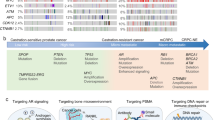
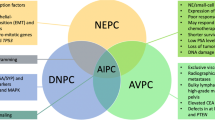
Key Points
-
Castration-resistant prostate cancer (CRPC) is associated with a poor prognosis and poses considerable therapeutic challenges.
-
Recent genetic and technological advances have provided insights into prostate cancer biology and enabled the identification of novel drug targets and potent molecularly targeted therapeutics for the disease.
-
Promising targets in CRPC include the androgen receptor and its variants, key signalling pathways such as phosphoinositide 3-kinase (PI3K)–AKT and WNT signalling, and DNA repair defects.
-
The therapeutic landscape of CRPC is evolving, with an increased focus on research into tumour heterogeneity, immuno-oncology, minimally invasive circulating tissue biomarkers, and modern clinical trial designs.
-
The use of state-of-the-art, high-throughput, genomic platforms enabling patient stratification will permit optimization of the development of current and future drugs for CRPC.
Abstract
Castration-resistant prostate cancer (CRPC) is associated with a poor prognosis and poses considerable therapeutic challenges. Recent genetic and technological advances have provided insights into prostate cancer biology and have enabled the identification of novel drug targets and potent molecularly targeted therapeutics for this disease. In this article, we review recent advances in prostate cancer target identification for drug discovery and discuss their promise and associated challenges. We review the evolving therapeutic landscape of CRPC and discuss issues associated with precision medicine as well as challenges encountered with immunotherapy for this disease. Finally, we envision the future management of CRPC, highlighting the use of circulating biomarkers and modern clinical trial designs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Siegel, R., Naishadham, D. & Jemal, A. Cancer statistics, 2013. CA Cancer J. Clin. 63, 11–30 (2013).
Prostate Cancer Trialists Collaborative Group. Maximum androgen blockade in advanced prostate cancer: an overview of 22 randomised trials with 3283 deaths in 5710 patients. Prostate Cancer Trialists' Collaborative Group. Lancet 346, 265–269 (1995).
Taylor, B. S. et al. Integrative genomic profiling of human prostate cancer. Cancer Cell 18, 11–22 (2010).
Yap, T. A., Gerlinger, M., Futreal, P. A., Pusztai, L. & Swanton, C. Intratumor heterogeneity: seeing the wood for the trees. Sci. Transl Med. 4, 127ps110 (2012).
Yap, T. A., Zivi, A., Omlin, A. & de Bono, J. S. The changing therapeutic landscape of castration-resistant prostate cancer. Nat. Rev. Clin. Oncol. 8, 597–610 (2011).
Berger, M. F. et al. The genomic complexity of primary human prostate cancer. Nature 470, 214–220 (2011). An important paper demonstrating the genomic complexity of prostate cancer.
Baca, S. C. et al. Punctuated evolution of prostate cancer genomes. Cell 153, 666–677 (2013).
Kumar, A. et al. Exome sequencing identifies a spectrum of mutation frequencies in advanced and lethal prostate cancers. Proc. Natl Acad. Sci. USA 108, 17087–17092 (2011).
Grasso, C. S. et al. The mutational landscape of lethal castration-resistant prostate cancer. Nature 487, 239–243 (2012). An important paper describing the mutational landscape of CRPC.
Barbieri, C. E. et al. Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nat. Genet. 44, 685–689 (2012).
Ozsolak, F. & Milos, P. M. RNA sequencing: advances, challenges and opportunities. Nat. Rev. Genet. 12, 87–98 (2011).
Sreekumar, A. et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 457, 910–914 (2009).
Tucker, T., Marra, M. & Friedman, J. M. Massively parallel sequencing: the next big thing in genetic medicine. Am. J. Hum. Genet. 85, 142–154 (2009).
Stratton, M. R. Exploring the genomes of cancer cells: progress and promise. Science 331, 1553–1558 (2011).
Mu, P. et al. TP53 and RB1 alterations promote reprogramming and antiandrogen resistance in advanced prostate cancer [abstract]. Cancer Res. 75 (Suppl.), LB-056 (2015).
Zhao, J., Zhang, Z., Liao, Y. & Du, W. Mutation of the retinoblastoma tumor suppressor gene sensitizes cancers to mitotic inhibitor induced cell death. Am. J. Cancer Res. 4, 42–52 (2014).
Robinson, D. et al. Integrative clinical genomics of advanced prostate cancer. Cell 161, 1215–1228 (2015). An important study of integrative clinical genomics of advanced prostate cancer.
Robbins, C. M. et al. Copy number and targeted mutational analysis reveals novel somatic events in metastatic prostate tumors. Genome Res. 21, 47–55 (2011).
Beltran, H. et al. Targeted next-generation sequencing of advanced prostate cancer identifies potential therapeutic targets and disease heterogeneity. Eur. Urol. 63, 920–926 (2013).
Fairley, J. A., Gilmour, K. & Walsh, K. Making the most of pathological specimens: molecular diagnosis in formalin-fixed, paraffin embedded tissue. Curr. Drug Targets 13, 1475–1487 (2012).
Holcomb, I. N. et al. Genomic alterations indicate tumor origin and varied metastatic potential of disseminated cells from prostate cancer patients. Cancer Res. 68, 5599–5608 (2008).
Ryan, C. J. & Tindall, D. J. Androgen receptor rediscovered: the new biology and targeting the androgen receptor therapeutically. J. Clin. Oncol. 29, 3651–3658 (2011).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01385293 (2015).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01741753 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01485861 (2015).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01884285 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01458067 (2016).
Carver, B. S. et al. Reciprocal feedback regulation of PI3K and androgen receptor signaling in PTEN-deficient prostate cancer. Cancer Cell 19, 575–586 (2011).
Yap, T. et al. Final results of a translational phase l study assessing a QOD schedule of the potent AKT inhibitor MK-2206 incorporating predictive, pharmacodynamic (PD), and functional imaging biomarkers [abstract]. J. Clin. Oncol. 29 (Suppl.) 3001 (2011).
Tomlins, S. A. et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310, 644–648 (2005). An important study identifying TMPRSS2 and ETS genes in prostate cancer.
Holzbeierlein, J. et al. Gene expression analysis of human prostate carcinoma during hormonal therapy identifies androgen-responsive genes and mechanisms of therapy resistance. Am. J. Pathol. 164, 217–227 (2004).
Scher, H. I. & Sawyers, C. L. Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J. Clin. Oncol. 23, 8253–8261 (2005).
Stanbrough, M. et al. Increased expression of genes converting adrenal androgens to testosterone in androgen-independent prostate cancer. Cancer Res. 66, 2815–2825 (2006).
Yu, X. et al. Foxa1 and Foxa2 interact with the androgen receptor to regulate prostate and epididymal genes differentially. Ann. NY Acad. Sci. 1061, 77–93 (2005).
Robinson, J. L. et al. Elevated levels of FOXA1 facilitate androgen receptor chromatin binding resulting in a CRPC-like phenotype. Oncogene (2013).
Hornberg, E. et al. Expression of androgen receptor splice variants in prostate cancer bone metastases is associated with castration-resistance and short survival. PLoS ONE 6, e19059 (2011).
