Abstract
Infective endocarditis (IE) is a rare, life-threatening disease that has long-lasting effects even among patients who survive and are cured. IE disproportionately affects those with underlying structural heart disease and is increasingly associated with health care contact, particularly in patients who have intravascular prosthetic material. In the setting of bacteraemia with a pathogenic organism, an infected vegetation may form as the end result of complex interactions between invading microorganisms and the host immune system. Once established, IE can involve almost any organ system in the body. The diagnosis of IE may be difficult to establish and a strategy that combines clinical, microbiological and echocardiography results has been codified in the modified Duke criteria. In cases of blood culture-negative IE, the diagnosis may be especially challenging, and novel microbiological and imaging techniques have been developed to establish its presence. Once diagnosed, IE is best managed by a multidisciplinary team with expertise in infectious diseases, cardiology and cardiac surgery. Antibiotic prophylaxis for the prevention of IE remains controversial. Efforts to develop a vaccine that targets common bacterial causes of IE are ongoing, but have not yet yielded a commercially available product.
Similar content being viewed by others
Introduction
Infective endocarditis (IE) is a disease that affects multiple systems and results from infection, usually bacterial, of the endocardial surface of the heart. It has been recognized as a pathological entity for hundreds of years and as an infectious process since the nineteenth century1. In his landmark 1885 Gulstonian Lectures on malignant endocarditis, Sir William Osler presented a unifying theory in which susceptible patients developed ‘mycotic’ growths on their valves followed by “transference to distant parts of microorganisms” (Ref. 2). The intervening 130 years have witnessed dramatic growth in our understanding of IE as well as fundamental changes in the disease itself. Medical progress, novel at-risk populations and the emergence of antimicrobial resistance have led to new clinical manifestations of IE. In this Primer, we review our current understanding of IE epidemiology, pathophysiology, aspects of diagnosis and clinical care, and speculate on future developments in IE and its management.
Epidemiology
IE is a relatively rare but life-threatening disease. In a systematic review of the global burden of IE, crude incidence ranged from 1.5 to 11.6 cases per 100,000 person-years, with high-quality data available from only ten — mostly high-income — countries3. Untreated, mortality from IE is uniform. Even with the best-available therapy, contemporary mortality rates from IE are approximately 25%4.
Demography
The mean age of patients with IE has increased considerably in the past several decades. For example, the median age of patients with IE presenting to Johns Hopkins Hospital was <30 years in 1926 (Ref. 5). By contrast, more than half of contemporary patients with IE are >50 years of age, and approximately two-thirds of cases occur in men4,6. Multiple factors have contributed to this changing age distribution in high-income countries. First, the cardiac risk factors that predispose patients to IE have shifted in many high-income countries from rheumatic heart disease, which is primarily seen in young adults, to degenerative valvular disease, which is principally encountered in the elderly. Second, the age of the population has increased steadily. Third, the relatively new entity of health care-associated IE, which disproportionately affects older adults, has emerged secondary to the introduction of new therapeutic modalities, such as intravascular catheters, hyperalimentation lines, cardiac devices and dialysis shunts.
Risk factors
Almost any type of structural heart disease can predispose to IE. Rheumatic heart disease was the most frequent underlying lesion in the past, and the mitral valve was the most commonly involved site7. In developed countries, the proportion of cases related to rheumatic heart disease has declined to ≤5% in the past two decades4. However, in developing countries, rheumatic heart disease remains the most common predisposing cardiac condition for IE8.
Prosthetic valves and cardiac devices (such as permanent pacemakers and cardioverter defibrillators) are significant risk factors for IE. Rates of implantation of these devices have increased dramatically in the past several decades. Consequently, prosthetic valves and cardiac devices are involved in a growing proportion of IE cases9. For example, in a recent cohort of 2,781 adults in 25 countries with definite IE, one-fifth had a prosthetic valve and 7% had a cardiac device4.
Congenital heart disease also confers increased risk of IE. In the same study mentioned above4, 12% of the 2,781 patients with definite IE had underlying congenital heart disease. However, because this cohort was assembled largely from referral centres with cardiac surgery programmes, this rate probably overestimates the association between congenital heart disease and IE in the general population. Mitral valve prolapse has been reported as the predominant predisposing structural abnormality in 7–30% of native-valve IE (NVIE) in developing countries10. In one case–control study, mitral prolapse was associated with IE with an odds ratio of 8.2 (95% CI: 2.4–28.4)11. In developed countries, degenerative cardiac lesions assume greatest importance in the 30–40% of patients with IE who do not have known valvular disease12. For example, in an autopsy series, mitral valve annular calcification was noted in 14% of patients with IE who were >65 years of age, which is a higher rate than that of the general population12,13.
Other factors that predispose to IE include injection drug use (IDU), human immunodeficiency virus infection and extensive health care system contact4,6. Health care-associated IE in particular has been rising in the past several decades, especially in developed countries6. For example, one-third of a recent prospective, multinational cohort of 1,622 patients with NVIE and no history of IDU had health care-associated IE14.
Microbiology
As patient risk factors change, the microbiology of IE also shifts. Although streptococci and staphylococci have collectively accounted for approximately 80% of IE cases, the proportion of these two organisms varies by region (Fig. 1) and has changed over time. The emergence of health care-associated IE has been accompanied by an increase in the prevalence of Staphylococcus aureus15 and coagulase-negative staphylococci14,16, whereas the proportion of IE due to viridans group streptococci (VGS) has declined16. Enterococci are the third leading cause of IE and are increasingly linked to health care contact17. Infections that involve Gram-negative and fungal pathogens in IE are rare and are primarily health care-associated when they do occur18,19.
The causative agents of infective endocarditis (IE) differ geographically. Data from Murdoch et al.4. CoNS, coagulase-negative staphylococci; HACEK, Haemophilus spp., Aggregatibacter spp., Cardiobacterium hominis, Eikenella corrodens and Kingella spp.; VGS, viridans group streptococci.
In approximately 10% of cases of IE, blood cultures are negative, most commonly due to patient receipt of antibiotics before the diagnostic work-up. ‘True’ culture-negative IE is caused by fastidious microorganisms that are difficult to isolate with conventional microbiological techniques. Highly specialized assays, such as serological testing and PCR using blood or valve biopsies, can ultimately suggest a causative pathogen in up to 60% of such cases20. Although the aetiology of true culture-negative IE varies with geographical and epidemiological factors, important causes include Coxiella burnetii (the causative agent of Q fever), Bartonella spp., Brucella spp. and Tropheryma whipplei4,20. Specific risk factors, such as contact with livestock or abattoirs (for Brucella spp. and Coxiella burnetii), homelessness or alcoholism (for Bartonella quintana), travel to the Middle East or the Mediterranean or consumption of unpasteurized dairy products (for Brucella spp.), contact with cats (for Bartonella henselae) or extensive health care contact in a patient with a prosthetic valve and negative blood cultures (for Aspergillus spp.) may be useful clues when evaluating potential IE cases.
Mechanisms/pathophysiology
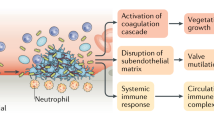
Experimentally, the normal valvular endothelium is resistant to bacterial colonization upon intravascular challenge21. Thus, the development of IE requires the simultaneous occurrence of several independent factors: alteration of the cardiac valve surface to produce a suitable site for bacterial attachment and colonization; bacteraemia with an organism that is capable of attaching to and colonizing valve tissue; and creation of the infected mass or ‘vegetation’ by ‘burying’ of the proliferating organism within a protective matrix of serum molecules (for example, fibrin) and platelets (Fig. 2).
a | Pathogens gain access to the bloodstream, for example, via an intravenous catheter, injection drug use or from a dental source. b | Pathogens adhere to an area of abnormal cardiac valve surface. c | Some pathogens, such as Staphylococcus aureus, obtain intracellular access to the valve endothelium. d | The infected vegetation is created by burying of the proliferating organism within a protective matrix of serum molecules. e | Vegetation particles can detach and disseminate to form emboli. These may lead to complications, such as ischaemic stroke, mycotic aneurysms and infarcts or abscesses at remote sites. IE, infective endocarditis. Figure from Ref. 204, Nature Publishing Group.
Non-bacterial thrombotic endocarditis
As noted above, IE rarely results from intravenous injections of bacteria unless the valvular surface is first perturbed21. In humans, equivalent damage to the valvular surface may result from various factors, including turbulent blood flow related to primary valvular damage from specific systemic disease states (such as rheumatic carditis), mechanical injury by catheters or electrodes, or injury arising from repeated injections of solid particles in IDU. This endothelial damage prompts the formation of fibrin–platelet deposits overlying interstitial oedema21, a pathophysiological entity first termed ‘non-bacterial thrombotic endocarditis’ (NBTE) by Gross and Friedberg in 1936 (Ref. 22). Serial scanning electron micrographs of such damaged valves of animals experimentally challenged with intravenous boluses of bacteria show bacterial adhesion to the NBTE surface within 24 hours following infection. This adhesion is followed by the generation of the fully developed infected vegetation upon further coverage of the bacteria with matrix molecules23.
Transient bacteraemia
Bloodstream infection is a prerequisite for the development of NVIE and probably the bulk of prosthetic-valve IE (PVIE) cases; in the latter setting, intraoperative contamination could account for valve infection. However, the minimum magnitude of bacteraemia (as measured by colony-forming units (CFU) per ml) required to cause IE is not known. Experimental models have typically used inocula of 105–108 CFU per ml either as a bolus dose or by continuous infusion over an extended period24. Low-grade bacteraemia (≥10 and <104 CFU per ml) seems to be common after mild mucosal trauma, such as with dental, gastrointestinal, urological or gynaecological procedures25. Bacteraemia is readily detectable in a majority of patients after dental procedures26 and after common daily activities, such as teeth brushing and chewing25. It is thus likely that low levels of bacteraemia, while commonplace, are usually insufficient to cause IE. In addition, many of the bacterial species present in the blood after mild mucosal trauma are not commonly implicated in cases of IE. For example, complement-mediated bactericidal activity eliminates most Gram-negative pathogens27. By contrast, organisms that are traditionally associated with IE (that is, S. aureus, Staphylococcus epidermidis, VGS, enterococci and Pseudomonas aeruginosa) adhere more readily to canine aortic leaflets in vitro than do pathogens that are less common causes of IE28. Even within the same species, there may be differences in the propensity to cause IE. Specific clonal complexes of S. aureus, for example, are associated with an increased risk of IE29. Similarly, members of the Streptococcus mitis-oralis group predominate as a cause of IE among the many members of VGS30.
Microorganism–NBTE interaction
Once bacteraemia has been established with a typical IE-inducing pathogen, the next step is adherence of the organism to the fibrin–platelet matrices of NBTE. The importance of this step was demonstrated in a study of dental extraction in rats with periodontitis. In this study, group G streptococci were responsible for 83% of IE episodes despite causing a minority of episodes of bacteraemia. In an in vitro model, these organisms were associated with increased adhesion to fibrin–platelet matrices compared with other species31. Adhesion to NBTE is also an important step in fungal IE. Whereas Candida krusei adheres poorly and is a rare cause of IE in humans, Candida albicans adheres to NBTE in vitro, readily produces experimental IE32 and is the most common cause of IE among candidal species19.
Mechanisms of bacterial adherence to the endocardium. Although binding of the pathogen to NBTE seems to be a common step in establishing IE, the mechanism by which this occurs may vary considerably. Some organisms seem to bind to components of damaged endothelium or NBTE, such as fibronectin, laminin and collagen33,34. Other organisms may bind directly to, or be internalized by, endothelial cells. This seems to be an important mechanism by which S. aureus infects cardiac valves35,36 (Fig. 3). In this model, adhesion is mediated by S. aureus-specific surface proteins that bind to fibrinogen, such as clumping factor (Clf) and coagulase37,38. It seems that a ‘cooperativity’ exists between the binding of fibrinogen and fibronectin in the induction of S. aureus IE, in which both adhesins mediate the initial attachment to vegetations, but fibronectin binding is crucial for the persistence of organisms at the valvular endothelial site39. Additional virulence factors, such as α-toxin, then mediate persistence and proliferation within maturing vegetations40.
a | Valve colonization as a consequence of mechanical injury. (1) Non-bacterial thrombotic endocarditis. (2) Bacteria bind to coagulum and colonize it during transient bacteraemia. Adhered monocytes release tissue factor and cytokines. (3) More platelets are attracted and become activated, and the vegetation grows. (4) Endothelial cells are infected and can be lysed by bacterial products or bacteria can persist inside the cells. b | Valve colonization as a consequence of an inflammatory endothelial lesion. (1) Activated endothelial cells express integrins that promote the local deposition of fibronectin; bacteria, such as S. aureus, adhere to this protein. (2) Bacteria are internalized and endothelial cells release tissue factor and cytokines, causing blood clotting and promoting the extension of inflammation and vegetation formation. (3) Infected endothelial cells can be lysed by bacterial products or bacteria can persist inside the cells. IE, infective endocarditis. Figure adapted from Ref. 204, Nature Publishing Group.
In addition, it seems that a key factor in adherence of oral streptococci to NBTE is dextran, which is a complex bacterial-derived extracellular polysaccharide41,42. Other proposed virulence factors that mediate streptococcal adhesion include FimA, which is a surface protein that functions as an adhesin in the oral cavity43,44, the sialic acid-binding adhesin Hsa45 and a phage-encoded bacterial adhesin that mediates a complex interaction between bacteria, fibrinogen and platelets46–48.
Platelet aggregation and evolution of the vegetation. Following bacterial colonization of the valve, the vegetation enlarges by further cycles of fibrin–platelet deposition and bacterial proliferation (Fig. 2). Some strains of bacteria are potent stimulators of platelet aggregation and the platelet release reaction (that is, degranulation)49. In general, IE-producing strains of staphylococci and streptococci more actively aggregate platelets than do other bacteria that less frequently produce IE. Streptococcus sanguinis promotes platelet aggregation via two bacterial cell surface antigens50. S. aureus seems to be able to bind to platelets via platelet-derived von Willebrand factor or directly to the von Willebrand factor receptor51,52.
