Abstract
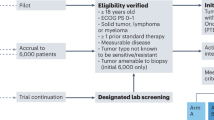
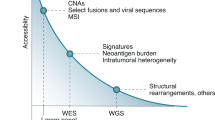
The recent surge in high-throughput sequencing of cancer genomes has supported an expanding molecular classification of cancer. These studies have identified putative predictive biomarkers signifying aberrant oncogene pathway activation and may provide a rationale for matching patients with molecularly targeted therapies in clinical trials. Here, we discuss some of the challenges of adapting these data for rare cancers or molecular subsets of certain cancers, which will require aligning the availability of investigational agents, rapid turnaround of clinical grade sequencing, molecular eligibility and reconsidering clinical trial design and end points.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Demeure, M. J. et al. Cancer of the ampulla of Vater: analysis of the whole genome sequence exposes a potential therapeutic vulnerability. Genome Med. 4, 56 (2012).
Mardis, E. R. A decade's perspective on DNA sequencing technology. Nature 470, 198–203 (2011).
Hudson, T. J. et al. International network of cancer genome projects. Nature 464, 993–998 (2010).
Ledford, H. Big science: the cancer genome challenge. Nature 464, 972–974 (2010).
Committee on a Framework for Development a New Taxonomy of Disease & the National Research Council. Toward Precision Medicine: Building a Knowlege Network for Biomedical Research and a New Taxonomy of Disease (National Academies Press, 2011).
MacConaill, L. E. et al. Profiling critical cancer gene mutations in clinical tumor samples. PLoS ONE 4, e7887 (2009).
Dias-Santagata, D. et al. Rapid targeted mutational analysis of human tumours: a clinical platform to guide personalized cancer medicine. EMBO Mol. Med. 2, 146–158 (2010).
Gnirke, A. et al. Solution hybrid selection with ultra-long oligonucleotides for massively parallel targeted sequencing. Nature Biotech. 27, 182–189 (2009).
Lipson, D. et al. Identification of new ALK and RET gene fusions from colorectal and lung cancer biopsies. Nature Med. 18, 382–384 (2012).
Wagle, N. et al. High-throughput detection of actionable genomic alterations in clinical tumor samples by targeted, massively parallel sequencing. Cancer Discov. 2, 82–93 (2012).
Gargis, A. S. et al. Assuring the quality of next-generation sequencing in clinical laboratory practice. Nature Biotech. 30, 1033–1036 (2012).
Beadling, C. et al. Combining highly multiplexed PCR with semiconductor-based sequencing for rapid cancer genotyping. J. Mol. Diagn. 15, 171–176 (2013).
Committee on the Review of Omics-Based Tests for Predicting Patient Outcomes in Clinical Trials, Board on Health Care Services, Board on Health Sciences Policy & The Institute of Medicine. Evolution of Translational Omics: Lessons Learned and the Path Forward (eds Christine, M., Micheel, S. J. N. & Omenn, G. S.) (National Academies Press, 2012).
Meyerson, M., Gabriel, S. & Getz, G. Advances in understanding cancer genomes through second-generation sequencing. Nature Rev. Genet. 11, 685–696 (2010).
Jia, W. et al. SOAPfuse: an algorithm for identifying fusion transcripts from paired-end RNA-Seq data. Genome Biol. 14, R12 (2013).
Koboldt, D. C. et al. VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res. 22, 568–576 (2012).
Sathirapongsasuti, J. F. et al. Exome sequencing-based copy-number variation and loss of heterozygosity detection: exome CNV. Bioinformatics 27, 2648–2654 (2011).
Cibulskis, K. et al. Sensitive detection of somatic point mutations in impure and heterogeneous cancer samples. Nature Biotech. 31, 213–219 (2013).
Prahallad, A. et al. Unresponsiveness of colon cancer to BRAF(V600E) inhibition through feedback activation of EGFR. Nature 483, 100–103 (2012).
Cooper, G. M. & Shendure, J. Needles in stacks of needles: finding disease-causal variants in a wealth of genomic data. Nature Rev. Genet. 12, 628–640 (2011).
Forbes, S. A. et al. COSMIC: mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 39, D945–D950 (2011).
