Abstract
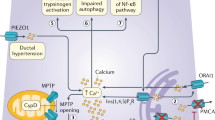
Chronic pancreatitis is defined as a pathological fibro-inflammatory syndrome of the pancreas in individuals with genetic, environmental and/or other risk factors who develop persistent pathological responses to parenchymal injury or stress. Potential causes can include toxic factors (such as alcohol or smoking), metabolic abnormalities, idiopathic mechanisms, genetics, autoimmune responses and obstructive mechanisms. The pathophysiology of chronic pancreatitis is fairly complex and includes acinar cell injury, acinar stress responses, duct dysfunction, persistent or altered inflammation, and/or neuro-immune crosstalk, but these mechanisms are not completely understood. Chronic pancreatitis is characterized by ongoing inflammation of the pancreas that results in progressive loss of the endocrine and exocrine compartment owing to atrophy and/or replacement with fibrotic tissue. Functional consequences include recurrent or constant abdominal pain, diabetes mellitus (endocrine insufficiency) and maldigestion (exocrine insufficiency). Diagnosing early-stage chronic pancreatitis is challenging as changes are subtle, ill-defined and overlap those of other disorders. Later stages are characterized by variable fibrosis and calcification of the pancreatic parenchyma; dilatation, distortion and stricturing of the pancreatic ducts; pseudocysts; intrapancreatic bile duct stricturing; narrowing of the duodenum; and superior mesenteric, portal and/or splenic vein thrombosis. Treatment options comprise medical, radiological, endoscopic and surgical interventions, but evidence-based approaches are limited. This Primer highlights the major progress that has been made in understanding the pathophysiology, presentation, prevalence and management of chronic pancreatitis and its complications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Whitcomb, D. C. et al. Chronic pancreatitis: an international draft consensus proposal for a new mechanistic definition. Pancreatology 16, 218–224 (2016).
Steer, M. L., Waxman, I. & Freedman, S. Chronic pancreatitis. N. Engl. J. Med. 332, 1482–1490 (1995).
Löhr, J. M. et al. United European Gastroenterology evidence-based guidelines for the diagnosis and therapy of chronic pancreatitis (HaPanEU). United European Gastroenterol. J. 5, 153–199 (2017). Current state-of-the-art guidelines for the diagnosis and therapy of chronic pancreatitis.
Muniraj, T., Aslanian, H. R., Farrell, J. & Jamidar, P. A. Chronic pancreatitis, a comprehensive review and update. Part I: epidemiology, etiology, risk factors, genetics, pathophysiology, and clinical features. Dis. Mon. 60, 530–550 (2014).
Peery, A. F. et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology 143, 1179–1187.e3 (2012).
Yadav, D., Timmons, L., Benson, J. T., Dierkhising, R. A. & Chari, S. T. Incidence, prevalence, and survival of chronic pancreatitis: a population-based study. Am. J. Gastroenterol. 106, 2192–2199 (2011). This is one of the few systematic, population-based studies on the epidemiology of chronic pancreatitis.
Levy, P. et al. Estimation of the prevalence and incidence of chronic pancreatitis and its complications. Gastroenterol. Clin. Biol. 30, 838–844 (2006).
Levy, P., Dominguez-Munoz, E., Imrie, C., Lohr, M. & Maisonneuve, P. Epidemiology of chronic pancreatitis: burden of the disease and consequences. United European Gastroenterol. J. 2, 345–354 (2014).
Lowenfels, A. B. et al. Racial factors and the risk of chronic pancreatitis. Am. J. Gastroenterol. 94, 790–794 (1999).
Yadav, D. & Lowenfels, A. B. The epidemiology of pancreatitis and pancreatic cancer. Gastroenterology 144, 1252–1261 (2013).
Majumder, S. & Chari, S. T. Chronic pancreatitis. Lancet 387, 1957–1966 (2016).
Hirota, M. et al. The seventh nationwide epidemiological survey for chronic pancreatitis in Japan: clinical significance of smoking habit in Japanese patients. Pancreatology 14, 490–496 (2014).
Wilcox, C. M. et al. Racial differences in the clinical profile, causes, and outcome of chronic pancreatitis. Am. J. Gastroenterol. 111, 1488–1496 (2016).
Romagnuolo, J. et al. Clinical profile, etiology, and treatment of chronic pancreatitis in North American women: analysis of a large multicenter cohort. Pancreas 45, 934–940 (2016).
Yadav, D., Muddana, V. & O’Connell, M. Hospitalizations for chronic pancreatitis in Allegheny County, Pennsylvania, USA. Pancreatology 11, 546–552 (2011).
Schwarzenberg, S. J. et al. Pediatric chronic pancreatitis is associated with genetic risk factors and substantial disease burden. J. Pediatr. 166, 890–896.e1 (2015).
Giefer, M. J. et al. Early-onset acute recurrent and chronic pancreatitis is associated with PRSS1 or CTRC gene mutations. J. Pediatr. 186, 95–100 (2017).
Masamune, A. et al. Nationwide epidemiological survey of early chronic pancreatitis in Japan. J. Gastroenterol. 52, 992–1000 (2017).
Otsuki, M. Chronic pancreatitis in Japan: epidemiology, prognosis, diagnostic criteria, and future problems. J. Gastroenterol. 38, 315–326 (2003).
Shimosegawa, T. et al. The revised Japanese clinical diagnostic criteria for chronic pancreatitis. J. Gastroenterol. 45, 584–591 (2010).
Cote, G. A. et al. Alcohol and smoking as risk factors in an epidemiology study of patients with chronic pancreatitis. Clin. Gastroenterol. Hepatol. 9, 266–273 (2011).
Frulloni, L. et al. Chronic pancreatitis: report from a multicenter Italian survey (PanCroInfAISP) on 893 patients. Dig. Liver Dis. 41, 311–317 (2009).
Yang, A. L., Vadhavkar, S., Singh, G. & Omary, M. B. Epidemiology of alcohol-related liver and pancreatic disease in the United States. Arch. Intern. Med. 168, 649–656 (2008).