Hu, R., Isaacs, W. B. & Luo, J. A snapshot of the expression signature of androgen receptor splicing variants and their distinctive transcriptional activities. Prostate 71, 1656–1667 (2011).
Antonarakis, E. S. et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N. Engl. J. Med. 371, 1028–1038 (2014).
Antonarakis, E. S., Nakazawa, M. & Luo, J. Resistance to androgen-pathway drugs in prostate cancer. N. Engl. J. Med. 371, 2234 (2014).
Antonarakis, E. S. et al. Androgen receptor splice variant 7 and efficacy of taxane chemotherapy in patients with metastatic castration-resistant prostate cancer. JAMA Oncol. 1, 582–591 (2015).
Onstenk, W. et al. Efficacy of cabazitaxel in castration-resistant prostate cancer is independent of the presence of AR-V7 in circulating tumor cells. Eur. Urol. 68, 939–945 (2015).
Ravindranathan, P. et al. Peptidomimetic targeting of critical androgen receptor-coregulator interactions in prostate cancer. Nat. Commun. 4, 1923 (2013).
Crea, F. et al. Identification of a long non-coding RNA as a novel biomarker and potential therapeutic target for metastatic prostate cancer. Oncotarget 5, 764–774 (2014).
Du, Z. et al. Integrative genomic analyses reveal clinically relevant long noncoding RNAs in human cancer. Nat. Struct. Mol. Biol. 20, 908–913 (2013).
Prensner, J. R. et al. RNA biomarkers associated with metastatic progression in prostate cancer: a multi-institutional high-throughput analysis of SChLAP1. Lancet Oncol. 15, 1469–1480 (2014).
Palanisamy, N. et al. Rearrangements of the RAF kinase pathway in prostate cancer, gastric cancer and melanoma. Nat. Med. 16, 793–798 (2010).
Ren, S. et al. RNA-seq analysis of prostate cancer in the Chinese population identifies recurrent gene fusions, cancer-associated long noncoding RNAs and aberrant alternative splicings. Cell Res. 22, 806–821 (2012).
Burkhardt, L. et al. CHD1 is a 5q21 tumor suppressor required for ERG rearrangement in prostate cancer. Cancer Res. 73, 2795–2805 (2013).
Liu, W. et al. Identification of novel CHD1-associated collaborative alterations of genomic structure and functional assessment of CHD1 in prostate cancer. Oncogene 31, 3939–3948 (2012).
Xu, K. et al. EZH2 oncogenic activity in castration-resistant prostate cancer cells is Polycomb-independent. Science 338, 1465–1469 (2012).
Fellmann, C. & Lowe, S. W. Stable RNA interference rules for silencing. Nat. Cell Biol. 16, 10–18 (2014).
Yegnasubramanian, V. in State of the Science Report: Highlights from the 20th Annual PCF Scientific Retreat (Prostate Cancer Foundation, 2013).
Plass, C. et al. Mutations in regulators of the epigenome and their connections to global chromatin patterns in cancer. Nat. Rev. Genet. 14, 765–780 (2013).
Duan, Z. et al. Developmental and androgenic regulation of chromatin regulators EZH2 and ANCCA/ATAD2 in the prostate via MLL histone methylase complex. Prostate 73, 455–466 (2013).
Asangani, I. A. et al. Therapeutic targeting of BET bromodomain proteins in castration-resistant prostate cancer. Nature 510, 278–282 (2014).
Gao, L. et al. Androgen receptor promotes ligand-independent prostate cancer progression through c-Myc upregulation. PloS ONE 8, e63563 (2013).
Attard, G., Ang, J. E., Olmos, D. & de Bono, J. S. Dissecting prostate carcinogenesis through ETS gene rearrangement studies: implications for anticancer drug development. J. Clin. Pathol. 61, 891–896 (2008).
Attard, G. et al. Duplication of the fusion of TMPRSS2 to ERG sequences identifies fatal human prostate cancer. Oncogene 27, 253–263 (2008).
Rhodes, D. R. et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6, 1–6 (2004).
Tomlins, S. A. et al. The role of SPINK1 in ETS rearrangement-negative prostate cancers. Cancer Cell 13, 519–528 (2008).
Rhodes, D. R. et al. Oncomine 3.0: genes, pathways, and networks in a collection of 18,000 cancer gene expression profiles. Neoplasia 9, 166–180 (2007).
Kaushik, A. K. et al. Metabolomic profiling identifies biochemical pathways associated with castration-resistant prostate cancer. J. Proteome Res. 13, 1088–1100 (2014).
Belanger, A., Pelletier, G., Labrie, F., Barbier, O. & Chouinard, S. Inactivation of androgens by UDP-glucuronosyltransferase enzymes in humans. Trends Endocrinol. Metab. 14, 473–479 (2003).
Shafi, A. A., Yen, A. E. & Weigel, N. L. Androgen receptors in hormone-dependent and castration-resistant prostate cancer. Pharmacol. Ther. 140, 223–238 (2013).
Ferraldeschi, R., Welti, J., Luo, J., Attard, G. & de Bono, J. S. Targeting the androgen receptor pathway in castration-resistant prostate cancer: progresses and prospects. Oncogene 34, 1745–1757 (2015).
Dar, J. A. et al. N-terminal domain of the androgen receptor contains a region that can promote cytoplasmic localization. J. Steroid Biochem. Mol. Biol. 139, 16–24 (2014).
Suzuki, H. et al. Codon 877 mutation in the androgen receptor gene in advanced prostate cancer: relation to antiandrogen withdrawal syndrome. Prostate 29, 153–158 (1996).
Hara, T., Kouno, J., Nakamura, K., Kusaka, M. & Yamaoka, M. Possible role of adaptive mutation in resistance to antiandrogen in prostate cancer cells. Prostate 65, 268–275 (2005).
van de Wijngaart, D. J. et al. Systematic structure–function analysis of androgen receptor Leu701 mutants explains the properties of the prostate cancer mutant L701H. J. Biol. Chem. 285, 5097–5105 (2010).
Thompson, J., Saatcioglu, F., Janne, O. A. & Palvimo, J. J. Disrupted amino- and carboxyl-terminal interactions of the androgen receptor are linked to androgen insensitivity. Mol. Endocrinol. 15, 923–935 (2001).
Sack, J. S. et al. Crystallographic structures of the ligand-binding domains of the androgen receptor and its T877A mutant complexed with the natural agonist dihydrotestosterone. Proc. Natl Acad. Sci. USA 98, 4904–4909 (2001).
Haile, S. & Sadar, M. D. Androgen receptor and its splice variants in prostate cancer. Cell. Mol. Life Sci. 68, 3971–3981 (2011).
Dehm, S. M., Schmidt, L. J., Heemers, H. V., Vessella, R. L. & Tindall, D. J. Splicing of a novel androgen receptor exon generates a constitutively active androgen receptor that mediates prostate cancer therapy resistance. Cancer Res. 68, 5469–5477 (2008).
Li, Y. et al. Androgen receptor splice variants mediate enzalutamide resistance in castration-resistant prostate cancer cell lines. Cancer Res. 73, 483–489 (2013).
Debes, J. D. & Tindall, D. J. Mechanisms of androgen-refractory prostate cancer. N. Engl. J. Med. 351, 1488–1490 (2004).
