Key Points
-
The Target Base and New Target Innovation in the Pharmaceutical Industry
-
This section describes the small number of targets that form the basis of the economics of the industry and how rare and valuable completely new targets are, both economically and for the treatment of unmet medical needs.
-
Genetics Works
-
This section summarizes the data from the knockout of the targets of the top 100 selling drugs of the year 2001.
-
Going Forward With Reverse Genetics
-
This section describes the prospective use of mouse knockout information to define the next targets for drug development.
-
Knocking Out the Druggable Genome
-
This section summarizes our efforts to knock out all druggable genes in order to discover new drug targets and estimates are made for the number of high quality targets the genome may contain.
Abstract
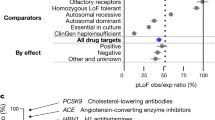
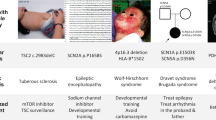
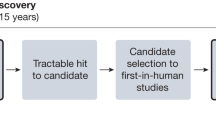
The biopharmaceutical industry is currently faced with a tremendous number of potential drug targets identified through the sequencing of the human genome. The challenge ahead is to delineate those targets with the greatest value for therapeutic intervention. Here, we critically evaluate mouse-knockout technology for target discovery and validation. A retrospective evaluation of the knockout phenotypes for the targets of the 100 best-selling drugs indicates that these phenotypes correlate well with known drug efficacy, illuminating a productive path forward for discovering future drug targets. Prospective mining of the druggable genome is being catalysed by large-scale mouse knockout programs combined with phenotypic screens focused on identifying targets that modulate mammalian physiology in a therapeutically relevant manner.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Drews, J. Biotechnology's metamorphosis into a drug discovery industry. Nature Biotechnol. 16, (Suppl) 22–24 (1998).
Hopkins, A. L. & Groom, C. R. The druggable genome. Nature Rev. Drug Discov. 1, 727–30 (2002). This paper describes the gene families that constitute the druggable genome from the perspective of chemists in the pharmaceutical industry.
PharmaLive.com. The Med Ad News 200- the world's best-selling medicines, May 2002. Engel Publishing Partners, Reprinted with permission from Med Ad News, (May 2002) (PharmaLive.com , West Trenton, NJ, 2002). This is a list of the top 200 selling pharmaceutical drugs of the year 2001.
FDA. FDA. (Center for Drug Evaluation and Research, Rockville, MD, CDER Report to the Nation, 1997– 2001). The annual CDER Report to the Nation describes the new molecular entities that have been approved by the FDA each year.
Vinson, M. C., Davis, W. M. & Waters, I. W. (Drug Topics, Montvale, NJ, New Drug Approvals of 1995–1997). Each year, Drug Topics publishes a review of the new drugs approved by the FDA and their mechanism of action.
Kaitin, K. I. & Manocchia, M. The new drug approvals of 1993, 1994, and 1995: trends in drug development. Am. J. Ther. 4, 46–54 (1997).
Scappini, B. et al. In vitro effects of STI 571-containing drug combinations on the growth of Philadelphia-positive chronic myelogenous leukemia cells. Cancer 94, 2653–2662 (2002).
Walke, D. W. et al. In vivo drug target discovery: identifying the best targets from the genome. Curr. Opin. Biotechnol. 12, 626–631 (2001).
Abuin, A., Holt, K. H., Platt, K. A., Sands, A. T. & Zambrowicz, B. P. Full-speed mammalian genetics: in vivo target validation in the drug discovery process. Trends Biotechnol. 20, 36–42 (2002).
BusinessCommunicationsCompany. (Business Communications Company, Inc., Norwalk, CT, 2002).
Spicer, Z. et al. Stomachs of mice lacking the gastric H,K-ATPase α-subunit have achlorhydria, abnormal parietal cells, and ciliated metaplasia. J. Biol. Chem. 275, 21555–21565 (2000). This is an example of a target knockout modelling a small-molecule antagonist of the same target.