Lowenfels, A. B. et al. Pancreatitis and the risk of pancreatic cancer. International Pancreatitis Study Group. N. Engl. J. Med. 328, 1433–1437 (1993). This is the first epidemiological study showing that chronic pancreatitis is a risk factor for pancreatic cancer.
Grocock, C. J. et al. The variable phenotype of the p. A16V mutation of cationic trypsinogen (PRSS1) in pancreatitis families. Gut 59, 357–363 (2010).
Howes, N. et al. Clinical and genetic characteristics of hereditary pancreatitis in Europe. Clin. Gastroenterol. Hepatol. 2, 252–261 (2004).
Whitcomb, D. C. et al. Multicenter approach to recurrent acute and chronic pancreatitis in the United States: the North American Pancreatitis Study 2 (NAPS2). Pancreatology 8, 520–531 (2008).
Conwell, D. L. et al. Validation of demographics, etiology, and risk factors for chronic pancreatitis in the USA: a report of the North American Pancreas Study (NAPS) group. Dig. Dis. Sci. 62, 2133–2140 (2017).
Apte, M. V., Pirola, R. C. & Wilson, J. S. Mechanisms of alcoholic pancreatitis. J. Gastroenterol. Hepatol. 25, 1816–1826 (2010).
Nordback, I. et al. The recurrence of acute alcohol-associated pancreatitis can be reduced: a randomized controlled trial. Gastroenterology 136, 848–855 (2009).
Pandol, S. J. & Raraty, M. Pathobiology of alcoholic pancreatitis. Pancreatology 7, 105–114 (2007).
Yadav, D. & Whitcomb, D. C. The role of alcohol and smoking in pancreatitis. Nat. Rev. Gastroenterol. Hepatol. 7, 131–145 (2010).
Yadav, D. et al. Alcohol consumption, cigarette smoking, and the risk of recurrent acute and chronic pancreatitis. Arch. Intern. Med. 169, 1035–1045 (2009). The North American Pancreatitis Study data showing a threshold for alcoholic pancreatitis, the independent risk of smoking, and the additive effects of alcohol and smoking.
Lin, Y., Tamakoshi, A., Hayakawa, T., Ogawa, M. & Ohno, Y. Associations of alcohol drinking and nutrient intake with chronic pancreatitis: findings from a case–control study in Japan. Am. J. Gastroenterol. 96, 2622–2627 (2001).
Takeyama, Y. Long-term prognosis of acute pancreatitis in Japan. Clin. Gastroenterol. Hepatol. 7, S15–S17 (2009).
Rebours, V. et al. Smoking and the course of recurrent acute and chronic alcoholic pancreatitis: a dose-dependent relationship. Pancreas 41, 1219–1224 (2012).
Andriulli, A. et al. Smoking as a cofactor for causation of chronic pancreatitis: a meta-analysis. Pancreas 39, 1205–1210 (2010).
Wittel, U. A. et al. Chronic pancreatic inflammation induced by environmental tobacco smoke inhalation in rats. Am. J. Gastroenterol. 101, 148–159 (2006).
Thrower, E. Pathologic cellular events in smoking-related pancreatitis. Cancers 7, 723–735 (2015).
Whitcomb, D. C. Genetic risk factors for pancreatic disorders. Gastroenterology 144, 1292–1302 (2013).
Cohn, J. A. et al. Increased risk of idiopathic chronic pancreatitis in cystic fibrosis carriers. Hum. Mutat. 26, 303–307 (2005).
Threadgold, J. et al. The N34S mutation of SPINK1 (PSTI) is associated with a familial pattern of idiopathic chronic pancreatitis but does not cause the disease. Gut 50, 675–681 (2002).
Bertin, C. et al. Pancreas divisum is not a cause of pancreatitis by itself but acts as a partner of genetic mutations. Am. J. Gastroenterol. 107, 311–317 (2012).
Okazaki, K. et al. International consensus for the treatment of autoimmune pancreatitis. Pancreatology 17, 1–6 (2016).
Sah, R. P. et al. Differences in clinical profile and relapse rate of type 1 versus type 2 autoimmune pancreatitis. Gastroenterology 139, 140–148 (2010).
Shimosegawa, T. et al. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas 40, 352–358 (2011).
Rebours, V. et al. The natural history of hereditary pancreatitis: a national series. Gut 58, 97–103 (2009).
Masamune, A. Genetics of pancreatitis: the 2014 update. Tohoku J. Exp. Med. 232, 69–77 (2014).
Keim, V. et al. Clinical characterization of patients with hereditary pancreatitis and mutations in the cationic trypsinogen gene. Am. J. Med. 111, 622–626 (2001).
Nemeth, B. C. & Sahin-Toth, M. Human cationic trypsinogen (PRSS1) variants and chronic pancreatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 306, G466–G473 (2014).
Whitcomb, D. C. et al. Hereditary pancreatitis is caused by a mutation in the cationic trypsinogen gene. Nat. Genet. 14, 141–145 (1996).
Schnur, A., Beer, S., Witt, H., Hegyi, P. & Sahin-Toth, M. Functional effects of 13 rare PRSS1 variants presumed to cause chronic pancreatitis. Gut 63, 337–343 (2014).
Khalid, A. et al. A 93 year old man with the PRSS1 R122H mutation, low SPINK1 expression, and no pancreatitis: insights into phenotypic non-penetrance. Gut 55, 728–731 (2006).
Aoun, E. et al. Pathways to injury in chronic pancreatitis: decoding the role of the high-risk SPINK1 N34S haplotype using meta-analysis. PLoS ONE 3, e2003 (2008).
Masson, E., Chen, J. M., Scotet, V., Le Marechal, C. & Ferec, C. Association of rare chymotrypsinogen C (CTRC) gene variations in patients with idiopathic chronic pancreatitis. Hum. Genet. 123, 83–91 (2008).
Rosendahl, J. et al. Chymotrypsin C (CTRC) variants that diminish activity or secretion are associated with chronic pancreatitis. Nat. Genet. 40, 78–82 (2008).
Beer, S. et al. Comprehensive functional analysis of chymotrypsin C (CTRC) variants reveals distinct loss-of-function mechanisms associated with pancreatitis risk. Gut 62, 1616–1624 (2013).
