Key Points
-
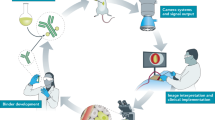
Near-infrared (NIR) fluorescence imaging has been demonstrated to be feasible during cancer surgery using available imaging systems and contrast agents
-
Clinical applicability has been described in sentinel lymph-node mapping, tumour imaging, visualization of vital structures and imaging of vascularization and perfusion
-
NIR fluorescence image-guided surgery has properties that make it a good candidate for clinical acceptance, as it fulfils a clinical need, is versatile and is fast
-
Targeted contrast agents, necessary for full evaluation of this technique, are in advanced stages of clinical approval
-
As novel contrast agents are developed and camera systems are optimized, the technique should prove its true clinical value
-
Well-designed outcomes studies have yet to be performed in the field
Abstract
Paradigm shifts in surgery arise when surgeons are empowered to perform surgery faster, better and less expensively than current standards. Optical imaging that exploits invisible near-infrared (NIR) fluorescent light (700–900 nm) has the potential to improve cancer surgery outcomes, minimize the time patients are under anaesthesia and lower health-care costs largely by way of its improved contrast and depth of tissue penetration relative to visible light. Accordingly, the past few years have witnessed an explosion of proof-of-concept clinical trials in the field. In this Review, we introduce the concept of NIR fluorescence imaging for cancer surgery, examine the clinical trial literature to date and outline the key issues pertaining to imaging system and contrast agent optimization. Although NIR seems to be superior to many traditional imaging techniques, its incorporation into routine care of patients with cancer depends on rigorous clinical trials and validation studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Weissleder, R. & Pittet, M. J. Imaging in the era of molecular oncology. Nature 452, 580–589 (2008).
Frangioni, J. V. New technologies for human cancer imaging. J. Clin. Oncol. 26, 4012–4021 (2008).
Rizzo, M. et al. The effects of additional tumor cavity sampling at the time of breast-conserving surgery on final margin status, volume of resection, and pathologist workload. Ann. Surg. Oncol. 17, 228–234 (2010).
McLaughlin, S. A. Surgical management of the breast: breast conservation therapy and mastectomy. Surg. Clin. North Am. 93, 411–428 (2013).
Kubben, P. L. et al. Intraoperative MRI-guided resection of glioblastoma multiforme: a systematic review. Lancet Oncol. 12, 1062–1070 (2011).
Chicoine, M. R. M. et al. Implementation and preliminary clinical experience with the use of ceiling mounted mobile high field intraoperative magnetic resonance imaging between two operating rooms. Acta Neurochir. Suppl. 109, 97–102 (2010).
Mislow, J. M., Golby, A. J. & Black, P. M. Origins of intraoperative MRI. Neurosurg. Clin. N. Am. 20, 137–146 (2009).
Gioux, S., Choi, H. S. & Frangioni, J. V. Image-guided surgery using invisible near-infrared light: fundamentals of clinical translation. Mol. Imaging 9, 237–255 (2010).
Schaafsma, B. E. et al. The clinical use of indocyanine green as a near-infrared fluorescent contrast agent for image-guided oncologic surgery. J. Surg. Oncol. 104, 323–332 (2011).
Chance, B. Near-infrared images using continuous, phase-modulated, and pulsed light with quantitation of blood and blood oxygenation. Ann. NY Acad. Sci. 838, 29–45 (1998).
Frangioni, J. V. In vivo near-infrared fluorescence imaging. Curr. Opin. Chem. Biol. 7, 626–634 (2003).
Weissleder, R., Tung, C. H., Mahmood, U. & Bogdanov, A. Jr. In vivo imaging of tumors with protease-activated near-infrared fluorescent probes. Nat. Biotechnol. 17, 375–378 (1999).
Choi, H. S. et al. Targeted zwitterionic near-infrared fluorophores for improved optical imaging. Nat. Biotechnol. 31, 148–153 (2013).
Olson, E. S. et al. Activatable cell penetrating peptides linked to nanoparticles as dual probes for in vivo fluorescence and MR imaging of proteases. Proc. Natl Acad. Sci. USA 107, 4311–4316 (2010).
Ohnishi, S. et al. Organic alternatives to quantum dots for intraoperative near-infrared fluorescent sentinel lymph node mapping. Mol. Imaging 4, 172–181 (2005).