Waldron, L., Simpson, P., Parmigiani, G. & Huttenhower, C. Report on emerging technologies for translational bioinformatics: a symposium on gene expression profiling for archival tissues. BMC Cancer 12, 124 (2012).
Kerick, M. et al. Targeted high throughput sequencing in clinical cancer settings: formaldehyde fixed-paraffin embedded (FFPE) tumor tissues, input amount and tumor heterogeneity. BMC Med. Genomics 4, 68 (2011).
Olson, E. M., Lin, N. U., Krop, I. E. & Winer, E. P. The ethical use of mandatory research biopsies. Nature Rev. Clin. Oncol. 8, 620–625 (2011).
Gerlinger, M. et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366, 883–892 (2012).
Swanton, C. Intratumor heterogeneity: evolution through space and time. Cancer Res. 72, 4875–4882 (2012).
Gorre, M. E. et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293, 876–880 (2001).
Carver, B. S. et al. Reciprocal feedback regulation of PI3K and androgen receptor signaling in PTEN-deficient prostate Cancer. Cancer Cell 19, 575–586 (2011).
Mao, M. et al. Resistance to BRAF inhibition in BRAF-mutant colon cancer can be overcome with PI3K inhibition or demethylating agents. Clin. Cancer Res. 19, 657–667 (2013).
Greger, J. G. et al. Combinations of BRAF, MEK, and PI3K/mTOR inhibitors overcome acquired resistance to the BRAF inhibitor GSK2118436 dabrafenib, mediated by NRAS or MEK mutations. Mol. Cancer Ther. 11, 909–920 (2012).
Flaherty, K. T. et al. Combined BRAF and MEK inhibition in melanoma with BRAF V600 mutations. N. Engl. J. Med. 367, 1694–1703 (2012).
Ding, L. et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature 481, 506–510 (2012).
Welch, J. S. et al. The origin and evolution of mutations in acute myeloid leukemia. Cell 150, 264–278 (2012).
Ahmed, J. et al. CancerResource: a comprehensive database of cancer-relevant proteins and compound interactions supported by experimental knowledge. Nucleic Acids Res. 39, D960–D967 (2011).
Roychowdhury, S. et al. Personalized oncology through integrative high-throughput sequencing: a pilot study. Sci. Transl. Med. 3, 111ra121 (2011).
Turner, N. & Grose, R. Fibroblast growth factor signalling: from development to cancer. Nature Rev. Cancer 10, 116–129 (2010).
Kwak, E. L. et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N. Engl. J. Med. 363, 1693–1703 (2010).
Le Tourneau, C. et al. Designs and challenges for personalized medicine studies in oncology: focus on the SHIVA trial. Target. Oncol. 7, 253–265 (2012).
Herlyn, M. New dream team for melanoma therapy. Pigment Cell Melanoma Res. 25, 279–280 (2012).
Gautschi, O. et al. A patient with BRAF V600E lung adenocarcinoma responding to vemurafenib. J. Thorac. Oncol. 7, e23–e24 (2012).
Printz, C. “Stand up to cancer” focuses on accelerating promising research: campaign has raised approximately $180 million. Cancer 117, 1557–1559 (2011).
Wang, Z., Gerstein, M. & Snyder, M. RNA-Seq: a revolutionary tool for transcriptomics. Nature Rev. Genet. 10, 57–63 (2009).
Dietrich, S. et al. BRAF inhibition in refractory hairy-cell leukemia. N. Engl. J. Med. 366, 2038–2040 (2012).
Simon, R. Clinical trials for predictive medicine: new challenges and paradigms. Clin. Trials 7, 516–524 (2010).
Simon, R. Clinical trial designs for evaluating the medical utility of prognostic and predictive biomarkers in oncology. Per. Med. 7, 33–47 (2010).
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001).
Horstmann, E. et al. Risks and benefits of phase 1 oncology trials, 1991 through 2002. N. Engl. J. Med. 352, 895–904 (2005).
Sharma, M. R. & Schilsky, R. L. Role of randomized Phase III trials in an era of effective targeted therapies. Nature Reviews Clin. Oncol. 9, 208–214 (2011).