Scarff, K. L., Judd, L. M., Toh, B. H., Gleeson, P. A. & Van Driel, I. R. Gastric H(+),K(+)-adenosine triphosphatase β subunit is required for normal function, development, and membrane structure of mouse parietal cells. Gastroenterology 117, 605–668 (1999).
Kobayashi, T. et al. Abnormal functional and morphological regulation of the gastric mucosa in histamine H2 receptor-deficient mice. J. Clin. Invest. 105, 1741–1749 (2000).
Wu, C. S., Lim, S. K., D'Agati, V. & Costantini, F. Generation of committed erythroid BFU-E and CFU-E progenitors does not require erythropoietin or the erythropoietin receptor. Cell 83, 59–67 (1996).
Lieschke, G. J. et al. Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 84, 1737–1746 (1994). This is an example of a knockout of a therapeutic protein showing the opposite phenotype to the effect produced by treatment with the same therapeutic protein.
Banu, Y. & Watanabe, T. Augmentation of antigen receptor-mediated responses by histamine H1 receptor signaling. J. Exp. Med. 189, 673–682 (1999).
Jutel, M. et al. Histamine regulates T-cell and antibody responses by differential expression of H1 and H2 receptors. Nature 413, 420–425 (2001).
Yanai, K. et al. Behavioural characterization and amounts of brain monoamines and their metabolites in mice lacking histamine H1 receptors. Neuroscience 87, 479–487 (1998).
Inoue, I. et al. Impaired locomotor activity and exploratory behavior in mice lacking histamine H1 receptors. Proc. Natl Acad. Sci. USA 93, 13316–13320 (1996).
Yanai, K., Son, L. Z., Endou, M., Sakurai, E. & Watanabe, T. Targeting disruption of histamine H1 receptors in mice: behavioral and neurochemical characterization. Life Sci. 62, 1607–1610 (1998).
Tilley, S. L., Coffman, T. M. & Koller, B. H. Mixed messages: modulation of inflammation and immune responses by prostaglandins and thromboxanes. J. Clin. Invest. 108, 15–23 (2001).
Morteau, O. Prostaglandins and inflammation: the cyclooxygenase controversy. Arch. Immunol. Ther. Exp. 48, 473–480 (2000).
Austin, S. C. & Funk, C. D. Insight into prostaglandin, leukotriene, and other eicosanoid functions using mice with targeted gene disruptions. Prostaglandins Other Lipid Mediat. 58, 231–252 (1999).
Langenbach, R., Loftin, C., Lee, C. & Tiano, H. Cyclooxygenase knockout mice: models for elucidating isoform-specific functions. Biochem. Pharmacol. 58, 1237–1246 (1999).
Williams, C. S., Mann, M. & DuBois, R. N. The role of cyclooxygenases in inflammation, cancer, and development. Oncogene 18, 7908–7916 (1999).
Langenbach, R. et al. Prostaglandin synthase 1 gene disruption in mice reduces arachidonic acid-induced inflammation and indomethacin-induced gastric ulceration. Cell 83, 483–492 (1995).
Langenbach, R., Loftin, C. D., Lee, C. & Tiano, H. Cyclooxygenase-deficient mice. A summary of their characteristics and susceptibilities to inflammation and carcinogenesis. Ann. NY Acad. Sci. 889, 52–61 (1999).
Reddy, S. T., Tiano, H. F., Langenbach, R., Morham, S. G. & Herschman, H. R. Genetic evidence for distinct roles of COX-1 and COX-2 in the immediate and delayed phases of prostaglandin synthesis in mast cells. Biochem. Biophys. Res. Commun. 265, 205–210 (1999).
Morteau, O. et al. Impaired mucosal defense to acute colonic injury in mice lacking cyclooxygenase-1 or cyclooxygenase-2. J. Clin. Invest. 105, 469–478 (2000).
Ballou, L. R., Botting, R. M., Goorha, S., Zhang, J. & Vane, J. R. Nociception in cyclooxygenase isozyme-deficient mice. Proc. Natl Acad. Sci. USA 97, 10272–10276 (2000).
Chulada, P. C. et al. Cycloxygenase-1 and-2 deficiency decrease spontaneous intestinal adenomas in the Min mouse. Proc. Am. Assoc. Cancer Res. 39, 195 (1998).