Feldman, B. J. & Feldman, D. The development of androgen-independent prostate cancer. Nat. Rev. Cancer 1, 34–45 (2001).
Mateo, J., Smith, A., Ong, M. & de Bono, J. S. Novel drugs targeting the androgen receptor pathway in prostate cancer. Cancer Metastasis Rev. 33, 567–579 (2014).
Lam, J. S., Leppert, J. T., Vemulapalli, S. N., Shvarts, O. & Belldegrun, A. S. Secondary hormonal therapy for advanced prostate cancer. J. Urol. 175, 27–34 (2006).
Tran, C. et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science 324, 787–790 (2009).
Clegg, N. J. et al. ARN-509: a novel antiandrogen for prostate cancer treatment. Cancer Res. 72, 1494–1503 (2012).
Ferraldeschi, R., Sharifi, N., Auchus, R. J. & Attard, G. Molecular pathways: Inhibiting steroid biosynthesis in prostate cancer. Clin. Cancer Res. 19, 3353–3359 (2013).
Li, Z. et al. Conversion of abiraterone to D4A drives anti-tumour activity in prostate cancer. Nature 523, 347–351 (2015).
Andersen, R. J. et al. Regression of castrate-recurrent prostate cancer by a small-molecule inhibitor of the amino-terminus domain of the androgen receptor. Cancer Cell 17, 535–546 (2010).
Myung, J. K. et al. An androgen receptor N-terminal domain antagonist for treating prostate cancer. J. Clin. Invest. 123, 2948–2960 (2013).
Brand, L. J. et al. EPI-001 is a selective peroxisome proliferator-activated receptor-gamma modulator with inhibitory effects on androgen receptor expression and activity in prostate cancer. Oncotarget 6, 3811–3824 (2015).
Montgomery, R. B. et al. A phase 1/2 open-label study of safety and antitumor activity of EPI-506, a novel AR N-terminal domain inhibitor, in men with metastatic castration-resistant prostate cancer (mCRPC) with progression after enzalutamide or abiraterone. J. Clin. Oncol. 33 (Suppl.), TPS5072 (2015).
Dalal, K. et al. Selectively targeting the DNA-binding domain of the androgen receptor as a prospective therapy for prostate cancer. J. Biol. Chem. 289, 26417–26429 (2014).
Knapp, R. T. et al. BAG-1 diversely affects steroid receptor activity. Biochem. J. 441, 297–303 (2012).
Ferraldeschi, R. et al. In vitro and in vivo antitumor activity of the next generation HSP90 inhibitor, AT13387, in both hormone-sensitive and castration-resistant prostate cancer models. Cancer Res. 73 (Suppl.), 2433 (2013).
Solit, D. B., Scher, H. I. & Rosen, N. Hsp90 as a therapeutic target in prostate cancer. Semin. Oncol. 30, 709–716 (2003).
Solit, D. B. et al. 17-Allylamino-17- demethoxygeldanamycin induces the degradation of androgen receptor and HER-2/neu and inhibits the growth of prostate cancer xenografts. Clin. Cancer Res. 8, 986–993 (2002).
Hieronymus, H. et al. Gene expression signature-based chemical genomic prediction identifies a novel class of HSP90 pathway modulators. Cancer Cell 10, 321–330 (2006).
Ferraldeschi, R. et al. Second-generation HSP90 inhibitor onalespib blocks mRNA splicing of androgen receptor variant 7 in prostate cancer cells. Cancer Res. 76, 2731–2742 (2016).
Pacey, S. et al. A phase I study of the heat shock protein 90 inhibitor alvespimycin (17-DMAG) given intravenously to patients with advanced solid tumors. Clin. Cancer Res. 17, 1561–1570 (2011).
Oh, W. K. et al. Multicenter phase II trial of the heat shock protein 90 inhibitor, retaspimycin hydrochloride (IPI-504), in patients with castration-resistant prostate cancer. Urology 78, 626–630 (2011).
Iyer, G. et al. A phase I trial of docetaxel and pulse-dose 17-allylamino-17-demethoxygeldanamycin in adult patients with solid tumors. Cancer Chemother. Pharmacol. 69, 1089–1097 (2012).
Ferraldeschi, R. et al. A Phase 1/2 study of AT13387, a heat shock protein 90 (Hsp90) inhibitor in combination with abiraterone acetate (AA) and prednisone (P) in patients with castration-resistant prostate cancer (mCRPC) no longer responding to AA. Ann. Oncol. 25 (Suppl. 4), iv255–iv279 (2014).
Libertini, S. J. et al. Evidence for calpain-mediated androgen receptor cleavage as a mechanism for androgen independence. Cancer Res. 67, 9001–9005 (2007).
Yamashita, S. et al. ASC-J9 suppresses castration-resistant prostate cancer growth through degradation of full-length and splice variant androgen receptors. Neoplasia 14, 74–83 (2012).
McClurg, U. L. & Robson, C. N. Deubiquitinating enzymes as oncotargets. Oncotarget 6, 9657–9668 (2015).
McClurg, U. L., Summerscales, E. E., Harle, V. J., Gaughan, L. & Robson, C. N. Deubiquitinating enzyme Usp12 regulates the interaction between the androgen receptor and the Akt pathway. Oncotarget 5, 7081–7092 (2014).
Mostaghel, E. A. et al. Intraprostatic androgens and androgen-regulated gene expression persist after testosterone suppression: therapeutic implications for castration-resistant prostate cancer. Cancer Res. 67, 5033–5041 (2007).
Bohrer, L. R. et al. FOXO1 binds to the TAU5 motif and inhibits constitutively active androgen receptor splice variants. Prostate 73, 1017–1027 (2013).
Dehm, S. M., Regan, K. M., Schmidt, L. J. & Tindall, D. J. Selective role of an NH2-terminal WxxLF motif for aberrant androgen receptor activation in androgen depletion independent prostate cancer cells. Cancer Res. 67, 10067–10077 (2007).
Dehm, S. M. & Tindall, D. J. Alternatively spliced androgen receptor variants. Endocr. Relat. Cancer 18, R183–R196 (2011).
Reid, A. H. et al. Molecular characterisation of ERG, ETV1 and PTEN gene loci identifies patients at low and high risk of death from prostate cancer. Br. J. Cancer 102, 678–684 (2010).
Chen, Z. et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436, 725–730 (2005).
Carver, B. S. et al. Aberrant ERG expression cooperates with loss of PTEN to promote cancer progression in the prostate. Nat. Genet. 41, 619–624 (2009).
Clegg, N. J. et al. MYC cooperates with AKT in prostate tumorigenesis and alters sensitivity to mTOR inhibitors. PLoS ONE 6, e17449 (2011).
Di Mitri, D. et al. Tumour-infiltrating Gr-1+ myeloid cells antagonize senescence in cancer. Nature 515, 134–137 (2014).
Schwartz, S. et al. Feedback suppression of PI3Kα signaling in PTEN-mutated tumors is relieved by selective inhibition of PI3Kβ. Cancer Cell 27, 109–122 (2015).
Rajan, P. et al. Next-generation sequencing of advanced prostate cancer treated with androgen-deprivation therapy. Eur. Urol. 66, 32–39 (2013).
Liu, J. et al. Targeting Wnt-driven cancer through the inhibition of Porcupine by LGK974. Proc. Natl Acad. Sci. USA 110, 20224–20229 (2013).
