Abstract
Irritable bowel syndrome (IBS) is a functional gastrointestinal disease with a high population prevalence. The disorder can be debilitating in some patients, whereas others may have mild or moderate symptoms. The most important single risk factors are female sex, younger age and preceding gastrointestinal infections. Clinical symptoms of IBS include abdominal pain or discomfort, stool irregularities and bloating, as well as other somatic, visceral and psychiatric comorbidities. Currently, the diagnosis of IBS is based on symptoms and the exclusion of other organic diseases, and therapy includes drug treatment of the predominant symptoms, nutrition and psychotherapy. Although the underlying pathogenesis is far from understood, aetiological factors include increased epithelial hyperpermeability, dysbiosis, inflammation, visceral hypersensitivity, epigenetics and genetics, and altered brain–gut interactions. IBS considerably affects quality of life and imposes a profound burden on patients, physicians and the health-care system. The past decade has seen remarkable progress in our understanding of functional bowel disorders such as IBS that will be summarized in this Primer.
Similar content being viewed by others
Introduction
Irritable bowel syndrome (IBS) is a functional bowel disorder (that is, not associated with structural or biochemical abnormalities that are detectable with the current routine diagnostic tools) characterized by abdominal pain or discomfort, stool irregularities and bloating (Box 1). Symptoms can be debilitating in many individuals, but may be mild or moderate in other patients. In addition, IBS is often associated with other somatic comorbidities (for example, pain syndromes, overactive bladder and migraine), psychiatric conditions (including depression and anxiety) and visceral sensitivity. The population prevalence of IBS is high (∼11%) and the condition has considerable consequences for quality of life (QOL) that are comparable to other chronic diseases, such as diabetes mellitus and hepatitis. IBS is diagnosed based on symptoms, and a distinction is made between the following subtypes of IBS: IBS with pain or discomfort and predominant constipation (IBS-C), IBS with diarrhoea (IBS-D), mixed IBS (IBS-M) and unsubtyped IBS (IBS-U) (Fig. 1). Moreover, other diseases (including other functional gastrointestinal diseases, such as functional dyspepsia and gastroesophageal reflux disease) that may cause the typical IBS symptoms should be excluded. Although a substantial proportion of patients will experience spontaneous remission over time, there is currently no treatment that cures IBS; relief of symptoms is the most that can be achieved.
A two-dimensional graph of the four possible irritable bowel syndrome (IBS) subtypes according to bowel form at a particular point in time, and the percentage of time this bowel form has to be present to meet the criteria for IBS with constipation (IBS-C), IBS with diarrhoea (IBS-D), mixed-type IBS (IBS-M) and unsubtyped IBS (IBS-U). Adapted with permission from Ref. 119, American Gastroenterology Association.
IBS is a multifactorial disease. Hence, the underlying pathogenesis is considered complex and the precise molecular pathophysiology is far from understood. Several functional alterations have been described, such as altered visceral sensitivity, functional brain alterations, bowel motility and secretory dysfunctions, and somatic and psychiatric comorbidities. Furthermore, gastrointestinal abnormalities — such as immune activation, gut dysbiosis (microbial imbalance), impaired mucosal functions, nerve sensitization, post-infectious plasticity, altered expression and release of mucosal and immune mediators, and altered gene expression profiles — have been associated with IBS. However, a coherent link between particular pathologies and IBS symptoms is yet to be established.
Moreover, results from studies assessing the contribution of most of the proposed pathological factors are inconsistent and the particular aetiology is often not related to particular gut symptoms. For example, some studies have found evidence for gut micro-inflammation in IBS, whereas others could not confirm this finding, despite similar gastrointestinal symptoms. Such discrepancies, which also apply to the other biomarker candidates (not only to inflammation), strongly suggest the existence of IBS subpopulations, which, despite the similarity in gut symptoms, can be defined and distinguished by their pathophysiology and in-depth assessments of clinical and molecular biomarker clusters. The same heterogeneity is evident with respect to clinical diagnosis and management. Indeed, medical treatment, nutritional intervention and psychotherapy lack consistent and homogeneous efficacy, but can be effective in some subgroups.
This Primer summarizes recent progress in our understanding of IBS prevalence, comorbidities, QOL and the putative roles of inflammation, genetics, the intestinal microbiota and the brain–gut axis in IBS pathogenesis. Furthermore, we will discuss the current diagnostic approach and highlight the therapeutic options in IBS, including drugs, nutrition and psychotherapy.
Epidemiology
Global prevalence and incidence
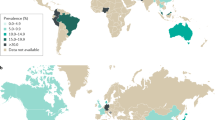
Prevalence rates of IBS vary between 1.1% and 45%, based on population studies from countries worldwide (Fig. 2; Supplementary information S1 (table)), with a pooled global prevalence of 11.2% (95% CI: 9.8–12.8)1. Prevalence rates of 5–10% are reported for most European countries, the United States and China1. Population statistics for IBS in most African and many Asian countries are unavailable, which might point to the inability to differentiate between infectious diarrhoea and IBS in tropical countries, especially in those nations with poor health-care systems or limited patient access to medical care, or to less attention of the health-care system for functional disorders, once an acute infection has been excluded2.
Pooled prevalence data per country are colour-coded. Data from Ref. 1 are supplemented by studies from another nine countries (see Supplementary-information S1 (table)). IBS, irritable bowel syndrome; N/A, not applicable.
Gathering subtype-specific prevalence information is complex. IBS subtypes overlap considerably in terms of symptoms, and patients vary over time in terms of their predominant symptoms, and thus switch subtype3. The few population studies that have differentiated between IBS subtypes suggest that, in countries with a total IBS prevalence of ∼10%, IBS-C and IBS-D each account for one-third of the affected population4. Incidence rates of IBS (that is, the annual occurrence of new cases) are not reported for most countries, but a few long-term surveys (≥10 years) in the United States allow for an estimation of the annual incidence in the range of 1–2%5. At the same time, disappearance rates of 2% have been reported6, indicating spontaneous disease remission.
Association between IBS and other disorders
Not only do IBS subtypes overlap6 but population-based studies also report a substantial overlap of ≥20% with other functional gastrointestinal disorders of the upper and lower gastrointestinal system: functional dyspepsia, heartburn, gastroesophageal reflux disease and nausea on the one hand7, and diarrhoea, incontinence, pelvic floor dyssynergia and constipation on the other hand8. An overlap of IBS with inflammatory bowel diseases (IBDs; including Crohn disease and ulcerative colitis) during remission phases has been proposed9 but is not mutually agreed on10.
Other IBS-associated disorders (Fig. 3) include functional non-gastrointestinal syndromes, such as urological chronic pelvic pain syndrome (this term includes interstitial cystitis and chronic prostatitis), vulvodynia, overactive bladder, prostatic pain syndrome, premenstrual syndrome, sexual (including erectile) dysfunction, chronic pelvic pain, fibromyalgia syndrome, chronic fatigue syndrome, migraine, eating disorders, nutritional intolerances and others11. All of these syndromes considerably overlap with IBS in population studies to a degree that is often beyond what is expected based on the prevalence rates of the individual diseases. Given that many of these conditions are only diagnosed in specialized centres, it has been questioned as to whether some of these conditions — for example, IBS and chronic pelvic pain — are one and the same disease12.
A model of irritable bowel syndrome (IBS) and its associations with other clinical, intestinal, extra-intestinal and psychiatric conditions. For each of the listed disorders, overlap with IBS symptoms has been reported in the literature11. The different components should be viewed as layers of complexity: the IBS subtypes are part of the group of functional bowel disorders, these are part of all kinds of functional disorders and these again are part of a ‘layer’ of psychiatric disorders. GERD, gastroesophageal reflux disease; IBS-C, IBS with constipation; IBS-D, IBS with diarrhoea; IBS-M, mixed-type IBS; IBS-U, unsubtyped IBS; PMS, premenstrual syndrome.
In addition, most epidemiological studies note the presence of psychiatric comorbidities (such as anxiety, depression, somatization or neuroticism) not only for IBS but also for these IBS-associated diseases. Again, the rates are above the expected levels for IBS and the population prevalence of these symptoms13. Thus, the entire disease entity (IBS, functional gastrointestinal disorders and other functional non-gastrointestinal disorders) has been included in the term ‘somatic symptom disorder’ in the Diagnostic and Statistical Manual of Mental Disorders 5th Edition (DSM-5)14 and in psychiatric or psychosomatic clinical management15. Patients with IBS who were treated by psychiatrists frequently did not receive adequate attention with respect to their gastrointestinal symptoms before the release of DSM-5.
Risk factors for IBS
The best-documented risk factor for IBS is female sex, which is associated with an odds ratio of 1.67 (95% CI: 1.53–1.82) across many population-based studies16, with explanations varying between sex-different health care, consultation behaviour and biological functions (for example, hormonal regulation of gut functions). The incidence of IBS decreases with advancing age (>50 years)1, but is similar in children and adolescents compared with adults and does not necessarily transmit from childhood to adulthood17. However, family aggregation has been reported18 that is driven by genetics19 as well as by social learning20. Box 2 lists the personal, disease, psychosocial and social factors that have been found to be associated with increased risk of IBS, although some of these factors have only been identified in individual studies21 or have been found to vary between countries and settings.
Post-infectious IBS
Several studies have shown an association between IBS and preceding gastrointestinal infections of bacterial, viral or other origin22,23. The pooled odds ratio is 7.3 (95% CI: 4.7–11.1) for the development of IBS after infectious gastroenteritis24, with a median prevalence of ∼10%22. This association seems to differ with respect to epidemic infectious events that affect many people at the same time and individual infections, such as travellers' diarrhoea. That is, prevalence data are reported to be higher (15–30%) in epidemic events22 and lower (5–10%) following travellers' diarrhoea23; these differences are presumably due to different reporting biases in these populations. Thus, a median prevalence of 10% might better reflect the true prevalence of post-infectious IBS than the extreme values reported in individual studies. Risk factors for the development of post-infectious IBS are female sex, younger age, the severity of the initial infection and premorbid psychological conditions22–24. Based on symptoms alone, post-infectious IBS cannot be distinguished from IBS without an infectious origin, but inflammatory biomarkers may. The most valid distinction may be a sudden onset that is well remembered by the patient and is associated with fever, bloody stools and a positive laboratory stool test for an infective agent.
Mechanisms/pathophysiology
Although the aetiology of IBS remains largely undetermined, our understanding of the potential mechanisms involved in gut dysfunction, visceral sensation and symptom generation is rapidly advancing. Growing evidence suggests that, in IBS, the epithelial barrier, gut microbiota, food antigens and bile acids elicit abnormal responses in the key regulators of sensorimotor functions, including the hypothalamus–pituitary–adrenal (HPA) axis, the immune system, the brain–gut axis and the enteric nervous system (ENS) (Fig. 4). Accordingly, these factors might have a role as potential biomarkers of disease (Box 3). In addition to these putative biomarkers, psychological factors (‘psychomarkers’) such as depression and anxiety, which are known to respond to abdominal symptoms (bottom-up), and psychosocial factors (‘stress’) that influence physiological (intestinal) functions, such as motility and visceral sensitivity (top-down), have been acknowledged and will be discussed in more detail.
Although the aetiology of irritable bowel syndrome (IBS) has not yet been completely elucidated, various factors have a role, including composition of the gut microbiota, intestinal permeability, immune cell reactivity and sensitivity of the enteric nervous system, the brain–gut axis (spinal, vagal or pelvic pathways) or the brain. The figure highlights those mediators that are probably involved in IBS pathology. The plus symbols indicate whether a mediator activates or inhibits its target cell; those in parentheses denote actions established in animal models and those without parentheses are effects demonstrated in humans (human tissue). 5-HT, 5-hydroxytryptamine (also known as serotonin); CGRP, calcitonin gene-related peptide; GDNF, glial cell-derived neurotrophic factor; IL, interleukin; PAR2, proteinase-activated receptor 2; TNF, tumour necrosis factor.
The epithelial barrier
The epithelial gut lining represents an enormous surface that is in constant contact with the environment and with billions of bacteria that constantly challenge the intestinal immune system. Increased intestinal permeability is considered an early event in IBS that leads to low-grade immune cell infiltration of the gut mucosa25. Indeed, increased epithelial permeability has been primarily described in post-infectious IBS in general and in IBS-D in particular, although some reports have also shown that IBS-C and IBS-M might also involve an increase in epithelial permeability25. Evidence for the presence of this remodelling in IBS has been provided by electron microscopy, which has detected enlarged spaces between epithelial cells and cytoskeletal condensation in gut biopsies of patients with IBS-D26. In addition, Ussing chamber experiments, which measure epithelial membrane properties on colonic mucosal biopsies, have shown excessive passage of macromolecules from the luminal to the basolateral side of gut tissue in biopsies obtained from patients with IBS compared with asymptomatic controls, hence providing the functional correlate for the described structural epithelial barrier defects27.
Morphological and functional changes in intestinal permeability are related to abnormal gene and protein expression of tight junction proteins, including a reduction in the expression of occludin and zonula occludens protein 1 (Refs 25,28). These findings have recently been corroborated by genetic and epigenetic findings in tight junction proteins claudin 1, claudin 2 and cingulin, as outlined below. Tight junction changes are probably the result of both bacterial-mediated and proteasome-mediated degradation triggered by low-grade inflammation29. Accordingly, inflammatory mediators including eicosanoids, histamine and proteases increase intestinal permeability. This may involve the participation of ENS neurons, which may amplify these effects27,30.
Increased intestinal permeability has been linked to diarrhoea and pain severity26, suggesting that this mechanism might have a role in symptom generation in IBS. Although the exact causes underlying the ‘leaky’ gut barrier in IBS remain elusive, it has been postulated that numerous factors could be involved, including genetics, epigenetics, dysbiosis and food allergies25. Confocal laser endomicroscopy of the duodenal mucosa of patients with IBS after challenge with food to which the patients reported intolerance showed epithelial breaks and increased intervillous spaces, indicative of increased intestinal permeability. These studies suggest a causative effect of food in the increased epithelial permeability in IBS31.
Bile acids
A subset of patients with features compatible with IBS-D present with increased levels of total faecal bile acids caused by increased excretion and synthesis of serum C4 (7α-hydroxy-4-cholesten-3-one; a surrogate for bile acid synthesis), which in turn influences bowel habit by accelerating colonic transit and inducing diarrhoea and visceral hypersensitivity in IBS32–34. Of note, genes involved in bile acid metabolism and function have been reported to be associated with colonic transit in IBS-D, as outlined below.
Immune response
It has been argued that the immune system participates in the pathophysiology of IBS based on the clinical observation that infectious gastroenteritis is a strong risk factor for the development of IBS24. Additional clinical support comes from the evidence that about one-third of patients with IBD in remission experience IBS-like symptoms35. These inferential data have been subsequently enriched by quantitative immunohistochemistry data showing increased infiltration of T cells and mast cells in the mucosa of the small and large intestine of some patients with IBS36.
Two randomized controlled trials (RCTs)37,38 in patients with IBS demonstrated that the anti-inflammatory agent mesalazine was not superior to placebo in alleviating IBS symptoms, although both studies clearly indicated that subgroups, particularly patients with post-infectious IBS, had sustained symptomatic responses. Thus, these studies confirm the hypothesis that immune activation has a considerable role in some patients with IBS.
Although mucosal immunocyte numbers are not always increased in IBS, there is strong functional and molecular evidence of an increased state of activation of immune cells in about half of patients with IBS36. Data from several studies point to the importance of mast cells as key components of inducing and maintaining low-grade immune activation in IBS36. For instance, higher proportions of mast cells were found in a degranulating state in colonic biopsies from patients with IBS than in control samples, suggesting that increased activation of mast cells is involved in the condition39. In addition, biopsy supernatants from patients with IBS contained higher amounts of mast cell mediators, including proteases and histamine36 as well as polyunsaturated fatty acid metabolites40, than controls. Mucosal immune activation is coupled with altered gene expression of several components of the host mucosal immune response to microbial pathogens (see below), suggesting that the microbiota might contribute to the observed immune activation36.
