for intelligence and finds that educational attainment and income predict 7% of the variance in intelligence in an independent sample.
Manolio, T. A. et al. Finding the missing heritability of complex diseases. Nature 461, 747–753 (2009).
Plomin, R. et al. Common DNA markers can account for more than half of the genetic influence on cognitive abilities. Psychol. Sci. 24, 562–568 (2013).
Boyle, E. A., Li, Y. I. & Pritchard, J. K. An expanded view of complex traits: from polygenic to omnigenic. Cell 169, 1177–1186 (2017).
Plomin, R. Blueprint: How DNA Makes Us Who We Are (Allen Lane/Penguin, in the press). This book describes genetic research on behaviour from twin studies to the DNA revolution and its implications for science and society.
Honzik, M. P., Macfarlane, J. W. & Allen, L. The stability of mental test performance between two and eighteen years. J. Exp. Educ. 17, 309–324 (1948).
Haworth, C. M. et al. A twin study of the genetics of high cognitive ability selected from 11,000 twin pairs in six studies from four countries. Behav. Genet. 39, 359–370 (2009).
Plomin, R. & Deary, I. J. Genetics and intelligence differences: five special findings. Mol. Psychiatry 20, 98–108 (2015). This article highlights five genetic findings that are special to intelligence differences, including one not mentioned in this Review — assortative mating is much greater for intelligence than for other traits.
Briley, D. A. & Tucker-Drob, E. M. Explaining the increasing heritability of cognitive ability across development: a meta-analysis of longitudinal twin and adoption studies. Psychol. Sci. 24, 1704–1713 (2013).
Selzam, S. et al. Predicting educational achievement from DNA. Mol. Psychiatry 22, 267–272 (2017). This study shows that EA2 predicts 9% of the variance in tested educational achievement at age 16, which was the strongest GPS prediction of a behavioural trait at that time.
Plomin, R. & Kovas, Y. Generalist genes and learning disabilities. Psychol. Bull. 131, 592–617 (2005).
Selzam, S. et al. Genome-wide polygenic scores predict reading performance throughout the school years. Sci. Stud. Read. 21, 334–349 (2017).
Carrion-Castillo, A. et al. Evaluation of results from genome-wide studies of language and reading in a novel independent dataset. Genes Brain Behav. 15, 531–541 (2016).
Krapohl, E. et al. Phenome-wide analysis of genome-wide polygenic scores. Mol. Psychiatry 21, 1188–1193 (2015).
Marioni, R. E. et al. Common genetic variants explain the majority of the correlation between height and intelligence: the generation Scotland study. Behav. Genet. 44, 91–96 (2014).
Williams, K. M. et al. Phenotypic and genotypic correlation between myopia and intelligence. Sci. Rep. 7, 45977 (2017).
Hill, W. D. et al. Age-dependent pleiotropy between general cognitive function and major psychiatric disorders. Biol. Psychiatry 80, 266–273 (2016).
Bulik-Sullivan, B. K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Plomin, R., Haworth, C. M. & Davis, O. S. Common disorders are quantitative traits. Nat. Rev. Genet. 10, 872–878 (2009).
Spain, S. L. et al. A genome-wide analysis of putative functional and exonic variation associated with extremely high intelligence. Mol. Psychiatry 21, 1145–1151 (2016).
Zabaneh, D. et al. A genome-wide association study for extremely high intelligence. Mol. Psychiatry https://doi.org/10.1038/mp.2017.121 (2017). This GWAS of intelligence uses a novel strategy to increase power — a case–control design in which the subjects were individuals with extremely high IQ from the top 0.0003 of the population (mean IQ of 170).
Reichenberg, A. et al. Discontinuity in the genetic and environmental causes of the intellectual disability spectrum. Proc. Natl Acad. Sci. USA 113, 1098–1103 (2016).
Vissers, L. E., Gilissen, C. & Veltman, J. A. Genetic studies in intellectual disability and related disorders. Nat. Rev. Genet. 17, 9–18 (2016).
Plomin, R. & Daniels, D. Why are children in the same family so different from one another? Behav. Brain Sci. 10, 1–16 (1987).
Tucker-Drob, E. M. & Bates, T. C. Large cross-national differences in gene × socioeconomic status interaction on intelligence. Psychol. Sci. 27, 138–149 (2016).
Hanscombe, K. B. et al. Socioeconomic status (SES) and children's intelligence (IQ): in a UK-representative sample SES moderates the environmental, not genetic, effect on IQ. PLOS ONE 7, e30320 (2012).