Waaler, J. et al. A novel tankyrase inhibitor decreases canonical Wnt signaling in colon carcinoma cells and reduces tumor growth in conditional APC mutant mice. Cancer Res. 72, 2822–2832 (2012).
Riffell, J. L., Lord, C. J. & Ashworth, A. Tankyrase-targeted therapeutics: expanding opportunities in the PARP family. Nat. Rev. Drug Discov. 11, 923–936 (2012).
Janku, F. et al. Phase I study of WNT974, a first-in-class Porcupine inhibitor, in advanced solid tumors. Mol. Cancer Ther. 14 (Suppl. 2), C45 (2015).
Beltran, H. DNA mismatch repair in prostate cancer. J. Clin. Oncol. 31, 1782–1784 (2013).
Ceccaldi, R. et al. A unique subset of epithelial ovarian cancers with platinum sensitivity and PARP inhibitor resistance. Cancer Res. 75, 628–634 (2015).
Mateo, J. et al. DNA-repair defects and olaparib in metastatic prostate cancer. N. Engl. J. Med. 373, 1697–1708 (2015). An important report showing the high response rate of patients with CRPC with DNA repair defects to the PARP inhibitor olaparib.
Brenner, J. C. et al. Mechanistic rationale for inhibition of poly(ADP-ribose) polymerase in ETS gene fusion-positive prostate cancer. Cancer Cell 19, 664–678 (2011).
Schiewer, M. J. et al. Dual roles of PARP-1 promote cancer growth and progression. Cancer Discov. 2, 1134–1149 (2012).
Pritchard, C. C. et al. Inherited DNA-repair gene mutations in men with metastatic prostate cancer. N. Engl. J. Med. 6 July 2016 [epub ahead of print].
Van Allen, E. M. et al. Genomic correlates of response to CTLA-4 blockade in metastatic melanoma. Science 350, 207–211 (2015). An excellent study of genomic markers of response to CTLA4 blockade.
Le, D. T. et al. PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 372, 2509–2520 (2015). An important paper demonstrating that MMR status predicts clinical benefit of immune checkpoint blockade with pembrolizumab.
Graff, J. N., Puri, S., Bifulco, C. B., Fox, B. A. & Beer, T. M. Sustained complete response to CTLA-4 blockade in a patient with metastatic, castration-resistant prostate cancer. Cancer Immunol. Res. 2, 399–403 (2014).
Hollenhorst, P. C., McIntosh, L. P. & Graves, B. J. Genomic and biochemical insights into the specificity of ETS transcription factors. Annu. Rev. Biochem. 80, 437–471 (2011).
Yu, J. et al. An integrated network of androgen receptor, polycomb, and TMPRSS2–ERG gene fusions in prostate cancer progression. Cancer Cell 17, 443–454 (2010).
Tomlins, S. A. et al. Role of the TMPRSS2–ERG gene fusion in prostate cancer. Neoplasia 10, 177–188 (2008).
Carver, B. S. et al. ETS rearrangements and prostate cancer initiation. Nature 457, E1; discussion E2–E3 (2009).
Zong, Y. et al. ETS family transcription factors collaborate with alternative signaling pathways to induce carcinoma from adult murine prostate cells. Proc. Natl Acad. Sci. USA 106, 12465–12470 (2009).
Yeh, J. E., Toniolo, P. A. & Frank, D. A. Targeting transcription factors: promising new strategies for cancer therapy. Curr. Opin. Oncol. 25, 652–658 (2013).
Nhili, R. et al. Targeting the DNA-binding activity of the human ERG transcription factor using new heterocyclic dithiophene diamidines. Nucleic Acids Res. 41, 125–138 (2013).
Shao, L. et al. Highly specific targeting of the TMPRSS2/ERG fusion gene using liposomal nanovectors. Clin. Cancer Res. 18, 6648–6657 (2012).
Vainio, P. et al. Arachidonic acid pathway members PLA2G7, HPGD, EPHX2, and CYP4F8 identified as putative novel therapeutic targets in prostate cancer. Am. J. Pathol. 178, 525–536 (2011).
Vainio, P. et al. Phospholipase PLA2G7, associated with aggressive prostate cancer, promotes prostate cancer cell migration and invasion and is inhibited by statins. Oncotarget 2, 1176–1190 (2011).
Iljin, K. et al. High-throughput cell-based screening of 4910 known drugs and drug-like small molecules identifies disulfiram as an inhibitor of prostate cancer cell growth. Clin. Cancer Res. 15, 6070–6078 (2009).
Erkizan, H. V. et al. A small molecule blocking oncogenic protein EWS-FLI1 interaction with RNA helicase A inhibits growth of Ewing's sarcoma. Nat. Med. 15, 750–756 (2009).
Rahim, S. et al. YK-4-279 inhibits ERG and ETV1 mediated prostate cancer cell invasion. PLoS ONE 6, e19343 (2011).
Aytes, A. et al. ETV4 promotes metastasis in response to activation of PI3-kinase and Ras signaling in a mouse model of advanced prostate cancer. Proc. Natl Acad. Sci. USA 110, E3506–E3515 (2013).
Lotan, T. L. et al. ERG gene rearrangements are common in prostatic small cell carcinomas. Mod. Pathol. 24, 820–828 (2011).
Beltran, H. et al. Molecular characterization of neuroendocrine prostate cancer and identification of new drug targets. Cancer Discov. 1, 487–495 (2011).
Mosquera, J. M. et al. Concurrent AURKA and MYCN gene amplifications are harbingers of lethal treatment-related neuroendocrine prostate cancer. Neoplasia 15, 1–10 (2013).
Brockmann, M. et al. Small molecule inhibitors of Aurora-A induce proteasomal degradation of N-Myc in childhood neuroblastoma. Cancer Cell 24, 75–89 (2013).
Tannock, I. F. et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 351, 1502–1512 (2004). Phase III trial demonstrating survival benefit of docetaxel plus prednisolone.
de Bono, J. S. et al. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 364, 1995–2005 (2011). Phase III trial showing survival benefit of abiraterone after chemotherapy.
Ryan, C. J. et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N. Engl. J. Med. 368, 138–148 (2012). Phase III trial demonstrating survival benefit of abiraterone before chemotherapy.
Scher, H. I. et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 367, 1187–1197 (2012). Phase III trial showing survival benefit of enzalutamide.
Beer, T. M. et al. Enzalutamide in men with chemotherapy-naive metastatic prostate cancer (mCRPC): results of phase III PREVAIL study. J. Clin. Oncol. 32, (Suppl. 4), LBA1 (2014).
de Bono, J. S. et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet 376, 1147–1154 (2010). Phase III trial showing survival benefit of cabazitaxel.
Parker, C. et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N. Engl. J. Med. 369, 213–223 (2013). Positive phase III trial of radium-223.
Kantoff, P. W. et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N. Engl. J. Med. 363, 411–422 (2010). Positive phase III trial of sipuleucel-T.
Sweeney, C. J. et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N. Engl. J. Med. 373, 737–746 (2015). An important study showing that administration of docetaxel at the beginning of androgen-deprivation therapy results in longer overall survival.
