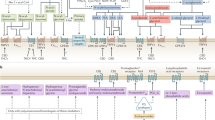
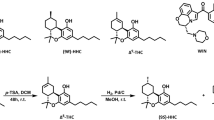
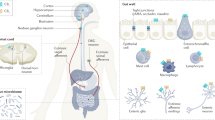
Abstract
The endocannabinoid signalling system was discovered because receptors in this system are the targets of compounds present in psychotropic preparations of Cannabis sativa. The search for new therapeutics that target endocannabinoid signalling is both challenging and potentially rewarding, as endocannabinoids are implicated in numerous physiological and pathological processes. Hundreds of mediators chemically related to the endocannabinoids, often with similar metabolic pathways but different targets, have complicated the development of inhibitors of endocannabinoid metabolic enzymes but have also stimulated the rational design of multi-target drugs. Meanwhile, drugs based on botanical cannabinoids have come to the clinical forefront, synthetic agonists designed to bind cannabinoid receptor 1 with very high affinity have become a societal threat and the gut microbiome has been found to signal in part through the endocannabinoid network. The current development of drugs that alter endocannabinoid signalling and how this complex system could be pharmacologically manipulated in the future are described in this Opinion article.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Alexander, S. P. Therapeutic potential of cannabis-related drugs. Prog. Neuropsychopharmacol. Biol. Psychiatry 64, 157–166 (2016).
Loewe, S. [Active principals of the cannabis and the pharmacology of the cannabinols]. Naunyn. Schmiedebergs Arch. Exp. Pathol. Pharmakol. 211, 175–193 (1950).
Mechoulam, R. & Shvo, Y. Hashish. I. The structure of cannabidiol. Tetrahedron 19, 2073–2078 (1963).
Gaoni, R. & Mechoulam, R. Isolation, structure and partial synthesis of an active constituent of hashish. J. Am. Chem. Soc. 86, 1646–1647 (1964).
Mechoulam, R. et al. Chemical basis of hashish activity. Science 169, 611–612 (1970).
Varvel, S. A. et al. Δ9-tetrahydrocannbinol accounts for the antinociceptive, hypothermic, and cataleptic effects of marijuana in mice. J. Pharmacol. Exp. Ther. 314, 329–337 (2005).
Matsuda, L. A. et al. Structure of a cannabinoid receptor and functional expression of a cloned cDNA. Nature 346, 561–564 (1990).
Munro, S., Thomas, K. L. & Abushaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 365, 31–65 (1993).
Howlett, A. C. in Cannabinoids: Handbook of Experimental Pharmacology (ed. Pertwee R. G.) 53–79 (Springer, Berlin, 2005).
Devane, W. A. et al. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258, 1946–1949 (1992).
Mechoulam, R. et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem. Pharmacol. 50, 83–90 (1995).
Sugiura, T. et al. 2-Arachidonoylglycerol: a possible endogenous cannabinoid receptor ligand in brain. Biochem. Biophys. Res. Commun. 215, 89–97 (1995).
Di Marzo, V. & Fontana, A. Anandamide, an endogenous cannabinomimetic eicosanoid: 'killing two birds with one stone'. Prostaglandins Leukot. Essent. Fatty Acids 53, 1–11 (1995).
Okamoto, Y. et al. Molecular characterization of a phospholipase D generating anandamide and its congeners. J. Biol. Chem. 279, 5298–5305 (2004).
Bisogno, T. et al. Cloning of the first sn1-DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain. J. Cell Biol. 163, 463–468 (2003).
Cravatt, B. F. et al. Molecular characterization of an enzyme that degrades neuromodulatory fatty-acid amides. Nature 384, 83–87 (1996).
Dinh, T. P. et al. Brain monoglyceride lipase participating in endocannabinoid inactivation. Proc. Natl Acad. Sci. USA 99, 10819–10824 (2002).
Ligresti, A., De Petrocellis, L. & Di Marzo, V. From Phytocannabinoids to Cannabinoid Receptors and Endocannabinoids: Pleiotropic Physiological and Pathological Roles Through Complex Pharmacology. Physiol. Rev. 96, 1593–1659 (2016).
Di Marzo, V. Targeting the endocannabinoid system: to enhance or reduce? Nat. Rev. Drug Discov. 7, 438–455 (2008).
Devinsky, O. et al. Cannabidiol in patients with treatment-resistant epilepsy: an open-label interventional trial. Lancet Neurol. 15, 270–278 (2016).
Keating, G. M. Δ9-Tetrahydrocannabinol/Cannabidiol oromucosal spray (Sativexâ): a review in multiple sclerosis-related spasticity. Drugs 77, 563–574 (2017).
Lötsch, J., Weyer-Menkhoff, I. & Tegeder, I. Current evidence of cannabinoid-based analgesia obtained in preclinical and human experimental settings. Eur. J. Pain 22, 471–484 (2017).
Tambaro, S. & Bortolato, M. Cannabinoid-related agents in the treatment of anxiety disorders: current knowledge and future perspectives. Recent Pat. CNS Drug Discov. 7, 25–40 (2012).
Fernández-Ruiz, J., Romero, J. & Ramos, J. A. Endocannabinoids and neurodegenerative disorders: Parkinson's disease, Huntington's chorea, Alzheimer's disease, and others. Handb. Exp. Pharmacol. 231, 233–259 (2015).
Hasenoehrl, C. Storr, M. & Schicho, R. Cannabinoids for treating inflammatory bowel diseases: where are we and where do we go? Expert Rev. Gastroenterol. Hepatol. 11, 329–337 (2017).
Richey, J. M. & Woolcott, O. Re-visiting the endocannabinoid system and its therapeutic potential in obesity and associated diseases. Curr. Diab. Rep. 17, 99 (2017).
Roser, P., Vollenweider, F. X. & Kawohl, W. Potential antipsychotic properties of central cannabinoid (CB1) receptor antagonists. World J. Biol. Psychiatry 11, 208–219 (2010).
Puighermanal, E. et al. Cellular and intracellular mechanisms involved in the cognitive impairment of cannabinoids. Phil. Trans. R. Soc. B 367, 3254–3263 (2012).
Sharma, M. K. et al. Prospective therapeutic agents for obesity: molecular modification approaches of centrally and peripherally acting selective cannabinoid 1 receptor antagonists. Eur. J. Med. Chem. 79, 298–339 (2014).
Badowski, M. E. A review of oral cannabinoids and medical marijuana for the treatment of chemotherapy-induced nausea and vomiting: a focus on pharmacokinetic variability and pharmacodynamics. Cancer Chemother. Pharmacol. 80, 441–449 (2017).
Tam, J. et al. The therapeutic potential of targeting the peripheral endocannabinoid/CB1 receptor system. Eur. J. Intern. Med. 49, 23–29 (2018).
Fowler, C. J. The potential of inhibitors of endocannabinoid metabolism for drug development: a critical review. Handb. Exp. Pharmacol. 231, 95–128 (2015).