Neuroimmune interactions
Mucosal mediators isolated from biopsy samples from patients with IBS have been extensively studied to identify their effect on bowel physiology and sensory perception in isolated tissues or laboratory animals41. Compared with controls, mucosal mediators from patients with IBS evoked higher activation of visceral and somatic pain pathways when applied to intestinal preparations isolated from rodents42,43. Mast cells and enteroendocrine cells have been suggested to participate in this abnormal neural signalling, as indicated by the activation of human ENS neurons via mast cell-derived histamine, enteroendocrine cell-derived serotonin (also known as 5-hydroxytryptamine (5-HT)) and protease-dependent mechanisms30,42 (Fig. 5). Although most of the proteases are secreted by mast cells, some of the serine and cysteine proteases that are present at a higher level in the mucosa or stool of patients with IBS than controls might be of other, probably pancreatic or bacterial, origin. In line with these findings, serine proteases in faecal supernatants from individuals with IBS-D evoked colonic hypersensitivity to distension44. By contrast, faecal cysteine protease activity was augmented in some patients with IBS-C compared with controls and increased protease activity correlated with abdominal pain and impaired epithelial permeability45. Further work showed the implication of serine proteases that act on protease-activated nociceptors located on intestinal nerves conveying pain stimuli to the brain43. Importantly, mucosal mediators from patients with IBS and visceral hypersensitivity — but not from normosensitive patients with IBS — acutely activated spinal nociceptors when given to animal models46. In the same model, chronic exposure to soluble mediators from patients with IBS-D was shown to sensitize nociceptive neurons47, implying that chronicity is associated with long-lasting plasticity alterations.
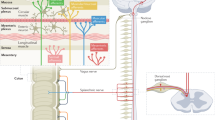
An intimate anatomical and functional association between enteric neurons, terminals from extrinsic nerves and cells of the enteric immune system is the basis for neuroimmune interactions in the gut wall. Functional signalling between nerves and immune cells mostly happens in the epithelial and submucosal layers where there is a high density of immune cells — in particular, T lymphocytes, mast cells and macrophages. The neuroimmune interactions are bidirectional. Enteric neurons, extrinsic nerves and glial cells respond to cytokines and mast cell mediators. Some patients with irritable bowel syndrome (IBS) have circulating autoantibodies against neuronal structures and antibodies that are generated as a response to antigen exposure from the lumen. Neurons can respond directly to antibodies through direct activation of channels or receptors. They also respond to antigens through pathways involving neuronal Toll-like receptor 3 (TLR3), TLR4 and TLR7. Direct signalling between microbiota and the host involves activation of neurons through polysaccharide A. These direct effects of luminal factors are very likely to be outnumbered by signalling between epithelial (in particular, enteroendocrine cells), immune and nerve cells. Neurons also express receptors for adenosine and ATP; both molecules are released in the gut wall under inflammatory or stress conditions. Reciprocally, nerves release factors that affect epithelial or immune cells. The best-documented effect is the activation of mast cells through the release of calcitonin gene-related peptide (CGRP) from extrinsic visceral afferents or enteric neurons. Conversely, acetylcholine (ACh) inhibits the activation of macrophages. Neurogenic inflammation, which is sometimes observed in animal models, is probably caused by the release of CGRP and substance P from extrinsic fibres followed by permeabilization of blood vessels. In addition, adipocytes in the lamina propria nestle against nerve fibres, and release of their pro-inflammatory mediators modulates nerve activity. The plus and minus symbols indicate whether a mediator activates or inhibits its target cell; those in parentheses denote actions established in animal models and those without parentheses are effects demonstrated in humans (human tissue). 5-HT, 5-hydroxytryptamine (also known as serotonin); CCK, cholecystokinin; CRF, corticotropin-releasing factor; IL-10, interleukin 10; NGF, nerve growth factor; PUFA, polyunsaturated fatty acid; TRP, transient receptor potential cation channel.
Attention has been directed to agonists of the transient receptor potential cation channels (TRPs), which have been implicated in the pathogenesis of sensory hyperalgesia. Colon tissue samples from patients with IBS have increased levels of specific polyunsaturated fatty acids, which stimulate sensory neurons from mice via the activation of TRP subfamily V member 4 (TRPV4) and generate visceral hypersensitivity40. The importance of those visceral afferents that express TRPs in IBS symptomatology is underscored by the finding that peripheral blood mononuclear cell (PBMC) supernatants from patients with IBS-D cause mechanical hypersensitivity of visceral afferents via tumour necrosis factor (TNF) and TRPA1; this was not observed if control supernatants were used48.
Recent data support the concept that the chronic release of factors with known effects on nerves in the intestinal milieu might not only have functional effects but could also affect the ENS and sensory fibres in a structural manner. For example, immunohistochemistry showed a 57.7% and 56.1% increase in mucosal neurons and neuronal outgrowth, respectively, in patients with IBS compared with healthy controls49. Indeed, the intestinal mucosa of patients with IBS contains increased levels of nerve growth factor (NGF), primarily in mucosal mast cells. Experimentally, the effect of NGF was demonstrated in primary cell cultures of the rat myenteric plexus and the neuroblastoma cell line SH-SY5Y, which showed an increase in neurite growth, and protein and mRNA expression of growth-associated protein 43 (GAP43; also known as neuromodulin) — a key neuronal growth protein — following exposure to supernatant obtained from mucosal biopsies of patients with IBS49.
Microbiota
The gastrointestinal microbiota is a diverse and numerous ecosystem that inhabits the entire gastrointestinal tract and has a systemic influence on our health. Owing to its enormous complexity and high interindividual variability, the microbiota is still in large part undefined regarding the scope of its contribution to human physiology and tolerable compositional variations under which normal functions are preserved50. The evidence for an involvement of altered gut microbiota composition in IBS pathophysiology has been accumulating (Box 4), but the aetiological role remains uncertain. The most prominent markers of IBS are derived from uncultured bacteria. Two groups of uncultured Clostridiales are significantly depleted in IBS51,52, and bacteria related to Ruminococcus torques (a species belonging to the Lachnospiraceae) are profoundly enriched in patients with IBS51,53,54 and levels positively correlate with bowel symptoms51,52,55. In addition, increased Firmicutes to Bacteroidetes ratios have been observed at the phylum level, at least in a subset of patients51 (for a recent review see Ref. 56). Given the provided evidence, the dysbiosis of microbiota in IBS has been acknowledged by the Rome Foundation Working Team57 as a plausible contributing factor to the disorder. Experiments with animal models have shown that colonization of germ-free animals with microbiota from patients with IBS can induce visceral hypersensitivity58, impair intestinal permeability and alter gastrointestinal transit time59 — indicating the importance and the possible aetiological role of the microbiota in IBS.
Although diet changes have an effect on the abundance of particular microbial groups, the microbiotic signature (in terms of present species) is very stable60. To observe a profound effect, the dietary change has to be dramatic (for example, vegans switching to high-fat and high-protein diets61). Dietary interventions (such as low dietary content of fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs; Box 5), or the addition of sweeteners (fructo-oligosaccharides) or fibre (psyllium)) can improve symptoms of some but not all patients with IBS. Future studies should evaluate the relevance of these microbial groups for IBS and could contribute to a better understanding of the role of the microbiota in the pathophysiology of IBS that is currently acknowledged for the following contexts.
Fermentation of non-digestible foods. An important role of the microbiota is degradation of non-digestible dietary components62. It is generally accepted that fermentation of carbohydrates is desirable because of the beneficial effects of the main fermentation products — short-chain fatty acids (SCFAs) — including energy supply to gastrointestinal epithelial cells, a decrease in inflammation and improvement in gut barrier function63. However, in patients with IBS, the presence of the resistant carbohydrates FODMAPs can provoke IBS symptoms64. This might be a result of overproduction or underproduction of relevant metabolites owing to the disturbed microbiotic balance, for example, due to an increased abundance of gas-producing and decreased abundance of gas-utilizing microorganisms. The quantity and composition of SCFAs in the gut differs between patients with IBS and healthy controls, although the available data are not always in agreement65,66. Moreover, the production of microbial SCFAs stimulates regulatory T cell differentiation and affects the balance between pro-inflammatory and anti-inflammatory mechanisms67, suggesting that inadequate levels of SCFAs could provoke low-grade intestinal inflammation as observed in patients with IBS68,69. Finally, studies of microbiota show that the abundance of several SCFA-producing bacteria — including Roseburia, Blautia and Veillonella70 — is significantly increased compared with the levels of these bacteria in healthy controls, providing a potential mechanistic basis for the development of IBS symptoms.
Other carbohydrate-utilizing gastrointestinal bacteria — namely, Dorea spp. — show significant increases in abundance in patients with IBS51; these are the main gas-producing bacteria in the human gastrointestinal tract71. The overproduction of gas is associated with IBS72 and this phenomenon could underlie flatulence and abdominal pain. The excessive production of gas can also cause faster colonic transit in patients with IBS-D, as the colons of these patients are more sensitive to increased intestinal volume than healthy controls73. Intestinal gases are efficiently removed by methanogenic archaea74, which seem to be depleted in patients with IBS51,52 and are negatively correlated with the presence of loose stools52. However, a significant increase in the abundance of this microbial group is characteristic of patients with slow transit and constipation75, whereas the degree of the methanogenic activity could be correlated with the severity of constipation in those with IBS-C76.
Another potential pathway for microbiotic involvement in IBS is protein degradation. The luminal contents of patients with IBS contain increased levels of proteases30, which could be due to the increased secretion of endogenous and microbial proteases in response to protein-rich nutrition (typical of western diets), but could also be due to insufficient endogenous protease degradation by the disturbed gastrointestinal microbial community77. Serine protease inhibitors are produced by many bacteria, including bifidobacteria78, and their activity could prevent the excessive proteolytic activity of intestinal content in IBS. The depletion of bifidobacteria has been noted in both faecal and mucosal samples of patients with IBS51,79, suggesting an important role for this bacterial genera in IBS. The fermentation of proteins generates numerous health-compromising substances80. Among these, hydrogen sulfide is a relevant toxin that impairs epithelial metabolism81 and can be further converted to tetrathionate, which stimulates the growth of tetrathionate-utilizing pathogens from Gammaproteobacteria82,83. The abundance of several Gammaproteobacteria significantly correlates with bowel symptoms in patients with IBS51,52, and also with the levels of the inflammatory markers interleukin 6 (IL-6) and IL-8 (Ref. 51) that are typically increased in IBS54.
Microbiota and 5-HT. 5-HT is an important metabolite that, among other functions, regulates gastrointestinal motility; disturbed levels of 5-HT seem to be relevant for IBS pathology84. As much as 90% of 5-HT is produced in enteroendocrine cells present in the gastrointestinal tract, and it has been recently shown that intestinal bacteria are needed for the stimulation of 5-HT synthesis. Attempts to identify microorganisms that are capable of 5-HT synthesis have shown that, in contrast to Bacteroides spp. and altered Schaedler flora (a community of eight bacterial strains), only specific spore-forming commensal bacteria have this feature. The majority of these spore-forming bacteria belong to the Clostridales class within the Firmicutes phylum. Two recent comprehensive studies51,85 revealed an increase of the Firmicutes phylum members on the account of the Bacteroidetes members in IBS. Given that the Clostridiales class within the Firmicutes phylum are the most diverse and the most abundant group of the microbiota70, it is not clear if the observed feature of the IBS microbiota is associated with 5-HT-mediated pathophysiology, but this possible link should certainly be further investigated.
Brain and behaviour
IBS is narrowly defined by recurrent abdominal pain and discomfort associated with altered bowel habits in the absence of an organic origin and/or explanation of symptoms. However, given that IBS is nearly always associated with increased anxiety and patients often show comorbidities with other chronic pain and psychiatric conditions, a more widespread dysregulation of the nervous and immune systems is probably implicated86.
The brain, the gut and its microbiota and the immune system show reciprocal associations in health and disease. On the one hand, the brain, via the autonomic nervous system and the HPA axis, can influence intestinal motility and fluid secretion87, intestinal epithelial permeability25,88,89, immune function90 and gut microbial composition91, all of which have been reported to be dysregulated in IBS. On the other hand, several of these peripheral alterations can influence brain structure and function either developmentally or in response to acute perturbations, setting up circular regulatory loops between the gut and the brain92.
In addition to its role in the bidirectional communications with the gut, the brain plays an essential part in assessing the salience of received or expected interoceptive (sensory) information93, determining how much of this information is amplified or tuned down, to what degree it is modulated by affect94 and how much of this interoceptive information from the gut is consciously perceived (visceral sensitivity). One of the best-studied behavioural aspects of IBS-related central processing of gut-related information involves a coping strategy referred to as catastrophizing, a term that refers to a bias towards prediction of a high likelihood of worst outcomes95. This measure strongly correlates with the severity of pain symptoms and is a primary treatment target in cognitive–behavioural therapy.
Multimodal brain imaging has made it possible to identify differences in functional (evoked and resting state) and structural (grey matter and white matter tracts) aspects of specific brain networks that provide a neurobiological substrate for previously observed affective and cognitive features of IBS (reviewed in Refs 92,96) (Fig. 6). These networks include the salience, attention, sensorimotor and emotional arousal networks. Profound sex-related differences in these networks have also been identified in both healthy individuals and patients with IBS (reviewed in Ref. 96). Cross-sectional correlations of brain networks with several clinical and non-brain biological parameters show a relationship between some of these brain signatures with IBS symptom severity and duration, a history of early adverse life events93, gut metabolite and microbial composition97, gene expression profiles in PBMCs98 and gene polymorphisms99. On the basis of these neurobiological findings, a comprehensive IBS pathophysiological model can be formulated (Fig. 6), which includes alterations in the appraisal of and selective attention to interoceptive signals (salience and attentional network), central sensory processing of interoceptive information (sensorimotor network) and engagement of emotional arousal associated with experience and expectation of gut sensations. This disease model not only identifies neurobiological correlates of well-characterized clinical and behavioural features of IBS but also provides a plausible explanation for the common coexistence of IBS with other chronic pain conditions and with increased trait anxiety.
Irritable bowel syndrome (IBS)-related pathways that are potential pharmacogenetic targets are marked in red when based on genetic findings and in blue when based on epigenetic findings; those in black are currently not seen as potential pharmacogenetic targets101. Various pathways might be affected in specific subgroups of patients with IBS: epithelial barrier (permeability), immune function, impaired bile acid metabolism and function, neuronal processing and signal transduction via spinal afferents from the periphery to the central nervous system in addition to the bidirectional crosstalk via the brain–gut axis, presumably contributing to psychological conditions such as anxiety, depression and somatization. Brain networks that have been associated with structural and functional alteration in IBS are depicted. ADRA, adrenoceptor-α; aINS, anterior insula; aMCC, anterior midcingulate cortex; CDC42, cell division cycle 42; CDH1, cadherin 1; CGN, cingulin; CLDN, claudin; COMT, catechol-O-methyltransferase; CRHR1, corticotropin-releasing hormone receptor 1; FGFR4, fibroblast growth factor receptor 4; GLUL, glutamate-ammonia ligase (also known as glutamine synthetase); GPBAR1, G protein-coupled bile acid receptor 1; GRID2IP, GRID2-interacting protein; HPA, hypothalamus–pituitary–adrenal; HTR, 5-hydroxytryptamine receptor; hypo, hypothalamus; IL, interleukin; KLB, Klotho-β; LCC, locus coeruleus complex; mir, microRNA; mPFC, medial prefrontal cortex; NKRF, nuclear factor-κB-repressing factor; NR3C1, nuclear receptor subfamily 3 group C member 1; NTS, solitary nucleus; OFC, orbitofrontal cortex; PAG, periaqueductal grey; PGR, progesterone receptor; SCN5A, sodium voltage-gated channel α-subunit 5; sgACC, subgenual anterior cingulate cortex; SLC6A4, solute carrier family 6 member 4; TNF, tumour necrosis factor; TNFSF15, TNF superfamily member 15; TRPV1, transient receptor potential cation channel subfamily V member 1.