Emerson, D. K. et al. A receptor-targeted fluorescent radiopharmaceutical for multireporter sentinel lymph node imaging. Radiology 265, 186–193 (2012).
Brouwer, O. R. et al. Feasibility of sentinel node biopsy in head and neck melanoma using a hybrid radioactive and fluorescent tracer. Ann. Surg. Oncol. 19, 1988–1994 (2012).
Hyde, D. et al. Hybrid FMT–CT imaging of amyloid-beta plaques in a murine Alzheimer's disease model. Neuroimage 44, 1304–1311 (2009).
Pham, W. et al. Crossing the blood-brain barrier: a potential application of myristoylated polyarginine for in vivo neuroimaging. Neuroimage 28, 287–292 (2005).
Deguchi, J.-O. et al. Inflammation in atherosclerosis: visualizing matrix metalloproteinase action in macrophages in vivo. Circulation 114, 55–62 (2006).
Sosnovik, D. E. et al. Fluorescence tomography and magnetic resonance imaging of myocardial macrophage infiltration in infarcted myocardium in vivo. Circulation 115, 1384–1391 (2007).
Wunder, A., Tung, C.-H., Müller-Ladner, U., Weissleder, R. & Mahmood, U. In vivo imaging of protease activity in arthritis: a novel approach for monitoring treatment response. Arthritis Rheum. 50, 2459–2465 (2004).
Choi, H. S. et al. Synthesis and in vivo fate of zwitterionic near-infrared fluorophores. Angew. Chem. Int. Ed. Engl. 50, 6258–6263 (2011).
Figueiredo, J.-L., Siegel, C., Nahrendorf, M. & Weissleder, R. Intraoperative near-infrared fluorescent cholangiography (NIRFC) in mouse models of bile duct injury. World J. Surg. 34, 336–343 (2010).
Whitney, M. A. et al. Fluorescent peptides highlight peripheral nerves during surgery in mice. Nat. Biotechnol. 29, 352–356 (2011).
Gibbs-Strauss, S. L. et al. Molecular imaging agents specific for the annulus fibrosus of the intervertebral disk. Mol. Imaging 9, 128–140 (2010).
Phillips, B. T. et al. Intraoperative perfusion techniques can accurately predict mastectomy skin flap necrosis in breast reconstruction. Plast. Reconstr. Surg. 129, 778e–788e (2012).
Hirche, C. et al. An experimental study to evaluate the Fluobeam 800 imaging system for fluorescence-guided lymphatic imaging and sentinel node biopsy. Surg. Innov. http://dx.doi.org/10.1177/1553350612468962.
Gotoh, K. et al. A novel image-guided surgery of hepatocellular carcinoma by indocyanine green fluorescence imaging navigation. J. Surg. Oncol. 100, 75–79 (2009).
Troyan, S. L. et al. The FLARE intraoperative near-infrared fluorescence imaging system: a first-in-human clinical trial in breast cancer sentinel lymph node mapping. Ann. Surg. Oncol. 16, 2943–2952 (2009).
Mieog, J. S. et al. Toward optimization of imaging system and lymphatic tracer for near-infrared fluorescent sentinel lymph node mapping in breast cancer. Ann. Surg. Oncol. 18, 2483–2491 (2011).
Crane, L. M. et al. Intraoperative multispectral fluorescence imaging for the detection of the sentinel lymph node in cervical cancer: a novel concept. Mol. Imaging Biol. 13, 1043–1049 (2011).
Yamauchi, K., Nagafuji, H., Nakamura, T., Sato, T. & Kohno, N. Feasibility of ICG fluorescence-guided sentinel node biopsy in animal models using the HyperEye Medical System. Ann. Surg. Oncol. 18, 2042–2047 (2011).
Cahill, R. A. et al. Near-infrared (NIR) laparoscopy for intraoperative lymphatic road-mapping and sentinel node identification during definitive surgical resection of early-stage colorectal neoplasia. Surg. Endosc. 26, 197–204 (2011).
van der Poel, H. G., Buckle, T., Brouwer, O. R., Valdés Olmos, R. A. & van Leeuwen, F. W. Intraoperative laparoscopic fluorescence guidance to the sentinel lymph node in prostate cancer patients: clinical proof of concept of an integrated functional imaging approach using a multimodal tracer. Eur. Urol. 60, 826–833 (2011).
Moroga, T. et al. Thoracoscopic segmentectomy with intraoperative evaluation of sentinel nodes for stage I non-small cell lung cancer. Ann. Thorac. Cardiovasc. Surg. 18, 89–94 (2012).