Lodola, A. et al. Fatty acid amide hydrolase inhibitors: a patent review (2009–2014). Expert Opin. Ther. Pat. 25, 1247–1266 (2015).
Huggins, J. P. et al. An efficient randomised, placebo-controlled clinical trial with the irreversible fatty acid amide hydrolase-1 inhibitor PF-04457845, which modulates endocannabinoids but fails to induce effective analgesia in patients with pain due to osteoarthritis of the knee. Pain 153, 1837–1846 (2012).
Kerbrat, A. et al. Acute neurologic disorder from an inhibitor of fatty acid amide hydrolase. N. Engl. J. Med. 375, 1717–1725 (2016).
van Esbroeck, A. C. M. et al. Activity-based protein profiling reveals off-target proteins of the FAAH inhibitor BIA 10–2474. Science 356, 1084–1087 (2017).
Nguyen, T. et al. Allosteric modulation: An alternate approach targeting the cannabinoid CB1 receptor. Med. Res. Rev. 37, 441–474 (2017).
Ignatowska-Jankowska, B. M. et al. A cannabinoid CB1 receptor-positive allosteric modulator reduces neuropathic pain in the mouse with no psychoactive effects. Neuropsychopharmacology 40, 2948–2959 (2015).
Slivicki, R. A. et al. Positive allosteric modulation of cannabinoid receptor type 1 suppresses pathological pain without producing tolerance or dependence. Biol. Psychiatry https://doi.org/10.1016/j.biopsych.2017.06.032 (2017).
Appiah, K. K. et al. Identification of small molecules that selectively inhibit diacylglycerol lipase-α activity. J. Biomol. Screen 19, 595–605 (2014).
Hsu, K.-L. et al. Optimization and characterization of a triazole urea inhibitor for diacylglycerol lipase beta (DAGL-β). Probe reports from the NIH molecular libraries program (National Center for Biotechnology Information, Bethesda, 2013).
Baggelaar, M. P. et al. Highly selective, reversible inhibitor identified by comparative chemoproteomics modulates diacylglycerol lipase activity in neurons. J. Am. Chem. Soc. 137, 8851–8857 (2015).
Greig, I. R. et al. Development of indole sulfonamides as cannabinoid receptor negative allosteric modulators. Bioorg. Med. Chem. Lett. 26, 4403–4407 (2016).
Bisogno, T. et al. A novel fluorophosphonate inhibitor of the biosynthesis of the endocannabinoid 2-arachidonoylglycerol with potential anti-obesity effects. Br. J. Pharmacol. 169, 784–793 (2013).
Janssen, F. J. & van der Stelt, M. Inhibitors of diacylglycerol lipases in neurodegenerative and metabolic disorders. Bioorg. Med. Chem. Lett. 26, 3831–3837 (2016).
Wilkerson, J. L. et al. Diacylglycerol lipase β inhibition reverses nociceptive behaviour in mouse models of inflammatory and neuropathic pain. Br. J. Pharmacol. 173, 1678–1692 (2016).
Navarro, G. et al. Targeting cannabinoid CB2 receptors in the central nervous system. Medicinal chemistry approaches with focus on neurodegenerative disorders. Front. Neurosci. 10, 406 (2016).
Morales, P. et al. Cannabinoid receptor 2 (CB2) agonists and antagonists: a patent update. Expert Opin. Ther. Pat. 26, 843–856 (2016).
Gruden, G. et al. Role of the endocannabinoid system in diabetes and diabetic complications. Br. J. Pharmacol. 173, 1116–1127 (2016).
Johnson, D. E. et al. Isolation, identification and synthesis of an endogenous arachidonic amide that inhibits calcium channel antagonist 1,4-dihydropyridine binding. Prostaglandins Leukot. Essent. Fatty Acids 48, 429–437 (1993).
Shimasue, K. et al. Effects of anandamide and arachidonic acid on specific binding of (+) -PN200-110, diltiazem and (−)-desmethoxyverapamil to L-type Ca2+ channel. Eur. J. Pharmacol. 296, 347–350 (1996).
Di Marzo, V. et al. Levels, metabolism, and pharmacological activity of anandamide in CB1 cannabinoid receptor knockout mice: evidence for non-CB1, non-CB2 receptor-mediated actions of anandamide in mouse brain. J. Neurochem. 75, 2434–2444 (2000).
Breivogel, C. S. et al. Evidence for a new G protein-coupled cannabinoid receptor in mouse brain. Mol. Pharmacol. 60, 155–163 (2001).
Monory, K. et al. Novel, not adenylyl cyclase-coupled cannabinoid binding site in cerebellum of mice. Biochem. Biophys. Res. Commun. 292, 231–235 (2002).
Offertáler, L. et al. Selective ligands and cellular effectors of a G protein-coupled endothelial cannabinoid receptor. Mol. Pharmacol. 63, 699–705 (2003).
Ryberg, E. et al. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol. 152, 1092–1101 (2007).
Lauckner, J. E. et al. GPR55 is a cannabinoid receptor that increases intracellular calcium and inhibits M current. Proc. Natl Acad. Sci. USA 105, 2699–2704 (2008).
McHugh, D. et al. Δ9-Tetrahydrocannabinol and N-arachidonyl glycine are full agonists at GPR18 receptors and induce migration in human endometrial HEC-1B cells. Br. J. Pharmacol. 165, 2414–2424 (2012).
Lee, J. W. et al. Orphan GPR110 (ADGRF1) targeted by N-docosahexaenoylethanolamine in development of neurons and cognitive function. Nat. Commun. 7, 13123 (2016).
Oka, S. et al. Identification of GPR55 as a lysophosphatidylinositol receptor. Biochem. Biophys. Res. Commun. 362, 928–934 (2007).
Finlay, D. B. et al. GPR18 undergoes a high degree of constitutive trafficking but is unresponsive to N-arachidonoyl glycine. PeerJ. 4, e1835 (2016).
Hanuš, L. O. et al. Phytocannabinoids: a unified critical inventory. Nat. Prod. Rep. 33, 1357–1392 (2016).
De Petrocellis, L., Nabissi, M., Santoni, G. & Ligresti, A. Actions and regulation of ionotropic cannabinoid receptors. Adv. Pharmacol. 80, 249–289 (2017).
Bisogno, T. et al. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br. J. Pharmacol. 134, 845–852 (2001).
Hu, S. S. et al. The biosynthesis of N-arachidonoyl dopamine (NADA), a putative endocannabinoid and endovanilloid, via conjugation of arachidonic acid with dopamine. Prostaglandins Leukot. Essent. Fatty Acids 81, 291–301 (2009).
Raboune, S. et al. Novel endogenous N-acyl amides activate TRPV1-4 receptors, BV-2 microglia, and are regulated in brain in an acute model of inflammation. Front. Cell. Neurosci. 8, 195 (2014).
De Petrocellis, L. et al. Plant-derived cannabinoids modulate the activity of transient receptor potential channels of ankyrin type-1 and melastatin type-8. J. Pharmacol. Exp. Ther. 325, 1007–1015 (2008).