Although these findings have identified disease-relevant brain alterations in patients with IBS, mechanistic and longitudinal studies are required to determine the causality between these factors. For example, are central sensorimotor alterations a consequence of increased signals from the gut, are they the consequence of dorsal horn sensitization by increased descending pain-facilitating signals or are they a genetically determined trait that predisposes individuals to IBS and might be present in asymptomatic relatives100? The correlation of gut microbial signatures and PBMC expression profiles with structural alterations in the sensorimotor network suggests a possible role of these peripheral factors in influencing the brain. Similarly, are the altered salience and attention network alterations a secondary response to the chronically increased perception of visceral signals or are they a primary abnormality that is responsible for the generation of aberrant endogenous pain modulation, as well as emotional and autonomic nervous system responses? Future studies will need to address the question of whether these brain signatures differ between subgroups of patients with IBS, such as male and female patients, patients with a history of early adversity, patients with different durations of symptoms and patients with post-infectious IBS.
Genetic and epigenetic data
The latest genetic and epigenetic findings support current models of IBS pathogenesis that suggest disturbed intestinal barrier function, immune response and neuronal signal transduction101 (Fig. 6). The data even point towards potential diagnostic biomarkers or therapeutic options (Box 3). For example, silencing the microRNA-29 (mir-29) family or amplifying mir-199a expression might have important therapeutic implications for selected patients with IBS and symptoms caused by increased intestinal permeability or hypersensitivity102,103.
Genetic data. Genetic studies to date range from family and twin studies to candidate gene approaches and, more recently, genome-wide association studies (GWAS). Regardless of enlarged sample sizes, increased statistical power and meta-analyses, genetic variants associated with IBS are still scarce and/or have not been replicated in independent cohorts. A recent paper summarizes all currently available genetic data that have been replicated101.
Polymorphisms or variants in several genes have been found to be associated with IBS. Genes encoding proteins involved in homeostasis of epithelial barrier function, such as cadherin 1 (CDH1) and cell division cycle 42 (CDC42), the immune system, such as IL6, IL10, TNF and TNF superfamily member 15 (TNFSF15; encoding cytokines and neuronal signal transduction) and others (such as neurexophilin 1 (NXPH1) and sodium voltage-gated channel α-subunit 5 (SCN5A)) have been replicated in several studies101. In 2014, a small pilot study reported an association between IBS and a locus on chromosome 10 (containing the protocadherin 15 (PCDH15) gene) in a discovery sample from Australia that could not be replicated in additional cohorts from Sweden and the United States104. Mutations in the following genes encoding proteins involved in the serotonergic system have also been shown to be associated with IBS: solute carrier family 6 member 4 (SLC6A4; also known as 5-HTTLPR or SERT), 5-HT receptor 3A (HTR3A), HTR3E and HTR4 (Ref. 101). A polymorphism in SLC6A4 has been found to be associated with altered brain responses, visualized through functional brain imaging following visceral pain stimuli in patients with IBS105. Furthermore, a functional polymorphism in HTR3A could be associated with altered amygdala responsiveness, anxiety and increased symptom score in IBS106. These findings underline the effect of polymorphic serotonergic and other genes in modulating gut-derived brain response in areas that process visceral perception and integrate autonomic control, salience and somatosensory and emotional central networks (Fig. 6).
Variants of genes encoding proteins that are involved in bile acid synthesis regulation (the Klotho-β (KLB) gene, the fibroblast growth factor receptor 4 (FGFR4) gene and the G protein-coupled bile acid receptor 1 (GPBAR1) gene) are associated with accelerated colonic transit in patients with IBS-D107,108. These variants also correlate with the colonic transit response to chenodeoxycholic acid (a bile acid used to treat constipation) in IBS-C109 and to colesevelam (a bile acid sequestrant used to treat diarrhoea) in patients with IBS-D110,111.
Finally, a locus at 7p22.1 in which the genes KDEL endoplasmic reticulum protein retention receptor 2 (KDELR2) and GRID2-interacting protein (GRID2IP) localize was significantly associated with IBS risk in the index GWAS (a large twin discovery sample from Sweden) and all replication cohorts in Europe, the United States and Australia112. However, the underlying molecular cause for this association finding has not been elucidated.
Epigenetic data. Even less insight into the role of epigenetics in IBS pathology is available compared to the genetic implications. To date, only a few miRNA studies have been performed. These studies reported on the differential expression profiles of miR-29a, miR-29b, miR-103, miR-16, miR-125b and miR-199a in the intestinal mucosa of patients with IBS-D. Upregulation of miR-29a and miR-29b was reported to accompany downregulation of the target genes encoding glutamine synthetase (GLUL)102, claudin 1 (CLDN1) and NF-κB-repressing factor (NKRF); CLDN1 and NKRF correlated with increased gut permeability103. In addition, decreased expression of miR-103, miR-16 and miR-125b correlated with the upregulation of the target genes encoding the tight junction proteins claudin 2 (CLDN2) and cingulin (CGN)113. In turn, a diminished miR-199 level correlated with an upregulation of TRPV1 and increased visceral sensitivity114. Moreover, variants residing in miRNA target regions of the 5-HT receptor genes HTR3E and HTR4B — namely, c.*76G>A and c.*61T>C — were found to be associated with IBS-D. Both variants were reported to impair miRNA regulation and to lead to disturbed expression regulation of miR-510 and miR-16, respectively115,116. One pilot study further indicated increased levels of circulating miR-150 and miR-342-3p in the blood of patients with IBS117. Of note, miR-150 has been described to be associated with IBD and pain, whereas miR-342-3p has been predicted to target genes that are relevant for pain signalling, colonic motility and smooth muscle function118.
Diagnosis, screening and prevention
The diagnosis of IBS relies on the patient fulfilling diagnostic criteria for IBS119 in conjunction with normal results on a limited number of additional tests and investigations used to rule out other diagnoses with reasonable certainty (Fig. 7). Although a substantial proportion of clinicians120 prefer a process of thorough exclusion of other diseases, the current recommendation is to base diagnosis on symptoms119. There is currently no valid biomarker for IBS121. The choice of the tests or investigations deemed necessary to rule out other conditions varies depending on the clinical situation and the symptom profile of the patient. In the majority of cases with a typical clinical history compatible with IBS, only a limited number of laboratory tests are recommended without any need to perform invasive investigations. Screening for IBS risk and for prevention of IBS development is currently not applicable, given the heterogeneity of the disease and the multiplicity of putative pathophysiological mechanisms.
Diagnostic criteria
As individual symptoms have poor sensitivity and specificity to diagnose IBS, diagnostic criteria incorporating a combination of symptoms have been developed, similar to the DSM system within psychiatry. The first attempt was the so-called Manning criteria, published in 1978 (Ref. 122). In this publication, several symptoms were shown to be more common in patients with IBS than in patients with another organic gastrointestinal disease. By combining these symptoms, IBS could be discriminated from other organic gastrointestinal diseases. The experience from the Manning criteria was then used to develop the Rome Foundation criteria, with three different versions over the past 15 years (Rome I, II and III); the latest criteria, the Rome III criteria, was published in 2006 (Refs 119,123,124). The updated Rome IV criteria are expected in May 2016. The sensitivity and specificity of the Rome criteria have been found to be 69–96% and 72–85%, respectively, in different studies, but a problem with these studies is how to define the gold standard for an IBS diagnosis121.
The common feature in all of these diagnostic criteria is abdominal pain and/or discomfort associated with abnormal bowel habit (diarrhoea (loose and frequent stools), constipation (hard and infrequent stools) or alternating constipation and diarrhoea). All of these criteria require a certain duration and frequency of the symptoms to fulfil the diagnostic criteria for IBS; that is, the symptoms should be chronic and recurring. Thus, the practical clinical use of the diagnostic criteria for IBS involves demonstrating through the clinical history the presence of a combination of these symptoms for ≥3 days per month in the past 3 months, with symptom onset ≥6 months before the diagnosis (Rome III criteria). However, it should be noted that patients with some organic gastrointestinal disease also meet these diagnostic criteria125 and, as such, the sensitivity and specificity of these criteria is suboptimal to distinguish the different disease entities125,126.
Clinical features
Besides the symptoms included in the diagnostic criteria, there are other clinical features that support a diagnosis of IBS, even though none of them is mandatory for an IBS diagnosis. One recent study found that variations in stool consistency and frequency or an unpredictable bowel pattern (‘irregularly irregular’) could be used to discriminate IBS-D adequately from organic gastrointestinal disease127. Moreover, abnormal stool frequency (>3 bowel movements per day or <3 bowel movements per week), excessive straining during defaecation, urgency (having to rush to the toilet), feelings of incomplete evacuation and mucus with bowel movements support an IBS diagnosis, but are nonspecific124. The same is true for postprandial worsening or exacerbation of symptoms, which is common in IBS128, but is also observed in other gastrointestinal diseases. The presence of other functional gastrointestinal diagnoses (such as functional dyspepsia)129, as well as reporting numerous functional non-gastrointestinal symptoms and syndromes (such as chronic fatigue, fibromyalgia, uro-gynaecological symptoms, muscle and joint pain and sleep disturbances)11,130 and psychological comorbidity (such as anxiety and depression)131, are all common and support an IBS diagnosis.
Physical examination
A physical examination should be part of the evaluation to reassure patients and also to help exclude another organic cause of the symptoms. Admittedly, an abdominal examination, which is part of the routine examination, rarely discloses a specific diagnosis (that is, abdominal tenderness is present in various diseases), but the absence of objective findings on a physical examination has been found to support a diagnosis of IBS132. A digital rectal examination is an important part of the physical examination and a useful tool to identify patients with dyssynergic defaecation, which is important to exclude in patients with constipation133,134 as well as to exclude rectal cancer. Perianal inspection should also be part of the examination to rule out perianal fistulas and other relevant anal pathology.
Laboratory tests
From the existing literature, it is not obvious which laboratory test to recommend in the diagnostic work-up of patients with IBS symptoms. Only serological tests for coeliac disease seem to be more likely to be abnormal in patients with symptoms compatible with IBS than in the general population135, even though a large multicentre trial failed to confirm this136. However, few studies have systematically evaluated the usefulness of laboratory tests in patients with potential IBS. A recent systematic review demonstrated that C-reactive protein (CRP) levels of ≤0.5 mg per dl or faecal calprotectin levels of ≤40 μg per g essentially exclude IBD in patients with IBS symptoms137. On the basis of the existing literature, it seems reasonable to perform a complete blood count and CRP measurement, as these are inexpensive and can be used to reassure the health-care provider and the patient. A thyroid profile can be included if the clinical suspicion of thyroid disease is high, a serological test for coeliac disease can be recommended in patients with non-constipated IBS and — if there is suspicion of an inflammatory process — a faecal calprotectin measurement can be added. Stool analyses to detect gastrointestinal infections can be considered if diarrhoea is predominant and difficult to treat, especially in regions where infectious diarrhoea is common138. As stated previously, there is currently no valid diagnostic biomarker, even though preliminary data have suggested that certain biomarkers or biomarker assays (Box 3) for clinical use might prove to be valid following further scientific investigation139,140.
Alarm features
Alarm features for IBS are symptoms that should raise the clinical concern of another gastrointestinal disease rather than IBS. Whether the use of alarm features (Box 6) improves the performance of diagnostic criteria for IBS is not totally clear125,141. However, from a clinical point of view, it seems reasonable to use these to select patients for further diagnostic testing, even though these may be present in a substantial proportion of patients without indicating a serious underlying condition in the gastrointestinal tract142. Alarm symptoms can necessitate further investigations to rule out another gastrointestinal disease before an IBS diagnosis can be recommended. Moreover, the predominance of diarrhoea, especially when watery and frequent, should alert the clinician to consider alternative diagnoses143.
Invasive investigations
In the majority of patients with symptoms compatible with IBS and normal routine laboratory tests but without alarm features144, no additional invasive investigations are needed and, importantly, performing investigations does not seem to improve patient satisfaction or QOL145,146.
Colonoscopy should be performed when alarm features prompt an investigation and when there is suspicion of an inflammatory condition in the gastrointestinal tract based on history or laboratory parameters (increased CRP or faecal calprotectin levels)137, or based on the indications for colorectal cancer screening in countries with population screening programmes147,148.
When the patient complains of watery diarrhoea as the predominant symptom, a colonoscopy with biopsies should also be considered to rule out microscopic colitis, especially in women >50 years of age143,149. Moreover, bile acid-induced diarrhoea has recently been found to be a very important differential diagnosis in patients with IBS symptoms with frequent, loose stools32,33, and a diagnostic test should be considered (75-homocholic acid taurine (75SeHCAT) test or serum C4 levels)150. Unfortunately, these tests are not available in all centres, therefore a therapeutic trial with a bile acid-binding agent is often used as an indirect, but far from perfect, assessment of bile acid-induced diarrhoea.
Carbohydrate malabsorption is another differential diagnosis in patients with IBS-D151–153, and lactose or fructose hydrogen breath tests can be considered154,155, but a trial period with dietary exclusion of the suspected carbohydrate for several weeks is often used instead.
If coeliac disease is suspected, based on a positive serological test or the clinical history, an upper gastrointestinal endoscopy with duodenal biopsies should be performed. Small intestinal bacterial overgrowth has been proposed to be common in IBS, but its prevalence and clinical importance is uncertain, therefore routine clinical testing for this cannot be advocated156,157, especially as valid tests with adequate sensitivity and specificity are lacking.
Management
Only a fraction of patients with IBS-like symptoms (∼50%) seek medical care158. Most of these patients will initially consult primary care physicians for their symptoms, and the factors that drive this consultation are symptom severity, especially pain, the occurrence of alarm symptoms (Box 6) and concerns that symptoms might indicate an underlying severe disease — for example, cancer159. Therefore, in many cases, gastrointestinal specialist care is needed to exclude diseases that can mimic IBS symptoms — for example, by endoscopy. Once a positive diagnosis of IBS has been established, clinical management can be carried out as well by primary care physicians and at substantially lower costs160.
Management of IBS involves an integrated approach, including the establishment of an effective patient–provider relationship, education, reassurance, dietary alterations, pharmacotherapy and behavioural and psychological treatment161. Owing to the fact that ∼50–70% of patients with IBS report additional somatic and psychological symptoms when they are asked161,162, a stepped-care approach including aspects of cognitive and interpersonal therapy is most appropriate15. The initial treatment strategy should be based on predominant symptoms and includes antispasmodics for abdominal pain, antidiarrhoeals for IBS-D and laxatives for IBS-C, whereas nutritional interventions and psychotherapy can be used in all subtypes.
Nutrition
Food ingestion is one of the most commonly reported factors that results in the exacerbation of symptoms among patients with IBS163,164. Postprandial symptoms per se and fear of their occurrence (anticipatory anxiety) contribute profoundly to reduced QOL in IBS128. Up until recently, food-related symptoms had received scant attention from clinical scientists, leaving patients to find their own way through the plethora of usually non-validated and untested diagnostic tests and dietary regimens, which could result in clinically relevant nutritional deficits165.
It has become evident that food intolerance (a physiological reaction to food allergens that is not associated with an immune response), and not classical IgE-mediated food allergy (which involves activation of the immune system), is the major mechanism responsible for symptomatic responses to certain foods165. This is not to say that immune responses to food or food components are irrelevant for IBS. For example, one study demonstrated that exposure of the small intestine to certain food antigens led to subtle ultrastructural changes in the duodenal mucosa of patients with IBS, but not in controls31. Another study also reported local immune responses to gluten among a group of non-coeliac patients with IBS166. Taken together, these observations leave the door open to the possibility that at least some patients with IBS may mount an, as yet to be defined, immunological response to certain dietary components, a response that seems to be confined to the mucosal immune system.