LaRusch, J. et al. The common chymotrypsinogen C (CTRC) variant G60G (C.180T) increases risk of chronic pancreatitis but not recurrent acute pancreatitis in a North American population. Clin. Transl Gastroenterol. 6, e68 (2015).
Bombieri, C. et al. Recommendations for the classification of diseases as CFTR-related disorders. J. Cyst. Fibros 10 (Suppl. 2), S86–S102 (2011).
Noone, P. G. et al. Cystic fibrosis gene mutations and pancreatitis risk: relation to epithelial ion transport and trypsin inhibitor gene mutations. Gastroenterology 121, 1310–1319 (2001).
Schneider, A. et al. Combined bicarbonate conductance-impairing variants in CFTR and SPINK1 variants are associated with chronic pancreatitis in patients without cystic fibrosis. Gastroenterology 140, 162–171 (2011).
LaRusch, J. et al. Mechanisms of CFTR functional variants that impair regulated bicarbonate permeation and increase risk for pancreatitis but not for cystic fibrosis. PLoS Genet. 10, e1004376 (2014).
Felderbauer, P. et al. Mutations in the calcium-sensing receptor: a new genetic risk factor for chronic pancreatitis? Scand. J. Gastroenterol. 41, 343–348 (2006).
Muddana, V. et al. Association between calcium sensing receptor gene polymorphisms and chronic pancreatitis in a US population: role of serine protease inhibitor Kazal 1type and alcohol. World J. Gastroenterol. 14, 4486–4491 (2008).
Witt, H. et al. Variants in CPA1 are strongly associated with early onset chronic pancreatitis. Nat. Genet. 45, 1216–1220 (2013).
Sahin-Toth, M. Genetic risk in chronic pancreatitis: the misfolding-dependent pathway. Curr. Opin. Gastroenterol. 33, 390–395 (2017).
Derikx, M. H. et al. Polymorphisms at PRSS1–PRSS2 and CLDN2–MORC4 loci associate with alcoholic and non-alcoholic chronic pancreatitis in a European replication study. Gut 64, 1426–1433 (2015).
Whitcomb, D. C. et al. Common genetic variants in the CLDN2 and PRSS1–PRSS2 loci alter risk for alcohol-related and sporadic pancreatitis. Nat. Genet. 44, 1349–1354 (2012).
Greer, J. B. et al. ABO blood group and chronic pancreatitis risk in the NAPS2 cohort. Pancreas 40, 1188–1194 (2011).
Weiss, F. U. et al. ABO blood type B and fucosyltransferase 2 non-secretor status as genetic risk factors for chronic pancreatitis. Gut 65, 353–354 (2016).
Brand, H., Diergaarde, B., O’Connell, M. R., Whitcomb, D. C. & Brand, R. E. Variation in the gamma-glutamyltransferase 1 gene and risk of chronic pancreatitis. Pancreas 42, 836–840 (2013).
Fjeld, K. et al. A recombined allele of the lipase gene CEL and its pseudogene CELP confers susceptibility to chronic pancreatitis. Nat. Genet. 47, 518–522 (2015).
Xiao, X. et al. A carboxyl ester lipase (CEL) mutant causes chronic pancreatitis by forming intracellular aggregates that activate apoptosis. J. Biol. Chem. 291, 23224–23236 (2016).
Fjeld, K. et al. Length of variable numbers of tandem repeats in the carboxyl ester lipase (CEL) gene may confer susceptibility to alcoholic liver cirrhosis but not alcoholic chronic pancreatitis. PLoS ONE 11, e0165567 (2016).
Kloppel, G. & Maillet, B. Pseudocysts in chronic pancreatitis: a morphological analysis of 57 resection specimens and 9 autopsy pancreata. Pancreas 6, 266–274 (1991).
Kloppel, G. & Maillet, B. The morphological basis for the evolution of acute pancreatitis into chronic pancreatitis. Virchows Arch. A Pathol. Anat. Histopathol. 420, 1–4 (1992).
Bhanot, U. K. & Moller, P. Mechanisms of parenchymal injury and signaling pathways in ectatic ducts of chronic pancreatitis: implications for pancreatic carcinogenesis. Lab. Invest. 89, 489–497 (2009).
Witt, H., Apte, M. V., Keim, V. & Wilson, J. S. Chronic pancreatitis: challenges and advances in pathogenesis, genetics, diagnosis, and therapy. Gastroenterology 132, 1557–1573 (2007).
Whitcomb, D. C. Hereditary pancreatitis: new insights into acute and chronic pancreatitis. Gut 45, 317–322 (1999).
Deng, X. et al. Chronic alcohol consumption accelerates fibrosis in response to cerulein-induced pancreatitis in rats. Am. J. Pathol. 166, 93–106 (2005).
Leung, P. S. & Chan, Y. C. Role of oxidative stress in pancreatic inflammation. Antioxid. Redox Signal. 11, 135–165 (2009).
Sendler, M. et al. Tumour necrosis factor alpha secretion induces protease activation and acinar cell necrosis in acute experimental pancreatitis in mice. Gut 62, 430–439 (2013).
Saito, I., Hashimoto, S., Saluja, A., Steer, M. L. & Meldolesi, J. Intracellular transport of pancreatic zymogens during caerulein supramaximal stimulation. Am. J. Physiol. 253, G517–G526 (1987).
Norton, I. D. et al. Chronic ethanol administration causes oxidative stress in the rat pancreas. J. Lab. Clin. Med. 131, 442–446 (1998).
Yamaguchi, M. et al. Bicarbonate-rich fluid secretion predicted by a computational model of guinea-pig pancreatic duct epithelium. J. Physiol. 595, 1947–1972 (2017).
Hall, P. A. & Lemoine, N. R. Rapid acinar to ductal transdifferentiation in cultured human exocrine pancreas. J. Pathol. 166, 97–103 (1992).
Houbracken, I. et al. Lineage tracing evidence for transdifferentiation of acinar to duct cells and plasticity of human pancreas. Gastroenterology 141, 731–741.e4 (2011).
Apte, M., Pirola, R. C. & Wilson, J. S. Pancreatic stellate cell: physiologic role, role in fibrosis and cancer. Curr. Opin. Gastroenterol. 31, 416–423 (2015).