Yamashita, S.-I. et al. Video-assisted thoracoscopic indocyanine green fluorescence imaging system shows sentinel lymph nodes in non-small-cell lung cancer. J. Thorac. Cardiovasc. Surg. 141, 141–144 (2011).
Borofsky, M. S. et al. Near-infrared fluorescence imaging to facilitate super-selective arterial clamping during zero-ischaemia robotic partial nephrectomy. BJU Int. 111, 604–610 (2012).
Spinoglio, G. et al. Real-time near-infrared (NIR) fluorescent cholangiography in single-site robotic cholecystectomy (SSRC): a single-institutional prospective study. Surg. Endosc. 27, 2156–2162 (2012).
The Flare Foundation. Lighting the way for surgery [online], (2013).
Achilefu, S. The insatiable quest for near-infrared fluorescent probes for molecular imaging. Angew. Chem. Int. Ed. Engl. 49, 9816–9818 (2010).
Bradley, R. S. & Thorniley, M. S. A review of attenuation correction techniques for tissue fluorescence. J. R. Soc. Interface 3, 1–13 (2006).
Themelis, G., Yoo, J. S., Soh, K.-S., Schulz, R. & Ntziachristos, V. Real-time intraoperative fluorescence imaging system using light-absorption correction. J. Biomed. Opt. 14, 064012 (2009).
Valdés, P. A. et al. Quantitative, spectrally-resolved intraoperative fluorescence imaging. Sci. Rep. 2, 798 (2012).
Saager, R. B., Cuccia, D. J., Saggese, S. S., Kelly, K. M. K. & Durkin, A. J. Quantitative fluorescence imaging of protoporphyrin IX through determination of tissue optical properties in the spatial frequency domain. J. Biomed. Opt. 16, 126013 (2011).
Matsui, A., Lee, B. T., Winer, J. H., Laurence, R. G. & Frangioni, J. V. Quantitative assessment of perfusion and vascular compromise in perforator flaps using a near-infrared fluorescence-guided imaging system. Plast. Reconstr. Surg. 124, 451–460 (2009).
Matsui, A., Winer, J. H., Laurence, R. G. & Frangioni, J. V. Predicting the survival of experimental ischaemic small bowel using intraoperative near-infrared fluorescence angiography. Br. J. Surg. 98, 1725–1734 (2011).
van der Vorst, J. R. et al. Near-infrared fluorescence-guided resection of colorectal liver metastases. Cancer http://dx.doi.org/10.1002/cncr.28203.
Mieog, J. S. D. et al. Image-guided tumor resection using real-time near-infrared fluorescence in a syngeneic rat model of primary breast cancer. Breast Cancer Res. Treat. 128, 679–689 (2011).
Winer, J. H. et al. Intraoperative localization of insulinoma and normal pancreas using invisible near-infrared fluorescent light. Ann. Surg. Oncol. 17, 1094–1100 (2010).
Verbeek, F. P. et al. Intraoperative near-infrared fluorescence-guided identification of the ureters using low-dose methylene blue: a first-in-human experience. J. Urol. 190, 574–579 (2013).
van der Vorst, J. R. et al. Near-infrared fluorescence imaging of a solitary fibrous tumor of the pancreas using methylene blue. World J. Gastrointest. Surg. 4, 180–184 (2012).
FDA. Product Insert: Indocyanine Green (IC-Green™) [online], (2013).
FDA. Product Insert: 5-Aminolevulinic Acid (Levulan®Kerastick™) [online], (1999).
Colditz, M. J. & Jeffree, R. L. Aminolevulinic acid (ALA)-protoporphyrin IX fluorescence guided tumour resection. Part 1: clinical, radiological and pathological studies. J. Clin. Neurosci. 19, 1471–1474 (2012).
Colditz, M. J., Leyen, K. V. & Jeffree, R. L. Aminolevulinic acid (ALA)-protoporphyrin IX fluorescence guided tumour resection. Part 2: theoretical, biochemical and practical aspects. J. Clin. Neurosci. 19, 1611–1616 (2012).
Stummer, W. et al. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol. 7, 392–401 (2006).
Moore, G. E., Peyton, W. T., French, L. A. & Walker, W. W. The clinical use of fluorescein in neurosurgery; the localization of brain tumors. J. Neurosurg. 5, 392–398 (1948).