Tiano, H. F. et al. Effects of cyclooxygenase deficiency on inflammation and papilloma formation in mouse skin. Proc. Am. Assoc. Cancer Res. 38, 257 (1998).
Morham, S. G. et al. Prostaglandin synthase 2 gene disruption causes severe renal pathology in the mouse. Cell 83, 473–482 (1995).
Li, S. et al. The febrile response to lipopolysaccharide is blocked in cyclooxygenase- 2(−/−), but not in cyclooxygenase-1(−/−) mice. Brain Res. 825, 86–94 (1999).
Myers, L. K. et al. The genetic ablation of cyclooxygenase 2 prevents the development of autoimmune arthritis. Arthritis Rheum. 43, 2687–2693 (2000).
Oshima, M. et al. Suppression of intestinal polyposis in Apc delta716 knockout mice by inhibition of cyclooxygenase 2 (COX-2). Cell 87, 803–809 (1996).
Irvin, C. G., Tu, Y. P., Sheller, J. R. & Funk, C. D. 5-Lipoxygenase products are necessary for ovalbumin-induced airway responsiveness in mice. Am. J. Physiol. 272, L1053–1058 (1997).
Peters-Golden, M. et al. Protection from pulmonary fibrosis in leukotriene-deficient mice. Am. J. Respir. Crit. Care Med. 165, 229–235 (2002).
Chen, X. S., Sheller, J. R., Johnson, E. N. & Funk, C. D. Role of leukotrienes revealed by targeted disruption of the 5- lipoxygenase gene. Nature 372, 179–182 (1994).
Maekawa, A., Austen, K. F. & Kanaoka, Y. Targeted gene disruption reveals the role of cysteinyl leukotriene 1 receptor in the enhanced vascular permeability of mice undergoing acute inflammatory responses. J. Biol. Chem. 277, 20820–20824 (2002).
Pasparakis, M., Alexopoulou, L., Episkopou, V. & Kollias, G. Immune and inflammatory responses in TNF α-deficient mice: a critical requirement for TNF α in the formation of primary B cell follicles, follicular dendritic cell networks and germinal centers, and in the maturation of the humoral immune response. J. Exp. Med. 184, 1397–1411 (1996). This is an example of a knockout of a therapeutic antibody product showing aspects of the phenotype produced by treatment with the therapeutic antibody that neutralizes the target.
Marino, M. W. et al. Characterization of tumor necrosis factor-deficient mice. Proc. Natl Acad. Sci. USA 94, 8093–8098 (1997).
Gu, J. J. et al. Inhibition of T lymphocyte activation in mice heterozygous for loss of the IMPDH II gene. J. Clin. Invest. 106, 599–606 (2000).
Schmid, W., Cole, T. J., Blendy, J. A. & Schutz, G. Molecular genetic analysis of glucocorticoid signalling in development. J. Steroid Biochem. Mol. Biol. 53, 33–35 (1995).
Reichardt, H. M., Tronche, F., Bauer, A. & Schutz, G. Molecular genetic analysis of glucocorticoid signaling using the Cre/loxP system. Biol. Chem. 381, 961–964 (2000).
Bueno, O. F., Brandt, E. B., Rothenberg, M. E. & Molkentin, J. D. Defective T cell development and function in calcineurin A β- deficient mice. Proc. Natl Acad. Sci. USA 99, 9398–9403 (2002).
Malleret, G., Hen, R., Guillou, J. L., Segu, L. & Buhot, M. C. 5-HT1B receptor knock-out mice exhibit increased exploratory activity and enhanced spatial memory performance in the Morris water maze. J. Neurosci. 19, 6157–6168 (1999).
Grailhe, R. et al. Increased exploratory activity and altered response to LSD in mice lacking the 5-HT(5A) receptor. Neuron 22, 581–591 (1999).
Tecott, L. H., Logue, S. F., Wehner, J. M. & Kauer, J. A. Perturbed dentate gyrus function in serotonin 5-HT2C receptor mutant mice. Proc. Natl Acad. Sci. USA 95, 15026–15031 (1998).