Although platelets are key components in the pathogenesis of IE, they also have a pivotal role in host defence against organism proliferation within the cardiac vegetation. For example, platelets phagocytose circulating staphylococci into engulfment vacuoles that fuse with α-granules. These α-granules contain antimicrobial peptides called platelet microbicidal proteins (PMPs). Depending on the intrinsic susceptibility of the specific strain of staphylococci to these bactericidal peptides, the organism is either killed within platelets or survives and disseminates using a ‘Trojan Horse’ mechanism53. Platelets also thwart bacterial proliferation within the vegetation by releasing antibacterial PMPs into the local vegetation environment54. Thus, resistance to PMPs (such as in S. aureus) contributes to virulence in IE. Finally, bacteria buried deep within the vegetation may exhibit a state of reduced metabolic activity based on an inability to uptake crucial nutrients55. This altered metabolic state promotes organism survival against selected antibiotics.
The invading microorganism, the endothelium and the monocyte interact in a complex manner in the pathogenesis of IE. After internalization by endothelial cells in vitro, microorganisms such as S. aureus evoke a potent pro-inflammatory chemokine response, including increased expression of IL-6, IL-8 and monocyte chemotactic peptide56. Monocytes are drawn into the endothelial cell microenvironment, where circulating bacteria may then bind directly to their surface, inducing the release of tissue thromboplastin (tissue factor)57. This release amplifies the procoagulant cascade that leads to the progressive evolution of the vegetation. As noted above, this same cascade also induces the antibacterial effect of PMP release by platelets within the vegetation matrix.
Biofilm formation. There is considerable debate concerning the role of ‘biofilm’ formation and the pathogenesis and/or outcomes of IE. It is clear that IE related to implantable cardiac devices can evoke peri-device biofilms. In these scenarios, biofilm formation contributes directly to the evolution of device-associated vegetation propagation. However, the contribution of biofilm formation to NVIE is not established. The most convincing data on the effect of biofilm formation in NVIE come from experimental studies in S. aureus IE. A series of studies over the past decade have linked the ability of S. aureus strains to produce biofilms in vitro and their ability to cause clinically ‘persistent’ methicillin-resistant S. aureus (MRSA) bacteraemia in humans (defined as >7 days of positive blood cultures despite the presence of vancomycin-susceptible isolates and adequate vancomycin treatment regimens)58,59. Of interest, clinically persistent MRSA bacteraemia strains produce significantly more biofilm in vitro when exposed to subinhibitory concentrations of vancomycin than do clinically resolving MRSA isolates58.
Quorum sensing. As IE vegetations contain large densities of organisms, the role of quorum sensing genetic regulation (that is, the regulation of gene expression on the basis of bacterial cell density) of virulence factors has been raised. Most data in this regard emanate from S. aureus, in this case, in the context of the quorum sensing regulon agr (accessory gene regulator). Of interest, in experimental IE, the ability of MRSA strains to evoke activation of agr early in the growth cycle correlates with the ability to cause vancomycin-persistent IE both clinically and in experimental IE. However, based on agr gene knockout studies, the ‘early activation’ profile of agr is, at most, a biomarker for persistent IE strains rather than being directly linked to this outcome pathogenetically58,60.
Immunopathological factors
IE results in stimulation of both humoral and cellular immunity, as manifested by hypergammaglobulinaemia, splenomegaly and the presence of macrophages in the peripheral blood. Several classes of circulating antibodies are produced in response to the continuous bacteraemia that typically characterizes IE. Opsonic antibodies, agglutinating antibodies, complement-fixing antibodies, cryoglobulins and antibodies directed against bacterial heat shock proteins and macroglobulins are produced by the host in an effort to control the ongoing infection61,62.
Effectiveness of antibody responses in IE. Animal studies suggest variable effectiveness of the antibody response to prevent IE. For example, rabbits immunized with heat-killed S. sanguinis plus Freud's adjuvant had a higher ID50 (that is, the S. sanguinis dose required to produce an infection in 50% of rabbits) than non-immunized controls after aortic valve trauma63. Antibodies against cell surface components reduce adhesion of C. albicans to fibrin and platelets in vitro and reduce the incidence of IE in vivo64. By contrast, whole-cell-induced antibodies to S. epidermidis and S. aureus did not prevent IE in immunized animals65. In addition, when administered in conjunction with antibiotic therapy, antibodies specific for the fibrinogen-binding protein ClfA increased bacterial clearance from vegetations66. Moreover, recent data indicate a possible role for vaccination against ClfA for the prevention of IE67, although human studies have not yielded an effective vaccine (see Outlook section).
Pathological antibodies. Rheumatoid factor (which is an anti-IgG IgM antibody) develops in about half of patients with IE of >6 weeks duration68 and decreases with antimicrobial therapy69. Although rheumatoid factor might contribute to pathogenesis by blocking IgG opsonizing activity, stimulating phagocytosis or accelerating microvascular damage, it does not seem to significantly contribute to immune complex glomerulonephritis associated with IE70. Antinuclear antibodies also occur in IE and may contribute to the musculoskeletal manifestations, fever or pleuritic pain71.
Immune complexes. Circulating immune complexes have been found in high titres in almost all patients with IE72. Deposition of immune complexes is implicated in IE-associated glomerulonephritis and may also cause some of the peripheral manifestations of IE, such as Osler's nodes (a skin manifestation of IE) and Roth spots (retinal haemorrhages). Pathologically, these lesions resemble an acute Arthus reaction, in which antigen–antibody complexes are deposited and lead to local vasculitis, although the finding of positive culture aspirates from Osler's nodes in one series suggests that these may be septic embolic phenomena73. Effective treatment leads to a prompt decrease in circulating immune complexes74, whereas therapeutic failures are characterized by rising titres of circulating immune complexes75.
Fastidious bacteria
Some organisms, such as the obligate intracellular pathogens C. burnetii and Bartonella spp., may cause IE by different pathophysiological mechanisms than those outlined above. In the case of C. burnetii, patients show a lack of macrophage activation, promoting intracellular survival of the organism and leading to the histopathological findings of empty or foamy macrophages that are suggestive of IE associated with Q fever76. Specific antibodies are produced, leading to immune complex formation. Affected valves exhibit subendothelial infection with smooth, nodular vegetations that microscopically demonstrate a mixture of fibrin deposits, necrosis and fibrosis without granulomas77.
Organ-specific pathology
As a systemic disease, IE results in characteristic pathological changes in multiple target organs78 (Fig. 4). Portions of the fibrin–platelet matrix of the vegetation may dislodge from the infected heart valve and travel with arterial blood until lodging in a vascular bed downstream from the heart. Such septic emboli can involve almost any organ system in the body and can manifest clinically in several ways. First, if the embolus is large enough to deprive adjacent tissue of oxygen where it lodges, infarction of the dependent tissues can result. This is the pathogenetic process for embolic strokes, myocardial infarctions and infarctions of the kidney, spleen, mesentery and skin. Second, bacteria embedded within the embolus can invade local tissues and create a visceral abscess. Last, extracardiac manifestations may also arise from immune complex deposition or from direct seeding of other tissues as a result of bacteraemia.
a | Roth spots on funduscopic examination. b | CT scans of an embolic stroke with haemorrhagic conversion and a pyogenic brain abscess. c | CT scan of multiple septic pulmonary emboli. d | CT scan of peripheral wedge-shaped splenic infarcts. e | Infarcts affecting multiple fingers. f | Explanted aortic valve leaflet with vegetation and perforation. g | Explanted mitral valve with vegetation. h | Pacemaker lead with vegetation. Ao, aorta; IE, infective endocarditis; LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle. Image in part a courtesy of W. B. Holland, Statesville, North Carolina, USA.
Cardiac manifestations. In the heart, the classic vegetation is usually in the line of closure of a valve leaflet on the atrial surface of atrioventricular valves (the mitral and tricuspid valves) or on the ventricular surface of semilunar valves (the aortic and pulmonary valves). Vegetations vary in size and can reach several centimetres in diameter. The infection may lead to perforation of a valve leaflet or rupture of the chordae tendineae, interventricular septum or papillary muscle. Valve ring abscesses with fistula formation in the myocardium or pericardial sac may result, especially with S. aureus. Finally, myocardial infarctions may occur as an embolic complication of IE, particularly in patients with aortic-valve IE79.
Renal manifestations. In patients with IE, the kidney may develop infarction due to emboli, abscess due to direct seeding by an embolus or an immune complex glomerulonephritis. Renal biopsies performed during active IE are uniformly abnormal, even in the absence of clinically overt renal disease80.
Neurovascular manifestations. Mycotic aneurysms are localized enlargements of arteries caused by infection of the artery wall and may be a feature of acute IE or may be detected months to years after successful treatment81. These aneurysms can arise via one of several mechanisms: direct bacterial invasion of the arterial wall with subsequent abscess formation; embolic occlusion of the vasa vasorum (which are small vessels that supply the walls of larger vessels); or immune complex deposition with resultant injury to the arterial wall81. Mycotic aneurysms tend to arise at bifurcation points, most commonly in the cerebral vessels, although almost any vascular bed can be affected. Cerebral aneurysms may be symptomatic, particularly if bleeding complications arise, but they may also be discovered in patients without neurological symptoms. For example, in one prospective series, 10 out of 130 consecutive patients with IE who underwent screening by cerebral MRI with angiography (a technique called magnetic resonance angiography) had clinically silent cerebral aneurysms82. Approximately 80% of all patients interrogated with magnetic resonance angiography in this study82 showed asymptomatic ‘microbleeds’ in small peripheral cerebral vessels; whether these microbleeds predict future risk of symptomatic intracerebral haemorrhage is unknown83.
Neurological manifestations of IE most frequently arise from cerebral emboli. These are clinically apparent in approximately 20–30% of patients with IE. However, if MRI of asymptomatic patients with IE is routinely undertaken, a majority will have evidence of cerebrovascular complications82,84. The incidence of stroke in IE is 4.82 cases per 1,000 patient-days in the first week of IE and drops rapidly after starting antibiotics85.
Splenic manifestations. Splenic infarcts are frequently found during autopsy of patients who died as a result of IE86 but may also be clinically occult. Splenic abscesses tend to be clinically apparent, with pain, fever and leukocytosis. Splenomegaly is found in about 10% of patients with IE in the developed world4 and is more common in patients with chronic IE (such as that caused by Q fever or VGS) than in acute cases, possibly as a consequence of prolonged immunological response.
Pulmonary manifestations. Thromboembolic showering — in which ‘showers’ of tiny emboli lodge within and occlude small vessels — can lead to the formation of septic pulmonary emboli, either with or without infarction. This phenomenon is a common complication of tricuspid-valve IE or other sources of microemboli, such as central venous catheters, that are located immediately ‘upstream’ of the lungs. Pneumonia, pleural effusions or empyema often accompany septic pulmonary emboli. Although septic pulmonary emboli most commonly appear as peripheral wedge-shaped densities on chest radiographs, rounded ‘cannonball’ lesions that mimic tumours may sometimes develop87.
Skin manifestations. Skin findings in IE include petechiae, cutaneous infarcts, Osler's nodes and Janeway lesions. At the microscopic level, Osler's nodes consist of arteriolar intimal proliferation with extension to venules and capillaries and may be accompanied by thrombosis and necrosis. A diffuse perivascular infiltrate composed of neutrophils and monocytes surrounds the dermal vessels. Immune complexes may be found within the lesions. Janeway lesions are caused by septic emboli and are characterized by the presence of bacteria, neutrophils, necrosis and subcutaneous haemorrhage88.
Ocular manifestations. Patients with IE may have Roth spots in their eyes. These immunological phenomena appear on funduscopic examination as retinal haemorrhages with a pale centre (Fig. 4). Microscopically, they consist of fibrin–platelet plugs or lymphocytes surrounded by oedema and haemorrhage in the nerve fibre layer of the retina89. In addition, direct bacteraemic seeding of the eye may occur, causing endophthalmitis (that is, inflammation of the lining of the intraocular cavities) involving the vitreous and/or aqueous humours90. Endophthalmitis is especially prevalent with S. aureus IE. For instance, in a prospective cohort of patients with S. aureus bacteraemia, 10 out of 23 (43%) patients who had IE also had ocular infection91.
Diagnosis, screening and prevention
Diagnosis
The diagnosis of IE typically requires a combination of clinical, microbiological and echocardiography results. Historically, and as is probably still the case in resource-limited settings, IE was diagnosed clinically based on classic findings of active valvulitis (such as cardiac murmur), embolic manifestations and immunological vascular phenomena in conjunction with positive blood cultures. These manifestations were the hallmarks of subacute or chronic infections, most often in young patients with rheumatic heart disease. However, in the modern era in developed countries, IE is usually an acute disease with few of these hallmarks because the epidemiology has shifted towards health care-associated IE, often with early presentations due to S. aureus. In this context, fever is the most common presenting symptom but is nonspecific4. The presence of other risk factors, such as IDU or the presence of intravascular prosthetic material, should increase the clinical suspicion for IE in a febrile patient. This clinical variability complicates efforts to identify patients with IE who would benefit from early effective antibiotics or cardiac valve surgery. The ability to reliably exclude IE is also important, both to avoid extended courses of unnecessary antibiotics and to focus diagnostic considerations onto other possibilities.
Diagnostic techniques. Blood culture is the most important initial laboratory test in the work-up of IE. Bacteraemia is usually continuous92 and the majority of patients with IE have positive blood cultures4. If antibiotic therapy has been administered before the collection of blood cultures, the rate of positive cultures declines93. Modern blood culture techniques now enable isolation of most pathogens that cause IE. For this reason, practices that were traditionally used to facilitate isolation of fastidious pathogens, such as the use of specific blood culture bottles or extending incubation beyond 5 days, are no longer generally recommended94. In cases of suspected IE when cultures are negative95, other microbiological testing approaches may be useful (Table 1). For example, serological testing is necessary for the diagnosis of Q fever, murine typhus96 and psittacosis97. In addition, Bartonella spp. can be isolated with special culture techniques98 and serological studies may also be helpful for identifying this pathogen. Culture of valvular tissue may yield a causative organism when blood cultures are negative, and microscopy for fastidious or intracellular pathogens may also be diagnostic76,99. Molecular techniques to recover specific DNA or 16S ribosomal RNA from valve tissue100 and blood or serum samples20 have been helpful in selected cases. Other investigative techniques have been reported (Table 1), although they are not widely available.