Lee, A. T. et al. Alcohol and cigarette smoke components activate human pancreatic stellate cells: implications for the progression of chronic pancreatitis. Alcohol. Clin. Exp. Res. 39, 2123–2133 (2015).
Bachem, M. G. et al. Identification, culture, and characterization of pancreatic stellate cells in rats and humans. Gastroenterology 115, 421–432 (1998).
Erkan, M. et al. StellaTUM: current consensus and discussion on pancreatic stellate cell research. Gut 61, 172–178 (2012).
Sendler, M. et al. Complement component 5 mediates development of fibrosis, via activation of stellate cells, in 2 mouse models of chronic pancreatitis. Gastroenterology 149, 765–776.e10 (2015).
Masamune, A. et al. Hypoxia stimulates pancreatic stellate cells to induce fibrosis and angiogenesis in pancreatic cancer. Am. J. Physiol. Gastrointest. Liver Physiol. 295, G709–G717 (2008).
Lankisch, P. G., Seidensticker, F., Lohr-Happe, A., Otto, J. & Creutzfeldt, W. The course of pain is the same in alcohol- and nonalcohol-induced chronic pancreatitis. Pancreas 10, 338–341 (1995).
Demir, I. E., Friess, H. & Ceyhan, G. O. Neural plasticity in pancreatitis and pancreatic cancer. Nat. Rev. Gastroenterol. Hepatol. 12, 649–659 (2015).
Drewes, A. M. et al. Pain in chronic pancreatitis: the role of neuropathic pain mechanisms. Gut 57, 1616–1627 (2008).
Poulsen, J. L., Olesen, S. S., Malver, L. P., Frokjaer, J. B. & Drewes, A. M. Pain and chronic pancreatitis: a complex interplay of multiple mechanisms. World J. Gastroenterol. 19, 7282–7291 (2013).
Schwartz, E. S. et al. Synergistic role of TRPV1 and TRPA1 in pancreatic pain and inflammation. Gastroenterology 140, 1283–1291.e2 (2011).
Zhu, Y. et al. Nerve growth factor modulates TRPV1 expression and function and mediates pain in chronic pancreatitis. Gastroenterology 141, 370–377 (2011).
Olesen, S. S. et al. Pain severity reduces life quality in chronic pancreatitis: implications for design of future outcome trials. Pancreatology 14, 497–502 (2014).
Rickels, M. R. et al. Detection, evaluation and treatment of diabetes mellitus in chronic pancreatitis: recommendations from PancreasFest 2012. Pancreatology 13, 336–342 (2013). A comprehensive review and recommendations for evaluating and managing diabetes mellitus in patients with chronic pancreatitis.
Andersen, B. N., Pedersen, N. T., Scheel, J. & Worning, H. Incidence of alcoholic chronic pancreatitis in Copenhagen. Scand. J. Gastroenterol. 17, 247–252 (1982).
Hoffmeister, A. et al. English language version of the S3-consensus guidelines on chronic pancreatitis: definition, aetiology, diagnostic examinations, medical, endoscopic and surgical management of chronic pancreatitis. Zeitschrift Gastroenterol. 53, 1447–1495 (2015).
Whitcomb, D. C. Peering Into the “black box” of the complex chronic pancreatitis syndrome. Pancreas 45, 1361–1364 (2016).
Johnson, C. D. et al. Qualitative assessment of the symptoms and impact of pancreatic exocrine insufficiency (PEI) to inform the development of a patient-reported outcome (PRO) instrument. Patienthttp://dx.doi.org/10.1007/s40271-017-0233-0 (2017).
DiMagno, E. P., Go, V. L. & Summerskill, W. H. Relations between pancreatic enzyme outputs and malabsorption in severe pancreatic insufficiency. N. Engl. J. Med. 288, 813–815 (1973).
Sanyal, R., Stevens, T., Novak, E. & Veniero, J. C. Secretin-enhanced MRCP: review of technique and application with proposal for quantification of exocrine function. AJR Am. J. Roentgenol. 198, 124–132 (2012).
Siegmund, E., Lohr, J. M. & Schuff-Werner, P. The diagnostic validity of non-invasive pancreatic function tests — a meta-analysis [German]. Zeitschrift Gastroenterol. 42, 1117–1128 (2004).
Keller, J., Aghdassi, A. A., Lerch, M. M., Mayerle, J. V. & Layer, P. Tests of pancreatic exocrine function — clinical significance in pancreatic and non-pancreatic disorders. Best Pract. Res. Clin. Gastroenterol. 23, 425–439 (2009).
Martinez-Moneo, E. et al. Deficiency of fat-soluble vitamins in chronic pancreatitis: a systematic review and meta-analysis. Pancreatology 16, 988–994 (2016).
Hoogenboom, S. A., Lekkerkerker, S. J., Fockens, P., Boermeester, M. A. & van Hooft, J. E. Systematic review and meta-analysis on the prevalence of vitamin D deficiency in patients with chronic pancreatitis. Pancreatology 16, 800–806 (2016).
Munigala, S., Agarwal, B., Gelrud, A. & Conwell, D. L. Chronic pancreatitis and fracture: a retrospective, population-based veterans administration study. Pancreas 45, 355–361 (2016).
Cui, Y. & Andersen, D. K. Pancreatogenic diabetes: special considerations for management. Pancreatology 11, 279–294 (2011).
Makuc, J. Management of pancreatogenic diabetes: challenges and solutions. Diabetes Metab. Syndr. Obes. 9, 311–315 (2016).
Kloppel, G. Chronic pancreatitis, pseudotumors and other tumor-like lesions. Mod. Pathol. 20 (Suppl. 1), S113–S131 (2007). This paper provides a fundamental pathology overview of chronic pancreatitis.
DeSouza, K. & Nodit, L. Groove pancreatitis: a brief review of a diagnostic challenge. Arch. Pathol. Lab. Med. 139, 417–421 (2015).
Esposito, I. et al. Mast cell distribution and activation in chronic pancreatitis. Hum. Pathol. 32, 1174–1183 (2001).