Folli, S. et al. Immunophotodiagnosis of colon carcinomas in patients injected with fluoresceinated chimeric antibodies against carcinoembryonic antigen. Proc. Natl Acad. Sci. USA 89, 7973–7977 (1992).
van Dam, G. M. et al. Intraoperative tumor-specific fluorescence imaging in ovarian cancer by folate receptor-α targeting: first in-human results. Nat. Med. 17, 1315–1319 (2011).
Morton, D. L. & Bostick, P. J. Will the true sentinel node please stand? Ann. Surg. Oncol. 6, 12–14 (1999).
Giuliano, A. E., Kirgan, D. M., Guenther, J. M. & Morton, D. L. Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Ann. Surg. 220, 391–398; discussion 398–401 (1994).
van den Berg, N. S. et al. Concomitant radio- and fluorescence-guided sentinel lymph node biopsy in squamous cell carcinoma of the oral cavity using ICG-(99m)Tc-nanocolloid. Eur. J. Nucl. Med. Mol. Imaging 39, 1128–1136 (2012).
van der Vorst, J. R. et al. Randomized comparison of near-infrared fluorescence imaging using indocyanine green and 99(m) technetium with or without patent blue for the sentinel lymph node procedure in breast cancer patients. Ann. Surg. Oncol. 19, 4104–4111 (2012).
Murawa, D., Hirche, C., Dresel, S. & Hünerbein, M. Sentinel lymph node biopsy in breast cancer guided by indocyanine green fluorescence. Br. J. Surg. 96, 1289–1294 (2009).
Hojo, T., Nagao, T., Kikuyama, M., Akashi, S. & Kinoshita, T. Evaluation of sentinel node biopsy by combined fluorescent and dye method and lymph flow for breast cancer. Breast 19, 210–213 (2010).
Kitai, T., Inomoto, T., Miwa, M. & Shikayama, T. Fluorescence navigation with indocyanine green for detecting sentinel lymph nodes in breast cancer. Breast Cancer 12, 211–215 (2005).
Sevick-Muraca, E. M. et al. Imaging of lymph flow in breast cancer patients after microdose administration of a near-infrared fluorophore: feasibility study. Radiology 246, 734–741 (2008).
Hirche, C., Murawa, D., Mohr, Z., Kneif, S. & Hünerbein, M. ICG fluorescence-guided sentinel node biopsy for axillary nodal staging in breast cancer. Breast Cancer Res. Treat. 121, 373–378 (2010).
Hirche, C. et al. Ultrastaging of colon cancer by sentinel node biopsy using fluorescence navigation with indocyanine green. Int. J. Colorectal Dis. 27, 319–324 (2012).
Ankersmit, M., van der Pas, M. H., van Dam, D. A. & Meijerink, W. J. Near infrared fluorescence lymphatic laparoscopy of the colon and mesocolon. Colorectal Dis. 13 (Suppl. 7), 70–73 (2011).
Hutteman, M. et al. Clinical translation of ex vivo sentinel lymph node mapping for colorectal cancer using invisible near-infrared fluorescence light. Ann. Surg. Oncol. 18, 1006–1014 (2011).
Schaafsma, B. E. et al. Ex vivo sentinel node mapping in colon cancer combining blue dye staining and fluorescence imaging. J. Surg. Res. 183, 253–257 (2013).
van der Pas, M. H. et al. Laparoscopic sentinel lymph node identification in patients with colon carcinoma using a near-infrared dye: description of a new technique and feasibility study. J. Laparoendosc. Adv. Surg. Tech. 23, 367–371 (2013).
Fujiwara, M., Mizukami, T., Suzuki, A. & Fukamizu, H. Sentinel lymph node detection in skin cancer patients using real-time fluorescence navigation with indocyanine green: preliminary experience. J. Plast. Reconstr. Aesthet. Surg. 62, e373–e378 (2009).
van der Vorst, J. R. et al. Dose optimization for near-infrared fluorescence sentinel lymph node mapping in patients with melanoma. Br. J. Dermatol. 168, 93–98 (2013).
van der Vorst, J. R. et al. Optimization of near-infrared fluorescent sentinel lymph node mapping in cervical cancer patients. Int. J. Gynecol. Cancer 21, 1472–1478 (2011).
Furukawa, N. et al. The usefulness of photodynamic eye for sentinel lymph node identification in patients with cervical cancer. Tumori 96, 936–940 (2010).