Echocardiography is the second cornerstone of diagnostic efforts and should be performed in all patients in whom IE is suspected101,102. Transthoracic echocardiography (TTE) may enable visualization of vegetations in many patients (Fig. 5). The sensitivity of TTE is variable103 and is highest in right-sided IE owing to the proximity of the tricuspid and pulmonic valves to the chest wall. Transoesophageal echocardiography (TOE) is more sensitive than TTE for the detection of vegetations and other intracardiac manifestations of IE, especially in the setting of prosthetic valves104. Thus, TTE and TOE are complementary imaging modalities. Both the 2015 European Society of Cardiology (ESC) and the 2015 American Heart Association (AHA) guidelines advocate echocardiography for all cases of suspected IE and encourage TOE for cases in which TTE is negative but suspicion for IE remains. These guidelines diverge regarding TOE in patients with positive TTE results. In this setting, the ESC guidelines recommend subsequent TOE in almost all cases to detect local valvular complications, such as abscess or fistula. By contrast, the AHA guidelines only advocate TOE for patients with a positive TTE if they are thought to be at high risk for such complications. Owing to its relative convenience, TTE is often performed first, although TOE may be the appropriate initial test in a difficult imaging candidate, such as an obese patient or a patient with a prosthetic valve. In addition, the timing of echocardiography is important. Echocardiography findings may be negative early in the disease course. Thus, repeat echocardiography after several days is recommended in patients in whom the initial echocardiography is negative but high suspicion for IE persists101,102. Intraoperative TOE can help to identify local complications and is recommended in all cases of IE that requires surgery. Patients with S. aureus bacteraemia should undergo echocardiography because of the high frequency of IE in this setting. TTE may be adequate in a carefully selected minority of patients who do not have a permanent intracardiac device, have sterile follow-up blood cultures within 4 days after the initial set, are not haemodialysis dependent, have nosocomial acquisition of bacteraemia, do not have secondary foci of infection and do not have clinical signs of IE105. To differentiate patients with S. aureus bacteraemia who are at high risk of IE from those at low risk, several scoring systems have been proposed106–110, although none have been prospectively evaluated.
a | Transthoracic echocardiography demonstrating native mitral valve vegetation. b | Cardiac MRI (systolic frame) demonstrating vegetations in the sub-tricuspid valve chordal apparatus with an adherent thrombus (white asterisk) and a posterior mitral valve leaflet (black asterisk). c | In this patient, infection of a prosthetic aortic valve was suspected but echocardiography was inconclusive. Using PET-CT, inflammatory leukocytes are visualized after taking up radiolabeled glucose, showing an area of active infection on the aortic valve (blue circle). Ao, aorta; IE, infective endocarditis; LV, left ventricle; Veg, vegetation. Image in part b courtesy of A. L. Crowley, Duke University School of Medicine, Durham, North Carolina, USA, and image in part c courtesy of N. E. Bruun, Gentofte Hospital, Copenhagen, Denmark.
Other imaging modalities have been evaluated for the diagnosis of IE in a preliminary manner. These include 3D TOE, cardiac CT, cardiac MRI (Fig. 5; Supplementary information S1 ((video)) and 18F-fluorodeoxyglucose PET-CT111–113 (Fig. 5). The use of multimodality imaging is likely to increase in the future if additive benefits can be demonstrated, and the 2015 ESC guidelines have incorporated these modalities into the diagnostic algorithm of PVIE102.
Diagnostic criteria. The original114 and subsequently modified Duke criteria115 provide the current gold-standard diagnostic strategy, which is both sensitive and specific for IE. The original Duke criteria were evaluated in multiple studies116–120 from geographically and clinically diverse populations, confirming their high sensitivity and specificity.
The modified Duke criteria stratify patients with suspected IE into three categories — ‘definite’, ‘possible’ and ‘rejected’ IE — on the basis of major and/or minor criteria (Box 1). Microbiological criteria form the first major criterion, with diagnostic weight accorded to bacteraemia with pathogens that typically cause IE. For organisms with a weaker association with IE, persistently positive blood cultures are required. The second major criterion is evidence of endocardial involvement, as demonstrated by echocardiography or findings of new valvular regurgitation. Minor criteria include a predisposing heart condition or IDU, fever, vascular phenomena, immunological phenomena or microbiological evidence that does not meet a major criterion.
Thus, IE diagnosis cannot be made on the basis of a single symptom, sign or diagnostic test. Rather, the diagnosis requires clinical suspicion, most commonly triggered by systemic illness in a patient with risk factors, followed by evaluation according to the diagnostic schema outlined in the modified Duke criteria. It is worth keeping in mind that the Duke criteria were originally developed to facilitate epidemiological and clinical research efforts and that the application of the criteria to the clinical practice setting is more difficult. The heterogeneity of patient presentations necessitates clinical judgement in addition to application of the criteria. Moreover, the criteria may be further modified as evidence accrues for new microbiological and imaging modalities. The 2015 ESC diagnostic algorithm has incorporated additional multimodal imaging (such as cardiac CT, PET-CT or leukocyte-labelled single-photon emission CT) for the challenging situation of ‘possible’ or ‘rejected’ PVIE by modified Duke criteria but with a persisting high level of suspicion for IE102.
Prevention
The substantial morbidity and mortality of IE has inspired efforts to prevent its occurrence in at-risk individuals. These prevention efforts have historically focused on oral health because VGS are normal oral flora and cause approximately 20% of IE cases16. Based on the assumption that dental procedures may lead to IE in patients with underlying cardiac disease, the AHA and other major society guidelines previously recommended prophylactic antibiotic therapy to prevent IE in patients with underlying cardiac conditions who underwent dental procedures121. More recently, however, this recommendation has come into question. There is now substantial evidence that transient bacteraemia is common with normal daily activities, including tooth brushing, flossing and chewing food, and the efficacy of antimicrobial prophylaxis is unknown25. In a departure from previous guidance, the 2002 French IE prophylaxis guidelines were the first to dramatically reduce dental prophylaxis indications122. The 2007 AHA guidelines reduced the recommended scope of cardiac conditions for which dental prophylaxis is reasonable to four clinical settings: patients with prosthetic valves or valve material; patients who had previous IE; patients with a subset of congenital heart disease; and cardiac transplantation recipients who develop cardiac valvulopathy121. Prophylaxis is no longer recommended for gastrointestinal or genitourinary procedures. Guidelines from the ESC similarly recommended using dental prophylaxis only for those with highest risk of developing IE102. Recommendations of the UK National Institute for Health and Clinical Excellence (NICE) were published in 2008 and were even more restrictive, advising against IE prophylaxis for any dental, gastrointestinal, genitourinary or respiratory tract procedures123.
Subsequent to the 2008 NICE guidelines, there was a highly significant 78.6% reduction in prescribing of antibiotic prophylaxis before dental procedures in the United Kingdom. With 2 years of follow-up data after guideline publication, there did not seem to be an appreciable increase in IE cases or deaths124. Similarly reassuring data were reported following the introduction of the revised French and AHA guidelines16,125. However, poor adherence to the AHA guidelines complicates interpretation of these results in the United States126.
Recently, increasing concerns have accompanied the availability of longer durations of follow-up. After extending the follow-up period in England through 2013, the number of IE cases seemed to increase significantly over the projected historical trend, leading to an estimated additional 35 more cases per month than would have been expected if prior prophylaxis rates had continued127. The increase was seen in patients in all risk categories as defined in the AHA and the ESC guidelines. However, this study did not contain organism-specific data, so it was not possible to tell whether this increase was due to VGS — which might plausibly have been prevented by dental prophylaxis — or to other pathogens, such as S. aureus. Subsequently, in the United States, a retrospective review of 457,052 IE-related hospitalizations in the Nationwide Inpatient Sample database suggested a similar trend, with an increase in IE hospitalization rates, both overall and due to organisms categorized as ‘streptococcal’, after the release of the new guidelines128. However, it was noted that enterococci were included in the streptococcal category and that the apparent increase in streptococcal IE might thus be owing to rising enterococcal IE rates129.
To date, at least nine population-based studies have examined IE rates before and after guideline changes (Table 2). Taken together, these data indicate that there may be both efficacy and risk (in the form of antibiotic-related adverse events) associated with antibiotic prophylaxis. Importantly, all of the available evidence derives from observational cohorts, with imprecise microbiological data. Furthermore, even if IE rates did increase following guideline changes, a causal relationship cannot be established. No prospective randomized controlled studies to assess the efficacy of prophylaxis have been performed, despite calls for such a trial for at least the past 25 years130. In a 2015 review of the prior 2008 guidelines, the NICE elected not to change any of the prior recommendations and reiterated the need for a randomized controlled trial comparing prophylaxis with no prophylaxis, including long-term follow-up for incident IE131.
In addition to dental prophylaxis, efforts at prevention of intravascular catheter-related bacteraemia may also reduce the incidence of health care-associated IE. Bacteraemia rates are reduced by quality improvement interventions, such as care bundles or checklists that consist of strict hand hygiene, use of full-barrier precautions during the insertion of central catheters, cleaning the skin with chlorhexidine, avoiding the femoral site if possible and removing unnecessary catheters132. Confirmatory data on the effect of these interventions on IE incidence are not available.
Management
In the modern era, management of IE typically requires a multidisciplinary team, including, at a minimum, an infectious disease specialist, a cardiologist and a cardiac surgeon133. All patients should receive antimicrobial therapy and a subset may benefit from cardiovascular surgical intervention.
General principles of antimicrobial therapy
The primary purpose of antimicrobial therapy is to eradicate infection. Several characteristics of infected vegetations pose particular challenges in this regard55, including high bacterial density (also called the ‘inoculum effect’)134, slow rates of bacterial growth in biofilms and low microorganism metabolic activity135. As a result, extended courses of parenteral therapy with bactericidal (or fungicidal) agents are typically required.
Duration of therapy. The duration of therapy must be sufficient to completely eradicate microorganisms within cardiac vegetations. Owing to poor penetration of antibiotics into these vegetations and the slowly bactericidal properties of some of the commonly used drugs (such as vancomycin), extended courses of antibiotics are usually required. When bactericidal activity is rapid, shorter courses may be feasible. For example, combination therapy with penicillin or ceftriaxone and an aminoglycoside is synergistic for VGS-associated IE, enabling effective courses as short as 2 weeks for susceptible strains101. Right-sided vegetations tend to have lower bacterial densities than left-sided vegetations and may also be amenable to shorter course therapy.
The duration of antimicrobial therapy is generally calculated from the first day on which blood cultures are negative. Blood cultures should be obtained every 24–72 hours until it is demonstrated that the bloodstream infection has cleared101,102. If operative valve tissue cultures are positive, an entire antimicrobial course should be considered following cardiovascular surgery.
Selection of the appropriate antimicrobial agent. Therapy should be targeted to the organism identified in blood cultures or serological studies. While awaiting microbiological results, an empirical regimen may be selected based on epidemiological and patient demographic features. As most IE cases are caused by Gram-positive bacteria, vancomycin is often an appropriate empirical choice. However, other empirical agents may also be appropriate based on local microbiology and susceptibility patterns. Detailed recommendations for antimicrobial treatment of specific pathogens are comprehensively addressed in recent treatment guidelines101,102,136. Key points are summarized in Table 3.
Considerations for prosthetic valves and implantable cardiac devices. For NVIE, treatment duration ranges from 2 weeks to 6 weeks, whereas a treatment duration of 6 weeks is usually used for PVIE. The antibiotics used for NVIE and PVIE are typically the same, with the exception of staphylococcal PVIE, for which the addition of rifampin and gentamicin is recommended.
Infections of cardiac implantable electronic devices (such as pacemakers and defibrillators) may occur with or without associated valvular IE137. Regardless of whether infection seems to involve the device lead alone (which is sometimes termed ‘lead endocarditis’), the valve alone, or both, complete device and lead removal is recommended138. There are limited clinical data to inform the optimal duration of antibiotic therapy for cardiac device infections; at least 4–6 weeks, using the same antibiotics as for valvular IE, are recommended for lead endocarditis138.
Organism-specific considerations
Staphylococci. The crucial distinction in selecting antibiotic therapy for S. aureus-associated IE is whether the isolate is methicillin-resistant (MRSA) or methicillin-susceptible (MSSA). Anti-staphylococcal β-lactam antibiotics are recommended whenever possible for MSSA-associated IE, as data from observational studies indicate worse outcomes for patients with MSSA bloodstream infections who are treated with vancomycin105,139. Whether it is necessary to use a β-lactam antibiotic as empirical therapy is unclear; small retrospective studies have suggested a potential benefit140. A more recent cohort study among >5,000 patients with MSSA bacteraemia suggested that β-lactams are superior for definitive therapy once MSSA has been identified, but not for empirical treatment139. Providers might avoid prescribing β-lactams to patients with reported penicillin allergies. However, among patients with a reported penicillin allergy, most do not have a true allergy when skin testing is performed141, and skin testing seemed to be cost-effective in decision analyses for treating MSSA bactaeremia142 and IE143.
For MRSA IE, vancomycin has historically been the antibiotic of choice and it remains a first-line therapy in treatment guidelines101,102,136,144. Recent reports have raised the concern that after decades of use, the vancomycin minimum inhibitory concentration (MIC) for S. aureus might be rising145. Increased vancomycin MICs, even among isolates still classified as susceptible, might be associated with worse outcomes in MRSA bacteraemia, although meta-analyses have reached different conclusions146,147. In a prospective cohort of 93 patients with left-sided MSSA IE who were treated with cloxacillin, high vancomycin MIC (≥1.5 mg per l) was associated with increased mortality, even though these patients did not receive vancomycin148. In light of this finding, it seems that a higher vancomycin MIC may be a surrogate marker for host-specific or pathogen-specific factors that lead to worse outcomes. Clinicians may consider the use of an alternative antibiotic for MRSA IE with a vancomycin MIC of ≥1.5 mg per l, but data are lacking to support a mortality benefit for alternative approaches. Ultimately, the clinical response of the patient should determine the continued use of vancomycin, independent of the MIC144.