Dagher, R. et al. Approval summary: imatinib mesylate in the treatment of metastatic and/or unresectable malignant gastrointestinal stromal tumors. Clin. Cancer Res. 8, 3034–3038 (2002).
Parkinson, D. R., Johnson, B. E. & Sledge, G. W. Making personalized cancer medicine a reality: challenges and opportunities in the development of biomarkers and companion diagnostics. Clin. Cancer Res. 18, 619–624 (2012).
Samuels, Y. et al. High frequency of mutations of the PIK3CA gene in human cancers. Science 304, 554 (2004).
Chalhoub, N. & Baker, S. J. PTEN and the PI3-kinase pathway in cancer. Annu. Rev. Pathol. 4, 127–150 (2009).
Cheung, L. W. et al. High frequency of PIK3R1 and PIK3R2 mutations in endometrial cancer elucidates a novel mechanism for regulation of PTEN protein stability. Cancer Discov. 1, 170–185 (2011).
Carpten, J. D. et al. A transforming mutation in the pleckstrin homology domain of AKT1 in cancer. Nature 448, 439–444 (2007).
Banerji, S. et al. Sequence analysis of mutations and translocations across breast cancer subtypes. Nature 486, 405–409 (2012).
Hollander, M. C., Blumenthal, G. M. & Dennis, P. A. PTEN loss in the continuum of common cancers, rare syndromes and mouse models. Nature Rev. Cancer 11, 289–301 (2011).
Sato, T., Nakashima, A., Guo, L., Coffman, K. & Tamanoi, F. Single amino-acid changes that confer constitutive activation of mTOR are discovered in human cancer. Oncogene 29, 2746–2752 (2010).
Chan, J. A. et al. Pathogenesis of tuberous sclerosis subependymal giant cell astrocytomas: biallelic inactivation of TSC1 or TSC2 leads to mTOR activation. J. Neuropathol. Exp. Neurol. 63, 1236–1242 (2004).
Hardt, M., Chantaravisoot, N. & Tamanoi, F. Activating mutations of TOR (target of rapamycin). Genes Cells 16, 141–151 (2011).
Davies, H. et al. Mutations of the BRAF gene in human cancer. Nature 417, 949–954 (2002).
Slamon, D. J. & Clark, G. M. Amplification of c-erbB-2 and aggressive human breast tumors? Science 240, 1795–1798 (1988).
Johnson, R. L. et al. Human homolog of patched, a candidate gene for the basal cell nevus syndrome. Science 272, 1668–1671 (1996).
Xie, J. et al. Activating Smoothened mutations in sporadic basal-cell carcinoma. Nature 391, 90–92 (1998).
Xie, J. et al. Mutations of the PATCHED gene in several types of sporadic extracutaneous tumors. Cancer Res. 57, 2369–2372 (1997).
Gherardi, E., Birchmeier, W., Birchmeier, C. & Vande Woude, G. Targeting MET in cancer: rationale and progress. Nature Rev. Cancer 12, 89–103 (2012).
Quintas-Cardama, A., Kantarjian, H., Cortes, J. & Verstovsek, S. Janus kinase inhibitors for the treatment of myeloproliferative neoplasias and beyond. Nature Rev. Drug Discov. 10, 127–140 (2011).
Lapenna, S. & Giordano, A. Cell cycle kinases as therapeutic targets for cancer. Nature Rev. Drug Discov. 8, 547–566 (2009).
Lens, S. M., Voest, E. E. & Medema, R. H. Shared and separate functions of polo-like kinases and aurora kinases in cancer. Nature Rev. Cancer 10, 825–841 (2010).
Ryan, C. J. & Tindall, D. J. Androgen receptor rediscovered: the new biology and targeting the androgen receptor therapeutically. J. Clin. Oncol. 29, 3651–3658 (2011).
Stirewalt, D. L. & Radich, J. P. The role of FLT3 in haematopoietic malignancies. Nature Rev. Cancer 3, 650–665 (2003).
Shangary, S. & Wang, S. Targeting the MDM2–p53 interaction for cancer therapy. Clin. Cancer Res. 14, 5318–5324 (2008).