Iannotti, F. A. et al. Nonpsychotropic plant cannabinoids, cannabidivarin (CBDV) and cannabidiol (CBD), activate and desensitize transient receptor potential vanilloid 1 (TRPV1) channels in vitro: potential for the treatment of neuronal hyperexcitability. ACS Chem. Neurosci 5, 1131–1141 (2014).
De Petrocellis, L. et al. Regulation of transient receptor potential channels of melastatin type 8 (TRPM8): effect of cAMP, cannabinoid CB1 receptors and endovanilloids. Exp. Cell Res. 313, 1911–1920 (2007).
Lam, P. M., McDonald, J. & Lambert, D. G. Characterization and comparison of recombinant human and rat TRPV1 receptors: effects of exo- and endocannabinoids. Br. J. Anaesth. 94, 649–656 (2005).
Di Marzo, V. et al. Hypolocomotor effects in rats of capsaicin and two long chain capsaicin homologues. Eur. J. Pharmacol. 25 420, 123–131 (2001).
Starowicz, K. et al. Full inhibition of spinal FAAH leads to TRPV1-mediated analgesic effects in neuropathic rats and possible lipoxygenase-mediated remodeling of anandamide metabolism. PLOS ONE 8, e60040 (2013).
Maione, S. et al. Elevation of endocannabinoid levels in the ventrolateral periaqueductal grey through inhibition of fatty acid amide hydrolase affects descending nociceptive pathways via both cannabinoid receptor type 1 and transient receptor potential vanilloid type-1 receptors. J. Pharmacol. Exp. Ther. 316, 969–982 (2006).
Morgese, M. G. et al. Anti-dyskinetic effects of cannabinoids in a rat model of Parkinson's disease: role of CB1 and TRPV1 receptors. Exp. Neurol. 208, 110–119 (2007).
Benito, C. et al. β-Amyloid exacerbates inflammation in astrocytes lacking fatty acid amide hydrolase through a mechanism involving PPAR-α, PPAR-γ and TRPV1, but not CB1 or CB2 receptors. Br. J. Pharmacol. 166, 1474–1489 (2012).
Schmid, H. H. Pathways and mechanisms of N-acylethanolamine biosynthesis: can anandamide be generated selectively? Chem. Phys. Lipids 108, 71–87 (2000).
Jin, X. H. et al. Discovery and characterization of a Ca2+-independent phosphatidylethanolamine N-acyltransferase generating the anandamide precursor and its congeners. J. Biol. Chem. 282, 3614–3623 (2007).
Ogura, Y. et al. A calcium-dependent acyltransferase that produces N-acyl phosphatidylethanolamines. Nat. Chem. Biol. 12, 669–671 (2016).
Tsuboi, K. et al. Molecular characterization of N-acylethanolamine-hydrolyzing acid amidase, a novel member of the choloylglycine hydrolase family with structural and functional similarity to acid ceramidase. J. Biol. Chem. 280, 11082–11092 (2005).
Fowler, C. J. Oleamide: a member of the endocannabinoid family? Br. J. Pharmacol. 141, 195–196 (2004).
Saghatelian, A. et al. A FAAH-regulated class of N-acyl taurines that activates TRP ion channels. Biochemistry 45, 9007–9015 (2006).
Labar, G. et al. Crystal structure of the human monoacylglycerol lipase, a key actor in endocannabinoid signaling. Chembiochem 11, 218–227 (2010).
Navia-Paldanius, D., Savinainen, J. R. & Laitinen, J. T. Biochemical and pharmacological characterization of human α/β-hydrolase domain containing 6 (ABHD6) and 12 (ABHD12). J. Lipid Res. 53, 2413–2424 (2012).
Kozak, K. R., Prusakiewicz, J. J. & Marnett, L. J. Oxidative metabolism of endocannabinoids by COX-2. Curr. Pharm. Des. 10, 659–667 (2004).
Ueda, N. et al. Lipoxygenase-catalyzed oxygenation of arachidonylethanolamide, a cannabinoid receptor agonist. Biochim. Biophys. Acta 1254, 127–134 (1995).
Snider, N. T., Walker, V. J. & Hollenberg, P. F. Oxidation of the endogenous cannabinoid arachidonoyl ethanolamide by the cytochrome P450 monooxygenases: physiological and pharmacological implications. Pharmacol. Rev. 62, 136–154 (2010).
Granchi, C. et al. A patent review of Monoacylglycerol Lipase (MAGL) inhibitors Expert Opin. Ther. Pat. 27, 1341–1351 (2017).
Bedse, G. et al. Therapeutic endocannabinoid augmentation for mood and anxiety disorders: comparative profiling of FAAH, MAGL and dual inhibitors. Transl Psychiatry. 8, 92 (2018).
Feledziak, M. et al. Inhibitors of the endocannabinoid-degrading enzymes, or how to increase endocannabinoid's activity by preventing their hydrolysis. Recent Pat. CNS Drug Discov. 7, 49–70 (2012).
Sagar, D. R., Kendall, D. A. & Chapman, V. Inhibition of fatty acid amide hydrolase produces PPARα-mediated analgesia in a rat model of inflammatory pain. Br. J. Pharmacol. 155, 1297–1306 (2008).
Luchicchi, A. et al. Effects of fatty acid amide hydrolase inhibition on neuronal responses to nicotine, cocaine and morphine in the nucleus accumbens shell and ventral tegmental area: involvement of PPAR-alpha nuclear receptors. Addict. Biol. 15, 277–288 (2010).
Kawahara, H., Drew, G. M., Christie, M. J., Vaughan, C. W. Inhibition of fatty acid amide hydrolase unmasks CB1 receptor and TRPV1 channel-mediated modulation of glutamatergic synaptic transmission in midbrain periaqueductal grey. Br. J. Pharmacol. 163, 1214–1222 (2011).
Hansen, H. S. et al. GPR119 as a fat sensor. Trends Pharmacol. Sci. 33, 374–381 (2012).
Wilkerson, J. L. et al. The endocannabinoid hydrolysis inhibitor SA-57: Intrinsic antinociceptive effects, augmented morphine-induced antinociception, and attenuated heroin seeking behavior in mice. Neuropharmacology 114, 156–167 (2017).
Ghosh, S. et al. The monoacylglycerol lipase inhibitor JZL184 suppresses inflammatory pain in the mouse carrageenan model. Life Sci. 92, 498–505 (2013).
Bandoh, K. et al. Lysophosphatidic acid (LPA) receptors of the EDG family are differentially activated by LPA species. Structure-activity relationship of cloned LPA receptors. FEBS Lett. 478, 159–165 (2000).
Nakane, S. et al. 2-Arachidonoyl-sn-glycero-3-phosphate, an arachidonic acid-containing lysophosphatidic acid: occurrence and rapid enzymatic conversion to 2-arachidonoyl-sn-glycerol, a cannabinoid receptor ligand, in rat brain. Arch. Biochem. Biophys. 402, 51–58 (2002).