Plomin, R. & Bergeman, C. S. The nature of nurture: genetic influence on “environmental” measures. Behav. Brain Sci. 14, 373–386 (1991).
Belsky, D. W. et al. The genetics of success. Psychol. Sci. 27, 957–972 (2016).
Krapohl, E. et al. Widespread covariation of early environmental exposures and trait-associated polygenic variation. Proc. Natl Acad. Sci. USA 114, 11727–11732 (2017).
Smith-Woolley, E. et al. Differences in exam performance between pupils attending different school types mirror the genetic differences between them. NPJ Sci. Learn. (in the press).
Ayorech, Z., Krapohl, E., Plomin, R. & von Stumm, S. Genetic influence on intergenerational educational attainment. Psychol. Sci. 28, 1302–1310 (2017). This paper describes both twin analyses and EA2 GPSs that show genetic influence on intergenerational EA.
Behavior Genetics Association 46th Annual Meeting Abstracts. Rimfeld, K., Trzaskowski, M., Esko, T., Metspalu, A. & Plomin, R. Genetic influence on educational attainment and occupational status during and after the Soviet era in Estonia [abstract]. Behav. Genet. 46, 803 (2016).
Plomin, R. & DeFries, J. C. Genetics and intelligence: recent data. Intelligence 4, 15–24 (1980).
McEwen, J. E. et al. The ethical, legal, and social implications program of the National Human Genome Research Institute: reflections on an ongoing experiment. Annu. Rev. Genom. Hum. Genet. 15, 481–504 (2014).
Bouregy, S., Grigorenko, E. L., Latham, S. R. & Tan, M. Genetics, Ethics and Education (Cambridge Univ. Press, 2017).
Conley, D. & Fletcher, J. The Genome Factor: What the Social Genomics Revolution Reveals about Ourselves, our History, and the Future (Princeton Univ. Press, 2017).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences (Lawrence Erlbaum Associates, 1977).
Gottfredson, L. S. Mainstream science on intelligence. Wall Street Journal (13 December 1994).
Carroll, J. B. Human Cognitive Abilities: A Survey of Factor-Analytic Studies (Cambridge Univ. Press, 1993).
Spearman, C. 'General Intelligence' objectively determined and measured. Am. J. Psychol. 15, 201–292 (1904).
Jensen, A. R. The g Factor: The Science of Mental Ability (Praeger, 1998).
Deary, I. J. Intelligence. Annu. Rev. Psychol. 63, 453–482 (2012). This article is an authoritative overview of intelligence research.
Gow, A. J. et al. Stability and change in intelligence from age 11 to ages 70, 79, and 87: the Lothian Birth Cohorts of 1921 and 1936. Psychol. Ageing 26, 232–240 (2011).
Schaie, K. W. Developmental Influences on Adult Intelligence: The Seattle Longitudinal Study (Oxford Univ. Press, 2005).
Brinch, C. N. & Galloway, T. A. Schooling in adolescence raises IQ scores. Proc. Natl Acad. Sci. USA 109, 425–430 (2012).
Protzko, J. Does the raising IQ–raising g distinction explain the fadeout effect? Intelligence 56, 65–71 (2016).
Duyme, M., Dumaret, A.-C. & Tomkiewicz, S. How can we boost IQs of “dull children”?: a late adoption study. Proc. Natl Acad. Sci. USA 96, 8790–8794 (1999).
Melby-Lervåg, M. & Hulme, C. Is working memory training effective? A meta-analytic review. Dev. Psychol. 49, 270–291 (2013).
Puma, M. et al. Head Start Impact Study Final Report. Administration for Children and Families https://www.acf.hhs.gov/sites/default/files/opre/hs_impact_study_final.pdf (2010).
Plomin, R. & Simpson, M. A. The future of genomics for developmentalists. Dev. Psychopathol. 25, 1263–1278 (2013).
Pasaniuc, B. & Price, A. L. Dissecting the genetics of complex traits using summary association statistics. Nat. Rev. Genet. 18, 117–127 (2017).
Vilhjálmsson, B. J. et al. Modeling linkage disequilibrium increases accuracy of polygenic risk scores. Am. J. Hum. Genet. 97, 576–592 (2015).
Euseden, J. et al. PRSice: polygenic risk score software. Bioinformatics 31, 1466–1468 (2015).
Hill, W. D. et al. Molecular genetic contributions to social deprivation and household income in UK Biobank. Curr. Biol. 26, 3083–3089 (2016).
Turley, P. et al. MTAG: Multi-Trait Analysis of GWAS. Preprint at https://doi.org/10.1101/118810 (2017).