How does one explain food-related symptoms in IBS? Given the primacy of food ingestion as a stimulus to most gastrointestinal functions, postprandial pain and rectal urgency in IBS could simply reflect an exaggeration of a normal physiological phenomenon. Exaggerated motor responses to food and, especially to lipids, have also been demonstrated in the small intestine in IBS167. Furthermore, tryptophan, the 5-HT precursor, and related compounds present in some foods could modulate psychological comorbidities and gastrointestinal symptoms in IBS168. Food-related symptoms could also be mediated through interactions between our diet, the products of digestion and the gut microbiota. Products of bacterial metabolism, such as deconjugated bile salts, SCFAs and gases, could exert potent effects on colonic physiology and thereby induce symptoms.
Although patients with IBS readily incriminate specific food items as those that are especially likely to precipitate symptoms, only 11–27% of those are correctly identified when confirmed in formal, blinded food challenge studies169. The limitations of dietary surveys and the poor reproducibility of reported food intolerances notwithstanding, some food items are reported as being more problematic: wheat, fruit and vegetables170. Current enthusiasm for diets low in FODMAPs is consistent with these observations.
Fibre and fibre-based supplements accelerate colon transit, increase stool bulk and facilitate its passage, resulting in an increase in stool frequency. These effects translate into clinically meaningful benefits for people with chronic constipation and IBS-C. Indeed, fibre and products based on synthetic fibre-like substances became a cornerstone in the management of IBS. However, RCTs found that not all patients gained relief and some even complained of exacerbation of their symptoms (including pain, bloating and distension). Recent meta-analyses and systematic reviews have shed some light on this issue by showing that fibres are heterogeneous and the consumption of soluble fibres such as psyllium, calcium polycarbophil and ispaghula bring symptomatic benefits, whereas insoluble fibres, represented by bran, are ineffective in patients with IBS169.
Interest in the use of low FODMAP diets (Box 5) in patients with IBS is increasing. RCTs have confirmed some beneficial effects of low FODMAP diets on IBS symptoms171, but they were not superior to conventional dietary advice when directly compared172. There are some limitations; studies to date have been small and, as has been the case with many studies of dietary interventions in IBS, suffer from some methodological limitations173. Furthermore, low FODMAP diets are complex, may require supervision by a qualified dietician and involve the elimination of many food items commonly regarded as components of a ‘healthy’ diet. Some initial investigations suggest that the low FODMAP diet may suppress the growth of bacterial species commonly regarded as important components of healthy microbiota, such as bifidobacteria174. Included in the FODMAP category are some molecules, such as lactose, fructose and sorbitol; some patients with IBS may benefit from the removal of one of these substances alone175. Predicting responders is difficult, as commonly used challenge tests, such as the lactose or fructose breath hydrogen test, do not seem to be of value175,176.
The concept of ‘non-coeliac gluten sensitivity’ has been advanced to explain instances of IBS-type symptoms that develop in individuals who do not satisfy diagnostic criteria for the diagnosis of coeliac disease (that is, positive serology and appropriate changes in small intestinal morphology)177. This remains an unsettled and contentious issue with some studies reporting that, when tested in a blinded manner, gluten did induce the usual IBS symptoms in some patients with IBS178. Others argue that gluten contributes little to IBS symptomatology, but that fructans (FODMAPs contained in wheat), and not gluten, are the culprits of wheat-related problems. Results of clinical trials assessing the role of gluten exposure in IBS pathology have therefore, not surprisingly, yielded mixed results179,180. Although gluten-free diets are currently enjoying considerable popularity among patients with IBS and the population at large in the United States, the rationale for gluten exclusion in IBS has yet to be firmly established.
Patients with IBS commonly consume any one or combinations of a wide variety of dietary supplements ranging from vitamins to ‘digestive enzymes’, antioxidants and essential oils. Few, if any, of these have been subjected to rigorous study. Prebiotics (non-digestible food ingredients that beneficially affect the host by selectively stimulating the growth and activity of one species or a limited number of species of bacteria in the colon) and probiotics (live microbial food ingredients that alter the microflora and confer health benefit) have also been used for decades in IBS in the absence of supportive data. Prebiotics and probiotics are now subjected to more-rigorous studies, as they might contribute to altered microbiota in IBS181,182. Although these studies must be interpreted with care, a recent meta-analysis does suggest efficacy for probiotics (as a category) in IBS183. However, high-quality RCTs remain few in number and available data provide scant information to assist the consumer in choosing a particular product to alleviate symptoms184 or to make a recommendation on prebiotics or synbiotics (a combination of a prebiotic and a probiotic) in IBS185.
Drug therapy
Broadly speaking, the current therapeutic armamentarium in IBS aims to alter predominant problematic bowel habits and/or visceral pain. However, an emerging area is manipulation of the gastrointestinal microbiota.
Antispasmodic drugs. Pain in IBS is mediated through central and peripheral mechanisms, and is in part the result of smooth muscle spasms. The mode of action of antispasmodic drugs is probably their ability to antagonize the binding of acetylcholine to the muscarinic receptor at the neuromuscular junction, with smooth muscle relaxation as a consequence186. Some studies have demonstrated a beneficial effect of otilonium bromide and hyoscine over placebo, with a number needed to treat (NNT) of four patients187. An adverse effect of anti-muscarinic agents is constipation because of their strong inhibition of intraluminal fluid secretion186. Accordingly, these drugs are best used in patients without constipation and should be taken 20 minutes before meals to ease postprandial symptoms. Peppermint oil, which also inhibits smooth muscle contraction albeit by calcium channel blockade, is beneficial in reducing IBS symptoms188. A recent RCT in patients with IBS-D and IBS-M demonstrated that a novel formulation of peppermint oil, designed to cause a sustained release within the small bowel, was superior to placebo in causing a reduction in total symptoms189.
Low-dose antidepressants. Antidepressants, such as tricyclic antidepressants (TCAs) or selective serotonin reuptake inhibitors (SSRIs), are recommended by existing guidelines for the treatment of pain in patients who are refractory to antispasmodics and dietary alterations190. However, these drugs are not licensed anywhere in the world for the treatment of patients with IBS, and their use is off-label. Given the lack of licensed indication, the rationale for using such drugs should be discussed in detail with patients. The exact analgesic mechanism of action of low-dose antidepressants is incompletely understood but is considered to be both peripheral, via alterations of histaminergic and/or cholinergic transmission within the gastrointestinal tract, and central, via modulation of both ascending visceral sensory afferents and central transmission191. SSRIs are generally well tolerated. Adverse effects such as constipation, dry mouth, drowsiness and fatigue are reported with TCAs. TCAs may be particularly effective for treating pain in patients with IBS-D, but are less suitable for patients who have IBS-C.
Laxatives and motility accelerants. In those with constipation, simple laxatives such as senna and docusate are often effective in managing symptoms. However, the use of lactulose is not recommended as it is often poorly tolerated by patients with IBS because of worsening of bloating and discomfort. Linaclotide, a minimally absorbed guanylyl cyclase C agonist peptide (Fig. 8), can be used as second-line therapy after laxatives have failed in patients with IBS-C and symptoms have lasted for >1 year. Linaclotide has a dual action through increasing intraluminal fluid secretion thereby giving its laxation effect but also an analgesic effect via modulation of colonic nociceptors192, and its effects caused reduced abdominal pain, bloating and bowel symptoms in two well-designed Phase III RCTs193,194. Lubiprostone, a minimally absorbed, locally active, bicyclic fatty acid derivative of prostaglandin E1, activates type 2 chloride channels on the enterocyctic apical membrane, thereby stimulating fluid secretion. Lubiprostone has been shown to improve global intestinal symptoms in IBS-C195. 5-HT4 receptor agonists (such as prucalopride), which promote gut motility through the activation of the serotoninergic pathways, have been shown to be effective in increasing complete spontaneous bowel movements in patients with chronic constipation196.
Drugs currently used for the treatment of irritable bowel syndrome (IBS) (orange boxes) target nerve activity, epithelial functions or the contractile state of the smooth muscle layers. Several drugs act by enhancing the activity of chloride channels to increase fluid secretion into the intestinal lumen as a consequence. Other mechanisms of action include modulation of visceral sensitivity at a central or peripheral level. Finally, drugs act to modulate signal transduction at the neuromuscular junction or alter motility by direct myogenic actions. The plus and minus symbols indicate whether a mediator activates or inhibits its target cell; those in parentheses denote actions established in animal models and those without parentheses are effects demonstrated in humans (human tissue). 5-HT, 5-hydroxytryptamine (also known as serotonin); ACh, acetylcholine; CFTR, cystic fibrosis transmembrane conductance regulator; CIC2, chloride channel protein 2; GC-C, guanylyl cyclase C; VIP, vasoactive intestinal peptide.
Antidiarrhoeals. The μ-opioid receptor agonist loperamide is frequently used as a first-line agent in IBS-D and improves diarrhoea by inducing peristalsis, which prolongs the gastrointestinal transit time. As loperamide does not cross the blood–brain barrier, central adverse effects are limited. Its main benefit is reducing stool frequency and defaecation urgency, and improving the consistency of the stool197. Eluxadoline, a mixed μ-opioid receptor agonist and δ-opioid receptor antagonist, has been evaluated in a Phase III RCT, although safety concerns have been expressed concerning the excess rates of pancreatitis198.
5-HT3 receptor antagonists, such as alosetron, ramosetron and ondansetron, are effective in the management of IBS-D symptoms. The mechanism of action of 5-HT3 receptor antagonists is complex and incompletely understood, but is considered to proceed through inhibition of the ascending excitatory component of the peristaltic reflex and of the high amplitude propagating contractions within the gastrointestinal tract199. However, a central effect of 5-HT3 receptor antagonists on pain cannot be excluded200. Safety concerns, with respect to ischaemic colitis, have been confined to alosetron, which subsequently led to restrictions in its prescription201. Consequently, other 5-HT3 receptor antagonists have been investigated, with ondansetron202 and ramosetron demonstrating efficacy in RCTs203.
Manipulation of the microbiota. Given the burgeoning evidence of the role of the microbiota in IBS, both antibiotics and probiotics have been evaluated. The non-absorbable antibiotic, rifaximin, has been demonstrated to cause a reduction in symptoms, with a NNT of approximately 11 patients, although it is not clear whether repeated courses of treatment are needed204. The mechanisms by which rifaximin exerts its positive effects on IBS symptoms are incompletely understood and may include modulation of the gut microbiota, but also direct effects on local micro-inflammation. Rifaximin is approved for use in the United States, but has not yet received regulatory approval in Europe. Probiotics can reduce pain and symptom severity, although recent meta-analyses have highlighted that inconsistencies in study design render definitive recommendations problematic183,184,205; again, it is unclear whether probiotics act on IBS symptoms through direct modulation of the microbiota, indirect via the gut immune system or otherwise.
Others. A proportion of patients use herbal supplements either as single herbs or in combination. Four weeks of treatment with iberogast, which is a mixture of nine plant extracts, improved abdominal pain and QOL in a double-blind RCT of 208 patients with all types of IBS206. Although the mechanism of action is poorly understood, it is probably multifaceted via acetylcholine, 5-HT and opioid receptors in the gastrointestinal tract207. Although herbal remedies represent a promising intervention, further rigorously designed larger RCTs in the subtypes of IBS are needed.
Psychotherapy
The biopsychosocial model of IBS suggests that abdominal symptoms secondarily influence anxiety and depression (bottom-up) and psychosocial factors influence physiological factors, such as motor function, sensory threshold and stress reactivity of the gut (top-down)208.
Treatment concepts that target these psychosocial factors of patients with IBS should be based on evidence-based models that take the following three components into account: altered peripheral regulation of gut function, altered brain–gut signalling and reducing psychological distress, including general hypervigilance and a general mindset of catastrophizing209. Such models might be helpful as a basis of patient education and a target for effective treatments. To further improve treatment programmes, we have to learn more about IBS-specific interactions and the role of stress and visceral sensitivity for clearer evidence on which group of patients might benefit from which treatment approach. In addition, it should be noted that patients with IBS often experience additional functional symptoms, pointing to the complexity of the condition15.
The effect of IBS symptoms on patients' feelings of shame, fearfulness and embarrassment is well established; patients report being poorly understood by their physicians, as well as by their family members and friends210. Patients who experience a positive therapeutic physician–patient relationship have fewer IBS-related follow-up visits211.
International treatment guidelines for IBS have advocated for a graded treatment approach212,213. The National Institute of Health and Care Excellence (NICE) guidelines advise that patients whose symptoms do not respond to pharmacological treatments after 12 months and who develop a continuing symptom profile (refractory IBS) should be considered for referral to cognitive–behavioural therapy (CBT), hypnotherapy (gut-directed hypnosis) or other psychological therapy, such as psychodynamic (interpersonal) therapy and mindfulness-based therapy190.
Box 7 describes the four major psychological-based therapies for patients with IBS. Several meta-analyses have been performed in the field of psychological and behavioural therapies (including studies in stress reduction and relaxation) that took 45 RCTs into account with a total of 3,325 patients with IBS of all subtypes (Table 1). Overall, the NNT for psychological therapies is four patients (95% CI: 3–5) and, therefore, better than the majority of drugs214. In a stepped-care approach (beginning with the least intensive or invasive treatment and stepping up or down depending on the needs of the patients), a psychology-based self-aid (educational) approach has been shown recently in a meta-analysis as an effective treatment option for all subtypes of IBS250. Compared with control treatments, a medium effect size was demonstrated on decreased symptom severity and a large effect size on increased patient's QOL.
The best evidence is available for CBT. Although CBT is not routinely available in primary care, it can be accessed in some local hospitals and health-care systems. There are medium-to-large significant pooled effect sizes for an improvement of IBS symptoms using CBT with a medium significant pooled effect size for QOL and a small-to-medium pooled effect size for psychological comorbid symptoms. The NNT for CBT is only three patients, with a limited variance between the RCTs. Nevertheless, to date there is no evidence of a superiority of CBT compared with other psychological treatments in IBS.
Validation of psychodynamic (interpersonal) therapy, gut-directed hypnosis and mindfulness-based therapy (Box 7) has only been done in a very limited number of tertiary treatment centres and the generalization of these treatment approaches is limited. Finally, mindfulness-based therapy for IBS shows some promising initial results, particularly in the subgroup of female patients with IBS215. Very limited data on multicomponent therapies and on the combination of antidepressants and psychological treatments are available169. Overall, there is a lack of reports of adverse effects of psychological and behavioural treatment approaches and treatment resistance in patients with IBS. Psychological therapies have also regularly not distinguished between IBS subtypes and, thus, might have missed differential indications and advantages and disadvantages.
Quality of life
In the field of medicine, general QOL and disease-specific QOL are distinguished. General QOL is a measure of the entire health perception of a person. Representative general QOL can be assessed using the Medical Outcome Study 36-item Short-Form Health Survey (SF-36)216 or the EuroQOL survey217. SF-36 is the most popular instrument that can evaluate physical functioning, physical role, bodily pain, general health perceptions, vitality, social functioning, emotional role and mental health216. Disease-specific QOL is a measure of life disturbance that is specifically caused by the disease218,219.
QOL in patients with IBS is greatly disturbed. Patients with IBS showed impaired general QOL with lower values on all SF-36 subscales except physical functioning than healthy controls in one study220, whereas lower values on the SF-36 subscales in patients with IBS (except physical functioning, physical role and emotional role) than in healthy controls were observed in another study221. All subscales of SF-36, except the physical functioning and physical role domains, were lower in patients with IBS than in healthy controls regardless of culture222. The degree of disturbance of general QOL in patients with IBS has been shown to be worse in several subcategories than in those with gastroesophageal reflux disease, diabetes mellitus or severe chronic kidney disease220. Finally, a study has shown that patients with IBS had more disturbed general QOL in physical role, bodily pain, general health perceptions and social functioning than non-consulters with IBS (individuals who do not seek treatment)221.