Nomura, D. K. et al. Endocannabinoid hydrolysis generates brain prostaglandins that promote neuroinflammation. Science 334, 809–813 (2011).
Nomura, D. K. et al. Monoacylglycerol lipase exerts dual control over endocannabinoid and fatty acid pathways to support prostate cancer. Chem. Biol. 18, 846–856 (2011).
Gatta, L. et al. Discovery of prostamide F2α and its role in inflammatory pain and dorsal horn nociceptive neuron hyperexcitability. PLOS ONE 7, e31111 (2012).
Alhouayek, M., Masquelier, J. & Muccioli, G. G. Controlling 2-arachidonoylglycerol metabolism as an anti-inflammatory strategy. Drug Discov. Today 19, 295–304 (2014).
Valdeolivas, S. et al. The inhibition of 2-arachidonoyl-glycerol (2-AG) biosynthesis, rather than enhancing striatal damage, protects striatal neurons from malonate-induced death: a potential role of cyclooxygenase-2-dependent metabolism of 2-AG. Cell Death Dis. 4, e862 (2013).
Brüser, A. et al. Prostaglandin E2 glyceryl ester is an endogenous agonist of the nucleotide receptor P2Y6. Sci. Rep. 7, 2380 (2017).
Hu, S. S. et al. Prostaglandin E2 glycerol ester, an endogenous COX-2 metabolite of 2-arachidonoylglycerol, induces hyperalgesia and modulates NFκB activity. Br. J. Pharmacol. 153, 1538–1549 (2008).
Cristino, L. et al. Orexin/endocannabinoid/leptin interaction affects hypothalamic tau phosphorilation by glycogen synthase kinase-3β activation. 10th FENS Forum of Neuroscience (Copenhagen, Denmark, 2016).
Gillum, M. P. et al. N-Acylphosphatidylethanolamine, a gut- derived circulating factor induced by fat ingestion, inhibits food intake. Cell 135, 813–824 (2008).
Weber, A. et al. Formation of prostamides from anandamide in FAAH knockout mice analyzed by HPLC with tandem mass spectrometry. J. Lipid Res. 45, 757–763 (2004).
Moreira, F. A., Kaiser, N., Monory, K. & Lutz, B. Reduced anxiety-like behaviour induced by genetic and pharmacological inhibition of the endocannabinoid-degrading enzyme fatty acid amide hydrolase (FAAH) is mediated by CB1 receptors. Neuropharmacology 54, 141–150 (2008).
Navia-Paldanius, D., et al. Increased tonic cannabinoid CB1R activity and brain region-specific desensitization of CB1R Gi/o signaling axis in mice with global genetic knockout of monoacylglycerol lipase. Eur. J. Pharm. Sci. 77, 180–188 (2015).
Imperatore, R., et al. Genetic deletion of monoacylglycerol lipase leads to impaired cannabinoid receptor CB1R signaling and anxiety-like behavior. J. Neurochem. 135, 799–813 (2015).
Huang, S. M. et al. Identification of a new class of molecules, the arachidonyl amino acids, and characterization of one member that inhibits pain. J. Biol. Chem. 276, 42639–42644 (2001).
Kohno, M. et al. Identification of N-arachidonylglycine as the endogenous ligand for orphan G-protein-coupled receptor GPR18. Biochem. Biophys. Res. Commun. 347, 827–832 (2006).
Chemin, J. et al. Direct inhibition of T-type calcium channels by the endogenous cannabinoid anandamide. EMBO J. 20, 7033–7040 (2001).
Cazade, M. et al. Cross-modulation and molecular interaction at the Cav3.3 protein between the endogenous lipids and the T-type calcium channel antagonist TTA-A2. Mol. Pharmacol. 85, 218–225 (2014).
Mann, A. et al. Palmitoyl Serine: An endogenous neuroprotective endocannabinoid-like entity after traumatic brain injury. J. Neuroimmune Pharmacol. 10, 356–363 (2015).
Smoum, R. et al. Oleoyl serine, an endogenous N-acyl amide, modulates bone remodeling and mass. Proc. Natl Acad. Sci. USA 107, 17710–17715 (2010).
Bisogno, T. et al. N-Acyl-dopamines: novel synthetic CB(1) cannabinoid-receptor ligands and inhibitors of anandamide inactivation with cannabimimetic activity in vitro and in vivo. Biochem. J. 351, 817–824 (2010).
Huang, S. M. et al. An endogenous capsaicin-like substance with high potency at recombinant and native vanilloid VR1 receptors. Proc. Natl Acad. Sci. USA 99, 8400–8405 (2002).
Chu, C. J. et al. N-Oleoyldopamine, a novel endogenous capsaicin-like lipid that produces hyperalgesia. J. Biol. Chem. 278, 13633–13639 (2003).
Ross, H. R., Gilmore, A. J. & Connor, M. Inhibition of human recombinant T-type calcium channels by the endocannabinoid N-arachidonoyl dopamine. Br. J. Pharmacol. 156, 740–750 (2009).
Barbara, G. et al. T-Type calcium channel inhibition underlies the analgesic effects of the endogenous lipoamino acids. J. Neurosci. 29, 13106–13114 (2009).
Verhoeckx, K. C. et al. Presence, formation and putative biological activities of N-acyl serotonins, a novel class of fatty-acid derived mediators, in the intestinal tract. Biochim. Biophys. Acta 1811, 578–586 (2011).
Bisogno, T. et al. Arachidonoylserotonin and other novel inhibitors of fatty acid amide hydrolase. Biochem. Biophys. Res. Commun. 248, 515–522 (1998).
Maione, S. et al. Analgesic actions of N-arachidonoyl-serotonin, a fatty acid amide hydrolase inhibitor with antagonistic activity at vanilloid TRPV1 receptors. Br. J. Pharmacol. 150, 766–781 (2007).
Gilmore, A. J. et al. Inhibition of human recombinant T-type calcium channels by N-arachidonoyl 5-HT. Br. J. Pharmacol. 167, 1076–1088 (2012).
Dempsey, D. R. et al. Identification of an arylalkylamine N-acyltransferase from Drosophila melanogaster that catalyzes the formation of long-chain N-acylserotonins. FEBS Lett. 588, 594–599 (2014).
Bradshaw, H. B., Raboune, S. & Hollis, J. L. Opportunistic activation of TRP receptors by endogenous lipids: exploiting lipidomics to understand TRP receptor cellular communication. Life Sci. 92, 404–409 (2013).
Connor, M., Vaughan, C. W. & Vandenberg, R. J. N-Acyl amino acids and N-acyl neurotransmitter conjugates: neuromodulators and probes for new drug targets. Br. J. Pharmacol. 160, 1857–1871 (2010).
Di Marzo, V. & Wang, J. (eds) The Endocannabinoidome: The World of Endocannabinoids and Related Mediators (Elsevier Academic Press, London, 2015).