James, N. D. et al. Docetaxel and/or zoledronic acid for hormone-naïve prostate cancer: first overall survival results from STAMPEDE (NCT00268476). J. Clin. Oncol. 33, (Suppl.) 5001 (2015).
Masson, S. & Bahl, A. Metastatic castrate-resistant prostate cancer: dawn of a new age of management. BJU Int. 110, 1110–1114 (2012).
Lorente, D., Mateo, J., Perez-Lopez, R., de Bono, J. S. & Attard, G. Sequencing of agents in castration-resistant prostate cancer. Lancet Oncol. 16, e279–e292 (2015).
Gerlinger, M. et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366, 883–892 (2012). A seminal paper demonstrating intratumour heterogeneity and branched evolution using multiregion sequencing.
Iyer, G. et al. Genome sequencing identifies a basis for everolimus sensitivity. Science 338, 221 (2012).
McArdle, P. A., McMillan, D. C., Sattar, N., Wallace, A. M. & Underwood, M. A. The relationship between interleukin-6 and C-reactive protein in patients with benign and malignant prostate disease. Br. J. Cancer 91, 1755–1757 (2004).
Davidsson, S. et al. CD4 helper T cells, CD8 cytotoxic T cells, and FOXP3+ regulatory T cells with respect to lethal prostate cancer. Mod. Pathol. 26, 448–455 (2013).
Ness, N. et al. Infiltration of CD8+ lymphocytes is an independent prognostic factor of biochemical failure-free survival in prostate cancer. Prostate 74, 1452–1461 (2014).
Gannon, P. O. et al. Characterization of the intra-prostatic immune cell infiltration in androgen-deprived prostate cancer patients. J. Immunol. Methods 348, 9–17 (2009).
Tse, B. W. et al. From bench to bedside: immunotherapy for prostate cancer. Biomed. Res. Int. 2014, 981434 (2014).
Sheikh, N. A. et al. Sipuleucel-T immune parameters correlate with survival: an analysis of the randomized phase 3 clinical trials in men with castration-resistant prostate cancer. Cancer Immunol. Immunother. 62, 137–147 (2013).
Robert, C. et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 372, 320–330 (2015).
Brahmer, J. et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N. Engl. J. Med. 373, 123–135 (2015).
Borghaei, H. et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N. Engl. J. Med. 373, 1627–1639 (2015).
Wing, K. et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science 322, 271–275 (2008).
Small, E. J. et al. A pilot trial of CTLA-4 blockade with human anti-CTLA-4 in patients with hormone-refractory prostate cancer. Clin. Cancer Res. 13, 1810–1815 (2007).
Slovin, S. F. et al. Ipilimumab alone or in combination with radiotherapy in metastatic castration-resistant prostate cancer: results from an open-label, multicenter phase I/II study. Ann. Oncol. 24, 1813–1821 (2013).
Kwon, E. D. et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 15, 700–712 (2014).
Attard, G. et al. Heterogeneity and clinical significance of ETV1 translocations in human prostate cancer. Br. J. Cancer 99, 314–320 (2008).
Attard, G. & de Bono, J. S. Prostate cancer: PSA as an intermediate end point in clinical trials. Nature Rev. Urol. 6, 473–475 (2009).
Kantoff, P. W. et al. Overall survival analysis of a phase II randomized controlled trial of a Poxviral-based PSA-targeted immunotherapy in metastatic castration-resistant prostate cancer. J. Clin. Oncol. 28, 1099–1105 (2010).
Attard, G. et al. Hormone-sensitive prostate cancer: a case of ETS gene fusion heterogeneity. J. Clin. Pathol. 62, 373–376 (2009).
Yap, T. A., Sandhu, S. K., Workman, P. & de Bono, J. S. Envisioning the future of early anticancer drug development. Nat. Rev. Cancer 10, 514–523 (2010).
Andre, F. et al. Prioritizing targets for precision cancer medicine. Ann. Oncol. 25, 2295–2303 (2014).
Logothetis, C. J. et al. Effect of abiraterone acetate and prednisone compared with placebo and prednisone on pain control and skeletal-related events in patients with metastatic castration-resistant prostate cancer: exploratory analysis of data from the COU-AA-301 randomised trial. Lancet Oncol. 13, 1210–1217 (2012).
Yap, T. A., Lorente, D., Omlin, A., Olmos, D. & de Bono, J. S. Circulating tumor cells: a multifunctional biomarker. Clin. Cancer Res. 20, 2553–2568 (2014).
Danila, D. C., Fleisher, M. & Scher, H. I. Circulating tumor cells as biomarkers in prostate cancer. Clin. Cancer Res. 17, 3903–3912 (2011).
Azad, A. A. et al. Androgen receptor gene aberrations in circulating cell-free DNA: biomarkers of therapeutic resistance in castration-resistant prostate cancer. Clin. Cancer Res. 21, 2315–2324 (2015).
Carreira, S. et al. Tumor clone dynamics in lethal prostate cancer. Sci. Transl Med. 6, 254ra125 (2014).
Romanel, A. et al. Plasma AR and abiraterone-resistant prostate cancer. Sci. Transl Med. 7, 312re310 (2015).
Laxman, B. et al. A first-generation multiplex biomarker analysis of urine for the early detection of prostate cancer. Cancer Res. 68, 645–649 (2008).
Kharaziha, P. et al. Molecular profiling of prostate cancer derived exosomes may reveal a predictive signature for response to docetaxel. Oncotarget 6, 21740–21754 (2015).
Duijvesz, D., Luider, T., Bangma, C. H. & Jenster, G. Exosomes as biomarker treasure chests for prostate cancer. Eur. Urol. 59, 823–831 (2011).
Attard, G. et al. Characterization of ERG, AR and PTEN gene status in circulating tumor cells from patients with castration-resistant prostate cancer. Cancer Res. 69, 2912–2918 (2009).
Gao, D. et al. Organoid cultures derived from patients with advanced prostate cancer. Cell 159, 176–187 (2014).
Lawrence, M. G. et al. A preclinical xenograft model of prostate cancer using human tumors. Nat. Protoc. 8, 836–848 (2013).
Dawson, S. J. et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N. Engl. J. Med. 368, 1199–1209 (2013).
Heitzer, E. et al. Tumor-associated copy number changes in the circulation of patients with prostate cancer identified through whole-genome sequencing. Genome Med. 5, 30 (2013).
Ross, R. W. et al. A whole-blood RNA transcript-based prognostic model in men with castration-resistant prostate cancer: a prospective study. Lancet Oncol. 13, 1105–1113 (2012).
Olmos, D. et al. Prognostic value of blood mRNA expression signatures in castration-resistant prostate cancer: a prospective, two-stage study. Lancet Oncol. 13, 1114–1124 (2012).
Miyamoto, D. T. et al. RNA-Seq of single prostate CTCs implicates noncanonical Wnt signaling in antiandrogen resistance. Science 349, 1351–1356 (2015).
Rodon, J. et al. Challenges in initiating and conducting personalized cancer therapy trials: perspectives from WINTHER, a Worldwide Innovative Network (WIN) Consortium trial. Ann. Oncol. 26, 1791–1798 (2015).
Smith, A. D., Roda, D. & Yap, T. A. Strategies for modern biomarker and drug development in oncology. J. Hematol. Oncol. 7, 70 (2014).