QOL seems to be the same among IBS subtypes. However, disease-specific QOL, as measured with the IBS-QOL in patients with IBS-D or IBS-M, was worse than in patients with IBS-C in one study222. In this study, increased food avoidance in patients with IBS-D and IBS-M may have been responsible for the lower QOL222, but there are controversial reports218.
In severe IBS, both gastrointestinal symptoms and psychiatric comorbidity independently contribute to disturbed QOL223 (Fig. 9). Another study revealed that the QOL of patients with IBS was more influenced by the extraintestinal symptoms — such as tiring easily, low in energy, the feeling that there is something seriously wrong with their body, feeling tense, feeling nervous, feeling hopeless, difficulty sleeping and low sexual interest — than by gastrointestinal symptoms224. The psychological and psychosocial dimensions of food ingestion might also have a role. Eating with family and friends is probably the most common form of social interaction worldwide. An inability to participate in such a fundamental component of social intercourse because of a fear of pain, urgency, diarrhoea or distension occurring during or immediately after a meal can be devastating and can result in social isolation210.
The genome and epigenome partially determine (‘filter’) the response of an individual to external stressors (psychosocial factors) and internal stressors (ingested food or microbiota). These, together with social support, appraisal, emotion and coping behaviours against stressors, determine the stress response affecting the brain–gut interactions. This response might involve regional brain activation, changes in autonomic and neuroendocrine function, which might lead to many of the clinical manifestations observed in irritable bowel syndrome (IBS), including visceral hypersensitivity, alteration in gastrointestinal motility, increased mucosal permeability and low-grade inflammation. These gastrointestinal symptoms and other extra-intestinal manifestations (such as multiple somatic symptoms and psychiatric comorbidities) impair the quality of life (QOL) of patients with IBS.
Systematic reviews have clarified that improvement of IBS-related pain by treatment results in better QOL in patients with IBS225. The disease-specific IBS-QOL and IBS-QOL questionnaires can measure the efficacy of treatment, especially long-term therapies226. Although the SF-36 can also detect the efficacy of long-term treatment (>1 year), it is less sensitive than the IBS-QOL. Both measures struggle to detect drug or psychotherapy efficacy in the short term (<1 month)226,227, but IBS-QOL is sensitive enough to detect efficacy for mid-term (3 months) treatment203. A therapeutic gain of ≥14 points in IBS-QOL denotes a clinically meaningful change. Even if primary end points based on cardinal symptoms of IBS are similar between treatments, a treatment resulting in better QOL may be preferred by patients over another treatment that does not improve QOL.
Outlook
The field of research into IBS has expanded considerably over the past decade with many new studies, in part driven by the development of new therapeutic agents. This trajectory seems likely to continue as patients with IBS account for a substantial proportion of all gastrointestinal consultations, and many questions in the field remain unanswered (Box 8).
Patient stratification and biomarkers
Many classes of drugs have been evaluated by RCTs in IBS and these have often produced disappointingly small differences from placebo187,214,228. These small differences conceal the fact that some patients benefit from the drugs. Proper stratification of patients by relevant underlying disease mechanism has been an issue, therefore many trials use unselected patients with IBS, independent of the underlying disease mechanisms and clinical presentations. The use of 5-HT receptor-modulating drugs has taught the research community that restricting 5-HT3 receptor antagonists to patients without constipation improved their effectiveness with significant differences from placebo229,230, owing to the fact that 5-HT3 receptor antagonists slow transit and aggravate constipation. However, RCTs rarely measure transit as a requirement for trial entry, which depends on symptoms recorded in daily symptom diaries. The use of more-objective biomarkers to select patients for RCTs would be expected to improve the effect size and reduce the number needed to test to show a significant difference from placebo.
The lack of reproducible, widely available biomarkers that reflect the targets of ‘older’ drugs has been a considerable limitation. Antispasmodics are a good example of such drugs that have fallen out of favour because we cannot reliably identify those with excessive motor activity who might be expected to respond. Future novel non-invasive motility assessments, such as MRI231, capsule endoscopy232 and the pressure-sensitive, temperature-sensitive and pH-sensitive SmartPill (Given Imaging Ltd, Yoqneam, Israel)233 (which can measure intestinal contractions), hold the possibility of identifying such patients in the future.
Although individual genetic markers seem likely to be associated with only quite modest increases in risk for IBS, they might be important predictors of drug sensitivity in particular pathways. 5-HT3 receptor antagonists are good examples of drugs with a wide range of sensitivities such that effective doses for one patient can produce unacceptable constipation in another. This finding may be due to a combination of important functional polymorphisms in genes involved in 5-HT synthesis (tryptophan hydroxylase 1 (TPH1)), those involved in 5-HT reuptake via the 5-HT transporter (SLC6A4) and polymorphisms in the 5-HT3 receptor genes (which alter sensitivity). Several small studies have suggested significant differences in responder status to one 5-HT3 receptor antagonist, ramosetron, according to polymorphisms in TPH1 (Ref. 234) and to another 5-HT3 receptor antagonist, alosetron, according to polymorphisms in SLC6A4 (Ref. 235). However, these studies are underpowered and have not yet been reproduced202. By analogy with other complex disorders236, the effect of any one individual polymorphism may be limited but combining polymorphisms that predict low 5-HT production with rapid uptake and low receptor sensitivity would be expected to be associated with higher odds ratios for success of 5-HT manipulation. Future studies should be powered to examine this notion such that the dose can be tailored to individual patients. Similarly, polymorphisms in the FGF19–FGFR4 pathway, which controls bile acid synthesis107,108, influence colonic transit and should be explored to see if different combinations alter sensitivity to bile acid sequestrants or bile acid transporter inhibitors.
Mode of action of food intolerances
Dietary restrictions such as low FODMAP diets (Box 5) are another example in which implementation of an effective treatment is hampered by lack of biomarkers to predict response or reliably identify the key component (or components) of food that are responsible for symptoms. Although poorly absorbed fermentable carbohydrates can undoubtedly cause symptoms in some patients, visceral sensitivity is the key to why some individuals experience symptoms and some do not237, at least in the case of lactose malabsorption. However, no trial of lactose exclusion in IBS has used measures of sensitivity to stratify patients. While rectal barostat tests to assess visceral sensitivity are difficult, although not impossible to standardize across centres, alternatives might be to use simple cutaneous pressure or thermal stimulation238. More remotely, somatization questionnaires concerning non-gastrointestinal symptoms such as headache, backache, dyspnoea and palpitations have been shown to correlate, albeit weakly, with rectal distension pressure thresholds for pain239.
The physical form of food is another key variable whose importance is yet to be defined. Many of the dietary components implicated in IBS symptoms are actually consumed as solids and hence delivered into the duodenum more slowly after trituration by antral contractions. The rapid entry of osmotically active poorly absorbed substrates — mainly in liquid form — such as lactose in a patient with lactose malabsorption240,241 or mannitol in healthy volunteers241 result in a rapid influx of water into the small intestine, which probably stimulates transit and rapid delivery into the colon. This leads to the virtually instantaneous generation of gas242, mainly hydrogen, given that the microbiota are unable to fully metabolize the sudden excess of substrate. Furthermore, distension of the ascending colon generates propulsive colonic motility, which a sensitized individual may experience as cramps; a slower delivery in a solid matrix may be better tolerated. Future studies should define how the physical form of FODMAPs alters their tolerability, which would allow a less restrictive diet that may be easier to follow and, hence, more widely adopted than at present.
Functional effect of changes in microbiota
Many studies have found profound differences in the microbiota of selected patients with IBS, but the agreement on the involved species between studies is poor57. Given the very large number of different species that have overlapping metabolic capabilities and functional effects, focusing on function may be more helpful than just identifying the species present.
Analysis of urine and stool metabolites, including bile acids and endogenous tryptase, may provide simpler biomarkers of function that could predict responsiveness to microbiota manipulation. Thus, low levels of butyrate, a SCFA, might encourage the provision of prebiotics that favour butyrate-producing bacteria, such as Eubacterium rectale and Roseburia cecicola. Future studies should also take into account the important role of transit time and its variability. The challenge of rapid transit favours organisms with either enhanced growth capacity or those that adhere to the mucosa to deal with rapid flow within the colon243, although, these results need to be replicated and studied in more detail to enable dissection of the extent to which differences in microbiota are the cause or the effect of rapid transit. Better insight might also enable the tailoring of diet to the existing microbiota in a patient, based on their metabolic capabilities and response to a substrate provided in the diet.
References
Lovell, R. M. & Ford, A. C. Global prevalence of and risk factors for irritable bowel syndrome: a meta-analysis. Clin. Gastroenterol. Hepatol. 10, 712–721.e4 (2012). This meta-analysis covers epidemiological population-based data across 90 studies in 33 countries worldwide and reports not only the pooled prevalences but also several risk factors common to all studies.
Gwee, K. A. Irritable bowel syndrome in developing countries — a disorder of civilization or colonization? Neurogastroenterol. Motil. 17, 317–324 (2005).
Olafsdottir, L. B., Gudjonsson, H., Jonsdottir, H. H. & Thjodleifsson, B. Stability of the irritable bowel syndrome and subgroups as measured by three diagnostic criteria — a 10-year follow-up study. Aliment. Pharmacol. Ther. 32, 670–680 (2010).
Hungin, A. P., Whorwell, P. J., Tack, J. & Mearin, F. The prevalence, patterns and impact of irritable bowel syndrome: an international survey of 40,000 subjects. Aliment. Pharmacol. Ther. 17, 643–650 (2003).
Halder, S. L. et al. Natural history of functional gastrointestinal disorders: a 12-year longitudinal population-based study. Gastroenterology 133, 799–807 (2007).
Ford, A. C., Forman, D., Bailey, A. G., Axon, A. T. & Moayyedi, P. Irritable bowel syndrome: a 10-yr natural history of symptoms and factors that influence consultation behavior. Am. J. Gastroenterol. 103, 1229–1239 (2008).
Yarandi, S. S., Nasseri-Moghaddam, S., Mostajabi, P. & Malekzadeh, R. Overlapping gastroesophageal reflux disease and irritable bowel syndrome: increased dysfunctional symptoms. World J. Gastroenterol. 16, 1232–1238 (2010).
Ford, A. C. et al. Characteristics of functional bowel disorder patients: a cross-sectional survey using the Rome III criteria. Aliment. Pharmacol. Ther. 39, 312–321 (2014).
Long, M. D. & Drossman, D. A. Inflammatory bowel disease, irritable bowel syndrome, or what?: A challenge to the functional-organic dichotomy. Am. J. Gastroenterol. 105, 1796–1798 (2010).
Keohane, J. et al. Irritable bowel syndrome-type symptoms in patients with inflammatory bowel disease: a real association or reflection of occult inflammation? Am. J. Gastroenterol. 105, 1789–1794 (2010).
Whitehead, W. E. et al. Comorbidity in irritable bowel syndrome. Am. J. Gastroenterol. 102, 2767–2776 (2007).
Matheis, A., Martens, U., Kruse, J. & Enck, P. Irritable bowel syndrome and chronic pelvic pain: a singular or two different clinical syndrome? World J. Gastroenterol. 13, 3446–3455 (2007).
Janssens, K. A., Zijlema, W. L., Joustra, M. L. & Rosmalen, J. G. Mood and anxiety disorders in chronic fatigue syndrome, fibromyalgia, and irritable bowel syndrome: results from the LifeLines Cohort study. Psychosom. Med. 77, 449–457 (2015).
Barsky, A. J. Assessing the new DSM-5 diagnosis of somatic symptom disorder. Psychosom. Med. 78, 2–4 (2016).
Henningsen, P., Zipfel, S. & Herzog, W. Management of functional somatic syndromes. Lancet 369, 946–955 (2007).
Lovell, R. M. & Ford, A. C. Effect of gender on prevalence of irritable bowel syndrome in the community: systematic review and meta-analysis. Am. J. Gastroenterol. 107, 991–1000 (2012).
Goodwin, L., White, P. D., Hotopf, M., Stansfeld, S. A. & Clark, C. Life course study of the etiology of self-reported irritable bowel syndrome in the 1958 British birth cohort. Psychosom. Med. 75, 202–210 (2013).
Saito, Y. A. et al. Familial aggregation of irritable bowel syndrome: a family case–control study. Am. J. Gastroenterol. 105, 833–841 (2010).
Bengtson, M. B., Aamodt, G., Vatn, M. H. & Harris, J. R. Co-occurrence of IBS and symptoms of anxiety or depression, among Norwegian twins, is influenced by both heredity and intrauterine growth. BMC Gastroenterol. 15, 9 (2015).
van Tilburg, M. A. et al. Psychosocial mechanisms for the transmission of somatic symptoms from parents to children. World J. Gastroenterol. 21, 5532–5541 (2015).
Koloski, N. A. et al. Identification of early environmental risk factors for irritable bowel syndrome and dyspepsia. Neurogastroenterol. Motil. 27, 1317–1325 (2015).
Schwille-Kiuntke, J., Frick, J. S., Zanger, P. & Enck, P. Post-infectious irritable bowel syndrome — a review of the literature. Z. Gastroenterol. 49, 997–1003 (2011).
Schwille-Kiuntke, J., Mazurak, N. & Enck, P. Systematic review with meta-analysis: post-infectious irritable bowel syndrome after travellers' diarrhoea. Aliment. Pharmacol. Ther. 41, 1029–1037 (2015).
Thabane, M., Kottachchi, D. T. & Marshall, J. K. Systematic review and meta-analysis: the incidence and prognosis of post-infectious irritable bowel syndrome. Aliment. Pharmacol. Ther. 26, 535–544 (2007). This is so far the largest meta-analysis of 18 reports of IBS occurrence following an acute gastrointestinal viral or bacterial infection (called post-infectious IBS) and its potential risk factors and predictors.
Bischoff, S. C. et al. Intestinal permeability — a new target for disease prevention and therapy. BMC Gastroenterol. 14, 189 (2014).
Martinez, C. et al. Diarrhoea-predominant irritable bowel syndrome: an organic disorder with structural abnormalities in the jejunal epithelial barrier. Gut 62, 1160–1168 (2013).
Piche, T. et al. Impaired intestinal barrier integrity in the colon of patients with irritable bowel syndrome: involvement of soluble mediators. Gut 58, 196–201 (2009).
Bertiaux-Vandaele, N. et al. The expression and the cellular distribution of the tight junction proteins are altered in irritable bowel syndrome patients with differences according to the disease subtype. Am. J. Gastroenterol. 106, 2165–2173 (2011).
Coeffier, M. et al. Increased proteasome-mediated degradation of occludin in irritable bowel syndrome. Am. J. Gastroenterol. 105, 1181–1188 (2010).
Buhner, S. et al. Activation of human enteric neurons by supernatants of colonic biopsy specimens from patients with irritable bowel syndrome. Gastroenterology 137, 1425–1434 (2009). This paper reports the relevance of a novel technique to identify biomarkers of micro-inflammation in IBS by using supernatants from biopsies of patients with IBS applied in vitro to isolated neurons of the ENS of animals.
Fritscher-Ravens, A. et al. Confocal endomicroscopy shows food-associated changes in the intestinal mucosa of patients with irritable bowel syndrome. Gastroenterology 147, 1012–1020.e4 (2014).
Slattery, S. A., Niaz, O., Aziz, Q., Ford, A. C. & Farmer, A. D. Systematic review with meta-analysis: the prevalence of bile acid malabsorption in the irritable bowel syndrome with diarrhoea. Aliment. Pharmacol. Ther. 42, 3–11 (2015).