Daptomycin is approved by the US FDA for the treatment of adults with S. aureus bactaeraemia and right-sided IE and is an alternative to vancomycin for MRSA IE101. The FDA-approved dose for IE is 6 mg per kg per day, but many authorities use higher doses (such as 8–10 mg per kg per day) owing to concerns for treatment-emergent resistance, which occurred in approximately 5% of patients (7 out of 120 daptomycin-treated patients) in the phase III trial comparing daptomycin to standard therapy for S. aureus bacteraemia and IE149. Daptomycin seems to be safe and effective at these higher doses150–152.
Gentamicin is not recommended for staphylococcal NVIE101 because it is associated with nephrotoxicity and does not have robust data to support clinical benefit153. Similarly, rifampin is also not recommended as an adjunct therapy for NVIE101 because it has been associated with adverse effects154 and prolonged bacteraemia155 and should be avoided in staphylococcal NVIE unless there is another indication for its use, such as concurrent osteoarticular infection. For staphylococcal PVIE, weak evidence supports the use of both gentamicin and rifampin156. A large trial examining the role of adjunctive rifampin for S. aureus bacteraemia has recently completed enrolment157.
Observational data have been reported for other antibiotic combinations. For example, ceftaroline is a cephalosporin antibiotic active against MRSA and has been used as salvage therapy for IE alone or in combination with other anti-staphylococcal antibiotics158,159. Other combinations have shown in vitro synergy and have limited human data in MRSA bacteraemia, such as vancomycin or daptomycin paired with β-lactams or with trimethoprim-sulfamethoxazole, daptomycin plus fosfomycin, or fosfomycin combined with β-lactams160,161. Recommended treatment regimens for coagulase-negative staphylococci are the same as those for S. aureus101,102.
Streptococci. Standard treatment for streptococcal IE is a β-lactam antibiotic (such as penicillin, amoxicillin or ceftriaxone) for 4 weeks. The addition of an aminoglycoside may enable a shorter 2-week course of therapy when administered once daily in combination with ceftriaxone for streptococcal NVIE101,162. For streptococcal isolates with an increased penicillin or ceftriaxone MIC, gentamicin should be added101.
Enterococci. From the early days of the antibiotic era, clinicians noted that penicillin worked less well for enterococci than for streptococci and combination therapy with an aminoglycoside was therefore recommended163. Although this has remained the standard approach, increasing rates of aminoglycoside resistance and the toxicity associated with this class of antibiotics have spurred efforts to find alternative therapeutic options.
Recent data indicate that the combination of ampicillin and ceftriaxone may be effective for IE due to ampicillin-susceptible Enterococcus faecalis, particularly in patients with aminoglycoside resistance, or in whom there is concern for nephrotoxicity with an aminoglycoside164,165. Vancomycin-resistant enterococcal IE is fortunately rare but has been successfully treated with linezolid166 and daptomycin152. If daptomycin is used, high-dose therapy may be considered101.
Other organisms. HACEK group organisms (Haemophilus spp., Aggregatibacter spp., Cardiobacterium hominis, Eikenella corrodens and Kingella spp.) were historically treated with ampicillin. However, β-lactamase-producing strains are increasingly problematic and susceptibility testing may fail to identify these strains167. Thus, HACEK organisms should be considered ampicillin-resistant and ceftriaxone is the preferred treatment. A duration of 4 weeks of therapy is generally sufficient for these organisms101.
IE due to non-HACEK Gram-negative bacilli is rare18. Consequently, optimal management strategies are not defined. Cardiac surgery combined with prolonged antibiotic therapy is considered a reasonable strategy in many cases101.
Fungal IE is also rare but outcomes are poor. Valve surgery is often used, but this approach is not clearly associated with improved outcomes168. Following initial parenteral therapy with an amphotericin-based regimen or an echinocandin, indefinite azole therapy is recommended, particularly if valve surgery is not performed169,170.
Culture-negative IE. Culture-negative IE cases are particularly challenging to manage. Although sterile blood cultures are most commonly due to patient receipt of antibiotics before obtaining blood cultures, they may also arise from inadequate microbiological techniques, infection with fastidious organisms or non-infectious causes of valvular vegetations, such as marantic or Libman–Sacks IE. Choosing an antibiotic regimen in these cases requires balancing the need for empirical therapy for all the likely pathogens with the potential adverse effects of using multiple antibiotics. Investigation for ‘true’ culture-negative IE (that is, for uncommon pathogens that do not grow in routine blood cultures) may yield an aetiology in these cases.
Surgery
The rate of early valve replacement or repair has increased over time4 in keeping with the prevailing opinion that surgery is a key component of the management of many complicated IE cases. However, the evidence base for this practice is decidedly mixed. A single randomized trial demonstrated a significant reduction in the composite outcome of in-hospital deaths and embolic events with early surgery171. While clearly transformational, study generalizability was nonetheless questioned. Study subjects were younger, healthier and infected with less-virulent pathogens (for example, VGS) than contemporary patients with IE encountered in general practice172. For most patients with IE, recommendations for surgery are based on observational studies and expert opinion.
The principal consensus indications for valve surgery are heart failure, uncontrolled infection and prevention of embolic events in patients at high risk of IE. Uncontrolled infection may be related to paravalvular complications, such as abscess, an enlarging vegetation or dehiscence of a prosthetic valve. In addition, uncontrolled infection may be manifested by ongoing systemic illness with persistent fevers and positive blood cultures despite appropriate antibiotic therapy. As larger left-sided vegetations are more likely to lead to embolic events, IE with a vegetation of >10 mm in length is a relative indication for surgical intervention.
The timing of cardiac surgery for patients with IE and neurovascular complications remains controversial. A large prospective cohort study of 857 patients with IE complicated by ischaemic stroke without haemorrhagic conversion found that no patient benefit was gained from delaying surgery173. By contrast, patients with embolic stroke complicated by haemorrhagic conversion sustained higher mortality when surgery was performed within 4 weeks of the haemorrhagic event than with later surgery (75% versus 40%, respectively)174. On the basis of these observational data, the AHA currently recommends that valve surgery may be considered in patients with IE who also have stroke or subclinical cerebral emboli without delay if intracranial haemorrhage has been excluded by imaging studies and neurological damage is not severe (such as coma). In patients with major ischaemic stroke or intracranial haemorrhage, AHA guidelines currently state that delaying valve surgery for at least 4 weeks is reasonable101.
Valve surgery was traditionally recommended for difficult-to-treat pathogens, such as Pseudomonas aeruginosa, fungal organisms and β-lactam-resistant staphylococci. However, these pathogen-specific recommendations for surgery have been recently called into question in favour of an individualized decision-making approach based on haemodynamic and structural indications168,175.
Other adjunctive therapies
Anticoagulation. Patients with PVIE who are receiving oral anticoagulants may be at an increased risk of death from cerebral haemorrhage176. Antiplatelet therapies are not currently recommended for IE. A single randomized trial examined the role of 325 mg of aspirin daily for patients with IE. The incidence of embolic events was similar between aspirin-treated and placebo-treated patients, and there was a non-significant increase in the rate of cerebral bleeding episodes177. However, there are several limitations to this study, including the dose of aspirin used and the delayed initiation of aspirin. For patients with another indication for antiplatelet therapy, it may be reasonable to continue the antiplatelet agent unless bleeding complications develop. Similarly, it is not recommended to initiate anticoagulant therapy such as warfarin for the purpose of treating IE. In patients with IE who have another indication for anticoagulation therapy, such as a mechanical valve, data are contradictory on whether to continue anticoagulation during acute therapy176,178, and bridging therapy with heparin products has not been studied.
Management of metastatic foci. Metastatic foci of infection frequently complicate IE. As with any infection, recognition of these foci of infection is important so that targeted interventions, such as drainage of abscesses or removal of infected prosthetic material, may be undertaken. This is of crucial importance in patients who require valve surgery because a persistent source of infection may serve as a source from which a recently placed prosthetic valve or annuloplasty ring becomes infected101,102. Some metastatic foci, such as vertebral osteomyelitis, may require additional antibiotic therapy beyond what is typically indicated for IE179. There is currently insufficient evidence to recommend specific imaging strategies to look for metastatic foci in all patients with IE.
Care at completion of therapy
Most patients with IE in the modern era are cured and attention can eventually turn to a follow-up plan. Elements of follow-up may include an echocardiogram at the completion of antimicrobial therapy to establish a new baseline for subsequent comparison, referral to a drug cessation programme for patients with IDUs and a thorough dental evaluation. A comprehensive search for the initial portal of pathogen entry may be undertaken so that this can be addressed to minimize repeat episodes of IE. In a prospective single-centre experience, a systematic search revealed the likely source in 74% of 318 patients180. Routine blood cultures at completion of antibiotic therapy are not recommended given a very low rate of positivity in patients with no signs of active infection. Patients should also be monitored for complications of IE, including relapse, incident heart failure and complications of antibiotic therapy, such as audiological toxicity from aminoglycosides or incident Clostridium difficile infection.
Quality of life
In addition to the stress associated with being diagnosed with a potentially lethal infection, patients with IE routinely experience prolonged hospitalizations and adverse reactions to treatment, and undergo multiple invasive procedures. For instance, treatment for left-sided IE requires extended courses of intravenous antibiotics, which involves long-term venous access and probably diminishes patient quality of life (QOL). To what extent these factors may impair the QOL of patients after they are discharged from the hospital is not well known, as only a few studies have addressed these issues181–183. In addition, life-threatening illness may cause post-traumatic stress disorder (PTSD), which has been shown to impair patient well-being in survivors of various life-threatening infectious diseases.
In one study of QOL in survivors of left-sided NVIE, 55 out of 86 eligible adults completed questionnaires 3 and 12 months after discharge from hospital and 12 more patients completed the 12-month questionnaires only. The health-related QOL was measured using the 36-Item Short-Form Health Survey and the PTSD questionnaires. In this study, 41 out of 55 patients (75%) and 36 out of 67 patients (54%) still had physical symptoms 3 and 12 months after the end of antimicrobial treatment, respectively. The most prevalent symptoms were weakness of the limbs (51%), fatigue (47%) and concentration disorders (35%). One year after discharge, 7 out of 64 patients (11%) were still suffering from PTSD. The 37 patients who were ≤60 years of age at the time of IE were questioned about their employment status. Before IE, 30 (81%) patients were employed and working. At 3 and 12 months, 16 out of 31 patients (52%) and 24 out of 37 patients (65%) were working again, respectively182. Given the low number of patients evaluated, the effect of factors, such as causative microorganisms or valve surgery, on QOL and on the rate of PTSD could not be assessed. In one study conducted in patients without IE who had undergone mitral valve surgery, the type of surgery (replacement versus repair) had no effect on the QOL of patients184.
Whether a comprehensive cardiac rehabilitation programme (which typically involves exercise and information sessions) may improve the QOL of patients surviving IE is currently being explored through a randomized clinical trial (the CopenHeart IE study)185, in which 150 patients treated for left-sided (NVIE or PVIE) or cardiac device IE will be randomized to either cardiac rehabilitation or usual care186.
In a qualitative evaluation of 11 patients recovering from IE, Rasmussen et al.187 described the innovative concept of ‘insufficient living’. Some patients described an ‘altered life’ period as a phase of adaptation to a new life situation, which some perceived as manageable and temporary, whereas others found it to be extremely distressing and prolonged. Patients also described a ‘shocking weakness’ feeling that was experienced physically, cognitively and emotionally. These feelings subsided quickly for a few, whereas most patients experienced a persisting weakness and felt frustrated about the prolonged recovery phase. Finally, patients expressed that support from relatives and health care professionals, as well as one's own actions, were important in facilitating recovery. This original study emphasized the need for research in follow-up care to support a patient's ability to cope with potential physical and psychoemotional consequences of IE187.
Given the scarcity of data on the subject, future studies are needed to define the effect of IE on patient QOL. Potential priorities for future research in IE QOL are listed in Box 2.
Outlook
Treatment
Future treatments for IE will emphasize pragmatism. For example, an effective treatment strategy for left-sided IE that avoids long-term venous access would be an important advance. At least two randomized trials are testing the effectiveness and safety of replacing part of the standard intravenous antibiotic course with a ‘step-down’ strategy to oral antibiotics188,189. In addition, two newly approved anti-staphylococcal antibiotics — dalbavancin and oritavancin — might eventually represent alternatives to the current standard intravenous treatment strategies for IE.
Along these lines, the POET study uses a non-inferiority, multicentre, prospective, randomized, open-label study design to test the hypothesis that partial oral antibiotic treatment is as safe and effective as parenteral therapy in left-sided IE188. A total of 400 stable patients with streptococcal, staphylococcal or enterococcal aortic or mitral valve IE will be randomized to receive a full 4–6 weeks of intravenous antibiotics or to receive oral antibiotics after a minimum of 10 days of parenteral therapy. Patients will be followed up for 6 months after completion of antibiotic therapy. The primary end point is a composite of all-cause mortality, unplanned cardiac surgery, embolic events and relapse of positive blood cultures with the primary pathogen. A non-inferiority margin of 10% is proposed.
The RODEO study, using the same primary end point, will also evaluate the impact of switching to oral therapy for left-sided IE189. In this study, 324 subjects with IE due to MSSA will receive at least 10 days of intravenous antibiotic therapy, then will be randomized to complete a full 4–6 weeks of intravenous therapy or to receive oral levofloxacin and rifampin for at least 14 additional days.