Ramboz, S. et al. Serotonin receptor 1A knockout: an animal model of anxiety-related disorder. Proc. Natl Acad. Sci. USA 95, 14476–14481 (1998).
Parks, C. L., Robinson, P. S., Sibille, E., Shenk, T. & Toth, M. Increased anxiety of mice lacking the serotonin1A receptor. Proc. Natl Acad. Sci. USA 95, 10734–10739 (1998).
Heisler, L. K. et al. Elevated anxiety and antidepressant-like responses in serotonin 5-HT1A receptor mutant mice. Proc. Natl Acad. Sci. USA 95, 15049–15054 (1998).
Ramboz, S. et al. 5-HT1B receptor knock out—behavioral consequences. Behav. Brain Res. 73, 305–312 (1996).
Mayorga, A. J. et al. Antidepressant-like behavioral effects in 5-hydroxytryptamine(1A) and 5- hydroxytryptamine(1B) receptor mutant mice. J. Pharmacol. Exp. Ther. 298, 1101–1107 (2001).
Dulawa, S. C., Grandy, D. K., Low, M. J., Paulus, M. P. & Geyer, M. A. Dopamine D4 receptor-knock-out mice exhibit reduced exploration of novel stimuli. J. Neurosci. 19, 9550–9556 (1999).
Kelly, M. A. et al. Locomotor activity in D2 dopamine receptor-deficient mice is determined by gene dosage, genetic background, and developmental adaptations. J. Neurosc.i 18, 3470–3479 (1998).
Xu, M. et al. Dopamine D3 receptor mutant mice exhibit increased behavioral sensitivity to concurrent stimulation of D1 and D2 receptors. Neuron 19, 837–848 (1997).
Smith, D. R. et al. Behavioural assessment of mice lacking D1A dopamine receptors. Neuroscience 86, 135–146 (1998).
Xu, M. et al. Dopamine D1 receptor mutant mice are deficient in striatal expression of dynorphin and in dopamine-mediated behavioral responses. Cell 79, 729–742 (1994).
Rubinstein, M. et al. Mice lacking dopamine D4 receptors are supersensitive to ethanol, cocaine, and methamphetamine. Cell 90, 991–1001 (1997).
Giros, B., Jaber, M., Jones, S. R., Wightman, R. M. & Caron, M. G. Hyperlocomotion and indifference to cocaine and amphetamine in mice lacking the dopamine transporter. Nature 379, 606–612 (1996).
Xu, F. et al. Mice lacking the norepinephrine transporter are supersensitive to psychostimulants. Nature Neurosci. 3, 465–471 (2000).
Homanics, G. E. et al. Mice devoid of γ-aminobutyrate type A receptor β3 subunit have epilepsy, cleft palate, and hypersensitive behavior. Proc. Natl Acad. Sci. USA 94, 4143–4148 (1997).
Sora, I. et al. Opiate receptor knockout mice define μ receptor roles in endogenous nociceptive responses and morphine-induced analgesia. Proc. Natl Acad. Sci. USA 94, 1544–1549 (1997). This is an example of a target knockout producing the opposite phenotype compared to a small molecule agonist of the same target.
Schomberg, D. W. et al. Targeted disruption of the estrogen receptor-α gene in female mice: characterization of ovarian responses and phenotype in the adult. Endocrinology 140, 2733–2744 (1999).
Dupont, S. et al. Effect of single and compound knockouts of estrogen receptors α (ERα) and β (ERβ) on mouse reproductive phenotypes. Development 127, 4277–4291 (2000).
Vidal, O. et al. Estrogen receptor specificity in the regulation of skeletal growth and maturation in male mice. Proc. Natl Acad. Sci. USA 97, 5474–5479. (2000).
Krege, J. H. et al. Generation and reproductive phenotypes of mice lacking estrogen receptor β. Proc. Natl Acad. Sci. USA 95, 15677–15682 (1998).
Dunford, J. E. et al. Structure-activity relationships for inhibition of farnesyl diphosphate synthase in vitro and inhibition of bone resorption in vivo by nitrogen-containing bisphosphonates. J. Pharmacol. Exp. Ther. 296, 235–242 (2001).