Piscitelli, F. et al. Effect of dietary krill oil supplementation on the endocannabinoidome of metabolically relevant tissues from high-fat-fed mice. Nutr. Metab. (Lond.) 8, 51 (2011).
Witkamp, R. Fatty acids, endocannabinoids and inflammation. Eur. J. Pharmacol. 785, 96–107 (2016).
Piazza, P. V., Cota, D. & Marsicano, G. The CB1 receptor as the cornerstone of exostasis. Neuron 93, 1252–1274 (2017).
Hasenoehrl, C. et al. G protein-coupled receptor GPR55 promotes colorectal cancer and has opposing effects to cannabinoid receptor 1. Int. J. Cancer 142, 121–132 (2018).
Ramer, R. & Hinz, B. Cannabinoids as anticancer drugs. Adv. Pharmacol. 80, 397–436 (2017).
Tsujiuchi, T. et al. Lysophosphatidic acid receptors in cancer pathobiology. Histol. Histopathol. 29, 313–321 (2014).
Velasco, M., O'Sullivan, C. & Sheridan, G. K. Lysophosphatidic acid receptors (LPARs): Potential targets for the treatment of neuropathic pain. Neuropharmacology 113, 608–617 (2017).
Deliu, E. et al. The lysophosphatidylinositol receptor GPR55 modulates pain perception in the periaqueductal gray. Mol. Pharmacol. 88, 265–272 (2015).
McKillop, A. M. et al. Evaluation of the insulin releasing and antihyperglycaemic activities of GPR55 lipid agonists using clonal β-cells, isolated pancreatic islets and mice. Br. J. Pharmacol. 170, 978–990 (2013).
Piras, A. et al. Effects of dietary CLA on n-3 HUFA score and N-acylethanolamides biosynthesis in the liver of obese Zucker rats. Prostaglandins Leukot. Essent. Fatty Acids 98, 15–19 (2015).
Melis, M. & Pistis, M. Targeting the interaction between fatty acid ethanolamides and nicotinic receptors: therapeutic perspectives. Pharmacol. Res. 86, 42–49 (2014).
Silvestri, C. et al. Anandamide-derived prostamide F2α negatively regulates adipogenesis. J. Biol. Chem. 288, 23307–22332 (2013).
Bisogno, T., Piscitelli, F. & Di Marzo, V. Lipidomic methodologies applicable to the study of endocannabinoids and related compounds: Endocannabinoidomics. Eur. J. Lipid Sci. Technol. 111, 53–63 (2008).
Piscitelli, F. in The Endocannabinoidome: The World of Endocannabinoids and Related Mediators ( eds Di Marzo, V. & Wang, J. ) 137–152 (Elsevier Academic Press, London, 2015).
Di Marzo, V. et al. Interactions between synthetic vanilloids and the endogenous cannabinoid system. FEBS Lett. 436, 449–454 (1998).
Ligresti, A. et al. Exploiting nanotechnologies and TRPV1 channels to investigate the putative anandamide membrane transporter. PLOS ONE 5, e10239 (2010).
Chicca, A. et al. Chemical probes to potently and selectively inhibit endocannabinoid cellular reuptake. Proc. Natl Acad. Sci. USA 114, E5006–E5015 (2017).
Zygmunt, P. M. et al. The anandamide transport inhibitor AM404 activates vanilloid receptors. Eur. J. Pharmacol. 396, 39–42 (2000).
Högestätt, E. D. et al. Conversion of acetaminophen to the bioactive N-acylphenolamine AM404 via fatty acid amide hydrolase-dependent arachidonic acid conjugation in the nervous system. J. Biol. Chem. 280, 31405–31412 (2005).
Sharma, C. V. et al. First evidence of the conversion of paracetamol to AM404 in human cerebrospinal fluid. J. Pain Res. 10, 2703–2709 (2017).
Ohashi, N. et al. Acetaminophen metabolite N-acylphenolamine induces analgesia via transient receptor potential vanilloid 1 receptors expressed on the primary afferent terminals of C-fibers in the spinal dorsal horn. Anesthesiology 127, 355–371 (2017).
Eberhardt, M. J. et al. Reactive metabolites of acetaminophen activate and sensitize the capsaicin receptor TRPV1. Sci. Rep. 7, 12775 (2017).
Ottani, A. et al. The analgesic activity of paracetamol is prevented by the blockade of cannabinoid CB1 receptors. Eur. J. Pharmacol. 531, 280–281 (2006).
Costa, B. et al. The dual fatty acid amide hydrolase/TRPV1 blocker, N-arachidonoyl-serotonin, relieves carrageenan-induced inflammation and hyperalgesia in mice. Pharmacol.Res. 61, 537–546 (2010).
Malek, N. et al. The multiplicity of spinal AA-5-HT anti-nociceptive action in a rat model of neuropathic pain. Pharmacol. Res. 111, 251–263 (2016).
Micale, V. et al. Anxiolytic effects in mice of a dual blocker of fatty acid amide hydrolase and transient receptor potential vanilloid type-1 channels. Neuropsychopharmacology 34, 593–606 (2009).
Navarria, A. et al. The dual blocker of FAAH/TRPV1 N-arachidonoylserotonin reverses the behavioral despair induced by stress in rats and modulates the HPA-axis. Pharmacol. Res. 87, 151–159 (2014).
Bashashati, M. et al. Targeting fatty acid amide hydrolase and transient receptor potential vanilloid-1 simultaneously to modulate colonic motility and visceral sensation in the mouse: A pharmacological intervention with N-arachidonoyl-serotonin (AA-5-HT). Neurogastroenterol. Motil. https://doi.org/10.1111/nmo.13148 (2017).
Maione, S. et al. Piperazinyl carbamate fatty acid amide hydrolase inhibitors and transient receptor potential channel modulators as “dual-target” analgesics. Pharmacol. Res. 76, 98–105 (2013).
Malek, N. et al. A multi-target approach for pain treatment: dual inhibition of fatty acid amide hydrolase and TRPV1 in a rat model of osteoarthritis. Pain 156, 890–903 (2015).
Marsch, R. et al. Reduced anxiety, conditioned fear, and hippocampal long-term potentiation in transient receptor potential vanilloid type 1 receptor-deficient mice. J. Neurosci. 27, 832–839 (2007).
Abdelhamid, R. E. et al. Depressive behavior in the forced swim test can be induced by TRPV1 receptor activity and is dependent on NMDA receptors. Pharmacol. Res. 79, 21–27 (2014).
Alhouayek, M. & Muccioli, G. G. COX-2-derived endocannabinoid metabolites as novel inflammatory mediators. Trends Pharmacol. Sci. 35, 284–292 (2014).
Ligresti, A. et al. Prostamide F2α receptor antagonism combined with inhibition of FAAH may block the pro-inflammatory mediators formed following selective FAAH inhibition. Br. J. Pharmacol. 171, 1408–1419 (2014).
Fowler, C. J. et al. Inhibitory properties of ibuprofen and its amide analogues towards the hydrolysis and cyclooxygenation of the endocannabinoid anandamide. J. Enzyme Inhib. Med. Chem. 28, 172–182 (2013).