Shrikhande, S. V. et al. Comparison of histological features and inflammatory cell reaction in alcoholic, idiopathic and tropical chronic pancreatitis. Br. J. Surg. 90, 1565–1572 (2003). This paper is one of the few comparative studies of non-autoimmune chronic pancreatitis.
Singhi, A. D. et al. The histopathology of PRSS1 hereditary pancreatitis. Am. J. Surg. Pathol. 38, 346–353 (2014).
Chari, S. T. et al. Histopathologic and clinical subtypes of autoimmune pancreatitis: the Honolulu consensus document. Pancreas 39, 549–554 (2010). Consensus criteria for histological diagnosis of autoimmune pancreatitis.
Esposito, I. et al. Autoimmune pancreatocholangitis, non-autoimmune pancreatitis and primary sclerosing cholangitis: a comparative morphological and immunological analysis. PLoS ONE 3, e2539 (2008). This paper is one of the few comparative studies of autoimmune pancreatitis.
Kloppel, G., Detlefsen, S., Chari, S. T., Longnecker, D. S. & Zamboni, G. Autoimmune pancreatitis: the clinicopathological characteristics of the subtype with granulocytic epithelial lesions. J. Gastroenterol. 45, 787–793 (2010).
Pickartz, T., Mayerle, J. & Lerch, M. M. Autoimmune pancreatitis. Nat. Clin. Pract. Gastroenterol. Hepatol. 4, 314–323 (2007).
Conwell, D. L. et al. American Pancreatic Association Practice Guidelines in Chronic Pancreatitis: evidence-based report on diagnostic guidelines. Pancreas 43, 1143–1162 (2014).
Dimastromatteo, J., Brentnall, T. & Kelly, K. A. Imaging in pancreatic disease. Nat. Rev. Gastroenterol. Hepatol. 14, 97–109 (2016).
Sandrasegaran, K., Lin, C., Akisik, F. M. & Tann, M. State-of-the-art pancreatic MRI. AJR Am. J. Roentgenol. 195, 42–53 (2010). A comprehensive overview of MRI as a non-invasive morphological and functional imaging technique for pancreatic diseases.
Perez-Johnston, R., Sainani, N. I. & Sahani, D. V. Imaging of chronic pancreatitis (including groove and autoimmune pancreatitis). Radiol. Clin. North Am. 50, 447–466 (2012).
Lee, L. S. & Conwell, D. L. Update on advanced endoscopic techniques for the pancreas: endoscopic retrograde cholangiopancreatography, drainage and biopsy, and endoscopic ultrasound. Radiol. Clin. North Am. 50, 547–561 (2012).
Trikudanathan, G. et al. Evaluation of Rosemont criteria for non-calcific chronic pancreatitis (NCCP) based on histopathology — a retrospective study. Pancreatology 17, 63–69 (2016).
Trikudanathan, G. et al. Diagnostic performance of endoscopic ultrasound (EUS) for non-calcific chronic pancreatitis (NCCP) based on histopathology. Am. J. Gastroenterol. 111, 568–574 (2016).
Shi, Y., Glaser, K. J., Venkatesh, S. K., Ben-Abraham, E. I. & Ehman, R. L. Feasibility of using 3D MR elastography to determine pancreatic stiffness in healthy volunteers. J. Magn. Reson. Imaging 41, 369–375 (2015).
Kuwahara, T. et al. Quantitative evaluation of pancreatic tumor fibrosis using shear wave elastography. Pancreatology 16, 1063–1068 (2016).
Tirkes, T. et al. T1 mapping for diagnosis of mild chronic pancreatitis. J. Magn. Reson. Imaging 45, 1171–1176 (2017). This study describes T1-mapping as an emerging MRI technique for chronic pancreatitis.
Concia, M. et al. Diffusion-weighted magnetic resonance imaging of the pancreas: diagnostic benefit from an intravoxel incoherent motion model-based 3 b-value analysis. Invest. Radiol 49, 93–100 (2014).
Akisik, M. F. et al. Assessment of chronic pancreatitis: utility of diffusion-weighted MR imaging with secretin enhancement. Radiology 250, 103–109 (2009). This large cohort study demonstrates that advanced diffusion-weighted MRI techniques can differentiate mild from moderate to severe chronic pancreatitis.
Sugita, R. et al. Direct visualization of pancreatic juice flow using unenhanced MRI with spin labeling can be aid in diagnosing chronic pancreatitis. AJR Am. J. Roentgenol. 202, 1027–1034 (2014).
Sarner, M. & Cotton, P. B. Classification of pancreatitis. Gut 25, 756–759 (1984).
Buchler, M. W., Martignoni, M. E., Friess, H. & Malfertheiner, P. A proposal for a new clinical classification of chronic pancreatitis. BMC Gastroenterol. 9, 93 (2009).
Catalano, M. F. et al. EUS-based criteria for the diagnosis of chronic pancreatitis: the Rosemont classification. Gastrointest. Endosc. 69, 1251–1261 (2009).
Schneider, A., Lohr, J. M. & Singer, M. V. The M-ANNHEIM classification of chronic pancreatitis: introduction of a unifying classification system based on a review of previous classifications of the disease. J. Gastroenterol. 42, 101–119 (2007).
Budde, C. et al. Development of a prognostic scoring system for chronic pancreatitis (CPSS). Pancreatology 14 (Suppl. 1), S15 (2014).
Maisonneuve, P. et al. Cigarette smoking accelerates progression of alcoholic chronic pancreatitis. Gut 54, 510–514 (2005).
Layer, P. et al. The different courses of early- and late-onset idiopathic and alcoholic chronic pancreatitis. Gastroenterology 107, 1481–1487 (1994).
Alexakis, N. et al. Influence of opioid use on surgical and long-term outcome after resection for chronic pancreatitis. Surgery 136, 600–608 (2004).
Beger, H. G., Schlosser, W., Friess, H. M. & Buchler, M. W. Duodenum-preserving head resection in chronic pancreatitis changes the natural course of the disease: a single-center 26-year experience. Ann. Surg. 230, 512–519; discussion 519–523 (1999).
Lieb, J. G. 2nd & Forsmark, C. E. Review article: pain and chronic pancreatitis. Aliment. Pharmacol. Ther. 29, 706–719 (2009).