Thompson, K., Dunford, J. E., Ebetino, F. H. & Rogers, M. J. Identification of a bisphosphonate that inhibits isopentenyl diphosphate isomerase and farnesyl diphosphate synthase. Biochem. Biophys. Res. Commun. 290, 869–873 (2002).
Grove, J. E., Brown, R. J. & Watts, D. J. The intracellular target for the antiresorptive aminobisphosphonate drugs in Dictyostelium discoideum is the enzyme farnesyl diphosphate synthase. J. Bone. Miner. Res. 15, 971–981 (2000).
Bergstrom, J. D., Bostedor, R. G., Masarachia, P. J., Reszka, A. A. & Rodan, G. Alendronate is a specific, nanomolar inhibitor of farnesyl diphosphate synthase. Arch. Biochem. Biophys. 373, 231–241 (2000).
Zhou, G. et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 108, 1167–1174 (2001).
Abu-Elheiga, L., Matzuk, M. M., Abo-Hasema, K. A. H. & Wakil, S. J. Continuous fatty acid oxidation and reduced fat storage in mice lacking Acetyl-CoA carboxylase 2. Science 291, 2613–2616 (2001).
Leroux, L. et al. Compensatory responses in mice carrying a null mutation for Ins1 or Ins2. Diabetes 50, (Suppl 1) S150–153 (2001).
Joshi, R. L. et al. Targeted disruption of the insulin receptor gene in the mouse results in neonatal lethality. EMBO J. 15, 1542–1547 (1996).
Accili, D. et al. Early neonatal death in mice homozygous for a null allele of the insulin receptor gene. Nature Genet 12, 106–109 (1996).
Wang, J. et al. A mutation in the insulin 2 gene induces diabetes with severe pancreatic β-cell dysfunction in the Mody mouse. J. Clin. Invest. 103, 27–37 (1999).
Oyadomari, S. et al. Targeted disruption of the Chop gene delays endoplasmic reticulum stress-mediated diabetes. J. Clin. Invest. 109, 525–532 (2002).
Miles, P. D., Barak, Y., He, W., Evans, R. M. & Olefsky, J. M. Improved insulin-sensitivity in mice heterozygous for PPAR-γ deficiency. J. Clin. Invest. 105, 287–292 (2000).
Lowe, M. E., Kaplan, M. H., Jackson-Grusby, L., D'Agostino, D. & Grusby, M. J. Decreased neonatal dietary fat absorption and T cell cytotoxicity in pancreatic lipase-related protein 2-deficient mice. J. Biol. Chem. 273, 31215–31221 (1998).
Weng, W. et al. Intestinal absorption of dietary cholesteryl ester is decreased but retinyl ester absorption is normal in carboxyl ester lipase knockout mice. Biochemistry 38, 4143–4149 (1999).
Ito, M. et al. Regulation of blood pressure by the type 1A angiotensin II receptor gene. Proc. Natl Acad. Sci. USA 92, 3521–3525 (1995).
Sugaya, T. et al. Angiotensin II type 1a receptor-deficient mice with hypotension and hyperreninemia. J. Biol. Chem. 270, 18719–18722 (1995).
Esther, C. R., Jr. et al. Mice lacking angiotensin-converting enzyme have low blood pressure, renal pathology, and reduced male fertility. Lab. Invest. 74, 953–65 (1996).
Krege, J. H. et al. Male-female differences in fertility and blood pressure in ACE- deficient mice. Nature 375, 146–148 (1995).
Foster, C. J. et al. Molecular identification and characterization of the platelet ADP receptor targeted by thienopyridine antithrombotic drugs. J. Clin. Invest. 107, 1591–1598 (2001).
Dewerchin, M. et al. Blood coagulation factor X deficiency causes partial embryonic lethality and fatal neonatal bleeding in mice. Thromb. Haemost. 83, 185–190 (2000).
Chruscinski, A. et al. Differential distribution of β-adrenergic receptor subtypes in blood vessels of knockout mice lacking β1- or β2-adrenergic receptors. Mol. Pharmacol. 60, 955–962 (2001).