Bajor, A., Tornblom, H., Rudling, M., Ung, K. A. & Simren, M. Increased colonic bile acid exposure: a relevant factor for symptoms and treatment in IBS. Gut 64, 84–92 (2015).
Valentin, N. et al. Biomarkers for bile acid diarrhoea in functional bowel disorder with diarrhoea: a systematic review and meta-analysis. Guthttp://dx.doi.org/10.1136/gutjnl-2015-309889 (2015).
Barbara, G., Cremon, C. & Stanghellini, V. Inflammatory bowel disease and irritable bowel syndrome: similarities and differences. Curr. Opin. Gastroenterol. 30, 352–358 (2014).
Barbara, G. et al. The immune system in irritable bowel syndrome. J. Neurogastroenterol. Motil. 17, 349–359 (2011).
Barbara, G. et al. Randomised controlled trial of mesalazine in IBS. Gut 65, 82–90 (2016).
Lam, C. et al. A mechanistic multicentre, parallel group, randomised placebo-controlled trial of mesalazine for the treatment of IBS with diarrhoea (IBS-D). Gut 65, 91–99 (2016).
Barbara, G. et al. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology 126, 693–702 (2004).
Cenac, N. et al. Quantification and potential functions of endogenous agonists of transient receptor potential channels in patients with irritable bowel syndrome. Gastroenterology 149, 433–444.e7 (2015).
Nasser, Y., Boeckxstaens, G. E., Wouters, M. M., Schemann, M. & Vanner, S. Using human intestinal biopsies to study the pathogenesis of irritable bowel syndrome. Neurogastroenterol. Motil. 26, 455–469 (2014).
Barbara, G. et al. Mast cell-dependent excitation of visceral-nociceptive sensory neurons in irritable bowel syndrome. Gastroenterology 132, 26–37 (2007).
Cenac, N. et al. Role for protease activity in visceral pain in irritable bowel syndrome. J. Clin. Invest. 117, 636–647 (2007).
Annahazi, A. et al. Fecal proteases from diarrheic-IBS and ulcerative colitis patients exert opposite effect on visceral sensitivity in mice. Pain 144, 209–217 (2009).
Annahazi, A. et al. Luminal cysteine-proteases degrade colonic tight junction structure and are responsible for abdominal pain in constipation-predominant IBS. Am. J. Gastroenterol. 108, 1322–1331 (2013).
Buhner, S. et al. Neuronal activation by mucosal biopsy supernatants from irritable bowel syndrome patients is linked to visceral sensitivity. Exp. Physiol. 99, 1299–1311 (2014).
Valdez-Morales, E. E. et al. Sensitization of peripheral sensory nerves by mediators from colonic biopsies of diarrhea-predominant irritable bowel syndrome patients: a role for PAR2. Am. J. Gastroenterol. 108, 1634–1643 (2013).
Hughes, P. A. et al. Sensory neuro-immune interactions differ between irritable bowel syndrome subtypes. Gut 62, 1456–1465 (2013).
Dothel, G. et al. Nerve fiber outgrowth is increased in the intestinal mucosa of patients with irritable bowel syndrome. Gastroenterology 148, 1002–1011.e4 (2015).
Turnbaugh, P. J. et al. The human microbiome project: exploring the microbial part of ourselves in a changing world. Nature 449, 804–810 (2007).
Rajilic´-Stojanovic´, M. et al. Global and deep molecular analysis of microbiota signatures in fecal samples from patients with irritable bowel syndrome. Gastroenterology 141, 1792–1801 (2011).
Jalanka-Tuovinen, J. et al. Faecal microbiota composition and host-microbe cross-talk following gastroenteritis and in postinfectious irritable bowel syndrome. Gut 63, 1737–1745 (2014).
Saulnier, D. M. et al. Gastrointestinal microbiome signatures of pediatric patients with irritable bowel syndrome. Gastroenterology 141, 1782–1791 (2011).
Scully, P. et al. Plasma cytokine profiles in females with irritable bowel syndrome and extra-intestinal co-morbidity. Am. J. Gastroenterol. 105, 2235–2243 (2010).
Malinen, E. et al. Association of symptoms with gastrointestinal microbiota in irritable bowel syndrome. World J. Gastroenterol. 16, 4532–4540 (2010).
Rajilic´-Stojanovic´, M. et al. Intestinal microbiota and diet in IBS: causes, consequences, or epiphenomena? Am. J. Gastroenterol. 110, 278–287 (2015). This narrative review summarizes state-of-the-art knowledge on the role of the gut microbiota and its influence through dietary, probiotic and pharmacological manipulation.
Simrén, M. et al. Intestinal microbiota in functional bowel disorders: a Rome foundation report. Gut 62, 159–176 (2013).
Crouzet, L. et al. The hypersensitivity to colonic distension of IBS patients can be transferred to rats through their fecal microbiota. Neurogastroenterol. Motil. 25, e272–e282 (2013).
De Palma, G. et al. The adoptive transfer of anxiety and gut dysfunction from IBS patients to axenic mice through microbiota transplantation. Gastroenterology 146, S-845 (2014).
Rajilic´-Stojanovic´, M., Heilig, H. G., Tims, S., Zoetendal, E. G. & de Vos, W. M. Long-term monitoring of the human intestinal microbiota composition. Environ. Microbiol. 15, 1146–1159 (2013).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Cummings, J. H. & Macfarlane, G. T. Role of intestinal bacteria in nutrient metabolism. Clin. Nutr. 16, 3–11 (1997).
Hamer, H. M. et al. Review article: the role of butyrate on colonic function. Aliment. Pharmacol. Ther. 27, 104–119 (2008).
Shepherd, S. J., Parker, F. C., Muir, J. G. & Gibson, P. R. Dietary triggers of abdominal symptoms in patients with irritable bowel syndrome: randomized placebo-controlled evidence. Clin. Gastroenterol. Hepatol. 6, 765–771 (2008).
Treem, W. R., Ahsan, N., Kastoff, G. & Hyams, J. S. Fecal short-chain fatty acids in patients with diarrhea-predominant irritable bowel syndrome: in vitro studies of carbohydrate fermentation. J. Pediatr. Gastroenterol. Nutr. 23, 280–286 (1996).
Mortensen, P. B., Andersen, J. R., Arffmann, S. & Krag, E. Short-chain fatty acids and the irritable bowel syndrome: the effect of wheat bran. Scand. J. Gastroenterol. 22, 185–192 (1987).
Arpaia, N. et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 504, 451–455 (2013).
Spiller, R. C. Inflammation as a basis for functional GI disorders. Best Pract. Res. Clin. Gastroenterol. 18, 641–661 (2004).
Bercik, P., Verdu, E. F. & Collins, S. M. Is irritable bowel syndrome a low-grade inflammatory bowel disease? Gastroenterol. Clin. North Am. 34, 235–245 (2005).
Rajilic´-Stojanovic´, M. & de Vos, W. M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 38, 996–1047 (2014).
Taras, D., Simmering, R., Collins, M. D., Lawson, P. A. & Blaut, M. Reclassification of Eubacterium formicigenerans Holdeman and Moore 1974 as Dorea formicigenerans gen. nov., comb. nov., and description of Dorea longicatena sp. nov., isolated from human faeces. Int. J. Syst. Evol. Microbiol. 52, 423–428 (2002).
King, T. S., Elia, M. & Hunter, J. O. Abnormal colonic fermentation in irritable bowel syndrome. Lancet 352, 1187–1189 (1998).
Pritchard, S. E. et al. Fasting and postprandial volumes of the undisturbed colon: normal values and changes in diarrhea-predominant irritable bowel syndrome measured using serial MRI. Neurogastroenterol. Motil. 26, 124–130 (2014).
Pimentel, M., Gunsalus, R. P., Rao, S. S. C. & Zhang, H. Methanogens in human health and disease. Am. J. Gastroenterol. Suppl. 1, 28–33 (2012).
Attaluri, A., Jackson, M., Valestin, J. & Rao, S. S. C. Methanogenic flora is associated with altered colonic transit but not stool characteristics in constipation without IBS. Am. J. Gastroenterol. 105, 1407–1411 (2010).
Chatterjee, S., Park, S., Low, K., Kong, Y. & Pimentel, M. The degree of breath methane production in IBS correlates with the severity of constipation. Am. J. Gastroenterol. 102, 837–841 (2007).
Tooth, D. et al. Characterisation of faecal protease activity in irritable bowel syndrome with diarrhoea: origin and effect of gut transit. Gut 63, 753–760 (2014).
Ivanov, D. et al. A serpin from the gut bacterium Bifidobacterium longum inhibits eukaryotic elastase-like serine proteases. J. Biol. Chem. 281, 17246–17252 (2006).
Kerckhoffs, A. P. M. et al. Lower Bifidobacteria counts in both duodenal mucosa-associated and fecal microbiota in irritable bowel syndrome patients. World J. Gastroenterol. 15, 2887–2892 (2009).
Rajilic´-Stojanovic´, M. Function of the microbiota. Best Pract. Res. Clin. Gastroenterol. 27, 5–16 (2013).
Jørgensen, J. & Mortensen, P. Hydrogen sulfide and colonic epithelial metabolism. Dig. Dis. Sci. 46, 1722–1732 (2001).
Thiennimitr, P. et al. Intestinal inflammation allows Salmonella to use ethanolamine to compete with the microbiota. Proc. Natl Acad. Sci. USA 108, 17480–17485 (2011).
Weissfeld, A. S. & Sonnenwirth, A. C. Rapid isolation of Yersinia spp. from feces. J. Clin. Microbiol. 15, 508–510 (1982).
Spiller, R. Serotonin and GI clinical disorders. Neuropharmacology 55, 1072–1080 (2008).
Jeffery, I. B. et al. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 61, 997–1006 (2012).
Mayer, E. A. & Bushnell, M. C. in Functional Pain Syndromes: Presentation and Pathophysiology 531–565 (eds Mayer, E. A. & Bushnell, M. C. ) (IASP Press, 2009).
Mayer, E. A. et al. Functional GI disorders: from animal models to drug development. Gut 57, 384–404 (2008).
Camilleri, M. Peripheral mechanisms in irritable bowel syndrome. N. Engl. J. Med. 367, 1626–1635 (2012).
Keita, A. V. & Soderholm, J. D. The intestinal barrier and its regulation by neuroimmune factors. Neurogastroenterol. Motil. 22, 718–733 (2010).
Elenkov, I. J. & Chrousos, G. P. Stress system — organization, physiology and immunoregulation. Neuroimmunomodulation 13, 257–267 (2006).
Mayer, E. A., Savidge, T. & Shulman, R. J. Brain–gut microbiome interactions and functional bowel disorders. Gastroenterology 146, 1500–1512 (2014).
Mayer, E. A., Labus, J. S., Tillisch, K., Cole, S. W. & Baldi, P. Towards a systems view of IBS. Nat. Rev. Gastroenterol. Hepatol. 12, 592–605 (2015). This narrative review summarizes the current knowledge on brain networks in relation to genomic-, gastrointestinal-, immune- and gut microbiota-related parameters and develops a novel model to understand their importance in IBS.
Grupe, D. W. & Nitschke, J. B. Uncertainty and anticipation in anxiety: an integrated neurobiological and psychological perspective. Nat. Rev. Neurosci. 14, 488–501 (2013).
Elsenbruch, S. et al. Patients with irritable bowel syndrome have altered emotional modulation of neural responses to visceral stimuli. Gastroenterology 139, 1310–1319 (2010).
Lackner, J. M. & Gurtman, M. B. Pain catastrophizing and interpersonal problems: a circumplex analysis of the communal coping model. Pain 110, 597–604 (2004).
Mayer, E. A., Gupta, A., Kilpatrick, L. A. & Hong, J. Y. Imaging brain mechanisms in chronic visceral pain. Pain 156, S50–S63 (2015).
Labus, J. S. et al. Regional brain morphology is associated with gut microbial metabolites in irritable bowel syndrome (IBS). Gastroenterology 148, S-142 (2015).
Mayer, E. A. et al. Correlation between gene expression profiles in peripheral blood mononuclear cells and structural and functional brain networks in chronic visceral pain. Neuropsychopharmacology 39, S641–S642 (2014).
Orand, A. et al. Catecholaminergic gene polymorphisms are associated with GI symptoms and morphological brain changes in irritable bowel syndrome. PLoS ONE 10, e0135910 (2015).
Erpelding, N., Moayedi, M. & Davis, K. D. Cortical thickness correlates of pain and temperature sensitivity. Pain 153, 1602–1609 (2012).
Gazouli, M. et al. Lessons learned: resolving the enigma of genetic factors in irritable bowel syndrome. Nat. Rev. Gastroenterol. Hepatol. 13, 77–87 (2015). This narrative review summarizes the current knowledge about genetic and epigenetic findings in IBS and critically discusses the methodological limitations that have to be overcome to underline clinical relevance of the findings.
Zhou, Q., Souba, W. W., Croce, C. M. & Verne, G. N. MicroRNA-29a regulates intestinal membrane permeability in patients with irritable bowel syndrome. Gut 59, 775–784 (2010).
Zhou, Q. et al. MicroRNA 29 targets nuclearfactor-κB-repressing factor and claudin 1 to increase intestinal permeability. Gastroenterology 148, 158–169.e8 (2015).
Holliday, E. G. et al. Genome-wide association study identifies two novel genomic regions in irritable bowel syndrome. Am. J. Gastroenterol. 109, 770–772 (2014).
Fukudo, S. et al. Impact of serotonin transporter gene polymorphism on brain activation by colorectal distention. NeuroImage 47, 946–951 (2009).
Kilpatrick, L. A. et al. The HTR3A polymorphismc.–42C<T is associated with amygdala responsiveness in patients with irritable bowel syndrome. Gastroenterology 140, 1943–1951 (2011).
Wong, B. S. et al. A Klothoβ variant mediates protein stability and associates with colon transit in irritable bowel syndrome with diarrhea. Gastroenterology 140, 1934–1942 (2011).
Camilleri, M. et al. Genetic variation in GPBAR1 predisposes to quantitative changes in colonic transit and bile acid excretion. Am. J. Physiol. Gastrointest. Liver Physiol. 307, G508–G516 (2014).
Rao, A. S. et al. Chenodeoxycholate in females with irritable bowel syndrome-constipation: a pharmacodynamic and pharmacogenetic analysis. Gastroenterology 139, 1549–1558.e1 (2010).
Wong, B. S. et al. Pharmacogenetics of the effects of colesevelam on colonic transit in irritable bowel syndrome with diarrhea. Dig. Dis. Sci. 57, 1222–1226 (2012).
Camilleri, M. et al. Effect of increased bile acid synthesis or fecal excretion in irritable bowel syndrome-diarrhea. Am. J. Gastroenterol. 109, 1621–1630 (2014).
Ek, W. E. et al. Exploring the genetics of irritable bowel syndrome: a GWA study in the general population and replication in multinational case–control cohorts. Gut 64, 1774–1782 (2014).
Martinez, C. et al. Differential expression of miRNAs in the jejunal mucosa of IBS-D is involved in intestinal epithelial barrier dysfunction through modulation of specific tight junction proteins. United European Gastroenterol. J. 3 (Suppl. 5), A88 (2015).
Zhou, Q. et al. Decreased miR-199 augments visceral pain in patients with IBS through translational upregulation of TRPV1. Gut http://dx.doi.org/10.1136/gutjnl-2013-306464 (2015).
Wohlfarth, C. et al. Impaired miRNA regulation may lead to altered 5-HT4 receptor levels in IBS-D patients. United European Gastroenterol. J. 1 (Suppl. 1), A27 (2013).
Kapeller, J. et al. First evidence for an association of a functional variant in the microRNA-510 target site of the serotonin receptor-type 3E gene with diarrhea predominant irritable bowel syndrome. Hum. Mol. Genet. 17, 2967–2977 (2008).