Cipriano, M. et al. Inhibition of fatty acid amide hydrolase and cyclooxygenase by the N-(3-methylpyridin-2-yl)amide derivatives of flurbiprofen and naproxen. Eur. J. Pharmacol. 720, 383–390 (2013).
Migliore, M. et al. Potent multitarget FAAH-COX inhibitors: Design and structure-activity relationship studies. Eur. J. Med. Chem. 109, 216–237 (2016).
Duggan, K. C. et al. (R)-Profens are substrate-selective inhibitors of endocannabinoid oxygenation by COX-2. Nat. Chem. Biol. 7, 803–809 (2011).
Bishay, P. et al. R-Flurbiprofen reduces neuropathic pain in rodents by restoring endogenous cannabinoids. PLOS ONE 5, e10628 (2010).
Gouveia-Figueira, S. et al. Characterisation of (R)-2-(2-Fluorobiphenyl-4-yl)-N-(3-Methylpyridin-2-yl)Propanamide as a dual fatty acid amide hydrolase: Cyclooxygenase inhibitor. PLOS ONE 10, e0139212 (2015).
Chicca, A. et al. Functionalization of β-caryophyllene generates novel polypharmacology in the endocannabinoid system. ACS Chem. Biol. 9, 1499–1507 (2014).
Sasso, O. et al. Multitarget fatty acid amide hydrolase/cyclooxygenase blockade suppresses intestinal inflammation and protects against nonsteroidal anti-inflammatory drug-dependent gastrointestinal damage. FASEB J. 29, 2616–2627 (2015).
Ligresti, A. et al. Targeting pain and inflammation by combining two therapeutic modalities in a single molecule: inhibition of FAAH and antagonism of multiple prostanoid receptors. Proc. Bioactive Lipids Conf. (2015).
Pérez-Fernández, R. et al. Discovery of potent dual PPARα agonists/CB1 ligands. ACS Med. Chem. Lett. 2, 793–797 (2011).
Long, J. Z. et al. Dual blockade of FAAH and MAGL identifies behavioral processes regulated by endocannabinoid crosstalk in vivo. Proc. Natl Acad. Sci. USA 106, 20270–20275 (2009).
Ramesh, D. et al. Dual inhibition of endocannabinoid catabolic enzymes produces enhanced antiwithdrawal effects in morphine-dependent mice. Neuropsychopharmacology 38, 1039–1049 (2013).
Sakin, Y. S. et al. The effect of FAAH, MAGL, and dual FAAH/MAGL inhibition on inflammatory and colorectal distension-induced visceral pain models in rodents. Neurogastroenterol. Motil 27, 936–944 (2015).
Parker, L. A. et al. A comparison of novel, selective fatty acid amide hydrolase (FAAH), monoacyglycerol lipase (MAGL) or dual FAAH/MAGL inhibitors to suppress acute and anticipatory nausea in rat models. Psychopharmacology 233, 2265–2275 (2016).
Yesilyurt, O. et al. Systemic and spinal administration of FAAH, MAGL inhibitors and dual FAAH/MAGL inhibitors produce antipruritic effect in mice. Arch. Dermatol. Res. 308, 335–345 (2016).
Novotna, A. et al. A randomized, double-blind, placebo-controlled, parallel-group, enriched-design study of nabiximols* (Sativexâ), as add-on therapy, in subjects with refractory spasticity caused by multiple sclerosis. Eur. J. Neurol. 18, 1122–1131 (2011).
Comelli, F. et al. Antihyperalgesic effect of a Cannabis sativa extract in a rat model of neuropathic pain: mechanisms involved. Phytother. Res. 22, 1017–1024 (2008).
Marcu, J. P. et al. Cannabidiol enhances the inhibitory effects of Δ9-tetrahydrocannabinol on human glioblastoma cell proliferation and survival. Mol. Cancer Ther. 9, 180–189 (2010).
Valdeolivas, S. et al. Sativex-like combination of phytocannabinoids is neuroprotective in malonate-lesioned rats, an inflammatory model of Huntington's disease: role of CB1 and CB2 receptors. ACS Chem. Neurosci. 3, 400–406 (2012).
Johnson, J. R. et al. Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J. Pain Symptom Manage. 39, 167–179 (2010).
Lichtman, A. H. et al. Results of a double-blind, randomized, placebo-controlled study of nabiximols oromucosal spray as a adjunctive therapy in advanced cancer patients with chronic uncontrolled pain. J. Pain Symptom. Manage. 55, 179–188 (2018).
López-Sendón Moreno, J. L. et al. A double-blind, randomized, cross-over, placebo-controlled, pilot trial with Sativex in Huntington's disease. J. Neurol. 263, 1390–1400 (2016).
Baker, D. et al. Cannabinoids control spasticity and tremor in a multiple sclerosis model. Nature 404, 84–87 (2000).
Schubart, C. D. et al. Cannabis with high cannabidiol content is associated with fewer psychotic experiences. Schizophr. Res. 130, 216–221 (2011).
Englund, A. et al. Cannabidiol inhibits THC-elicited paranoid symptoms and hippocampal-dependent memory impairment. J. Psychopharmacol. 27, 19–27 (2013).
Gomes, F. V., Del Bel, E. A. & Guimarães, F. S. Cannabidiol attenuates catalepsy induced by distinct pharmacological mechanisms via 5-HT1A receptor activation in mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 46, 43–47 (2013).
Hilliard, A. et al. Evaluation of the effects of Sativex (THC BDS: CBD BDS) on inhibition of spasticity in a chronic relapsing experimental allergic autoimmune encephalomyelitis: a model of multiple sclerosis. ISRN Neurol. 2012, 802649 (2012).
McPartland, J. M. et al. Are cannabidiol and Δ9-tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review. Br. J. Pharmacol. 172, 737–753 (2015).
Wargent, E. T. et al. The cannabinoid Δ9-tetrahydrocannabivarin (THCV) ameliorates insulin sensitivity in two mouse models of obesity. Nutr. Diabetes 3, e68 (2013).
Silvestri, C. et al. Two non-psychoactive cannabinoids reduce intracellular lipid levels and inhibit hepatosteatosis. J. Hepatol. 62, 1382–1390 (2015).
Jadoon, K. A. et al. Efficacy and safety of cannabidiol and tetrahydrocannabivarin on glycemic and lipid parameters in patients with type 2 diabetes: a randomized, double-blind,placebo-controlled, parallel group pilot study. Diabetes Care 39, 1777–1786 (2016).
Leweke, F. et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry 2, e94 (2012).
McGuire, P. et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am. J. Psychiatry 175, 225–231 (2017).
Devinsky, O. et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N. Engl. J. Med. 376, 2011–2020 (2017).
Katona, I. Cannabis and endocannabinoid signaling in epilepsy. Handb. Exp. Pharmacol. 231, 285–316 (2015).
Turner, S. E. et al. Molecular pharmacology of phytocannabinoids. Prog. Chem. Org. Nat. Prod. 103, 61–101 (2017).