Naga Prasad, S. V., Nienaber, J. & Rockman, H. A. β-adrenergic axis and heart disease. Trends Genet. 17, S44–49 (2001).
Eckhart, A. D. & Koch, W. J. Transgenic studies of cardiac adrenergic receptor regulation. J. Pharmacol. Exp. Ther. 299, 1–5 (2001).
Kaumann, A. J., Engelhardt, S., Hein, L., Molenaar, P. & Lohse, M. Abolition of (-)-CGP 12177-evoked cardiostimulation in double β1/β2-adrenoceptor knockout mice. Obligatory role of β1-adrenoceptors for putative β4-adrenoceptor pharmacology. Naunyn. Schmiedebergs Arch. Pharmacol. 363, 87–93 (2001).
Rohrer, D. K., Chruscinski, A., Schauble, E. H., Bernstein, D. & Kobilka, B. K. Cardiovascular and metabolic alterations in mice lacking both β1- and β2-adrenergic receptors. J. Biol. Chem. 274, 16701–8. (1999).
Chruscinski, A. J. et al. Targeted disruption of the β2 adrenergic receptor gene. J. Biol. Chem. 274, 16694–700 (1999).
Rohrer, D. K. Physiological consequences of β-adrenergic receptor disruption. J. Mol. Med. 76, 764–772 (1998).
Lowell, B. B. Using gene knockout and transgenic techniques to study the physiology and pharmacology of β3-adrenergic receptors. Endocr. J. 45 (Suppl) S9–13 (1998).
Preitner, F. et al. Metabolic response to various β-adrenoceptor agonists in β3-adrenoceptor knockout mice: evidence for a new β-adrenergic receptor in brown adipose tissue. Br. J. Pharmacol. 124, 1684–1688 (1998).
Rohrer, D. K., Schauble, E. H., Desai, K. H., Kobilka, B. K. & Bernstein, D. Alterations in dynamic heart rate control in the β1-adrenergic receptor knockout mouse. Am. J. Physiol. 274, H1184–1193 (1998).
Kaumann, A. J. et al. (-)-CGP 12177 causes cardiostimulation and binds to cardiac putative β4-adrenoceptors in both wild-type and β3-adrenoceptor knockout mice. Mol. Pharmacol. 53, 670–675 (1998).
Rohrer, D. K. et al. The developmental and physiological consequences of disrupting genes encoding β1 and β2 adrenoceptors. Adv. Pharmacol. 42, 499–501 (1998).
Rohrer, D. K. et al. Targeted disruption of the mouse β1-adrenergic receptor gene: developmental and cardiovascular effects. Proc. Natl Acad. Sci. USA 93, 7375–7380 (1996).
Matsui, M. et al. Multiple functional defects in peripheral autonomic organs in mice lacking muscarinic acetylcholine receptor gene for the M3 subtype. Proc. Natl Acad. Sci. USA 97, 9579–9584 (2000).
Li, E., Sucov, H. M., Lee, K. F., Evans, R. M. & Jaenisch, R. Normal development and growth of mice carrying a targeted disruption of the α 1 retinoic acid receptor gene. Proc. Natl Acad. Sci. USA 90, 1590–1594 (1993).
Luo, J., Pasceri, P., Conlon, R. A., Rossant, J. & Giguere, V. Mice lacking all isoforms of retinoic acid receptor β develop normally and are susceptible to the teratogenic effects of retinoic acid. Mech. Dev. 53, 61–71 (1995).
Lohnes, D. et al. Function of retinoic acid receptor γ in the mouse. Cell 73, 643–658 (1993).
Kwan, K. Y. & Wang, J. C. Mice lacking DNA topoisomerase IIIβ develop to maturity but show a reduced mean lifespan. Proc. Natl Acad. Sci. USA 98, 5717–5721 (2001).
Rao, C. V. & Lei, Z. M. Consequences of targeted inactivation of LH receptors. Mol. Cell. Endocrinol. 187, 57–67 (2002).
O'Keefe, T. L., Williams, G. T., Davies, S. L. & Neuberger, M. S. Mice carrying a CD20 gene disruption. Immunogenetics 48, 125–132 (1998).