Delhaye, M. et al. Belgian consensus on chronic pancreatitis in adults and children: statements on diagnosis and nutritional, medical, and surgical treatment. Acta Gastroenterol. Belg. 77, 47–65 (2014).
Dumonceau, J. M. et al. Endoscopic treatment of chronic pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy 44, 784–800 (2012).
Talukdar, R., Murthy, H. V. & Reddy, D. N. Role of methionine containing antioxidant combination in the management of pain in chronic pancreatitis: a systematic review and meta-analysis. Pancreatology 15, 136–144 (2015).
Winstead, N. S. & Wilcox, C. M. Clinical trials of pancreatic enzyme replacement for painful chronic pancreatitis — a review. Pancreatology 9, 344–350 (2009).
Bhardwaj, P. et al. A randomized controlled trial of antioxidant supplementation for pain relief in patients with chronic pancreatitis. Gastroenterology 136, 149–159.e2 (2009).
Burton, F. et al. Use and perceived effectiveness of non-analgesic medical therapies for chronic pancreatitis in the United States. Aliment. Pharmacol. Ther. 33, 149–159 (2011).
Jadad, A. R. & Browman, G. P. The WHO analgesic ladder for cancer pain management. Stepping up the quality of its evaluation. JAMA 274, 1870–1873 (1995).
Wilder-Smith, C. H., Hill, L., Osler, W. & O’Keefe, S. Effect of tramadol and morphine on pain and gastrointestinal motor function in patients with chronic pancreatitis. Dig. Dis. Sci. 44, 1107–1116 (1999).
Drewes, A. M. et al. Differences between opioids: pharmacological, experimental, clinical and economical perspectives. Br. J. Clin. Pharmacol. 75, 60–78 (2013). This review documents the differences between opioids that are normally used to treat pain, and justifies the rationale for opioid rotation and individualized treatment.
Olesen, S. S. et al. Pharmacological pain management in chronic pancreatitis. World J. Gastroenterol. 19, 7292–7301 (2013).
Niemann, T., Madsen, L. G., Larsen, S. & Thorsgaard, N. Opioid treatment of painful chronic pancreatitis. Int. J. Pancreatol. 27, 235–240 (2000).
Olesen, S. S., Bouwense, S. A., Wilder-Smith, O. H., van Goor, H. & Drewes, A. M. Pregabalin reduces pain in patients with chronic pancreatitis in a randomized, controlled trial. Gastroenterology 141, 536–543 (2011).
Fregni, F. et al. Clinical effects and brain metabolic correlates in non-invasive cortical neuromodulation for visceral pain. Eur. J. Pain 15, 53–60 (2011).
Guarner, L., Navalpotro, B., Molero, X., Giralt, J. & Malagelada, J. R. Management of painful chronic pancreatitis with single-dose radiotherapy. Am. J. Gastroenterol. 104, 349–355 (2009).
Issa, Y., Ahmed Ali, U., Bouwense, S. A., van Santvoort, H. C. & van Goor, H. Preoperative opioid use and the outcome of thoracoscopic splanchnicectomy in chronic pancreatitis: a systematic review. Surg. Endosc. 28, 405–412 (2014).
Juel, J. et al. Acupuncture for pain in chronic pancreatitis: a single-blinded randomized crossover trial. Pancreas 46, 170–176 (2017).
Kapural, L., Cywinski, J. B. & Sparks, D. A. Spinal cord stimulation for visceral pain from chronic pancreatitis. Neuromodulation 14, 423–426; discussion 426–427 (2011).
Balliet, W. E. et al. Depressive symptoms, pain, and quality of life among patients with nonalcohol-related chronic pancreatitis. Pain Res. Treat 2012, 978646 (2012).
Gebhardt, S., Heinzel-Gutenbrunner, M. & Konig, U. Pain relief in depressive disorders: a meta-analysis of the effects of antidepressants. J. Clin. Psychopharmacol. 36, 658–668 (2016).
Lindkvist, B., Phillips, M. E. & Dominguez-Munoz, J. E. Clinical, anthropometric and laboratory nutritional markers of pancreatic exocrine insufficiency: prevalence and diagnostic use. Pancreatology 15, 589–597 (2015).
Whitcomb, D. C. et al. Pancrelipase delayed-release capsules (CREON) for exocrine pancreatic insufficiency due to chronic pancreatitis or pancreatic surgery: a double-blind randomized trial. Am. J. Gastroenterol. 105, 2276–2286 (2010).
Dominguez-Munoz, J. E., Iglesias-Garcia, J., Iglesias-Rey, M., Figueiras, A. & Vilarino-Insua, M. Effect of the administration schedule on the therapeutic efficacy of oral pancreatic enzyme supplements in patients with exocrine pancreatic insufficiency: a randomized, three-way crossover study. Aliment. Pharmacol. Ther. 21, 993–1000 (2005).
de la Iglesia-Garcia, D. et al. Efficacy of pancreatic enzyme replacement therapy in chronic pancreatitis: systematic review and meta-analysis. Gut 66, 1354–1355 (2017).
de-Madaria, E. et al. The Spanish Pancreatic Club's recommendations for the diagnosis and treatment of chronic pancreatitis: part 2 (treatment). Pancreatology 13, 18–28 (2013).
Ahmed Ali, U. et al. Endoscopic or surgical intervention for painful obstructive chronic pancreatitis. Cochrane Database Syst. Rev. 3, CD007884 (2015).
Dumonceau, J. M. et al. Treatment for painful calcified chronic pancreatitis: extracorporeal shock wave lithotripsy versus endoscopic treatment: a randomised controlled trial. Gut 56, 545–552 (2007).
Rosch, T. et al. Endoscopic treatment of chronic pancreatitis: a multicenter study of 1000 patients with long-term follow-up. Endoscopy 34, 765–771 (2002).
Seza, K. et al. A long-term controlled trial of endoscopic pancreatic stenting for treatment of main pancreatic duct stricture in chronic pancreatitis. Hepatogastroenterology 58, 2128–2131 (2011).
Ito, T. et al. Evidence-based clinical practice guidelines for chronic pancreatitis 2015. J. Gastroenterol. 51, 85–92 (2016).