Brodie, J. S., Di Marzo, V. & Guy, G. W. Polypharmacology shakes hands with complex aetiopathology. Trends Pharmacol. Sci. 36, 802–821 (2015).
Di Marzo, V. & Piscitelli, F. The endocannabinoid system and its modulation by phytocannabinoids. Neurotherapeutics 12, 692–698 (2015).
Pistis, M. & O'Sullivan, S. E. The role of nuclear hormone receptors in cannabinoid function. Adv. Pharmacol. 80, 291–328 (2017).
Paronis, C. A. et al. Δ9-Tetrahydrocannabinol acts as a partial agonist/antagonist in mice. Behav. Pharmacol. 23, 802–805 (2012).
Wiley, J. L., Marusich, J. A. & Huffman, J. W. Moving around the molecule: relationship between chemical structure and in vivo activity of synthetic cannabinoids. Life Sci. 97, 55–63 (2014).
Atwood, B. K. et al. JWH018, a common constituent of 'Spice' herbal blends, is a potent and efficacious cannabinoid CB receptor agonist. Br. J. Pharmacol. 160, 585–593 (2010).
Maccarrone, M., Guzmán, M., Mackie, K., Doherty, P. & Harkany, T. Programming of neural cells by (endo)cannabinoids: from physiological rules to emerging therapies. Nat. Rev. Neurosci. 15, 786–801 (2014).
Pacher, P., Steffens, S., Haskó, G., Schindler, T. H. & Kunos, G. Cardiovascular effects of marijuana and synthetic cannabinoids: the good, the bad, and the ugly. Nat. Rev. Cardiol. 15, 151–166 (2018).
Armenian, P. et al. Intoxication from the novel synthetic cannabinoids AB-PINACA and ADB-PINACA: A case series and review of the literature. Neuropharmacology 134, 82–91 (2017).
Debruyne, D. & Le Boisselier, R. Emerging drugs of abuse: current perspectives on synthetic cannabinoids. Subst. Abuse Rehabil. 6, 113–129 (2015).
Paul, A. B. M. et al. Teens and spice: a review of adolescent fatalities associated with synthetic cannabinoid use. J. Forens. Sci. 63, 1321–1324 (2017).
Hruba, L. & McMahon, L. R. Apparent affinity estimates and reversal of the effects of synthetic cannabinoids AM-2201, CP-47,497, JWH-122, and JWH-250 by rimonabant in Rhesus Monkeys. J. Pharmacol. Exp. Ther. 362, 278–286 (2017).
Martinez, K. B., Leone, V. & Chang, E. B. Microbial metabolites in health and disease: Navigating the unknown in search of function. J. Biol. Chem. 292, 8553–8559 (2017).
Rousseaux, C. et al. Lactobacillus acidophilus modulates intestinal pain and induces opioid and cannabinoid receptors. Nat. Med. 13, 35–37 (2007).
Karwad, M. A. et al. The role of CB1 in intestinal permeability and inflammation. FASEB J. 31, 3267–3277 (2017).
Kang, C. et al. Gut microbiota mediates the protective effects of dietary capsaicin against chronic low-grade inflammation and associated obesity induced by high-fat diet. MBio 8, e00470-17 (2017).
Schneeberger, M. et al. Akkermansia muciniphila inversely correlates with the onset of inflammation, altered adipose tissue metabolism and metabolic disorders during obesity in mice. Sci. Rep. 5, 16643 (2015).
Mehrpouya-Bahrami, P. et al. Blockade of CB1 cannabinoid receptor alters gut microbiota and attenuates inflammation and diet-induced obesity. Sci. Rep. 7, 15645 (2017).
Silvestri, C. & Di Marzo, V. The endocannabinoid system in energy homeostasis and the etiopathology of metabolic disorders. Cell Metab. 17, 475–490 (2013).
Wu, D. F. et al. Role of receptor internalization in the agonist-induced desensitization of cannabinoid type 1 receptors. J. Neurochem. 104, 1132–1143 (2008).
Cluny, N. L. et al. Prevention of diet-induced obesity effects on body weight and gut microbiota in mice treated chronically with Δ9-tetrahydrocannabinol. PLOS ONE 10, e0144270 (2015).
Geurts, L. et al. Adipose tissue NAPE-PLD controls fat mass development by altering the browning process and gut microbiota. Nat. Commun. 6, 6495 (2015).
Everard, A. et al. Intestinal epithelial MyD88 is a sensor switching host metabolism towards obesity according to nutritional status. Nat. Commun. 5, 5648 (2014).
Everard, A. et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl Acad. Sci. USA 110, 9066–9071 (2013).
Bruce-Keller, A. J., Salbaum, J. M. & Berthoud, H. R. Harnessing gut microbes for mental health: getting from here to there. Biol. Psychiatry 83, 214–223 (2018).
Guida, F. et al. Antibiotic-induced microbiota perturbation causes gut endocannabinoidome changes, hippocampal neuroglial reorganization and depression in mice. Brain Behav. Immun. 67, 230–245 (2018).
de Lago, E. et al. Effect of repeated systemic administration of selective inhibitors of endocannabinoid inactivation on rat brain endocannabinoid levels. Biochem. Pharmacol. 70, 446–452 (2005).
Cohen, L. J. et al. Commensal bacteria make GPCR ligands that mimic human signalling molecules. Nature 549, 48–53 (2017).
Karwad, M. A. et al. Oleoylethanolamine and palmitoylethanolamine modulate intestinal permeability in vitro via TRPV1 and PPARα. FASEB J. 31, 469–481 (2017).
McPartland, J. M., Guy, G. W. & Di Marzo, V. Care and feeding of the endocannabinoid system: a systematic review of potential clinical interventions that upregulate the endocannabinoid system. PLOS ONE 9, e89566 (2014).
Ferreira, R. C. M. et al. The involvement of the endocannabinoid system in the peripheral antinociceptive action of ketamine. J. Pain 19, 487–495 (2017).
Petrosino, S. & Di Marzo, V. The pharmacology of palmitoylethanolamide and first data on the therapeutic efficacy of some of its new formulations. Br. J. Pharmacol. 174, 1349–1365 (2017).
Gonsiorek, W. et al. Endocannabinoid 2-arachidonyl glycerol is a full agonist through human type 2 cannabinoid receptor: antagonism by anandamide. Mol. Pharmacol. 57, 1045–1050 (2000).
Iannotti, F. A. et al. The endocannabinoid 2-AG controls skeletal muscle cell differentiation via CB1 receptor-dependent inhibition of Kv7 channels. Proc. Natl Acad. Sci. USA 111, E2472–E2481 (2014).
Dhopeshwarkar, A. et al. Two Janus cannabinoids that are both CB2 agonists and CB1 antagonists. J. Pharmacol. Exp. Ther. 360, 300–311 (2017).
Bauer, M. et al. Identification and quantification of a new family of peptide endocannabinoids (Pepcans) showing negative allosteric modulation at CB1 receptors. J. Biol. Chem. 287, 36944–36967 (2012).