Mural, R. J. et al. A comparison of whole-genome shotgun-derived mouse chromosome 16 and the human genome. Science 296, 1661–1671 (2002).
Saftig, P. et al. Impaired osteoclastic bone resorption leads to osteopetrosis in cathepsin-K-deficient mice. Proc. Natl Acad. Sci. USA 95, 13453–13458 (1998).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Chen, A. S. et al. Inactivation of the mouse melanocortin-3 receptor results in increased fat mass and reduced lean body mass. Nature Genet. 26, 97–102 (2000).
Chen, Y. et al. Targeted disruption of the melanin-concentrating hormone receptor-1 results in hyperphagia and resistance to diet-induced obesity. Endocrinology 143, 2469–2477 (2002).
Marsh, D. J. et al. Melanin-concentrating hormone 1 receptor-deficient mice are lean, hyperactive, and hyperphagic and have altered metabolism. Proc. Natl Acad. Sci. USA 99, 3240–3245 (2002).
Ntambi, J. M. et al. Loss of stearoyl-CoA desaturase-1 function protects mice against adiposity. Proc. Natl Acad. Sci. USA 99, 11482–11486 (2002).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Related links
Related links
DATABASES
Cancer.Gov
Online Mendelian Inheritance in Man
Adenomatous polyposis of the colon
FURTHER INFORMATION
Encyclopedia of Life Sciences
Glossary
- CHRONIC MYELOID LEUKAEMIA
-
A leukaemia characterized by the presence of large numbers of abnormal mature granulocytes circulating in the blood.
- METABOLOMICS
-
The quantitative measurement of all low-molecular-weight metabolites in an organism's cells at a specified time under specific environmental conditions.
- GASTROESOPHAGEAL REFLUX DISEASE
-
A disorder in which there is recurrent return of stomach contents back up into the oesophagus, frequently causing heartburn, a symptom of irritation of the oesophagus by stomach acid.
- ERYTHROPOIESIS
-
Red blood cell development, in which a pluripotent stem cell produces, by a series of divisions, committed stem cells that give rise to cells that will divide only a few more times to produce mature erythrocytes.
- NEUTROPENIA
-
A decrease in neutrophil numbers in the peripheral blood.
- DECIDUALIZATION
-
Formation of the deciduas, the inner layer of the wall of the uterus, which envelops the embryo, forms a part of the placenta and is discharged with it.
- CARRAGEENAN
-
A sulphated cell-wall polysaccharide found in certain red algae, which contains repeating sulphated disaccharides of galactose, and sometimes anhydrogalactose, and is used to induce an inflammatory lesion when injected into experimental animals.
- TOPOISOMERASE
-
Enzymes that change the degree of supercoiling in DNA by cutting one or both strands.
- WERNER'S SYNDROME
-
A disorder causing accelerated aging consisting of scleroderma-like skin changes, bilateral juvenile cataracts, progeria, hypogonadism, and diabetes mellitus; it results from the autosomal recessive inheritance of a mutation in a topoisomerase gene.
- ORTHOLOGOUS GENE
-
Homologous gene in different species, the lineage of which derives from a common ancestral gene without gene duplication or horizontal transmission.
Rights and permissions
About this article
Cite this article
Zambrowicz, B., Sands, A. Knockouts model the 100 best-selling drugs—will they model the next 100?. Nat Rev Drug Discov 2, 38–51 (2003). https://doi.org/10.1038/nrd987
Issue Date:
DOI: https://doi.org/10.1038/nrd987
This article is cited by
-
High-throughput phenotyping reveals expansive genetic and structural underpinnings of immune variation
Nature Immunology (2020)
-
The virtuous cycle of human genetics and mouse models in drug discovery
Nature Reviews Drug Discovery (2019)
-
NOTUM inhibition increases endocortical bone formation and bone strength
Bone Research (2019)
-
INFRAFRONTIER: a European resource for studying the functional basis of human disease
Mammalian Genome (2016)
-
Identification of novel therapeutics for complex diseases from genome-wide association data
BMC Medical Genomics (2014)