Saxena, P. et al. A US multicenter study of safety and efficacy of fully covered self-expandable metallic stents in benign extrahepatic biliary strictures. Dig. Dis. Sci. 60, 3442–3448 (2015).
Schmidt, A. et al. Effective treatment of benign biliary strictures with a removable, fully covered, self-expandable metal stent: a prospective, multicenter European study. United European Gastroenterol. J. 5, 398–407 (2017).
Poley, J. W. et al. A prospective group sequential study evaluating a new type of fully covered self-expandable metal stent for the treatment of benign biliary strictures (with video). Gastrointest. Endosc. 75, 783–789 (2012).
Deviere, J. et al. Successful management of benign biliary strictures with fully covered self-expanding metal stents. Gastroenterology 147, 385–395 (2014).
Cote, G. A. et al. Effect of covered metallic stents compared with plastic stents on benign biliary stricture resolution: a randomized clinical trial. JAMA 315, 1250–1257 (2016).
Lerch, M. M., Stier, A., Wahnschaffe, U. & Mayerle, J. Pancreatic pseudocysts: observation, endoscopic drainage, or resection? Dtsch. Arztebl. Int. 106, 614–621 (2009).
Kraft, M. et al. Successful drainage of recurrent pancreatic pseudocyst via a transpapillary and transpancreatic approach, using a conventional cystotome. Endoscopy 44 (Suppl. 2) E186–E187 (2012).
Stecher, S. S. et al. Delayed severe bleeding complications after treatment of pancreatic fluid collections with lumen-apposing metal stents. Guthttp://dx.doi.org/10.1136/gutjnl-2016-313562 (2017).
Cahen, D. L. et al. Endoscopic versus surgical drainage of the pancreatic duct in chronic pancreatitis. N. Engl. J. Med. 356, 676–684 (2007).
Dite, P., Ruzicka, M., Zboril, V. & Novotny, I. A prospective, randomized trial comparing endoscopic and surgical therapy for chronic pancreatitis. Endoscopy 35, 553–558 (2003).
Cahen, D. L. et al. Long-term outcomes of endoscopic versus surgical drainage of the pancreatic duct in patients with chronic pancreatitis. Gastroenterology 141, 1690–1695 (2011).
Kleeff, J., Friess, H. & Buchler, M. W. Endoscopic versus surgical treatment for chronic pancreatitis. N. Engl. J. Med. 356, 2103; author reply 2103–2104 (2007).
Alexakis, N. et al. Duodenum- and spleen-preserving total pancreatectomy for end-stage chronic pancreatitis. Br. J. Surg. 90, 1401–1408 (2003).
Alexakis, N. et al. Major resection for chronic pancreatitis in patients with vascular involvement is associated with increased postoperative mortality. Br. J. Surg. 91, 1020–1026 (2004).
Jimenez, R. E., Fernandez-del Castillo, C., Rattner, D. W., Chang, Y. & Warshaw, A. L. Outcome of pancreaticoduodenectomy with pylorus preservation or with antrectomy in the treatment of chronic pancreatitis. Ann. Surg. 231, 293–300 (2000).
Muller, M. W. et al. Perioperative and follow-up results after central pancreatic head resection (Berne technique) in a consecutive series of patients with chronic pancreatitis. Am. J. Surg. 196, 364–372 (2008).
Riediger, H. et al. Long-term outcome after resection for chronic pancreatitis in 224 patients. J. Gastrointestinal Surg. 11, 949–959; discussion 959–960 (2007).
Chinnakotla, S. et al. Long-term outcomes of total pancreatectomy and islet auto transplantation for hereditary/genetic pancreatitis. J. Am. Coll. Surg. 218, 530–543 (2014).
Winny, M. et al. Insulin dependence and pancreatic enzyme replacement therapy are independent prognostic factors for long-term survival after operation for chronic pancreatitis. Surgery 155, 271–279 (2014).
Ahmed Ali, U. et al. Clinical outcome in relation to timing of surgery in chronic pancreatitis: a nomogram to predict pain relief. Arch. Surg. 147, 925–932 (2012).
van der Gaag, N. A. et al. Functional and medical outcomes after tailored surgery for pain due to chronic pancreatitis. Ann. Surg. 255, 763–770 (2012).
Ueda, J., Tanaka, M., Ohtsuka, T., Tokunaga, S. & Shimosegawa, T. Surgery for chronic pancreatitis decreases the risk for pancreatic cancer: a multicenter retrospective analysis. Surgery 153, 357–364 (2013).
Kleeff, J. et al. Evidence-based surgical treatments for chronic pancreatitis. Dtsch. Arztebl. Int. 113, 489–496 (2016).
Partington, P. F. & Rochelle, R. E. Modified Puestow procedure for retrograde drainage of the pancreatic duct. Ann. Surg. 152, 1037–1043 (1960).
Beger, H. G., Krautzberger, W. & Gogler, H. Resection of the head of the pancreas (cephalic pancreatectomy) wih conservation of the duodenum in chronic pancreatitis, tumours of the head of the pancreas and compression of the common bile duct (author's transl) [French]. Chirurgie 107, 597–604 (1981).
White, S. A. et al. Pancreas resection and islet autotransplantation for end-stage chronic pancreatitis. Ann. Surg. 233, 423–431 (2001).
Wilson, G. C. et al. Long-term outcomes after total pancreatectomy and islet cell autotransplantation: is it a durable operation? Ann. Surg. 260, 659–665; discussion 665–657 (2014).
Yin, Z., Sun, J., Yin, D. & Wang, J. Surgical treatment strategies in chronic pancreatitis: a meta-analysis. Arch. Surg. 147, 961–968 (2012).
Lu, W. P. et al. A meta-analysis of the long-term effects of chronic pancreatitis surgical treatments: duodenum-preserving pancreatic head resection versus pancreatoduodenectomy. Chinese Med. J. 126, 147–153 (2013).
Jawad, Z. A. et al. Short and long-term post-operative outcomes of duodenum preserving pancreatic head resection for chronic pancreatitis affecting the head of pancreas: a systematic review and meta-analysis. HPB (Oxford) 18, 121–128 (2016).