Corless, C. L., Barnett, C. M. & Heinrich, M. C. Gastrointestinal stromal tumours: origin and molecular oncology. Nature Rev. Cancer 11, 865–878 (2011).
Soda, M. et al. Identification of the transforming EML4–ALK fusion gene in non-small-cell lung cancer. Nature 448, 561–566 (2007).
Chen, Y. et al. Oncogenic mutations of ALK kinase in neuroblastoma. Nature 455, 971–974 (2008).
Bergethon, K. et al. ROS1 rearrangements define a unique molecular class of lung cancers. J. Clin. Oncol. 30, 863–870 (2012).
Matulonis, U. A. et al. High throughput interrogation of somatic mutations in high grade serous cancer of the ovary. PLoS ONE 6, e24433 (2011).
Weiss, G. J. et al. Paired tumor and normal whole genome sequencing of metastatic olfactory neuroblastoma. PLoS ONE 7, e37029 (2012).
Acknowledgements
S.R. is supported by Pelotonia, the American Cancer Society (grant MRSG-12-194-01), the Landon-Foundation AACR Innovator Award for Personalized Cancer Medicine, and a Young Investigator Award from the Prostate Cancer Foundation. Special thanks to J. Bush for reading the manuscript and A. Marsell for administrative support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
DATABASES
FURTHER INFORMATION
Glossary
- Amplicon-based sequencing
-
The use of PCR to selectively amplify small genomic regions for sequencing.
- Clinical Laboratory Improvements Amendment
-
(CLIA). A US regulatory standard that applies to all clinical laboratory testing.
- Depth of coverage
-
Also known as sequencing depth; the number of times a genome position has been sequenced to ensure data accuracy.
- Driver mutations
-
Mutations that are implicated in cancer biology and provide a growth advantage at some point during the development of cancer, causing positive selection for the mutation.
- Next-generation sequencing
-
(NGS). Also referred to as high-throughput sequencing and massively parallel sequencing. Refers to technologies that parallelize DNA sequencing effectively to produce millions of sequences in a rapid and cost-effective manner.
- Orthogonal platform
-
A second DNA sequencing technology used to confirm data obtained through next-generation sequencing.
- Passenger mutations
-
Mutations that do not contribute to cancer biology and do not appear to provide a growth advantage, but are carried along with driver mutations.
- Transcriptome sequencing
-
Also referred to as RNAseq or whole-transcriptome shotgun sequencing. The sequencing of cDNA generated from total RNA. Transcriptome sequencing can provide data on gene expression, alternatively spliced transcripts, non-coding RNA and gene fusions or rearrangements.
- Whole-exome sequencing
-
Also referred to as targeted exome capture. The selective application of next-generation sequencing to the coding regions of the genome using complementary oligonucleotide probes that selectively hybridize and capture the desired genomic regions of interest. Whole-exome sequencing represents approximately 20,000 genes or a little more than 1% of the whole genome, and is therefore a cheaper strategy than whole-genome sequencing. Targeted gene sequencing can be completed for a shorter defined list of genes: for example, for 200 to 1,000 or more cancer-related genes.
- Whole-genome sequencing
-
Complete sequencing of an organism's entire DNA sequence, including exons and non-coding genome regions.
Rights and permissions
About this article
Cite this article
Simon, R., Roychowdhury, S. Implementing personalized cancer genomics in clinical trials. Nat Rev Drug Discov 12, 358–369 (2013). https://doi.org/10.1038/nrd3979
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd3979
This article is cited by
-
Integrative multi-omics networks identify PKCδ and DNA-PK as master kinases of glioblastoma subtypes and guide targeted cancer therapy
Nature Cancer (2023)
-
Cancer Biomarkers: Status and Its Future Direction
Indian Journal of Surgery (2023)
-
Comprehensive genomic profiling for oncological advancements by precision medicine
Medical Oncology (2023)
-
Combination of furosemide, gold, and dopamine as a potential therapy for breast cancer
Functional & Integrative Genomics (2023)
-
Accucopy: accurate and fast inference of allele-specific copy number alterations from low-coverage low-purity tumor sequencing data
BMC Bioinformatics (2021)