Petrucci, V. et al. Pepcan-12 (RVD-hemopressin) is a CB2 receptor positive allosteric modulator constitutively secreted by adrenals and in liver upon tissue damage. Sci. Rep. 7, 9560 (2017).
Vallée, M. et al. Pregnenolone can protect the brain from cannabis intoxication. Science 343, 94–98 (2014).
Busquets-Garcia, A. et al. Pregnenolone blocks cannabinoid-induced acute psychotic-like states in mice. Mol. Psychiatry 22, 1594–1603 (2017).
Liang, Y. et al. Identification and pharmacological characterization of the prostaglandin FP receptor and FP receptor variant complexes. Br. J. Pharmacol. 154, 1079–1093 (2008).
Haj-Dahmane, S. et al. Fatty-acid-binding protein 5 controls retrograde endocannabinoid signaling at central glutamate synapses. Proc. Natl Acad. Sci. USA 115, 3482–3487 (2018).
Oddi, S. et al. Molecular identification of albumin and Hsp70 as cytosolic anandamide-binding proteins. Chem. Biol. 16, 624–632 (2009).
Herman, T. S. et al. Superiority of nabilone over prochlorperazine as an antiemetic in patients receiving cancer chemotherapy. N. Engl. J. Med. 300, 1295–1297 (1979).
Topol, E. J. et al. Rimonabant for prevention of cardiovascular events (CRESCENDO): a randomised, multicentre, placebo-controlled trial. Lancet 376, 517–523 (2010).
Ostenfeld, T. et al. A randomized, controlled study to investigate the analgesic efficacy of single doses of the cannabinoid receptor-2 agonist GW842166, ibuprofen or placebo in patients with acute pain following third molar tooth extraction. Clin. J. Pain 27, 668–676 (2011).
Odan, M. et al. Discovery of S-777469: an orally available CB2 agonist as an antipruritic agent. Bioorg. Med. Chem. Lett. 22, 2803–2806 (2012).
Tepper, M. A., Zurier, R. B. & Burstein, S. H. Ultrapure ajulemic acid has improved CB2 selectivity with reduced CB1 activity. Bioorg. Med. Chem. 22, 3245–3251 (2014).
Bird Rock Bio. Press release on namacizumab. Birdrock Bio http://www.birdrockbio.com/our-pipeline/namacizumab/
Dziadulewicz, E. K. et al. Naphthalen-1-yl-(4-pentyloxynaphthalen-1-yl)methanone: a potent, orally bioavailable human CB1/CB2 dual agonist with antihyperalgesic properties and restricted central nervous system penetration. J. Med. Chem. 50, 3851–3856 (2007).
Neomed. Press release on NEO1940. Neomed neomed.ca/en/projects/neo1940
Abide Therapeutics. Press release on ABX-1431. Abide Therapeutics http://abidetx.com/news/abide-therapeutics-presents-positive-data-from-a-phase-1b-study-of-abx-1431-in-tourette-syndrome-at-the-american-academy-of-neurology-70th-annual-meeting/
Meiri, E. et al. Efficacy of dronabinol alone and in combination with ondansetron versus ondansetron alone for delayed chemotherapy-induced nausea and vomiting. Curr. Med. Res. Opin 23, 533–543 (2007).
Thiele, E. A. et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 391, 1085–1096 (2018).
Gentry, C., Andersson, D. A. & Bevan, S. TRPA1 mediates the hypothermic action of acetaminophen. Sci. Rep. 5, 12771 (2015).
Maione, S. et al. Effects of metabolites of the analgesic agent dipyrone (metamizol) on rostral ventromedial medulla cell activity in mice. Eur. J. Pharmacol. 748, 115–122 (2015).
Qin, N. et al. TRPV2 is activated by cannabidiol and mediates CGRP release in cultured rat dorsal root ganglion neurons. J. Neurosci. 28, 6231–6238 (2008).
De Petrocellis, L. et al. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br. J. Pharmacol. 163, 1479–1494 (2011).
De Petrocellis, L. et al. Cannabinoid actions at TRPV channels: effects on TRPV3 and TRPV4 and their potential relevance to gastrointestinal inflammation. Acta Physiol. 204, 255–266 (2012).
Laprairie, R. B. et al. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br. J. Pharmacol. 172, 4790–4805 (2015).
Laun, A. S. & Song, Z. H. GPR3 and GPR6, novel molecular targets for cannabidiol. Biochem. Biophys. Res. Commun. 490, 17–21 (2017).
Maione, S. et al. Non-psychoactive cannabinoids modulate the descending pathway of antinociception in anaesthetized rats through several mechanisms of action. Br. J. Pharmacol. 162, 584–596 (2011).
Nadal, X. et al. Tetrahydrocannabinolic acid is a potent PPARγ agonist with neuroprotective activity. Br. J. Pharmacol. 174, 4263–4276 (2017).
Bolognini, D. et al. Cannabidiolic acid prevents vomiting in Suncus murinus and nausea-induced behaviour in rats by enhancing 5-HT1A receptor activation. Br. J. Pharmacol. 168, 1456–1470 (2013).
Cascio, M. G. et al. The phytocannabinoid, Δ9-tetrahydrocannabivarin, can act through 5-HT1A receptors to produce antipsychotic effects. Br. J. Pharmacol. 172, 1305–1318 (2015).
Rock, E. M. et al. Interaction between non-psychotropic cannabinoids in marihuana: effect of cannabigerol (CBG) on the anti-nausea or anti-emetic effects of cannabidiol (CBD) in rats and shrews. Psychopharmacology 215, 505–512 (2011).
Acknowledgements
The author dedicates this article to R. Mechoulam, the father of endocannabinoid research, on the occasion of his 88th birthday.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
V.D. acts as a consultant for, and is a recipient of research grants from, GW Pharmaceuticals, UK.
Related links
Rights and permissions
About this article
Cite this article
Di Marzo, V. New approaches and challenges to targeting the endocannabinoid system. Nat Rev Drug Discov 17, 623–639 (2018). https://doi.org/10.1038/nrd.2018.115
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd.2018.115
This article is cited by
-
Research progress in the management of vascular disease with cannabidiol: a review
Journal of Cardiothoracic Surgery (2024)
-
The intervention of cannabinoid receptor in chronic and acute kidney disease animal models: a systematic review and meta-analysis
Diabetology & Metabolic Syndrome (2024)
-
Association between the FAAH C385A variant (rs324420) and obesity-related traits: a systematic review
International Journal of Obesity (2024)
-
AM1172 (a hydrolysis-resistant endocannabinoid analog that inhibits anandamide cellular uptake) reduces the viability of the various melanoma cells, but it exerts significant cytotoxic effects on healthy cells: an in vitro study based on isobolographic analysis
Pharmacological Reports (2024)
-
The diet rapidly and differentially affects the gut microbiota and host lipid mediators in a healthy population
Microbiome (2023)