Dalbavancin and oritavancin, which are lipoglycopeptide-class antibiotics that were approved in 2014 by the FDA for the treatment of acute bacterial skin and skin structure infections (ABSSSIs), represent potential improvements to the current options of intravenous therapy for IE. An important property is their extremely long half-life, estimated to be 10–14 days190,191, which allows infrequent administration. Dalbavancin is approved by the FDA for the treatment of ABSSSIs using a single 1,500 mg dose or with a two-dose strategy: a 1,000 mg loading dose on day 1 followed by a 500 mg infusion 1 week later192,193. Oritavancin is approved for the treatment of ABSSSIs as a single 3-hour infusion of 1,200 mg194. These dosing strategies might ultimately avoid the need for home health or skilled nursing facility care for outpatient intravenous antibiotics. Although no data are currently available for the efficacy of such treatment strategies in IE, the pharmacokinetics of dalbavancin dosed at 1,000 mg of dalbavancin on day 1 followed by 500 mg weekly for 7 additional weeks seemed to be favourable in one phase I study190. In addition, dalbavancin was studied in catheter-associated bloodstream infection195. Therapies not requiring extended intravenous access, such as dalbavancin or oritavancin, could be especially advantageous in treating IE in patients with IDU or who have limited options for intravascular line placement.
Vaccines to prevent common bacterial causes of IE
The best way to treat IE is to prevent it. Although most efforts to date on IE prevention have focused on infection control and dental prophylaxis, considerable resources have also been invested in vaccine development that targets common bacterial causes of IE. Success has been mixed and none of these agents is currently commercially available. Nonetheless, future prevention strategies for some causes of IE are likely to include vaccines. Although vaccine candidates for pathogens such as VGS196 and C. albicans197 have been evaluated in animal models, human studies in vaccines that target the causes of IE have been primarily limited to P. aeruginosa, group B streptococcus and S. aureus.
Passive immunization strategies for staphylococcal infections. At least ten studies have tested vaccines or immunotherapeutics for the prevention or treatment of S. aureus infections, including bacteraemia (Table 4). Efforts to date have pursued two approaches: passive immunization with existing antibodies or active immunization by stimulating a host antibody response in a classic vaccine design. Two passive immunization strategies have been attempted: treatment of active staphylococcal infections as an adjunct in addition to standard treatment; and prevention of staphylococcal infections in patients deemed to be at high risk of developing infection. Each approach has strengths and limitations. Treatment strategies provide the design advantage of a relatively small sample size and relative ease of enrolment owing to provision of standard of care treatment in both arms, but will require demonstrating superiority over standard of care therapy for FDA approval. Although three immunotherapeutic compounds to date have been evaluated as treatment adjuncts in patients with S. aureus infection, none has demonstrated efficacy. A fourth compound, 514G3, is currently undergoing evaluation in a phase II safety and efficacy study in hospitalized patients with S. aureus bacteraemia198.
Three passive immunotherapeutic compounds have undergone clinical trials to prevent staphylococcal infections (aimed at both S. aureus and S. epidermidis) in neonates. None exhibited significant protection. Pagibaximab, a humanized murine chimeric monoclonal antibody that targets lipoteichoic acid in the cell wall of S. aureus, has shown an encouraging trend in outcomes in a phase II study but no significant protective effect in the registrational trial.
Active immunization strategies for staphylococcal infections. Two S. aureus vaccine candidates have been evaluated in phase III trials as active immunizations for S. aureus. A third registrational trial is underway199. All three trials focus on specific adult populations at high risk for S. aureus infection, including those undergoing haemodialysis (in the StaphVAX vaccine trial), cardiac surgery (in the V710 vaccine trial) and spinal surgery (the SA4Ag vaccine trial).
StaphVAX (Nabi Biopharmaceuticals, Rockville, Maryland, USA) is a bivalent vaccine of capsular polysaccharides 5 and 8 that was tested in 1,804 patients with a primary fistula or synthetic graft vascular access undergoing haemodialysis. Although receipt of StaphVAX was associated with a significant reduction in rates of S. aureus bacteraemia at 40 weeks post-vaccination (efficacy: 57%; P = 0.02), the study failed to demonstrate significantly reduced rates of S. aureus bacteraemia at the prespecified end point of 54 weeks post-vaccination200. Thus, a second trial of StaphVAX in 3,600 patients undergoing haemodialysis was undertaken. In this second study, the primary efficacy end point was set at 6 months. Unfortunately, this unpublished trial also failed to demonstrate protection against the development of S. aureus bacteraemia.
V710 is a vaccine that targets the cell wall constitutive iron-regulatory protein IsdB and was tested in patients undergoing median sternotomy. The study was terminated after approximately 8,000 patients were enrolled owing to lack of efficacy and also to a higher rate of multiple organ system failure-related deaths among patients who received V710. In post-hoc analyses, patients that received V710 and subsequently became infected with S. aureus were approximately five-times more likely to die than patients that received control and then became infected with S. aureus (23 versus 4.2 per 100 person-years)221. The reason for this increased mortality is unknown.
A phase IIb study of the SA4Ag vaccine has been initiated. This study aims to test the efficacy and safety of a vaccine that targets S. aureus infection in patients who are undergoing elective posterior instrumented lumbar spinal fusion surgery199. Unlike previous S. aureus vaccine approaches, this candidate vaccine includes four epitopes: ClfA, manganese transport protein C (MntC) and capsular polysaccharides 5 and 8.
At least two other S. aureus vaccine candidates are in late preclinical development. Candidate vaccine NDV-3 contains the amino-terminal portion of the C. albicans agglutinin-like sequence 3 protein (Als3p) formulated with an aluminium hydroxide adjuvant. Preclinical studies have demonstrated that the Als3p vaccine antigen protects mice from mucocutaneous and intravenous challenge with both C. albicans197 and S. aureus201. The vaccine has been shown to be safe and immunogenic in healthy adults202. Most recently, a multi-subunit vaccine that targets five known S. aureus virulence determinants — α-haemolysin (Hla), ess extracellular A (EsxA), EsxB, and the surface proteins ferric hydroxamate uptake D2 and conserved staphylococcal antigen 1A — was described. When formulated with a novel Toll-like receptor 7-dependent agonist, the five antigens provided high levels of T helper 1-driven protection against S. aureus in animal models203.
Conclusions
Although much has changed since Osler elucidated its fundamental disease mechanisms in the late 1800s, IE remains a disease of high morbidity and mortality with far-reaching effects on the QOL of survivors. In the near term, the epidemiology will continue to reflect the epidemiological and microbiological effect of health care contact. Improved algorithms for the diagnosis of IE will incorporate new microbiological techniques, especially for blood culture-negative cases. We can safely assume that imaging technology will continue to advance, and further research is needed to define which patients with suspected IE should undergo TOE and which patients may benefit from newer imaging modalities. Novel Gram-positive antibiotics are promising but as yet untested in IE. If proven to be effective, they might enable simpler and more-patient-friendly treatment regimens. It is likely that the debate around IE prophylaxis will continue until prophylaxis strategies are compared prospectively. Vaccine development has not yet yielded an effective and commercially available product, but numerous candidates are in the pipeline.
References
Contrepois, A. Towards a history of infective endocarditis. Med. Hist. 40, 25–54 (1996).
Osler, W. The Gulstonian Lectures, on malignant endocarditis. Br. Med. J. 1, 577–579 (1885).
Bin Abdulhak, A. A. et al. Global and regional burden of infective endocarditis, 1990–2010: a systematic review of the literature. Glob. Heart 9, 131–143 (2014).
Murdoch, D. R. et al. Clinical presentation, etiology, and outcome of infective endocarditis in the 21st century: the International Collaboration on Endocarditis-Prospective Cohort study. Arch. Intern. Med. 169, 463–473 (2009). This prospective cohort study of 2,781 adults with definite IE demonstrates that IE had shifted from a subacute disease of younger people with rheumatic valvular abnormalities to one in which the presentation is more acute and is characterized by a high rate of S. aureus infection in patients with previous health care exposure.
Thayer, W. Studies on bacterial (infective) endocarditis. Johns Hopkins Hosp. Rep. 22, 1–8 (1926).
Fowler, V. G. Jr et al. Staphylococcus aureus endocarditis: a consequence of medical progress. JAMA 293, 3012–3021 (2005). This prospective cohort study of 1,779 patients with definite IE demonstrates that S. aureus is the leading cause of IE in many regions of the world.
Rabinovich, S., Evans, J., Smith, I. M. & January, L. E. A long-term view of bacterial endocarditis. 337 cases 1924 to 1963. Ann. Intern. Med. 63, 185–198 (1965).
Watt, G. et al. Prospective comparison of infective endocarditis in Khon Kaen, Thailand and Rennes, France. Am. J. Trop. Med. Hyg. 92, 871–874 (2015).
Greenspon, A. J. et al. 16-year trends in the infection burden for pacemakers and implantable cardioverter-defibrillators in the United States 1993 to 2008. J. Am. Coll. Cardiol. 58, 1001–1006 (2011).
Thiene, G. & Basso, C. Pathology and pathogenesis of infective endocarditis in native heart valves. Cardiovasc. Pathol. 15, 256–263 (2006).
Clemens, J. D., Horwitz, R. I., Jaffe, C. C., Feinstein, A. R. & Stanton, B. F. A controlled evaluation of the risk of bacterial endocarditis in persons with mitral-valve prolapse. N. Engl. J. Med. 307, 776–781 (1982).
Kaye, D. Changing pattern of infective endocarditis. Am. J. Med. 78, 157–162 (1985).
Movahed, M. R., Saito, Y., Ahmadi-Kashani, M. & Ebrahimi, R. Mitral annulus calcification is associated with valvular and cardiac structural abnormalities. Cardiovasc. Ultrasound 5, 14 (2007).
Benito, N. et al. Health care-associated native valve endocarditis: importance of non-nosocomial acquisition. Ann. Intern. Med. 150, 586–594 (2009).
Federspiel, J. J., Stearns, S. C., Peppercorn, A. F., Chu, V. H. & Fowler, V. G. Jr Increasing US rates of endocarditis with Staphylococcus aureus: 1999–2008. Arch. Intern. Med. 172, 363–365 (2012).
Duval, X. et al. Temporal trends in infective endocarditis in the context of prophylaxis guideline modifications: three successive population-based surveys. J. Am. Coll. Cardiol. 59, 1968–1976 (2012).
Pericas, J. M. et al. Enterococcal endocarditis revisited. Future Microbiol. 10, 1215–1240 (2015).
Morpeth, S. et al. Non-HACEK Gram-negative bacillus endocarditis. Ann. Intern. Med. 147, 829–835 (2007).
Baddley, J. W. et al. Candida infective endocarditis. Eur. J. Clin. Microbiol. Infect. Dis. 2 = 7, 519–529 (2008).
Fournier, P. E. et al. Comprehensive diagnostic strategy for blood culture-negative endocarditis: a prospective study of 819 new cases. Clin. Infect. Dis. 51, 131–140 (2010).
Durack, D. T., Beeson, P. B. & Petersdorf, R. G. Experimental bacterial endocarditis. 3. Production and progress of the disease in rabbits. Br. J. Exp. Pathol. 54, 142–151 (1973).
Gross, L. & Friedberg, C. K. Nonbacterial thrombotic endocarditis: classification and general description. Arch. Intern. Med. 58, 620–640 (1936).
McGowan, D. A. & Gillett, R. Scanning electron microscopic observations of the surface of the initial lesion in experimental streptococcal endocarditis in the rabbit. Br. J. Exp. Pathol. 61, 164–171 (1980).
Veloso, T. R. et al. Use of a human-like low-grade bacteremia model of experimental endocarditis to study the role of Staphylococcus aureus adhesins and platelet aggregation in early endocarditis. Infect. Immun. 81, 697–703 (2013).
Forner, L., Larsen, T., Kilian, M. & Holmstrup, P. Incidence of bacteremia after chewing, tooth brushing and scaling in individuals with periodontal inflammation. J. Clin. Periodontol. 33, 401–407 (2006).
Lockhart, P. B. The risk for endocarditis in dental practice. Periodontol 2000 23, 127–135 (2000).
Durack, D. T. & Beeson, P. B. Protective role of complement in experimental Escherichia coli endocarditis. Infect. Immun. 16, 213–217 (1977).
Gould, K., Ramirez-Ronda, C. H., Holmes, R. K. & Sanford, J. P. Adherence of bacteria to heart valves in vitro. J. Clin. Invest. 56, 1364–1370 (1975).
Nienaber, J. J. et al. Methicillin-susceptible Staphylococcus aureus endocarditis isolates are associated with clonal complex 30 genotype and a distinct repertoire of enterotoxins and adhesins. J. Infect. Dis. 204, 704–713 (2011).
Simmon, K. E. et al. Phylogenetic analysis of viridans group streptococci causing endocarditis. J. Clin. Microbiol. 46, 3087–3090 (2008).
Moreillon, P., Overholser, C. D., Malinverni, R., Bille, J. & Glauser, M. P. Predictors of endocarditis in isolates from cultures of blood following dental extractions in rats with periodontal disease. J. Infect. Dis. 157, 990–995 (1988).
Scheld, W. M., Calderone, R. A., Alliegro, G. M. & Sande, M. A. Yeast adherence in the pathogenesis of Candida endocarditis. Proc. Soc. Exp. Biol. Med. 168, 208–213 (1981).
Scheld, W. M., Strunk, R. W., Balian, G. & Calderone, R. A. Microbial adhesion to fibronectin in vitro correlates with production of endocarditis in rabbits. Proc. Soc. Exp. Biol. Med. 180, 474–482 (1985).
Lowrance, J. H., Baddour, L. M. & Simpson, W. A. The role of fibronectin binding in the rat model of experimental endocarditis caused by Streptococcus sanguis. J. Clin. Invest. 86, 7–13 (1990).
Hamill, R. J., Vann, J. M. & Proctor, R. A. Phagocytosis of Staphylococcus aureus by cultured bovine aortic endothelial cells: model for postadherence events in endovascular infections. Infect. Immun. 54, 833–836 (1986).