McHorney, C. A., Ware, J. E. Jr & Raczek, A. E. The MOS 36-Item Short-Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med. Care 31, 247–263 (1993).
Mullady, D. K. et al. Type of pain, pain-associated complications, quality of life, disability and resource utilisation in chronic pancreatitis: a prospective cohort study. Gut 60, 77–84 (2011).
Amann, S. T. et al. Physical and mental quality of life in chronic pancreatitis: a case–control study from the North American Pancreatitis Study 2 cohort. Pancreas 42, 293–300 (2013).
Machicado, J. D. et al. Quality of life in chronic pancreatitis is determined by constant pain, disability/unemployment, current smoking, and associated co-morbidities. Am. J. Gastroenterol. 112, 633–642 (2017).
Fitzsimmons, D. et al. Development of a disease specific quality of life (QoL) questionnaire module to supplement the EORTC core cancer QoL questionnaire, the QLQ-C30 in patients with pancreatic cancer. EORTC Study Group on Quality of Life. Eur. J. Cancer 35, 939–941 (1999).
Wassef, W. et al. Pancreatitis Quality of Life Instrument: a psychometric evaluation. Am. J. Gastroenterol. 111, 1177–1186 (2016).
Fitzsimmons, D. et al. Symptoms and quality of life in chronic pancreatitis assessed by structured interview and the EORTC QLQ-C30 and QLQ-PAN26. Am. J. Gastroenterol. 100, 918–926 (2005).
Pezzilli, R. et al. The quality of life in patients with chronic pancreatitis evaluated using the SF-12 questionnaire: a comparative study with the SF-36 questionnaire. Dig. Liver Dis. 38, 109–115 (2006).
Gardner, T. B. et al. Chronic pancreatitis and its effect on employment and health care experience: results of a prospective American multicenter study. Pancreas 39, 498–501 (2010).
Mokrowiecka, A., Pinkowski, D., Malecka-Panas, E. & Johnson, C. D. Clinical, emotional and social factors associated with quality of life in chronic pancreatitis. Pancreatology 10, 39–46 (2010).
Etemad, B. & Whitcomb, D. C. Chronic pancreatitis: diagnosis, classification, and new genetic developments. Gastroenterology 120, 682–707 (2001).
Wilcox, C. M. et al. Chronic pancreatitis pain pattern and severity are independent of abdominal imaging findings. Clin. Gastroenterol. Hepatol. 13, 552–560 (2015). This negative study highlights that there is no relationship between the morphological changes in chronic pancreatitis and pain pattern, and hence challenges the rationale for endoscopic and surgical treatment.
Pitchumoni, C. S., Glasser, M., Saran, R. M., Panchacharam, P. & Thelmo, W. Pancreatic fibrosis in chronic alcoholics and nonalcoholics without clinical pancreatitis. Am. J. Gastroenterol. 79, 382–388 (1984).
van Geenen, E. J. et al. Smoking is related to pancreatic fibrosis in humans. Am. J. Gastroenterol. 106, 1161–1166 (2011).
Acknowledgements
This work was partially supported by the COST action BM1204 “EUPancreas: An integrated European platform for pancreas cancer research: from basic science to clinical and public health interventions for a rare disease” (eupancreas.com) to J.K., I.E. and J.P.N., and by the National Cancer Institute and the National Institute of Diabetes and Digestive and Kidney Diseases of the US National Institutes of Health under award number U01DK108306 (to D.C.W.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Introduction (D.C.W.); Epidemiology (T.G.); Mechanisms/pathophysiology (V.R. and D.C.W.); Diagnosis, screening and prevention (J.M., I.E. and F.A.); Management (A.M.D., J.K., J.P.N., T.S., M.M.L. and J.E.D.M.); Quality of life (A.M.D.); Outlook (D.C.W.); Overview of the Primer (J.K. and J.P.N.).
Corresponding author
Ethics declarations
Competing interests
A.M.D. has received unrestricted funds for research from Mundipharma, AstraZeneca and Grünenthal, and has participated in advisory boards and received speaker fees from Almirall, Shire, Kyowa Kirin, Norgina and Allergan. V.R. received speaker fees from Laboratoires Mayoly Spindler. J.E.D.M. has received unrestricted grants for research from Abbott Laboratories and Mylan, and has participated in advisory boards and received speaker fees from Abbott Laboratories and Mylan. J.P.N. reports a consulting or advisory role for Boehringer Ingelheim Pharma; Novartis Pharma AG; KAEL GemVax and Astellas, research funding from Taiho Pharma (Japan), PI, paid to institution; KAEL GemVax (Korea), PI, paid to Institution; AstraZeneca, PI, paid to institution; Pharma Nord, PI, paid to institution, and travel expenses from NUCANA, Amgen and Mylan. D.C.W. reports participating in advisory boards for AbbVie, serving as a consultant for Ariel Precision Medicine, Clinical Outcome Solutions and Regeneron, research support from the National Institutes of Health (USA), the Department of Defense (USA), Shire, the Wayne Fusaro Pancreatic Cancer Research Fund and UPMC (USA), and has equity in Ariel Precision Medicine. All other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Kleeff, J., Whitcomb, D., Shimosegawa, T. et al. Chronic pancreatitis. Nat Rev Dis Primers 3, 17060 (2017). https://doi.org/10.1038/nrdp.2017.60
Published:
DOI: https://doi.org/10.1038/nrdp.2017.60
This article is cited by
-
Familial chylomicronemia syndrome: case reports of siblings with deletions of the GPIHBP1 gene
BMC Endocrine Disorders (2024)
-
Pancreatic Endocrine Insufficiency
Current Surgery Reports (2024)
-
Exocrine Pancreatic Insufficiency Dosing Guidelines for Pancreatic Enzyme Replacement Therapy Vary Widely Across Disease Types
Digestive Diseases and Sciences (2024)
-
Single-Cell Sequencing Data Analysis Unveiled HDAC1 as the Therapeutic Target for Chronic Pancreatitis
Molecular Biotechnology (2024)
-
Adherence to pancreatic enzyme replacement therapy among patients with chronic pancreatitis in East China: a mixed methods study
Scientific Reports (2023)