Fourie, N. H. et al. Elevated circulating miR-150 and miR-342-3p in patients with irritable bowel syndrome. Exp. Mol. Pathol. 96, 422–425 (2014).
Gheinani, A. H., Burkhard, F. C. & Monastyrskaya, K. Deciphering microRNA code in pain and inflammation: lessons from bladder pain syndrome. Cell. Mol. Life Sci. 70, 3773–3789 (2013).
Longstreth, G. F. et al. Functional bowel disorders. Gastroenterology 130, 1480–1491 (2006).
Spiegel, B. M., Farid, M., Esrailian, E., Talley, J. & Chang, L. Is irritable bowel syndrome a diagnosis of exclusion?: A survey of primary care providers, gastroenterologists, and IBS experts. Am. J. Gastroenterol. 105, 848–858 (2010).
Sood, R., Law, G. R. & Ford, A. C. Diagnosis of IBS: symptoms, symptom-based criteria, biomarkers or ‘psychomarkers’? Nat. Rev. Gastroenterol. Hepatol. 11, 683–691 (2014). This narrative review describes how — based on current knowledge — symptoms, symptom-based criteria, biomarkers and psychomarkers, and their combinations may enable the diagnosis of IBS.
Manning, A. P., Thompson, W. G., Heaton, K. W. & Morris, A. F. Towards positive diagnosis of the irritable bowel. Br. Med. J. 2, 653–654 (1978).
Drossman, D. A., Thompson, W. G. & Talley, N. J. Identification of sub-groups of functional gastrointestinal disorders. Gastroenterol. Int. 3, 159–172 (1990).
Thompson, W. G. et al. Functional bowel disorders and functional abdominal pain. Gut 45 (Suppl. 2), II43–II47 (1999).
Ford, A. C. et al. Validation of the Rome III criteria for the diagnosis of irritable bowel syndrome in secondary care. Gastroenterology 145, 1262–1270.e1 (2013).
Whitehead, W. E. & Drossman, D. A. Validation of symptom-based diagnostic criteria for irritable bowel syndrome: a critical review. Am. J. Gastroenterol. 105, 814–820 (2010).
Pimentel, M. et al. New clinical method for distinguishing D-IBS from other gastrointestinal conditions causing diarrhea: the LA/IBS diagnostic strategy. Dig. Dis. Sci. 55, 145–149 (2010).
Bohn, L., Storsrud, S., Tornblom, H., Bengtsson, U. & Simren, M. Self-reported food-related gastrointestinal symptoms in IBS are common and associated with more severe symptoms and reduced quality of life. Am. J. Gastroenterol. 108, 634–641 (2013).
Ford, A. C., Marwaha, A., Lim, A. & Moayyedi, P. Systematic review and meta-analysis of the prevalence of irritable bowel syndrome in individuals with dyspepsia. Clin. Gastroenterol. Hepatol. 8, 401–409 (2010).
Vandvik, P. O., Wilhelmsen, I., Ihlebaek, C. & Farup, P. G. Comorbidity of irritable bowel syndrome in general practice: a striking feature with clinical implications. Aliment. Pharmacol. Ther. 20, 1195–1203 (2004).
Jerndal, P. et al. Gastrointestinal-specific anxiety: an important factor for severity of GI symptoms and quality of life in IBS. Neurogastroenterol. Motil. 22, 646–e179 (2010).
Kruis, W. et al. A diagnostic score for the irritable bowel syndrome. Its value in the exclusion of organic disease. Gastroenterology 87, 1–7 (1984).
Soh, J. S. et al. The diagnostic value of a digital rectal examination compared with high-resolution anorectal manometry in patients with chronic constipation and fecal incontinence. Am. J. Gastroenterol. 110, 1197–1204 (2015).
Tantiphlachiva, K., Rao, P., Attaluri, A. & Rao, S. S. C. Digital rectal examination is a useful tool for identifying patients with dyssynergia. Clin. Gastroenterol. Hepatol. 8, 955–960 (2010).
American College of Gastroenterology Task Force on Irritable Bowel Syndrome et al. An evidence-based position statement on the management of irritable bowel syndrome. Am. J. Gastroenterol. 104, S1–S35 (2009).
Cash, B. D. et al. The prevalence of celiac disease among patients with nonconstipated irritable bowel syndrome is similar to controls. Gastroenterology 141, 1187–1193 (2011).
Menees, S. B., Powell, C., Kurlander, J., Goel, A. & Chey, W. D. A meta-analysis of the utility of C-reactive protein, erythrocyte sedimentation rate, fecal calprotectin, and fecal lactoferrin to exclude inflammatory bowel disease in adults with IBS. Am. J. Gastroenterol. 110, 444–454 (2015).
Dickinson, B. & Surawicz, C. M. Infectious diarrhea: an overview. Curr. Gastroenterol. Rep. 16, 399 (2014).
Pimentel, M. et al. Development and validation of a biomarker for diarrhea-predominant irritable bowel syndrome in human subjects. PLoS ONE 10, e0126438 (2015).
Jones, M. P. et al. A biomarker panel and psychological morbidity differentiates the irritable bowel syndrome from health and provides novel pathophysiological leads. Aliment. Pharmacol. Ther. 39, 426–437 (2014).
Vanner, S. J. et al. Predictive value of the Rome criteria for diagnosing the irritable bowel syndrome. Am. J. Gastroenterol. 94, 2912–2917 (1999).
Whitehead, W. E. et al. Utility of red flag symptom exclusions in the diagnosis of irritable bowel syndrome. Aliment. Pharmacol. Ther. 24, 137–146 (2006).
Stotzer, P. O. et al. Are the definitions for chronic diarrhoea adequate? Evaluation of two different definitions in patients with chronic diarrhoea. United European Gastroenterol. J. 3, 381–386 (2015).
Gunnarsson, J. & Simren, M. Efficient diagnosis of suspected functional bowel disorders. Nat. Clin. Pract. Gastroenterol. Hepatol. 5, 498–507 (2008).
Begtrup, L. M. et al. A positive diagnostic strategy is noninferior to a strategy of exclusion for patients with irritable bowel syndrome. Clin. Gastroenterol. Hepatol. 11, 956–962.e1 (2013).
Spiegel, B. M. et al. Is a negative colonoscopy associated with reassurance or improved health-related quality of life in irritable bowel syndrome? Gastrointest. Endosc. 62, 892–899 (2005).
Cairns, S. R. et al. Guidelines for colorectal cancer screening and surveillance in moderate and high risk groups. Gut 59, 666–689 (2010).
Rex, D. K. et al. American College of Gastroenterology guidelines for colorectal cancer screening 2009 [corrected]. Am. J. Gastroenterol. 104, 739–750 (2009).
Chey, W. D. et al. The yield of colonoscopy in patients with non-constipated irritable bowel syndrome: results from a prospective, controlled US trial. Am. J. Gastroenterol. 105, 859–865 (2010).
Vijayvargiya, P., Camilleri, M., Shin, A. & Saenger, A. Methods for diagnosis of bile acid malabsorption in clinical practice. Clin. Gastroenterol. Hepatol. 11, 1232–1239 (2013).
Money, M. E. & Camilleri, M. Review: management of postprandial diarrhea syndrome. Am. J. Med. 125, 538–544 (2012).
Melchior, C., Gourcerol, G., Dechelotte, P., Leroi, A. M. & Ducrotte, P. Symptomatic fructose malabsorption in irritable bowel syndrome: a prospective study. United European Gastroenterol. J. 2, 131–137 (2014).
Misselwitz, B. et al. Lactose malabsorption and intolerance: pathogenesis, diagnosis and treatment. United European Gastroenterol. J. 1, 151–159 (2013).
Rana, S. V. & Malik, A. Breath tests and irritable bowel syndrome. World J. Gastroenterol. 20, 7587–7601 (2014).
Simren, M. & Stotzer, P. O. Use and abuse of hydrogen breath tests. Gut 55, 297–303 (2006).
Ford, A. C., Spiegel, B. M., Talley, N. J. & Moayyedi, P. Small intestinal bacterial overgrowth in irritable bowel syndrome: systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 7, 1279–1286 (2009).
Posserud, I., Stotzer, P. O., Bjornsson, E. S., Abrahamsson, H. & Simren, M. Small intestinal bacterial overgrowth in patients with irritable bowel syndrome. Gut 56, 802–808 (2007).
Heaton, K. W. et al. Symptoms of irritable bowel syndrome in a British urban community: consulters and nonconsulters. Gastroenterology 102, 1962–1967 (1992).
Ringstrom, G., Abrahamsson, H., Strid, H. & Simren, M. Why do subjects with irritable bowel syndrome seek health care for their symptoms? Scand. J. Gastroenterol. 42, 1194–1203 (2007).
Flik, C. E., Laan, W., Smout, A. J., Weusten, B. L. & de Wit, N. J. Comparison of medical costs generated by IBS patients in primary and secondary care in the Netherlands. BMC Gastroenterol. 15, 168 (2015).
Khan, S. & Chang, L. Diagnosis and management of IBS. Nat. Rev. Gastroenterol. Hepatol. 7, 565–581 (2010).
Hausteiner-Wiehle, C. & Henningsen, P. Irritable bowel syndrome: relations with functional, mental, and somatoform disorders. World J. Gastroenterol. 20, 6024–6030 (2014).
Posserud, I. et al. Symptom pattern following a meal challenge test in patients with irritable bowel syndrome and healthy controls. United European Gastroenterol. J. 1, 358–367 (2013).
Bohn, L., Storsrud, S. & Simren, M. Nutrient intake in patients with irritable bowel syndrome compared with the general population. Neurogastroenterol. Motil. 25, 23–30.e21 (2013).
Hayes, P. A., Fraher, M. H. & Quigley, E. M. Irritable bowel syndrome: the role of food in pathogenesis and management. Gastroenterol. Hepatol (N. Y.) 10, 164–174 (2014).
Wahnschaffe, U., Ullrich, R., Riecken, E. O. & Schulzke, J. D. Celiac disease-like abnormalities in a subgroup of patients with irritable bowel syndrome. Gastroenterology 121, 1329–1338 (2001).
Kellow, J. E., Miller, L. J., Phillips, S. F., Zinsmeister, A. R. & Charboneau, J. W. Altered sensitivity of the gallbladder to cholecystokinin octapeptide in irritable bowel syndrome. Am. J. Physiol. 253, G650–G655 (1987).
Kennedy, P. J. et al. Acute tryptophan depletion reduces kynurenine levels: implications for treatment of impaired visuospatial memory performance in irritable bowel syndrome. Psychopharmacology (Berl.) 232, 1357–1371 (2015).
Ford, A. C. et al. American College of Gastroenterology monograph on the management of irritable bowel syndrome and chronic idiopathic constipation. Am. J. Gastroenterol. 109, S2–S26 (2014).
Guo, Y. B. et al. Association between diet and lifestyle habits and irritable bowel syndrome: a case–control study. Gut Liver 9, 649–656 (2014).
Halmos, E. P., Power, V. A., Shepherd, S. J., Gibson, P. R. & Muir, J. G. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 146, 67–75.e5 (2014).
Bohn, L. et al. Diet low in FODMAPs reduces symptoms of irritable bowel syndrome as well as traditional dietary advice: a randomized controlled trial. Gastroenterology 149, 1399–1407.e2 (2015). In this paper, the data compare a diet low in FODMAPs with conventional dietary advice in IBS and report equal efficacy, thus counteracts the current enthusiasm with low FODMAP diets in patients with IBS.
Moayyedi, P. et al. The effect of dietary intervention on irritable bowel syndrome: a systematic review. Clin. Transl Gastroenterol. 6, e107 (2015).
Halmos, E. P. et al. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 64, 93–100 (2015).
Berg, L. K. et al. Effect of fructose-reduced diet in patients with irritable bowel syndrome, and its correlation to a standard fructose breath test. Scand. J. Gastroenterol. 48, 936–943 (2013).
Wilder-Smith, C. H., Materna, A., Wermelinger, C. & Schuler, J. Fructose and lactose intolerance and malabsorption testing: the relationship with symptoms in functional gastrointestinal disorders. Aliment. Pharmacol. Ther. 37, 1074–1083 (2013).
Ludvigsson, J. F. et al. The Oslo definitions for coeliac disease and related terms. Gut 62, 43–52 (2013).
Vazquez-Roque, M. I. et al. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: effects on bowel frequency and intestinal function. Gastroenterology 144, 903–911.e3 (2013).
Rodrigo, L., Blanco, I., Bobes, J. & de Serres, F. J. Effect of one year of a gluten-free diet on the clinical evolution of irritable bowel syndrome plus fibromyalgia in patients with associated lymphocytic enteritis: a case–control study. Arthritis Res. Ther. 16, 421 (2014).
Biesiekierski, J. R. et al. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 145, 320–328.e3 (2013).
Lee, K. N. & Lee, O. Y. Intestinal microbiota in pathophysiology and management of irritable bowel syndrome. World J. Gastroenterol. 20, 8886–8897 (2014).
Shanahan, F. & Quigley, E. M. Manipulation of the microbiota for treatment of IBS and IBD-challenges and controversies. Gastroenterology 146, 1554–1563 (2014).
Ford, A. C. et al. Efficacy of prebiotics, probiotics, and synbiotics in irritable bowel syndrome and chronic idiopathic constipation: systematic review and meta-analysis. Am. J. Gastroenterol. 109, 1547–1561 (2014).
Mazurak, N., Broelz, E., Storr, M. & Enck, P. Probiotic therapy in the irritable bowel syndrome — why is the evidence for clinical efficacy still poor and what can be done about it? J. Neurogastroeterol. Motil. 21, 471–485 (2015).
Hill, C. et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 11, 506–514 (2014).
Krueger, D. et al. Effect of hyoscine butylbromide (Buscopan®) on cholinergic pathways in the human intestine. Neurogastroenterol. Motil. 25, e530–e539 (2013).
Ford, A. C. et al. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ 337, a2313 (2008).
Ruepert, L. et al. Bulking agents, antispasmodics and antidepressants for the treatment of irritable bowel syndrome. Cochrane Database Syst. Rev. 8, CD003460 (2011).
Cash, B. D., Epstein, M. S. & Shah, S. M. A novel delivery system of peppermint oil is an effective therapy for irritable bowel syndrome symptoms. Dig. Dis. Sci. 61, 560–571 (2016).
National Institute of Health and Care Excellence. CG61. Irritable bowel syndrome in adults: diagnosis and management of irritable bowel syndrome in primary care. NICE[online], (2008).
Moret, C. & Briley, M. Antidepressants in the treatment of fibromyalgia. Neuropsychiatr. Dis. Treat. 2, 537–548 (2006).
Castro, J. et al. Linaclotide inhibits colonic nociceptors and relieves abdominal pain via guanylate cyclase-C and extracellular cyclic guanosine 3′,5′-monophosphate. Gastroenterology 145, 1334–1346.e11 (2013).
Chey, W. D. et al. Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am. J. Gastroenterol. 107, 1702–1712 (2012).
Rao, S. et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am. J. Gastroenterol. 107, 1714–1724 (2012).
Drossman, D. A. et al. Clinical trial: lubiprostone in patients with constipation-associated irritable bowel syndrome — results of two randomized, placebo-controlled studies. Aliment. Pharmacol. Ther. 29, 329–341 (2009).
Tack, J., van Outryve, M., Beyens, G., Kerstens, R. & Vandeplassche, L. Prucalopride (Resolor) in the treatment of severe chronic constipation in patients dissatisfied with laxatives. Gut 58, 357–365 (2009).
Efskind, P. S., Bernklev, T. & Vatn, M. H. A double-blind placebo-controlled trial with loperamide in irritable bowel syndrome. Scand. J. Gastroenterol. 31, 463–468 (1996).