Yao, J., Bone, R. C. & Sawhney, R. S. Differential effects of tumor necrosis factor-alpha on the expression of fibronectin and collagen genes in cultured bovine endothelial cells. Cell. Mol. Biol. Res. 41, 17–28 (1995).
McDevitt, D., Francois, P., Vaudaux, P. & Foster, T. J. Molecular characterization of the clumping factor (fibrinogen receptor) of Staphylococcus aureus. Mol. Microbiol. 11, 237–248 (1994).
Moreillon, P. et al. Role of Staphylococcus aureus coagulase and clumping factor in pathogenesis of experimental endocarditis. Infect. Immun. 63, 4738–4743 (1995).
Que, Y. A. et al. Fibrinogen and fibronectin binding cooperate for valve infection and invasion in Staphylococcus aureus experimental endocarditis. J. Exp. Med. 201, 1627–1635 (2005).
Bayer, A. S. et al. Hyperproduction of alpha-toxin by Staphylococcus aureus results in paradoxically reduced virulence in experimental endocarditis: a host defense role for platelet microbicidal proteins. Infect. Immun. 65, 4652–4660 (1997).
Scheld, W. M., Valone, J. A. & Sande, M. A. Bacterial adherence in the pathogenesis of endocarditis. Interaction of bacterial dextran, platelets, and fibrin. J. Clin. Invest. 61, 1394–1404 (1978).
Pelletier, L. L. Jr, Coyle, M. & Petersdorf, R. Dextran production as a possible virulence factor in streptococcal endocarditis. Proc. Soc. Exp. Biol. Med. 158, 415–420 (1978).
Burnette-Curley, D. et al. FimA, a major virulence factor associated with Streptococcus parasanguis endocarditis. Infect. Immun. 63, 4669–4674 (1995).
Viscount, H. B., Munro, C. L., Burnette-Curley, D., Peterson, D. L. & Macrina, F. L. Immunization with FimA protects against Streptococcus parasanguis endocarditis in rats. Infect. Immun. 65, 994–1002 (1997).
Takahashi, Y. et al. Contribution of sialic acid-binding adhesin to pathogenesis of experimental endocarditis caused by Streptococcus gordonii DL1. Infect. Immun. 74, 740–743 (2006).
Xiong, Y. Q., Bensing, B. A., Bayer, A. S., Chambers, H. F. & Sullam, P. M. Role of the serine-rich surface glycoprotein GspB of Streptococcus gordonii in the pathogenesis of infective endocarditis. Microb. Pathog. 45, 297–301 (2008).
Siboo, I. R., Chaffin, D. O., Rubens, C. E. & Sullam, P. M. Characterization of the accessory Sec system of Staphylococcus aureus. J. Bacteriol. 190, 6188–6196 (2008).
Mitchell, J., Siboo, I. R., Takamatsu, D., Chambers, H. F. & Sullam, P. M. Mechanism of cell surface expression of the Streptococcus mitis platelet binding proteins PblA and PblB. Mol. Microbiol. 64, 844–857 (2007).
Clawson, C. C., Rao, G. H. & White, J. G. Platelet interaction with bacteria. IV. Stimulation of the release reaction. Am. J. Pathol. 81, 411–420 (1975).
Herzberg, M. C. et al. The platelet interactivity phenotype of Streptococcus sanguis influences the course of experimental endocarditis. Infect. Immun. 60, 4809–4818 (1992).
Shenkman, B. et al. Complex interaction of platelets, Staphylococcus aureus and subendothelium: the role of GPIb and platelet adhesion. Blood 86, 2193–2193 (1995).
Hartleib, J. et al. Protein A is the von Willebrand factor binding protein on Staphylococcus aureus. Blood 96, 2149–2156 (2000).
Youssefian, T., Drouin, A., Masse, J. M., Guichard, J. & Cramer, E. M. Host defense role of platelets: engulfment of HIV and Staphylococcus aureus occurs in a specific subcellular compartment and is enhanced by platelet activation. Blood 99, 4021–4029 (2002).
Yeaman, M. R., Norman, D. C. & Bayer, A. S. Staphylococcus aureus susceptibility to thrombin-induced platelet microbicidal protein is independent of platelet adherence and aggregation in vitro. Infect. Immun. 60, 2368–2374 (1992).
Durack, D. T. & Beeson, P. B. Experimental bacterial endocarditis. II. Survival of a bacteria in endocardial vegetations. Br. J. Exp. Pathol. 53, 50–53 (1972).
Yao, L., Berman, J. W., Factor, S. M. & Lowy, F. D. Correlation of histopathologic and bacteriologic changes with cytokine expression in an experimental murine model of bacteremic Staphylococcus aureus infection. Infect. Immun. 65, 3889–3895 (1997).
Bancsi, M. J. L. M. F., Veltrop, M. H. A. M., Bertina, R. M. & Thompson, J. Role of phagocytosis in activation of the coagulation system in Streptococcus sanguis endocarditis. Infect. Immun. 64, 5166–5170 (1996).
Seidl, K. et al. Combinatorial phenotypic signatures distinguish persistent from resolving methicillin-resistant Staphylococcus aureus bacteremia isolates. Antimicrob. Agents Chemother. 55, 575–582 (2011).
Xiong, Y. Q. et al. Phenotypic and genotypic characteristics of persistent methicillin-resistant Staphylococcus aureus bacteremia in vitro and in an experimental endocarditis model. J. Infect. Dis. 199, 201–208 (2009).
Abdelhady, W. et al. Early agr activation correlates with vancomycin treatment failure in multi-clonotype MRSA endovascular infections. J. Antimicrob. Chemother. 70, 1443–1452 (2015).
Laxdal, T., Messner, R. P., Williams, R. C. & Quie, P. G. Opsonic agglutinating and complement-fixing antibodies in patients with subacute bacterial endocarditis. J. Lab. Clin. Med. 71, 638–653 (1968).
Qoronfleh, M. W., Weraarchakul, W. & Wilkinson, B. J. Antibodies to a range of Staphylococcus aureus and Escherichia coli heat-shock proteins in sera from patients with S. aureus endocarditis. Infect. Immun. 61, 1567–1570 (1993).
Scheld, W. M., Thomas, J. H. & Sande, M. A. Influence of preformed antibody on experimental Streptococcus sanguis endocarditis. Infect. Immun. 25, 781–785 (1979).
Scheld, W. M., Calderone, R. A., Brodeur, J. P. & Sande, M. A. Influence of preformed antibody on the pathogenesis of experimental Candida albicans endocarditis. Infect. Immun. 40, 950–955 (1983).
Greenberg, D. P., Ward, J. I. & Bayer, A. S. Influence of Staphylococcus aureus antibody on experimental endocarditis in rabbits. Infect. Immun. 55, 3030–3034 (1987).
Vernachio, J. et al. Anti-clumping factor A immunoglobulin reduces the duration of methicillin-resistant Staphylococcus aureus bacteremia in an experimental model of infective endocarditis. Antimicrob. Agents Chemother. 47, 3400–3406 (2003).
Scully, I. L. et al. Demonstration of the preclinical correlate of protection for Staphylococcus aureus clumping factor A in a murine model of infection. Vaccine 33, 5452–5457 (2015).
Williams, R. C. & Kunkel, H. G. Rheumatoid factors and their disappearance following therapy in patients with subacute bacterial endocarditis. Arthritis Rheum. 5, 126 (1962).
Sheagren, J. N., Tuazon, C. U., Griffin, C. & Padmore, N. Rheumatoid factor in acute bacterial endocarditis. Arthritis Rheum. 19, 887–890 (1976).
Phair, J. P. & Clarke, J. Immunology of infective endocarditis. Prog. Cardiovasc. Dis. 22, 137–144 (1979).
Bacon, P. A., Davidson, C. & Smith, B. Antibodies to Candida and autoantibodies in sub-acute bacterial endocarditis. Q. J. Med. 43, 537–550 (1974).
Bayer, A. S., Theofilopoulos, A. N., Dixon, F. J. & Guze, L. B. Circulating immune complexes in infective endocarditis. Clin. Res. 24, A451–A451 (1976).
Alpert, J. S., Krous, H. F., Dalen, J. E., O'Rourke, R. A. & Bloor, C. M. Pathogenesis of Osler's nodes. Ann. Intern. Med. 85, 471–473 (1976).
Cabane, J. et al. Fate of circulating immune complexes in infective endocarditis. Am. J. Med. 66, 277–282 (1979).
Kauffmann, R. H., Thompson, J., Valentijn, R. M., Daha, M. R. & Van Es, L. A. The clinical implications and the pathogenetic significance of circulating immune complexes in infective endocarditis. Am. J. Med. 71, 17–25 (1981).
Brouqui, P., Dumler, J. S. & Raoult, D. Immunohistologic demonstration of Coxiella burnetii in the valves of patients with Q fever endocarditis. Am. J. Med. 97, 451–458 (1994).
Brouqui, P. & Raoult, D. Endocarditis due to rare and fastidious bacteria. Clin. Microbiol. Rev. 14, 177–207 (2001).
Morris, A. J. et al. Gram stain, culture, and histopathological examination findings for heart valves removed because of infective endocarditis. Clin. Infect. Dis. 36, 697–704 (2003).
Manzano, M. C. et al. [Acute coronary syndrome in infective endocarditis]. Rev. Esp. Cardiol. 60, 24–31 (in Spanish) (2007).
Morel-Maroger, L., Sraer, J. D., Herreman, G. & Godeau, P. Kidney in subacute endocarditis. Pathological and immunofluorescence findings. Arch. Pathol. 94, 205–213 (1972).
Brown, S. L. et al. Bacteriologic and surgical determinants of survival in patients with mycotic aneurysms. J. Vasc. Surg. 1, 541–547 (1984).
Duval, X. et al. Effect of early cerebral magnetic resonance imaging on clinical decisions in infective endocarditis: a prospective study. Ann. Intern. Med. 152, 497–504 (2010).
Champey, J. et al. Value of brain MRI in infective endocarditis: a narrative literature review. Eur. J. Clin. Microbiol. Infect. Dis. 35, 159–168 (2016).
Snygg-Martin, U. et al. Cerebrovascular complications in patients with left-sided infective endocarditis are common: a prospective study using magnetic resonance imaging and neurochemical brain damage markers. Clin. Infect. Dis. 47, 23–30 (2008).
Dickerman, S. A. et al. The relationship between the initiation of antimicrobial therapy and the incidence of stroke in infective endocarditis: an analysis from the ICE Prospective Cohort Study (ICE-PCS). Am. Heart J. 154, 1086–1094 (2007).
Weinstein, L. & Schlesinger, J. J. Pathoanatomic, pathophysiologic and clinical correlations in endocarditis (second of two parts). N. Engl. J. Med. 291, 1122–1126 (1974).
Miro, J. M., del Rio, A. & Mestres, C. A. Infective endocarditis and cardiac surgery in intravenous drug abusers and HIV-1 infected patients. Cardiol. Clin. 21, 167–184 (2003).
Kerr, A. Jr & Tan, J. S. Biopsies of the Janeway lesion of infective endocarditis. J. Cutan. Pathol. 6, 124–129 (1979).
Loughrey, P. B., Armstrong, D. & Lockhart, C. J. Classical eye signs in bacterial endocarditis. QJM 108, 909–910 (2015).
Okada, A. A., Johnson, R. P., Liles, W. C., D'Amico, D. J. & Baker, A. S. Endogenous bacterial endophthalmitis. Report of a ten-year retrospective study. Ophthalmology 101, 832–838 (1994).
Jung, J. et al. Incidence and risk factors of ocular infection caused by Staphylococcus aureus bacteremia. Antimicrob. Agents Chemother. 60, 2012–2017 (2016).
Beeson, P. B., Brannon, E. S. & Warren, J. V. Observations on the sites of removal of bacteria from the blood in patients with bacterial endocarditis. J. Exp. Med. 81, 9–23 (1945).
Pazin, G. J., Saul, S. & Thompson, M. E. Blood culture positivity: suppression by outpatient antibiotic therapy in patients with bacterial endocarditis. Arch. Intern. Med. 142, 263–268 (1982).
Tattevin, P., Watt, G., Revest, M., Arvieux, C. & Fournier, P. E. Update on blood culture-negative endocarditis. Med. Mal. Infect. 45, 1–8 (2015).
Tunkel, A. R. & Kaye, D. Endocarditis with negative blood cultures. N. Engl. J. Med. 326, 1215–1217 (1992).
Austin, S. M., Smith, S. M., Co, B., Coppel, I. G. & Johnson, J. E. Serologic evidence of acute murine typhus infection in a patient with culture-negative endocarditis. Am. J. Med. Sci. 293, 320–323 (1987).
Shapiro, D. S. et al. Brief report: Chlamydia psittaci endocarditis diagnosed by blood culture. N. Engl. J. Med. 326, 1192–1195 (1992).
Raoult, D. et al. Diagnosis of 22 new cases of Bartonella endocarditis. Ann. Intern. Med. 125, 646–652 (1996).
Fenollar, F., Lepidi, H. & Raoult, D. Whipple's endocarditis: review of the literature and comparisons with Q fever, Bartonella infection, and blood culture-positive endocarditis. Clin. Infect. Dis. 33, 1309–1316 (2001).
Vollmer, T. et al. 23S rDNA real-time polymerase chain reaction of heart valves: a decisive tool in the diagnosis of infective endocarditis. Eur. Heart J. 31, 1105–1113 (2010).
Baddour, L. M. et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation 132, 1435–1486 (2015). This guideline from the AHA updates the previous 2005 guideline for the diagnosis and management of IE, including detailed antibiotic guidance.
Habib, G. et al. 2015 ESC guidelines for the management of infective endocarditis: the Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur. Heart J. 36, 3075–3128 (2015). This guideline from the ESC updates the previous 2009 guideline for the management of IE, including guidance on the prevention, diagnosis and treatment of IE.
Popp, R. L. Echocardiography (1). N. Engl. J. Med. 323, 101–109 (1990).
Erbel, R. et al. Improved diagnostic value of echocardiography in patients with infective endocarditis by transoesophageal approach. A prospective study. Eur. Heart J. 9, 43–53 (1988).