Audeh, M. W. et al. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: a proof-of-concept trial. Lancet 376, 245–251 (2010).
Tutt, A. et al. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and advanced breast cancer: a proof-of-concept trial. Lancet 376, 235–244 (2010).
de Bono, J. S. & Ashworth, A. Translating cancer research into targeted therapeutics. Nature 467, 543–549 (2010).
Shendure, J. & Ji, H. Next-generation DNA sequencing. Nat. Biotechnol. 26, 1135–1145 (2008).
Summerer, D. et al. Targeted high throughput sequencing of a cancer-related exome subset by specific sequence capture with a fully automated microarray platform. Genomics 95, 241–246 (2010).
Tayeh, M. K. et al. Targeted comparative genomic hybridization array for the detection of single- and multiexon gene deletions and duplications. Genet. Med. 11, 232–240 (2009).
Wang, Z., Gerstein, M. & Snyder, M. RNA-Seq: a revolutionary tool for transcriptomics. Nat. Rev. Genet. 10, 57–63 (2009).
Koboldt, D. C., Steinberg, K. M., Larson, D. E., Wilson, R. K. & Mardis, E. R. The next-generation sequencing revolution and its impact on genomics. Cell 155, 27–38 (2013).
De Lellis, L. et al. Analysis of extended genomic rearrangements in oncological research. Ann. Oncol. 18 (Suppl. 6), vi173–vi178 (2007).
Hurd, P. J. & Nelson, C. J. Advantages of next-generation sequencing versus the microarray in epigenetic research. Brief Funct. Genomic Proteomic 8, 174–183 (2009).
Bass, B. L. RNA editing by adenosine deaminases that act on RNA. Annu. Rev. Biochem. 71, 817–846 (2002).
Hansen, K. D., Brenner, S. E. & Dudoit, S. Biases in Illumina transcriptome sequencing caused by random hexamer priming. Nucleic Acids Res. 38, e131 (2010).
Kozarewa, I. et al. Amplification-free Illumina sequencing-library preparation facilitates improved mapping and assembly of (G+C)-biased genomes. Nat. Methods 6, 291–295 (2009).
Dohm, J. C., Lottaz, C., Borodina, T. & Himmelbauer, H. Substantial biases in ultra-short read data sets from high-throughput DNA sequencing. Nucleic Acids Res. 36, e105 (2008).
Liu, D. & Graber, J. H. Quantitative comparison of EST libraries requires compensation for systematic biases in cDNA generation. BMC Bioinformat. 7, 77 (2006).
Ozsolak, F. et al. Direct RNA sequencing. Nature 461, 814–818 (2009).
Bulusu, K. C. et al. canSAR: updated cancer research and drug discovery knowledgebase. Nucleic Acids Res. 42, D1040–D1047 (2014).
Patel, M. N., Halling-Brown, M. D., Tym, J. E., Workman, P. & Al-Lazikani, B. Objective assessment of cancer genes for drug discovery. Nat. Rev. Drug Discov. 12, 35–50 (2013).
Coffey, D. S. & Isaacs, J. T. Prostate tumor biology and cell kinetics — theory. Urology 17, 40–53 (1981).
Sfanos, K. S. et al. Human prostate-infiltrating CD8+ T lymphocytes are oligoclonal and PD-1+. Prostate 69, 1694–1703 (2009).
Mercader, M. et al. T cell infiltration of the prostate induced by androgen withdrawal in patients with prostate cancer. Proc. Natl Acad. Sci. USA 98, 14565–14570 (2001).
Quinn, D. I., Shore, N. D., Egawa, S., Gerritsen, W. R. & Fizazi, K. Immunotherapy for castration-resistant prostate cancer: progress and new paradigms. Urol. Oncol. 33, 245–260 (2015).
Motzer, R. J. et al. Nivolumab for metastatic renal cell carcinoma: results of a randomized phase II trial. J. Clin. Oncol. 33, 1430–1437 (2015).
Gerritsen, W. R. & Sharma, P. Current and emerging treatment options for castration-resistant prostate cancer: a focus on immunotherapy. J. Clin. Immunol. 32, 25–35 (2012).
Podrazil, M. et al. Phase I/II clinical trial of dendritic-cell based immunotherapy (DCVAC/PCa) combined with chemotherapy in patients with metastatic, castration-resistant prostate cancer. Oncotarget 6, 18192–18205 (2015).
Sonpavde, G. et al. Results of a phase I/II clinical trial of BPX-101, a novel drug-activated dendritic cell (DC) vaccine for metastatic castration-resistant prostate cancer (mCRPC). J. Clin. Oncol. 29 (Suppl. 7), 132 (2011).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01322490 (2015).
Noguchi, M. et al. A randomized phase II trial of personalized peptide vaccine plus low dose estramustine phosphate (EMP) versus standard dose EMP in patients with castration resistant prostate cancer. Cancer Immunol. Immunother. 59, 1001–1009 (2010).
Higano, C. et al. A phase III trial of GVAX immunotherapy for prostate cancer versus docetaxel plus prednisone in asymptomatic, castration-resistant prostate cancer (CRPC) [abstract]. Genitourinary Cancers Symposium 2009 http://meetinglibrary.asco.org/content/20543-64 (2009).
Small, E. et al. A phase III trial of GVAX immunotherapy for prostate cancer in combination with docetaxel versus docetaxel plus prednisone in symptomatic, castration-resistant prostate cancer (CRPC) [abstract]. Genitourinary Cancers Symposium 2009 http://meetinglibrary.asco.org/content/20295-64 (2009).
Attard, G. et al. Selective inhibition of CYP17 with abiraterone acetate is highly active in the treatment of castration-resistant prostate cancer. J. Clin. Oncol. 27, 3742–3748 (2009).
Attard, G., Reid, A. H., Dearnaley, D. & De Bono, J. S. New prostate cancer drug: prostate cancer's day in the sun. BMJ 337, a1249 (2008).
Varambally, S. et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 419, 624–629 (2002).
Kong, D. et al. Loss of let-7 up-regulates EZH2 in prostate cancer consistent with the acquisition of cancer stem cell signatures that are attenuated by BR-DIM. PloS ONE 7, e33729 (2012).
Bishop, J. L. et al. PD-L1 is highly expressed in enzalutamide resistant prostate cancer. Oncotarget 6, 234–242 (2015).
Crane, C. A. et al. PI(3) kinase is associated with a mechanism of immunoresistance in breast and prostate cancer. Oncogene 28, 306–312 (2009).
Attard, G., Reid, A. H., Olmos, D. & de Bono, J. S. Antitumor activity with CYP17 blockade indicates that castration-resistant prostate cancer frequently remains hormone driven. Cancer Res. 69, 4937–4940 (2009).
Attard, G. et al. A novel, spontaneously immortalized, human prostate cancer cell line, Bob, offers a unique model for pre-clinical prostate cancer studies. Prostate 69, 1507–1520 (2009).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01251861 (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02121639 (2014).
Ayhan, A., Ertunc, D., Tok, E. C. & Ayhan, A. Expression of the c-Met in advanced epithelial ovarian cancer and its prognostic significance. Int. J. Gynecol. Cancer 15, 618–623 (2005).
Reaper, P. M. et al. Selective killing of ATM- or p53-deficient cancer cells through inhibition of ATR. Nat. Chem. Biol. 7, 428–430 (2011).