Lembo, A. J. et al. Eluxadoline for irritable bowel syndrome with diarrhea. N. Engl. J. Med. 374, 242–253 (2016).
Clave, P. Treatment of IBS-D with 5-HT3 receptor antagonists versus spasmolytic agents: similar therapeutical effects from heterogeneous pharmacological targets. Neurogastroenterol. Motil. 23, 1051–1055 (2011).
Berman, S. M. et al. Condition-specific deactivation of brain regions by 5-HT3 receptor antagonist alosetron. Gastroenterology 123, 969–977 (2002).
Chang, L. et al. Incidence of ischemic colitis and serious complications of constipation among patients using alosetron: systematic review of clinical trials and post-marketing surveillance data. Am. J. Gastroenterol. 101, 1069–1079 (2006).
Garsed, K. et al. A randomised trial of ondansetron for the treatment of irritable bowel syndrome with diarrhoea. Gut 63, 1617–1625 (2014).
Fukudo, S., Ida, M., Akiho, H., Nakashima, Y. & Matsueda, K. Effect of ramosetron on stool consistency in male patients with irritable bowel syndrome with diarrhea. Clin. Gastroenterol. Hepatol. 12, 953–959.e4 (2014).
Pimentel, M. et al. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N. Engl. J. Med. 364, 22–32 (2011).
Didari, T., Mozaffari, S., Nikfar, S. & Abdollahi, M. Effectiveness of probiotics in irritable bowel syndrome: updated systematic review with meta-analysis. World J. Gastroenterol. 21, 3072–3084 (2015).
Madisch, A., Holtmann, G., Plein, K. & Hotz, J. Treatment of irritable bowel syndrome with herbal preparations: results of a double-blind, randomized, placebo-controlled, multi-centre trial. Aliment. Pharmacol. Ther. 19, 271–279 (2004).
Simmen, U. et al. Binding of STW 5 (Iberogast®) and its components to intestinal 5-HT, muscarinic M3, and opioid receptors. Phytomedicine 13 (Suppl. 5), 51–55 (2006).
Fond, G. et al. Anxiety and depression comorbidities in irritable bowel syndrome (IBS): a systematic review and meta-analysis. Eur. Arch. Psychiatry Clin. Neurosci. 264, 651–660 (2014).
Hungin, A. P. et al. Irritable bowel syndrome: an integrated explanatory model for clinical practice. Neurogastroenterol. Motil. 27, 750–763 (2015).
Drossman, D. A. et al. A focus group assessment of patient perspectives on irritable bowel syndrome and illness severity. Dig. Dis. Sci. 54, 1532–1541 (2009).
Owens, D. M., Nelson, D. K. & Talley, N. J. The irritable bowel syndrome: long-term prognosis and the physician-patient interaction. Ann. Intern. Med. 122, 107–112 (1995).
Spiller, R. et al. Guidelines on the irritable bowel syndrome: mechanisms and practical management. Gut 56, 1770–1798 (2007).
Fukudo, S. et al. Evidence-based clinical practice guidelines for irritable bowel syndrome. J. Gastroenterol. 50, 11–30 (2015).
Ford, A. C. et al. Effect of antidepressants and psychological therapies, including hypnotherapy, in irritable bowel syndrome: systematic review and meta-analysis. Am. J. Gastroenterol. 109, 1350–1365 (2014).
Gaylord, S. A. et al. Mindfulness training reduces the severity of irritable bowel syndrome in women: results of a randomized controlled trial. Am. J. Gastroenterol. 106, 1678–1688 (2011).
Ware, J. E. Jr & Sherbourne, C. D. The MOS 36-item Short-Form Health Survey (SF-36). I. Conceptual framework and item selection. Med. Care 30, 473–483 (1992).
EuroQoL-Group. EuroQol — a new facility for the measurement of health-related quality of life. Health Policy 16, 199–208 (1990).
Patrick, D. L., Drossman, D. A., Frederick, I. O., DiCesare, J. & Puder, K. L. Quality of life in persons with irritable bowel syndrome: development and validation of a new measure. Dig. Dis. Sci. 43, 400–411 (1998).
Hahn, B. A., Kirchdoerfer, L. J., Fullerton, S. & Mayer, E. Evaluation of a new quality of life questionnaire for patients with irritable bowel syndrome. Aliment. Pharmacol. Ther. 11, 547–552 (1997).
Gralnek, I. M., Hays, R. D., Kilbourne, A., Naliboff, B. & Mayer, E. A. The impact of irritable bowel syndrome on health-related quality of life. Gastroenterology 119, 654–660 (2000).
Halder, S. L. et al. Impact of functional gastrointestinal disorders on health-related quality of life: a population-based case–control study. Aliment. Pharmacol. Ther. 19, 233–242 (2004). This population-based, nested case–control study compares patients with IBS, patients with functional dyspepsia and patients with both diagnoses to healthy control individuals for health-related QOL (SF-36) and reports that most of the determinants of QOL are psychological in nature.
Singh, P. et al. Patients with irritable bowel syndrome-diarrhea have lower disease-specific quality of life than irritable bowel syndrome-constipation. World J. Gastroenterol. 21, 8103–8109 (2015).
Creed, F. et al. Health-related quality of life and health care costs in severe, refractory irritable bowel syndrome. Ann. Intern. Med. 134, 860–868 (2001).
Spiegel, B. M. et al. Clinical determinants of health-related quality of life in patients with irritable bowel syndrome. Arch. Intern. Med. 164, 1773–1780 (2004).
El-Serag, H. B., Olden, K. & Bjorkman, D. Health-related quality of life among persons with irritable bowel syndrome: a systematic review. Aliment. Pharmacol. Ther. 16, 1171–1185 (2002).
Fukudo, S., Hongo, M., Kaneko, H., Takano, M. & Ueno, R. Lubiprostone increases spontaneous bowel movement frequency and quality of life in patients with chronic idiopathic constipation. Clin. Gastroenterol. Hepatol. 13, 294–301.e5 (2015).
Fukudo, S., Hongo, M., Kaneko, H. & Ueno, R. Efficacy and safety of oral lubiprostone in constipated patients with or without irritable bowel syndrome: a randomized, placebo-controlled and dose-finding study. Neurogastroenterol. Motil. 23, 544–e205 (2011).
Ford, A. C. et al. Efficacy of 5-HT3 antagonists and 5-HT4 agonists in irritable bowel syndrome: systematic review and meta-analysis. Am. J. Gastroenterol. 104, 1831–1843 (2009).
Bardhan, K. D. et al. A double-blind, randomized, placebo-controlled dose-ranging study to evaluate the efficacy of alosetron in the treatment of irritable bowel syndrome. Aliment. Pharmacol. Ther. 14, 23–34 (2000).
Camilleri, M. et al. Efficacy and safety of alosetron in women with irritable bowel syndrome: a randomised, placebo-controlled trial. Lancet 355, 1035–1040 (2000).
Marciani, L. et al. Stimulation of colonic motility by oral PEG electrolyte bowel preparation assessed by MRI: comparison of split versus single dose. Neurogastroenterol. Motil. 26, 1426–1436 (2014).
Malagelada, C. et al. Classification of functional bowel disorders by objective physiological criteria based on endoluminal image analysis. Am. J. Physiol. Gastrointest. Liver Physiol. 309, G413–G419 (2015).
Rao, S. S. et al. Investigation of colonic and whole-gut transit with wireless motility capsule and radiopaque markers in constipation. Clin. Gastroenterol. Hepatol. 7, 537–544 (2009).
Shiotani, A. et al. Pilot study of biomarkers for predicting effectiveness of ramosetron in diarrhea-predominant irritable bowel syndrome: expression of S100A10 and polymorphisms of TPH1. Neurogastroenterol. Motil. 27, 82–91 (2015).
Camilleri, M. et al. Serotonin-transporter polymorphism pharmacogenetics in diarrhea-predominant irritable bowel syndrome. Gastroenterology 123, 425–432 (2002).
Barrett, J. C. et al. Genome-wide association defines more than 30 distinct susceptibility loci for Crohn's disease. Nat. Genet. 40, 955–962 (2008).
Yang, J. et al. Lactose intolerance in irritable bowel syndrome patients with diarrhoea: the roles of anxiety, activation of the innate mucosal immune system and visceral sensitivity. Aliment. Pharmacol. Ther. 39, 302–311 (2014).
Rodrigues, A. C., Nicholas Verne, G., Schmidt, S. & Mauderli, A. P. Hypersensitivity to cutaneous thermal nociceptive stimuli in irritable bowel syndrome. Pain 115, 5–11 (2005).
Dorn, S. D. et al. Increased colonic pain sensitivity in irritable bowel syndrome is the result of an increased tendency to report pain rather than increased neurosensory sensitivity. Gut 56, 1202–1209 (2007).
Christopher, N. L. & Bayless, T. M. Role of the small bowel and colon in lactose-induced diarrhea. Gastroenterology 60, 845–852 (1971).
Marciani, L. et al. Postprandial changes in small bowel water content in healthy subjects and patients with irritable bowel syndrome. Gastroenterology 138, 469–477.e1 (2010).
Florent, C. et al. Influence of chronic lactulose ingestion on the colonic metabolism of lactulose in man (an in vivo study). J. Clin. Invest. 75, 608–613 (1985).
Vandeputte, D. et al. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut 65, 57–62 (2015).
Andresen, V. et al. Irritable bowel syndrome — the main recommendations. Dtsch. Arztebl. Int. 108, 751–760 (2011).
Henrich, J. F. et al. Identifying effective techniques within psychological treatments for irritable bowel syndrome: a meta-analysis. J. Psychosomat. Res. 78, 205–222 (2015). This meta-analysis summarizes data from 48 RCTs, which tested psychological interventions in patients with IBS, and reports that, overall, improvement in gastrointestinal symptoms was strongly associated with improvement in psychological distress.
Creed, F. et al. The cost-effectiveness of psychotherapy and paroxetine for severe irritable bowel syndrome. Gastroenterology 124, 303–317 (2003).
Peters, S. L., Muir, J. G. & Gibson, P. R. Review article: gut-directed hypnotherapy in the management of irritable bowel syndrome and inflammatory bowel disease. Aliment. Pharmacol. Ther. 41, 1104–1115 (2015).
Hutton, J. Cognitive behaviour therapy for irritable bowel syndrome. Eur. J. Gastroenterol. Hepatol. 17, 11–14 (2005).
Hyphantis, T., Guthrie, E., Tomenson, B. & Creed, F. Psychodynamic interpersonal therapy and improvement in interpersonal difficulties in people with severe irritable bowel syndrome. Pain 145, 196–203 (2009).
Liegl, G., Plessen, C. Y., Leitner, A., Boeckle, M. & Pieh, C. Guided self-help interventions for irritable bowel syndrome: a systematic review and meta-analysis. Eur. J. Gastroenterol. Hepatol. 27, 1209–1221 (2015).
Acknowledgements
The research leading to these results has received funding from the People Programme of the European Union's Seventh Framework Programme under REA grant agreement no. 607652 (NeuroGUT), involving of some of the authors. Most of the authors are also engaged in and have received support of the international network COST Action BM1106 GENIEUR (GENes in Irritable Bowel Syndrome Research Network EURope, www.GENIEUR.eu).
Author information
Authors and Affiliations
Contributions
Introduction (P.E. and R.C.S.); Epidemiology (P.E. and J.S.-K.); Mechanisms/pathophysiology (G.B., B.N., M.R.-S., M.Schemann and E.A.M.); Diagnosis, screening and prevention (M.Simren); Management (E.M.M.Q., Q.A., A.D.F. and S.Z.); Quality of life (S.F.); Outlook (R.C.S.); Overview of Primer (P.E.).
Corresponding author
Ethics declarations
Competing interests
The authors have received unrestricted research grants from Alfa Wassermann (G.B.), Alimentary Health (E.M.M.Q.), Astellas (S.F.), Boehringer-Ingelheim (M.Schemann), Cadi Group (G.B.), Danone Research (M.Simren), Falk (G.B.), Ferring (M.Simren), IMA (G.B.), Italchimici (G.B.), Ironwood (R.C.S.), Kao (S.F.), Lesaffre (R.C.S.), Lotenzatto (G.B.), Ono Pharmaceuticals (Q.A. and S.F.), Parmalat (G.B.), Pfizer (Q.A.), Pretexin (Q.A.), Rhythme (E.M.M.Q.), Schwabe (M.Schemann), Shire (P.E.), Sofar (G.B.), Steigerwald (M.Schemann), SymbioPharm (P.E.), Therevance (E.M.M.Q.), Vivrant (E.M.M.Q.), Yakult (G.B.) and Zespri (G.B.), have served as a consultant/advisory board members for Actavis (E.M.M.Q.), Albireo (M.Simren), Afa Wassermann (G.B.), Alimentary Health (E.M.M.Q.), Allergan (G.B. and M.Simren), Almirall (A.D.F., E.M.M.Q., G.B., M.Simren, P.E. and R.C.S.), Astellas (S.F.), AstraZeneca (M.Simren and P.E.), Bionorika (M.Schemann), Boehringer-Ingelheim (P.E.), Chr Hansen (M.Simren), Commonwealth (E.M.M.Q. and G.B.), Danone (E.A.M., G.B., M.Simren and R.C.S.), Ferring (P.E.), Glycom (M.Simren), IM Sciences (E.M.M.Q.), Ironwood (G.B.), Italchimici (G.B.), Ipsen (R.C.S.), Kissei (S.F.), Melasci (G.B.), Menarini (G.B.), Noos (G.B.), Parmalat (G.B.), Nestlé (M.Simren), Salix (E.M.M.Q.), Shire (E.M.M.Q., M.Simren and P.E.), Sofar (G.B.), SymbioPharm (P.E.), Synergy (E.M.M.Q. and G.B.), Takeda (M.Schemann), Yakult (G.B.), Yuhan Corp (R.C.S.) and Zenspri (G.B.), and as a speaker for Abbott (S.F.), Alfa Wassermann (G.B.), Almirall (B.N., E.M.M.Q., G.B., M.Simren, P.E. and Q.A.), Astellas (S.F.), AstraZeneca (E.M.M.Q.), Chiesi (P.E.), Danone (G.B.), Falk (B.N. and P.E.), Gruenenthal (P.E. and Q.A.), Heel (P.E.), Ironwood (E.M.M.Q.), Melasci (G.B.), Menarini (G.B. and R.C.S.), Noos (G.B.), Shire (A.D.F., E.M.M.Q., G.B., M.Simren and P.E.), Sofar (G.B.), Steigerwald (M.Schemann), SymbioPharm (P.E.), Takeda (M.Simren), Tillotts (M.Simren) and Yakult (G.B.). J.S.-K., M.R.-S. and S.Z. declare no potential conflict of interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Enck, P., Aziz, Q., Barbara, G. et al. Irritable bowel syndrome. Nat Rev Dis Primers 2, 16014 (2016). https://doi.org/10.1038/nrdp.2016.14
Published:
DOI: https://doi.org/10.1038/nrdp.2016.14
This article is cited by
-
Symptom effects and central mechanism of acupuncture in patients with functional gastrointestinal disorders: a systematic review based on fMRI studies
BMC Gastroenterology (2024)
-
Efficacy and safety of peppermint oil for the treatment in Japanese patients with irritable bowel syndrome: a prospective, open-label, and single-arm study
BioPsychoSocial Medicine (2024)
-
Fermented mixed grain ameliorates chronic stress-induced depression-like behavior and memory deficit
Food Science and Biotechnology (2024)
-
Diagnosis and Pharmacological Management of Microscopic Colitis in Geriatric Care
Drugs & Aging (2024)
-
Upper gastrointestinal endoscopic findings in functional constipation and irritable bowel syndrome diagnosed using the Rome IV criteria: a cross-sectional survey during a medical check-up in Japan
BMC Gastroenterology (2023)