Holland, T. L., Arnold, C. & Fowler, V. G. Jr Clinical management of Staphylococcus aureus bacteremia: a review. JAMA 312, 1330–1341 (2014).
Kaasch, A. J. et al. Use of a simple criteria set for guiding echocardiography in nosocomial Staphylococcus aureus bacteremia. Clin. Infect. Dis. 53, 1–9 (2011).
Joseph, J. P. et al. Prioritizing echocardiography in Staphylococcus aureus bacteraemia. J. Antimicrob. Chemother. 68, 444–449 (2013).
Khatib, R. & Sharma, M. Echocardiography is dispensable in uncomplicated Staphylococcus aureus bacteremia. Medicine (Baltimore) 92, 182–188 (2013).
Palraj, B. R. et al. Predicting risk of endocarditis using a clinical tool (PREDICT): scoring system to guide use of echocardiography in the management of Staphylococcus aureus bacteremia. Clin. Infect. Dis. 61, 18–28 (2015).
Showler, A. et al. Use of transthoracic echocardiography in the management of low-risk Staphylococcus aureus bacteremia: results from a retrospective multicenter cohort study. JACC Cardiovasc. Imaging 8, 924–931 (2015).
Bruun, N. E., Habib, G., Thuny, F. & Sogaard, P. Cardiac imaging in infectious endocarditis. Eur. Heart J. 35, 624–632 (2014).
Pizzi, M. N. et al. Improving the diagnosis of infective endocarditis in prosthetic valves and intracardiac devices with 18F-fluordeoxyglucose positron emission tomography/computed tomography angiography: initial results at an infective endocarditis referral center. Circulation 132, 1113–1126 (2015).
Saby, L. et al. Positron emission tomography/computed tomography for diagnosis of prosthetic valve endocarditis: increased valvular 18F-fluorodeoxyglucose uptake as a novel major criterion. J. Am. Coll. Cardiol. 61, 2374–2382 (2013).
Durack, D. T., Lukes, A. S. & Bright, D. K. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am. J. Med. 96, 200–209 (1994).
Li, J. S. et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin. Infect. Dis. 30, 633–638 (2000).
Hoen, B. et al. Evaluation of the Duke criteria versus the Beth Israel criteria for the diagnosis of infective endocarditis. Clin. Infect. Dis. 21, 905–909 (1995).
Heiro, M., Nikoskelainen, J., Hartiala, J. J., Saraste, M. K. & Kotilainen, P. M. Diagnosis of infective endocarditis. Sensitivity of the Duke vs von Reyn criteria. Arch. Intern. Med. 158, 18–24 (1998).
Stockheim, J. A. et al. Are the Duke criteria superior to the Beth Israel criteria for the diagnosis of infective endocarditis in children? Clin. Infect. Dis. 27, 1451–1456 (1998).
Hoen, B. et al. The Duke criteria for diagnosing infective endocarditis are specific: analysis of 100 patients with acute fever or fever of unknown origin. Clin. Infect. Dis. 23, 298–302 (1996).
Sekeres, M. A. et al. An assessment of the usefulness of the Duke criteria for diagnosing active infective endocarditis. Clin. Infect. Dis. 24, 1185–1190 (1997).
Wilson, W. et al. Prevention of infective endocarditis: guidelines from the American Heart Association. J. Am. Dent. Assoc. 138, 739–760 (2007).
Danchin, N., Duval, X. & Leport, C. Prophylaxis of infective endocarditis: French recommendations 2002. Heart 91, 715–718 (2005).
Richey, R., Wray, D. & Stokes, T. Prophylaxis against infective endocarditis: summary of NICE guidance. BMJ 336, 770–771 (2008).
Thornhill, M. H. et al. Impact of the NICE guideline recommending cessation of antibiotic prophylaxis for prevention of infective endocarditis: before and after study. BMJ 342, d2392 (2011).
Pasquali, S. K. et al. Trends in endocarditis hospitalizations at US children's hospitals: impact of the 2007 American Heart Association Antibiotic Prophylaxis guidelines. Am. Heart J. 163, 894–899 (2012).
Lockhart, P. B., Hanson, N. B., Ristic, H., Menezes, A. R. & Baddour, L. Acceptance among and impact on dental practitioners and patients of American Heart Association recommendations for antibiotic prophylaxis. J. Am. Dent. Assoc. 144, 1030–1035 (2013).
Dayer, M. J. et al. Incidence of infective endocarditis in England, 2000–13: a secular trend, interrupted time-series analysis. Lancet 385, 1219–1228 (2015).
Pant, S. et al. Trends in infective endocarditis incidence, microbiology, and valve replacement in the United States from 2000 to 2011. J. Am. Coll. Cardiol. 65, 2070–2076 (2015).
Pericas, J. M. et al. Neglecting enterococci may lead to a misinterpretation of the consequences of last changes in endocarditis prophylaxis American Heart Association guidelines. J. Am. Coll. Cardiol. 66, 2156 (2015).
Duval, X. & Hoen, B. Prophylaxis for infective endocarditis: let's end the debate. Lancet 385, 1164–1165 (2015).
National Institute for Health and Care Excellence. Prophylaxis against infective endocarditis: antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures. NICEhttp://www.nice.org.uk/guidance/cg64 (2015).
Blot, K., Bergs, J., Vogelaers, D., Blot, S. & Vandijck, D. Prevention of central line-associated bloodstream infections through quality improvement interventions: a systematic review and meta-analysis. Clin. Infect. Dis. 59, 96–105 (2014).
Nishimura, R. A. et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 63, e57–e185 (2014).
LaPlante, K. L. & Rybak, M. J. Impact of high-inoculum Staphylococcus aureus on the activities of nafcillin, vancomycin, linezolid, and daptomycin, alone and in combination with gentamicin, in an in vitro pharmacodynamic model. Antimicrob. Agents Chemother. 48, 4665–4672 (2004).
Eng, R. H., Padberg, F. T., Smith, S. M., Tan, E. N. & Cherubin, C. E. Bactericidal effects of antibiotics on slowly growing and nongrowing bacteria. Antimicrob. Agents Chemother. 35, 1824–1828 (1991).
Gould, F. K. et al. Guidelines for the diagnosis and antibiotic treatment of endocarditis in adults: a report of the Working Party of the British Society for Antimicrobial Chemotherapy. J. Antimicrob. Chemother. 67, 269–289 (2012).
Athan, E. et al. Clinical characteristics and outcome of infective endocarditis involving implantable cardiac devices. JAMA 307, 1727–1735 (2012).
Baddour, L. M. et al. Update on cardiovascular implantable electronic device infections and their management: a scientific statement from the American Heart Association. Circulation 121, 458–477 (2010).
McDanel, J. S. et al. Comparative effectiveness of beta-lactams versus vancomycin for treatment of methicillin-susceptible Staphylococcus aureus bloodstream infections among 122 hospitals. Clin. Infect. Dis. 61, 361–367 (2015).
McConeghy, K. W., Bleasdale, S. C. & Rodvold, K. A. The empirical combination of vancomycin and a β-lactam for Staphylococcal bacteremia. Clin. Infect. Dis. 57, 1760–1765 (2013).
Bernstein, I. L. et al. Allergy diagnostic testing: an updated practice parameter. Ann. Allergy Asthma Immunol. 100, S1–S148 (2008).
Blumenthal, K. G., Parker, R. A., Shenoy, E. S. & Walensky, R. P. Improving clinical outcomes in patients with methicillin-sensitive Staphylococcus aureus bacteremia and reported penicillin allergy. Clin. Infect. Dis. 61, 741–749 (2015).
Dodek, P. & Phillips, P. Questionable history of immediate-type hypersensitivity to penicillin in Staphylococcal endocarditis: treatment based on skin-test results vers-us empirical alternative treatment — a decision analysis. Clin. Infect. Dis. 29, 1251–1256 (1999).
Liu, C. et al. Clinical practice guidelines by the infectious diseases society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin. Infect. Dis. 52, e18–e55 (2011).
Steinkraus, G., White, R. & Friedrich, L. Vancomycin MIC creep in non-vancomycin-intermediate Staphylococcus aureus (VISA), vancomycin-susceptible clinical methicillin-resistant S. aureus (MRSA) blood isolates from 2001–05. J. Antimicrob. Chemother. 60, 788–794 (2007).
van Hal, S. J., Lodise, T. P. & Paterson, D. L. The clinical significance of vancomycin minimum inhibitory concentration in Staphylococcus aureus infections: a systematic review and meta-analysis. Clin. Infect. Dis. 54, 755–771 (2012).
Kalil, A. C., Van Schooneveld, T. C., Fey, P. D. & Rupp, M. E. Association between vancomycin minimum inhibitory concentration and mortality among patients with Staphylococcus aureus bloodstream infections: a systematic review and meta-analysis. JAMA 312, 1552–1564 (2014).
Cervera, C. et al. Effect of vancomycin minimal inhibitory concentration on the outcome of methicillin-susceptible Staphylococcus aureus endocarditis. Clin. Infect. Dis. 58, 1668–1675 (2014).
Fowler, V. G. Jr et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N. Engl. J. Med. 355, 653–665 (2006). This randomized trial of daptomycin versus standard therapy for S. aureus bacteraemia and IE demonstrates that daptomycin at a dose of 6 mg per kg daily is not inferior to standard therapy. This led to FDA approval of daptomycin for the treatment of S. aureus bacteraemia and right-sided IE.
Carugati, M. et al. High-dose daptomycin therapy for left-sided infective endocarditis: a prospective study from the international collaboration on endocarditis. Antimicrob. Agents Chemother. 57, 6213–6222 (2013).
Kullar, R. et al. High-dose daptomycin for treatment of complicated Gram-positive infections: a large, multicenter, retrospective study. Pharmacotherapy 31, 527–536 (2011).
Kullar, R. et al. A multicentre evaluation of the effectiveness and safety of high-dose daptomycin for the treatment of infective endocarditis. J. Antimicrob. Chemother. 68, 2921–2926 (2013).
Cosgrove, S. E. et al. Initial low-dose gentamicin for Staphylococcus aureus bacteremia and endocarditis is nephrotoxic. Clin. Infect. Dis. 48, 713–721 (2009).
Riedel, D. J., Weekes, E. & Forrest, G. N. Addition of rifampin to standard therapy for treatment of native valve infective endocarditis caused by Staphylococcus aureus. Antimicrob. Agents Chemother. 52, 2463–2467 (2008).
Levine, D. P., Fromm, B. S. & Reddy, B. R. Slow response to vancomycin or vancomycin plus rifampin in methicillin-resistant Staphylococcus aureus endocarditis. Ann. Intern. Med. 115, 674–680 (1991).
Drinkovic, D., Morris, A. J., Pottumarthy, S., MacCulloch, D. & West, T. Bacteriological outcome of combination versus single-agent treatment for staphylococcal endocarditis. J. Antimicrob. Chemother. 52, 820–825 (2003).
Thwaites, G. et al. Adjunctive rifampicin to reduce early mortality from Staphylococcus aureus bacteraemia (ARREST): study protocol for a randomised controlled trial. Trials 13, 241 (2012).
Casapao, A. M. et al. Large retrospective evaluation of the effectiveness and safety of ceftaroline fosamil therapy. Antimicrob. Agents Chemother. 58, 2541–2546 (2014).
Fabre, V., Ferrada, M., Buckel, W. R., Avdic, E. & Cosgrove, S. E. Ceftaroline in combination with trimethoprim-sulfamethoxazole for salvage therapy of methicillin-resistant Staphylococcus aureus bacteremia and endocarditis. Open Forum Infect. Dis. 1, ofu046 (2014).
del Rio, A. et al. Fosfomycin plus β-lactams as synergistic bactericidal combinations for experimental endocarditis due to methicillin-resistant and glycopeptide-intermediate Staphylococcus aureus. Antimicrob. Agents Chemother. 60, 478–486 (2016).
Holubar, M., Meng, L. & Deresinski, S. Bacteremia due to methicillin-resistant Staphylococcus aureus: new therapeutic approaches. Infect. Dis. Clin. North Am. 30, 491–507 (2016).
Sexton, D. J. et al. Ceftriaxone once daily for four weeks compared with ceftriaxone plus gentamicin once daily for two weeks for treatment of endocarditis due to penicillin-susceptible streptococci. Endocarditis Treatment Consortium Group. Clin. Infect. Dis. 27, 1470–1474 (1998). This randomized, open-label study compares two treatment regimens for penicillin-susceptible streptococci: ceftriaxone for 4 weeks or ceftriaxone plus gentamicin for 2 weeks. Both were associated with clinical cure rates of >95% at 3 months follow-up and are now considered standard of care therapy.
Robbins, W. C. & Tompsett, R. Treatment of enterococcal endocarditis and bacteremia; results of combined therapy with penicillin and streptomycin. Am. J. Med. 10, 278–299 (1951).
Pericas, J. M. et al. Changes in the treatment of Enterococcus faecalis infective endocarditis in Spain in the last 15 years: from ampicillin plus gentamicin to ampicillin plus ceftriaxone. Clin. Microbiol. Infect. 20, O1075–O1083 (2014).
Fernandez-Hidalgo, N. et al. Ampicillin plus ceftriaxone is as effective as ampicillin plus gentamicin for treating Enterococcus faecalis infective endocarditis. Clin. Infect. Dis. 56, 1261–1268 (2013). This observational study shows that, among patients with E. faecalis IE treated with ampicillin plus ceftriaxone (n = 159), there were no differences in mortality, treatment failure or relapse rates when compared with those receiving gentamicin (n = 87). However, adverse events including renal failure were more common among patients treated with gentamicin.
Falagas, M. E., Manta, K. G., Ntziora, F. & Vardakas, K. Z. Linezolid for the treatment of patients with endocarditis: a systematic review of the published evidence. J. Antimicrob. Chemother. 58, 273–280 (2006).
Coburn, B. et al. Antimicrobial susceptibilities of clinical isolates of HACEK organisms. Antimicrob. Agents Chemother. 57, 1989–1991 (2013).