Zheng, J. et al. LD Hub: a centralized database and web interface to perform LD score regression that maximizes the potential of summary level GWAS data for SNP heritability and genetic correlation analysis. Bioinformatics 33, 272–279 (2017).
Yang, J. et al. Concepts, estimation and interpretation of SNP-based heritability. Nat. Genet. 49, 1304–1310 (2017).
Sullivan, P. F. et al. Psychiatric genomics: an update and an agenda. Am. J. Psychol. https://doi.org/10.1176/appi.ajp.2017.17030283 (2017).
Bacanu, S. A. Sharing extended summary data from contemporary genetic studies is unlikely to threaten subject privacy. PLOS ONE 12, e0179504 (2017).
Calvin, C. M. et al. Multivariate genetic analyses of cognition and academic achievement from two population samples of 174,000 and 166,000 school children. Behav. Genet. 42, 699–710 (2012).
Marioni, R. E. et al. Molecular genetic contributions to socioeconomic status and intelligence. Intelligence 44, 26–32 (2014).
Branigan, A. R., McCallum, K. J. & Freese, J. Variation in the heritability of educational attainment: An international meta-analysis. Soc. Forces 92, 109–140 (2013).
Krapohl, E. et al. The high heritability of educational achievement reflects many genetically influenced traits, not just intelligence. Proc. Natl Acad. Sci. USA 111, 15273–15278 (2014).
Haworth, C. M., Davis, O. S. & Plomin, R. Twins Early Development Study (TEDS): a genetically sensitive investigation of cognitive and behavioral development from childhood to young adulthood. Twin Res. Hum. Genet. 16, 117–125 (2013).
Acknowledgements
The authors gratefully acknowledge the ongoing contribution of the participants in the Twins Early Development Study (TEDS) and their families. TEDS is supported by a programme grant to R.P. from the UK Medical Research Council (MR/M021475/1 and previously G0901245), with additional support from the US National Institutes of Health (AG046938). The research reported here has also received funding from the European Research Council (ERC) under the European Union's Seventh Framework Programme (FP7/2007-2013) grant agreement 602768 and ERC grant agreement 295366. R.P. is also supported by a Medical Research Council Professorship award (G19/2). S.v.S. is supported by a Jacobs Foundation Research Fellowship award (2017–2019).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
PowerPoint slides
Glossary
- Twin studies
-
Studies comparing the resemblance of identical and fraternal twins to estimate genetic and environmental components of variance.
- Variance
-
An index of how spread out scores are in a study population, which is calculated as the average of the squared deviations from the mean.
- Genome-wide association studies
-
(GWAS). Studies that aim to identify loci throughout the genome associated with an observed trait or disorder.
- Heritability
-
The proportion of observed differences among individuals that can be attributed to inherited differences in genome sequence.
- Genome-wide polygenic scores
-
(GPSs). Genetic indices of a trait for each individual that are the sum across the genome of thousands of single-nucleotide polymorphisms (SNPs) of the individual's increasing alleles associated with the trait, usually weighted by the effect size of each SNP's association with the trait in genome-wide association studies.
- Candidate gene studies
-
Studies that focus on genes for which the function suggests that they are associated with a trait, in contrast to genome-wide association studies.
- Effect sizes
-
Proportions of variance of traits in the study population accounted for by a particular factor such as a genome-wide polygenic score.
- Single-nucleotide polymorphisms
-
(SNPs). Single base pair differences in inherited DNA sequence between individuals.
- Linkage disequilibrium (LD) score regression analysis
-
Analysis that, for each single-nucleotide polymorphism in a genome-wide association study (GWAS), regresses χ2 statistics from GWAS summary statistics against LD scores.
Rights and permissions
About this article
Cite this article
Plomin, R., von Stumm, S. The new genetics of intelligence. Nat Rev Genet 19, 148–159 (2018). https://doi.org/10.1038/nrg.2017.104
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg.2017.104
This article is cited by
-
Infrastructuring Educational Genomics: Associations, Architectures, and Apparatuses
Postdigital Science and Education (2024)
-
Genetic determinism, essentialism and reductionism: semantic clarity for contested science
Nature Reviews Genetics (2023)
-
Celebrating a Century of Research in Behavioral Genetics
Behavior Genetics (2023)
-
Do Children Think it is Important to Predict Learning and Behaviour Problems, and Do They Think Genetic Screening Has a Role to Play in This?
Journal of Autism and Developmental Disorders (2023)
-
The impact of twenty-first century skills on university students’ robotic achievements
Education and Information Technologies (2023)