Henriquez-Hernandez, L. A. et al. Single nucleotide polymorphisms in DNA repair genes as risk factors associated to prostate cancer progression. BMC Med. Genet. 15, 143 (2014).
Wang, S. et al. Ablation of the oncogenic transcription factor ERG by deubiquitinase inhibition in prostate cancer. Proc. Natl Acad. Sci. USA 111, 4251–4256 (2014).
Fong, P. C. et al. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N. Engl. J. Med. 361, 123–134 (2009).
Sandhu, S. K. et al. Poly (ADP-ribose) polymerase (PARP) inhibitors for the treatment of advanced germline BRCA2 mutant prostate cancer. Ann. Oncol. 24, 1416–1418 (2013).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01682772 (2014).
Ateeq, B. et al. Therapeutic targeting of SPINK1-positive prostate cancer. Sci. Transl Med. 3, 72ra17 (2011).
Rodrigues, L. U. et al. Coordinate loss of MAP3K7 and CHD1 promotes aggressive prostate cancer. Cancer Res. 75, 1021–1034 (2015).
Attard, G. et al. Prostate cancer. 387, 70–78 Lancet (2015).
Huang, S. et al. Recurrent deletion of CHD1 in prostate cancer with relevance to cell invasiveness. Oncogene 31, 4164–4170 (2012).
Bruxvoort, K. J. et al. Inactivation of Apc in the mouse prostate causes prostate carcinoma. Cancer Res. 67, 2490–2496 (2007).
Wyce, A. et al. Inhibition of BET bromodomain proteins as a therapeutic approach in prostate cancer. Oncotarget 4, 2419–2429 (2013).
Rathkopf, D. E. et al. Phase I study of ARN-509, a novel antiandrogen, in the treatment of castration-resistant prostate cancer. J. Clin. Oncol. 31, 3525–3530 (2013).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01946204 (2016).
Smith, M. R. et al. Final analysis of COMET-1: cabozantinib (Cabo) versus prednisone (Pred) in metastatic castration-resistant prostate cancer (mCRPC) patients (pts) previously treated with docetaxel (D) and abiraterone (A) and/or enzalutamide (E). J. Clin. Oncol. 33 (Suppl. 7), 139 (2015).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01574937 (2016).
Chi, K. N. et al. Phase III SYNERGY trial: docetaxel +/− custirsen and overall survival in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) and poor prognosis. J. Clin. Oncol. 33 (Suppl.), 5009 (2015).
Bartkova, J. et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434, 864–870 (2005).
Spisek, R. et al. Phase I/II clinical trials of dendritic cell-based immunotherapy in patients with the biochemical relapse of the prostate cancer. J. Clin. Oncol. 31 (Suppl.), e16002 (2013).
European Medicines Agency. EU Clinical Trials Register https://www.clinicaltrialsregister.eu/ctr-search/search?query=eudract_number:2011-004735-32 (2012).
Shore, N. D. et al. Phase III efficacy and safety trial of a new leuprolide acetate 3.75 mg depot formulation in prostate cancer patients. J. Clin. Oncol. 27 (Suppl.), e16152 (2009).
Dreicer, R. et al. Results from a phase 3, randomized, double-blind, multicenter, placebo-controlled trial of orteronel (TAK-700) plus prednisone in patients with metastatic castration-resistant prostate cancer (mCRPC) that has progressed during or following docetaxel-based therapy (ELM-PC 5 trial). J. Clin. Oncol. 32 (Suppl. 4), 7 (2014).
Bean, J. et al. MET amplification occurs with or without T790M mutations in EGFR mutant lung tumors with acquired resistance to gefitinib or erlotinib. Proc. Natl Acad. Sci. USA 104, 20932–20937 (2007).
Xu, F. et al. Phase I and biodistribution study of recombinant adenovirus vector-mediated herpes simplex virus thymidine kinase gene and ganciclovir administration in patients with head and neck cancer and other malignant tumors. Cancer Gene Ther. 16, 723–730 (2009).
US National Library of Medicine. ClinicalTrials.govhttps://www.clinicaltrials.gov/ct2/show/NCT01436968 (2016).
Araujo, J. C. et al. Docetaxel and dasatinib or placebo in men with metastatic castration-resistant prostate cancer (READY): a randomised, double-blind phase 3 trial. Lancet Oncol. 14, 1307–1316 (2013).
Pili, R. et al. Phase II randomized, double-blind, placebo-controlled study of tasquinimod in men with minimally symptomatic metastatic castrate-resistant prostate cancer. J. Clin. Oncol. 29, 4022–4028 (2011).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01234311 (2015).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01360840 (2015).
Festuccia, C. et al. Ozarelix, a fourth generation GnRH antagonist, induces apoptosis in hormone refractory androgen receptor negative prostate cancer cells modulating expression and activity of death receptors. Prostate 70, 1340–1349 (2010).
Bullrich, F. et al. ATM mutations in B-cell chronic lymphocytic leukemia. Cancer Res. 59, 24–27 (1999).
Tagawa, S. T. et al. Phase II study of lutetium-177-labeled anti-prostate-specific membrane antigen monoclonal antibody J591 for metastatic castration-resistant prostate cancer. Clin. Cancer Res. 19, 5182–5191 (2013).
Burgess, T. et al. Fully human monoclonal antibodies to hepatocyte growth factor with therapeutic potential against hepatocyte growth factor/c-Met-dependent human tumors. Cancer Res. 66, 1721–1729 (2006).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01812746 (2016).
Yu, E. Y. et al. The effect of low-dose GTx-758 on free testerone levels in men with metastatic castration resistant prostate cancer (mCRPC). J. Clin. Oncol. 32 (Suppl. 4), 60 (2014).
de Souza, P. L., Castillo, M. & Myers, C. E. Enhancement of paclitaxel activity against hormone-refractory prostate cancer cells in vitro and in vivo by quinacrine. Br. J. Cancer 75, 1593–1600 (1997).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT00417274 (2013).
Chi, K. N. et al. A phase 2 study of patupilone in patients with metastatic castration-resistant prostate cancer previously treated with docetaxel: Canadian Urologic Oncology Group study P07a. Ann. Oncol. 23, 53–58 (2012).
Denmeade, S. R. et al. Engineering a prostate-specific membrane antigen-activated tumor endothelial cell prodrug for cancer therapy. Sci. Transl Med. 4, 140ra186 (2012).
Carser, J. E. et al. BRCA1 protein expression as a predictor of outcome following chemotherapy in sporadic epithelial ovarian cancer (EOC). J. Clin. Oncol. 27 (Suppl.), 5527 (2009).
Higano, C. S. et al. Phase 1/2 dose-escalation study of a GM-CSF-secreting, allogeneic, cellular immunotherapy for metastatic hormone-refractory prostate cancer. Cancer 113, 975–984 (2008).
Kabbinavar, F. F. et al. An open-label phase II clinical trial of the RXR agonist IRX4204 in taxane-resistant, castration-resistant metastatic prostate cancer (CRPC). J. Clin. Oncol. 32 (Suppl.), 169 (2014).
Hong, D. S. et al. A phase 1 dose escalation, pharmacokinetic, and pharmacodynamic evaluation of eIF-4E antisense oligonucleotide LY2275796 in patients with advanced cancer. Clin. Cancer Res. 17, 6582–6591 (2011).