Arnold, C. J. et al. Candida infective endocarditis: an observational cohort study with a focus on therapy. Antimicrob. Agents Chemother. 59, 2365–2373 (2015).
Pappas, P. G. et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 48, 503–535 (2009).
Cornely, O. A. et al. ESCMID guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin. Microbiol. Infect. 18 (Suppl. 7), 19–37 (2012).
Kang, D. H. et al. Early surgery versus conventional treatment for infective endocarditis. N. Engl. J. Med. 366, 2466–2473 (2012). This trial, which is the only randomized study to examine the role of early surgery for IE, shows better outcomes with surgery. However, this trial included subjects who were younger, healthier and infected with less-virulent pathogens than most contemporary patients with IE.
Gordon, S. M. & Pettersson, G. B. Native-valve infective endocarditis — when does it require surgery? N. Engl. J. Med. 366, 2519–2521 (2012).
Barsic, B. et al. Influence of the timing of cardiac surgery on the outcome of patients with infective endocarditis and stroke. Clin. Infect. Dis. 56, 209–217 (2013). This study of 857 patients with IE complicated by ischaemic stroke without haemorrhagic conversion found that no survival benefit was gained from delaying surgery.
Garcia-Cabrera, E. et al. Neurological complications of infective endocarditis: risk factors, outcome, and impact of cardiac surgery: a multicenter observational study. Circulation 127, 2272–2284 (2013).
Chirouze, C. et al. Impact of early valve surgery on outcome of Staphylococcus aureus prosthetic valve infective endocarditis: analysis in the International Collaboration of Endocarditis-Prospective Cohort study. Clin. Infect. Dis. 60, 741–749 (2015).
Tornos, P. et al. Infective endocarditis due to Staphylococcus aureus: deleterious effect of anticoagulant therapy. Arch. Intern. Med. 159, 473–475 (1999).
Chan, K. L. et al. Effect of long-term aspirin use on embolic events in infective endocarditis. Clin. Infect. Dis. 46, 37–41 (2008).
Snygg-Martin, U. et al. Warfarin therapy and incidence of cerebrovascular complications in left-sided native valve endocarditis. Eur. J. Clin. Microbiol. Infect. Dis. 30, 151–157 (2011).
Park, K. H. et al. Optimal duration of antibiotic therapy in patients with hematogenous vertebral osteomyelitis at low risk and high risk of recurrence. Clin. Infect. Dis. 62, 1262–1269 (2016).
Delahaye, F. et al. Systematic search for present and potential portals of entry for infective endocarditis. J. Am. Coll. Cardiol. 67, 151–158 (2016).
Perrotta, S., Aljassim, O., Jeppsson, A., Bech-Hanssen, O. & Svensson, G. Survival and quality of life after aortic root replacement with homografts in acute endocarditis. Ann. Thorac. Surg. 90, 1862–1867 (2010).
Verhagen, D. W. et al. Health-related quality of life and posttraumatic stress disorder among survivors of left-sided native valve endocarditis. Clin. Infect. Dis. 48, 1559–1565 (2009).
Yeates, A. et al. Early and mid-term outcomes following surgical management of infective endocarditis with associated cerebral complications: a single centre experience. Heart Lung Circ. 19, 523–527 (2010).
Jokinen, J. J., Hippelainen, M. J., Pitkanen, O. A. & Hartikainen, J. E. Mitral valve replacement versus repair: propensity-adjusted survival and quality-of-life analysis. Ann. Thorac. Surg. 84, 451–458 (2007).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01512615 (2016).
Rasmussen, T. B. et al. A randomised clinical trial of comprehensive cardiac rehabilitation versus usual care for patients treated for infective endocarditis — the CopenHeartIE trial protocol. BMJ Open 2, e001929 (2012).
Rasmussen, T. B., Zwisler, A. D., Moons, P. & Berg, S. K. Insufficient living: experiences of recovery after infective endocarditis. J. Cardiovasc. Nurs. 30, E11–E19 (2015).
Iversen, K. et al. Partial oral treatment of endocarditis. Am. Heart J. 165, 116–122 (2013).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02701608 (2016).
Dunne, M. W. et al. Extended-duration dosing and distribution of dalbavancin into bone and articular tissue. Antimicrob. Agents Chemother. 59, 1849–1855 (2015).
Messina, J. A., Fowler, V. G. Jr & Corey, G. R. Oritavancin for acute bacterial skin and skin structure infections. Expert Opin. Pharmacother. 16, 1091–1098 (2015).
Boucher, H. W. et al. Once-weekly dalbavancin versus daily conventional therapy for skin infection. N. Engl. J. Med. 370, 2169–2179 (2014).
Dunne, M. W. et al. A randomized clinical trial of single-dose versus weekly dalbavancin for treatment of acute bacterial skin and skin structure infection. Clin. Infect. Dis. 62, 545–551 (2016).
Corey, G. R. et al. Single-dose oritavancin in the treatment of acute bacterial skin infections. N. Engl. J. Med. 370, 2180–2190 (2014).
Raad, I. et al. Efficacy and safety of weekly dalbavancin therapy for catheter-related bloodstream infection caused by Gram-positive pathogens. Clin. Infect. Dis. 40, 374–380 (2005).
Kitten, T., Munro, C. L., Wang, A. & Macrina, F. L. Vaccination with FimA from Streptococcus parasanguis protects rats from endocarditis caused by other viridans streptococci. Infect. Immun. 70, 422–425 (2002).
Spellberg, B. J. et al. Efficacy of the anti-Candida rAls3p-N or rAls1p-N vaccines against disseminated and mucosal candidiasis. J. Infect. Dis. 194, 256–260 (2006).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02357966 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02388165 (2016).
Shinefield, H. et al. Use of a Staphylococcus aureus conjugate vaccine in patients receiving hemodialysis. N. Engl. J. Med. 346, 491–496 (2002).
Spellberg, B. et al. The antifungal vaccine derived from the recombinant N terminus of Als3p protects mice against the bacterium Staphylococcus aureus. Infect. Immun. 76, 4574–4580 (2008).
Schmidt, C. S. et al. NDV-3, a recombinant alum-adjuvanted vaccine for Candida and Staphylococcus aureus, is safe and immunogenic in healthy adults. Vaccine 30, 7594–7600 (2012).
Bagnoli, F. et al. Vaccine composition formulated with a novel TLR7-dependent adjuvant induces high and broad protection against Staphylococcus aureus. Proc. Natl Acad. Sci. USA 112, 3680–3685 (2015).
Werdan, K. et al. Mechanisms of infective endocarditis: pathogen–host interaction and risk states. Nat. Rev. Cardiol. 11, 35–50 (2014).
Lepidi, H., Durack, D. T. & Raoult, D. Diagnostic methods current best practices and guidelines for histologic evaluation in infective endocarditis. Infect. Dis. Clin. North Am. 16, 339–361 (2002).
Marin, M. et al. Molecular diagnosis of infective endocarditis by real-time broad-range polymerase chain reaction (PCR) and sequencing directly from heart valve tissue. Medicine (Baltimore) 86, 195–202 (2007).
Lepidi, H., Coulibaly, B., Casalta, J. P. & Raoult, D. Autoimmunohistochemistry: a new method for the histologic diagnosis of infective endocarditis. J. Infect. Dis. 193, 1711–1717 (2006).
Imai, A. et al. Comprehensive metagenomic approach for detecting causative microorganisms in culture-negative infective endocarditis. Int. J. Cardiol. 172, e288–e289 (2014).
Benoit, M. et al. The transcriptional programme of human heart valves reveals the natural history of infective endocarditis. PLoS ONE 5, e8939 (2010).
Ahn, S. H. et al. Gene expression-based classifiers identify Staphylococcus aureus infection in mice and humans. PLoS ONE 8, e48979 (2013).
DeSimone, D. C. et al. Incidence of infective endocarditis caused by viridans group streptococci before and after publication of the 2007 American Heart Association's endocarditis prevention guidelines. Circulation 126, 60–64 (2012).
Bikdeli, B. et al. Trends in hospitalization rates and outcomes of endocarditis among Medicare beneficiaries. J. Am. Coll. Cardiol. 62, 2217–2226 (2013).
DeSimone, D. C. et al. Incidence of infective endocarditis due to viridans group streptococci before and after the 2007 American Heart Association's prevention guidelines: an extended evaluation of the Olmsted County, Minnesota, population and nationwide inpatient sample. Mayo Clin. Proc. 90, 874–881 (2015).
Mackie, A. S., Liu, W., Savu, A., Marelli, A. J. & Kaul, P. Infective endocarditis hospitalizations before and after the 2007 American Heart Association prophylaxis guidelines. Can. J. Cardiol. 32, 942–948 (2016).
Weems, J. J. Jr et al. Phase II, randomized, double-blind, multicenter study comparing the safety and pharmacokinetics of tefibazumab to placebo for treatment of Staphylococcus aureus bacteremia. Antimicrob. Agents Chemother. 50, 2751–2755 (2006).
Rupp, M. E. et al. Phase II, randomized, multicenter, double-blind, placebo-controlled trial of a polyclonal anti-Staphylococcus aureus capsular polysaccharide immune globulin in treatment of Staphylococcus aureus bacteremia. Antimicrob. Agents Chemother. 51, 4249–4254 (2007).
Benjamin, D. K. et al. A blinded, randomized, multicenter study of an intravenous Staphylococcus aureus immune globulin. J. Perinatol. 26, 290–295 (2006).
DeJonge, M. et al. Clinical trial of safety and efficacy of INH-A21 for the prevention of nosocomial staphylococcal bloodstream infection in premature infants. J. Pediatr. 151, 260–265.e1 (2007).
Weisman, L. E. et al. A randomized study of a monoclonal antibody (pagibaximab) to prevent staphylococcal sepsis. Pediatrics 128, 271–279 (2011).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT00646399 (2011).
Fowler, V. G. et al. Effect of an investigational vaccine for preventing Staphylococcus aureus infections after cardiothoracic surgery: a randomized trial. JAMA 309, 1368–1378 (2013).
Fowler, V. G. Jr & Proctor, R. A. Where does a Staphylococcus aureus vaccine stand?. Clin. Microbiol. Infect. 20 (Suppl. 5), 66–75 (2014).
Acknowledgements
This work was supported by the US National Institute of Allergy and Infectious Diseases at the US NIH (grants K24-AI093969 and R01-AI068804 to V.G.F.); T.L.H. is also supported by grant N01-AI-90023, for which V.G.F. is the principal investigator.
Author information
Authors and Affiliations
Contributions
Introduction (V.G.F. and T.L.H.); Epidemiology (B.H. and T.L.H.); Mechanisms/pathophysiology (A.S.B. and T.L.H.); Diagnosis, screening and prevention (J.M.M. and T.L.H.); Management (L.M.B. and T.L.H.); Quality of life (B.H.); Outlook (V.G.F. and T.L.H.); Overview of Primer (V.G.F. and T.L.H.).
Corresponding author
Ethics declarations
Competing interests
V.G.F. reports the following potential conflicts of interest: Chair of the Scientific Advisory Board for Merck V710 Staphylococcus aureus vaccine; paid consultant for Pfizer, Novartis, Galderma, Novadigm, Durata, Debiopharm, Genentech, Achaogen, Affinium, Medicines Co., Cerexa, Tetraphase, Trius, MedImmune, Bayer, Theravance, Cubist, Basilea, Affinergy, Arsanis and XBiotech; grants pending from MedImmune, Actavis/Forest/Cerexa, Pfizer, Merck/Cubist, Advanced Liquid Logics, Theravance, Novartis, Contrafect, Genentech and Karius; royalties from UpToDate; personal fees for the development or presentation of educational presentations from Green Cross, Cubist, Cerexa, Durata and Theravance; and a patent pending related to sepsis diagnostics. T.L.H. reports the following potential conflicts of interest: paid consultant for The Medicines Company and Basilea Pharmaceutica; and royalties from UpToDate. A.S.B. reports the following potential conflicts of interest: research grants from ContraFect and Theravance; and advisory board member for ContraFect. J.M.M. reports the following potential conflicts of interest: consulting honoraria and/or research grants from AbbVie, Bristol-Myers Squibb, Cubist, Genentech, Merck, Novartis, Gilead Sciences, ViiV Healthcare and Instituto de Salud Carlos III, Ministerio de Economía y Competitividad, Madrid (Spain)a personal intensification research grant #INT15/00168 during 2016. L.M.B. and B.H. declare no conflicts of interest.
Supplementary information
Supplementary information S1 Video 1
Infective endocarditis diagnosed by cardiac MRI. Cine cardiac MRI shows vegetations in the sub-tricuspid valve chordal apparatus and posterior mitral valve leaflet with moderate tricuspid and mitral regurgitation. Video courtesy of A. L. Crowley, Duke University School of Medicine, Durham, North Carolina, USA. (MP4 544 kb)
Rights and permissions
About this article
Cite this article
Holland, T., Baddour, L., Bayer, A. et al. Infective endocarditis. Nat Rev Dis Primers 2, 16059 (2016). https://doi.org/10.1038/nrdp.2016.59
Published:
DOI: https://doi.org/10.1038/nrdp.2016.59
This article is cited by
-
Association of COVID-19 with endocarditis in patients with cocaine or opioid use disorders in the US
Molecular Psychiatry (2023)
-
von Willebrand factor-binding protein (vWbp)-activated factor XIII and transglutaminase 2 (TG2) promote cross-linking between FnBPA from Staphylococcus aureus and fibrinogen
Scientific Reports (2023)
-
Epidemiology, microbiology, and outcomes of infective endocarditis in a tertiary center in Jordan
Wiener Medizinische Wochenschrift (2023)
-
Streptococcus canis prostatitis and endocarditis with thromboembolism in a dog with sertoli cell tumour in a cryptic testis and prostatic squamous metaplasia
Veterinary Research Communications (2023)
-
Transcatheter Aortic Valve Replacement for Aortic Valve Infective Endocarditis: A Systematic Review and Call for Action
Cardiology and Therapy (2023)