Liu, G., Chen, Y. H., Dipaola, R., Carducci, M. & Wilding, G. Phase II trial of weekly ixabepilone in men with metastatic castrate-resistant prostate cancer (E3803): a trial of the Eastern Cooperative Oncology Group. Clin. Genitourin Cancer 10, 99–105 (2012).
Antonarakis, E. S. et al. A phase 2 study of KX2-391, an oral inhibitor of Src kinase and tubulin polymerization, in men with bone-metastatic castration-resistant prostate cancer. Cancer Chemother. Pharmacol. 71, 883–892 (2013).
North, S. A., Graham, K., Bodnar, D. & Venner, P. A pilot study of the liposomal MUC1 vaccine BLP25 in prostate specific antigen failures after radical prostatectomy. J. Urol. 176, 91–95 (2006).
Chiorean, E. G. et al. A phase I study of olaratumab, an anti-platelet-derived growth factor receptor alpha (PDGFRα) monoclonal antibody, in patients with advanced solid tumors. Cancer Chemother. Pharmacol. 73, 595–604 (2014).
Chalmers, A. J. Poly(ADP-ribose) polymerase-1 and ionizing radiation: sensor, signaller and therapeutic target. Clin. Oncol. 16, 29–39 (2004).
Fizazi, K. et al. Activity and safety of ODM-201 in patients with progressive metastatic castration-resistant prostate cancer (ARADES): an open-label phase 1 dose-escalation and randomised phase 2 dose expansion trial. Lancet Oncol. 15, 975–985 (2014).
Brose, M. S. et al. Cancer risk estimates for BRCA1 mutation carriers identified in a risk evaluation program. J. Natl Cancer Inst. 94, 1365–1372 (2002).
Hotte, S. J. et al. Phase I trial of OGX-427, a 2′methoxyethyl antisense oligonucleotide (ASO), against heat shock protein 27 (Hsp27): final results. J. Clin. Oncol. 28 (Suppl.), 3077 (2010).
Chambers, A. F., Groom, A. C. & MacDonald, I. C. Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer 2, 563–572 (2002).
Hawkins, R. E. et al. Safety and tolerability of PCK3145, a synthetic peptide derived from prostate secretory protein 94 (PSP94) in metastatic hormone-refractory prostate cancer. Clin. Prostate Cancer 4, 91–99 (2005).
Meulenbeld, H. J. et al. Randomized phase II study of danusertib in patients with metastatic castration-resistant prostate cancer after docetaxel failure. BJU Int. 111, 44–52 (2013).
Anthony, S. P. et al. Pharmacodynamic activity demonstrated in phase I for PLX3397, a selective inhibitor of FMS and Kit. J. Clin. Oncol. 29 (Suppl.), 3093 (2011).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01499043 (2015).
Petrylak, D. P. et al. Prostate-specific membrane antigen antibody drug conjugate (PSMA ADC): a phase I trial in metastatic castration-resistant prostate cancer (mCRPC) previously treated with a taxane. J. Clin. Oncol. 31 (Suppl. 6), 119 (2013).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01695044 (2015).
Spratlin, J. L. et al. Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J. Clin. Oncol. 28, 780–787 (2010).
Acknowledgements
The Drug Development Unit of the Royal Marsden NHS Foundation Trust and The Institute of Cancer Research is supported in part by a programme grant from Cancer Research UK. Support is also provided by the Experimental Cancer Medicine Centre (to The Institute of Cancer Research) and the National Institute for Health Research Biomedical Research Centre (jointly to the Royal Marsden NHS Foundation Trust and The Institute of Cancer Research).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
All authors are current or former employees of The Institute of Cancer Research, London, UK, which has a commercial interest in the development of abiraterone acetate and phosphoinositide 3-kinase (PI3K) inhibitors, including pictilisib, and operates a rewards-to-discoverers scheme. T.A.Y. has received research funding from AstraZeneca and Vertex Pharmaceuticals; is or was a consultant or advisory board member for Pfizer and Merck Serono; and has received travel support from Bristol-Myers Squibb, Janssen and Merck. R.F. is an employee of Astex Pharmaceuticals. P.W. has received research funding from Astellas Pharma and Piramed Pharma, has ownership interest in Chroma Therapeutics and previously Piramed Pharma, and is or was a consultant or advisory board member for Chroma Therapeutics, Nextech Invest, NuEvolution and Piramed Pharma. J.S.d.B. has received consulting fees from Ortho Biotech Oncology Research and Development; consulting fees and travel support from Amgen, Astellas Pharma, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Dendreon, Enzon, Exelixis, Genentech, GlaxoSmithKline, Medivation, Merck, Novartis, Pfizer, Roche, Sanofi-Aventis, Supergen and Takeda; and grant support from AstraZeneca and Genentech.
Related links
Supplementary information
Supplementary information S1 (table)
Supplementary information (XLSX 31 kb)
Glossary
- Oncogenome
-
The collection of cancer-associated genes, epigenetics and transcripts.
- Massively parallel sequencing
-
A method of high-throughput DNA sequencing using multiple sequencing in parallel via spatially separated, clonally amplified DNA templates.
- Sequencing depth
-
The number of times a nucleotide is read during DNA sequencing. Deep sequencing requires a high number of reads.
- Base-calling
-
The identification of a particular base in a strand of nucleic acids.
- Sequence coverage
-
The average number of reads representing a given nucleotide in a reconstructed sequence.
- Actionable targets
-
Specific genomic events that potentially have important diagnostic, prognostic or therapeutic implications in subsets of patients with cancer and for specific therapies.
- Comparative genomic hybridization array
-
(aCGH). An analysis of copy number variation relative to the ploidy level of a set of genes in a test sample.
- Fluorescent in situ hybridization
-
(FISH). A technique using fluorescent nucleic acid probes to highlight only regions of the nucleic acid with a high degree of base sequence complementarity.
- DNA methyltransferase
-
An enzyme that catalyses the transfer of a methyl group to DNA (that is, a type of epigenetic modification).
- Microsatellite
-
Di-, tri- or tetranucleotide tandem repeats in DNA sequences.
Rights and permissions
About this article
Cite this article
Yap, T., Smith, A., Ferraldeschi, R. et al. Drug discovery in advanced prostate cancer: translating biology into therapy. Nat Rev Drug Discov 15, 699–718 (2016). https://doi.org/10.1038/nrd.2016.120
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd.2016.120
This article is cited by
-
The key cellular senescence related molecule RRM2 regulates prostate cancer progression and resistance to docetaxel treatment
Cell & Bioscience (2023)
-
Taraxasterol suppresses the proliferation and tumor growth of androgen-independent prostate cancer cells through the FGFR2-PI3K/AKT signaling pathway
Scientific Reports (2023)
-
Soft computing techniques for biomedical data analysis: open issues and challenges
Artificial Intelligence Review (2023)
-
Endoplasmic reticulum stress inhibits AR expression via the PERK/eIF2α/ATF4 pathway in luminal androgen receptor triple-negative breast cancer and prostate cancer
npj Breast Cancer (2022)
-
rAAV-based and intraprostatically delivered miR-34a therapeutics for efficient inhibition of prostate cancer progression
Gene Therapy (2022)