Crane, L. M. et al. Intraoperative near-infrared fluorescence imaging for sentinel lymph node detection in vulvar cancer: first clinical results. Gynecol. Oncol. 120, 291–295 (2011).
Hutteman, M. et al. Optimization of near-infrared fluorescent sentinel lymph node mapping for vulvar cancer. Am. J. Obstet. Gynecol. 206, 89.e1–89.e5 (2012).
Schaafsma, B. E. et al. Randomized comparison of near-infrared fluorescence lymphatic tracers for sentinel lymph node mapping of cervical cancer. Gynecol. Oncol. 127, 126–130 (2012).
Brouwer, O. R. et al. Comparing the hybrid fluorescent-radioactive tracer indocyanine green-99mTc-nanocolloid with 99mTc-nanocolloid for sentinel node identification: a validation study using lymphoscintigraphy and SPECT/CT. J. Nucl. Med. 53, 1034–1040 (2012).
Bredell, M. G. Sentinel lymph node mapping by indocyanin green fluorescence imaging in oropharyngeal cancer - preliminary experience. Head Neck Oncol. 2, 31 (2010).
Yamashita, S. et al. Sentinel node navigation surgery by thoracoscopic fluorescence imaging system and molecular examination in non-small cell lung cancer. Ann. Surg. Oncol. 19, 728–733 (2012).
Holloway, R. W. et al. Detection of sentinel lymph nodes in patients with endometrial cancer undergoing robotic-assisted staging: a comparison of colorimetric and fluorescence imaging. Gynecol. Oncol. 126, 25–29 (2012).
Tajima, Y. et al. Sentinel node mapping guided by indocyanine green fluorescence imaging during laparoscopic surgery in gastric cancer. Ann. Surg. Oncol. 17, 1787–1793 (2010).
Tajima, Y. et al. Sentinel node mapping guided by indocyanine green fluorescence imaging in gastric cancer. Ann. Surg. 249, 58–62 (2009).
Kubota, K. et al. Application of the HyperEye Medical System for esophageal cancer surgery: a preliminary report. Surg. Today 43, 215–220 (2013).
Hutteman, M. et al. Randomized, double-blind comparison of indocyanine green with or without albumin premixing for near-infrared fluorescence imaging of sentinel lymph nodes in breast cancer patients. Breast Cancer Res. Treat. 127, 163–170 (2011).
Buckle, T. et al. A self-assembled multimodal complex for combined pre- and intraoperative imaging of the sentinel lymph node. Nanotechnology 21, 355101 (2010).
Vera, D. R., Wallace, A. M., Hoh, C. K. & Mattrey, R. F. A synthetic macromolecule for sentinel node detection: (99m)Tc-DTPA-mannosyl-dextran. J. Nucl. Med. 42, 951–959 (2001).
Ting, R. et al. Fast 18F labeling of a near-infrared fluorophore enables positron emission tomography and optical imaging of sentinel lymph nodes. Bioconjug. Chem. 21, 1811–1819 (2010).
Leong, S. P. L. et al. A phase 2 study of (99m)Tc-tilmanocept in the detection of sentinel lymph nodes in melanoma and breast cancer. Ann. Surg. Oncol. 18, 961–969 (2011).
[No authors listed]. Agent approved for finding cancer lymph nodes. Cancer Discov. 3, OF8 (2013).
Keereweer, S. et al. Optical image-guided surgery—where do we stand? Mol. Imaging Biol. 13, 199–207 (2011).
Sevick-Muraca, E. M. Translation of near-infrared fluorescence imaging technologies: emerging clinical applications. Annu. Rev. Med. 63, 217–231 (2012).
Horowitz, N. S. et al. Laparoscopy in the near infrared with ICG detects microscopic tumor in women with ovarian cancer: 0078. Int. J. Gynecol. Cancer 16, 616–623 (2006).
Ishizawa, T. et al. Real-time identification of liver cancers by using indocyanine green fluorescent imaging. Cancer 115, 2491–2504 (2009).
Uchiyama, K. K. et al. Combined use of contrast-enhanced intraoperative ultrasonography and a fluorescence navigation system for identifying hepatic metastases. World J. Surg. 34, 2953–2959 (2010).
Peloso, A. et al. Combined use of intraoperative ultrasound and indocyanine green fluorescence imaging to detect liver metastases from colorectal cancer. HPB (Oxford) http://dx.doi.org/10.1111/hpb.12057.
Ishizuka, M. et al. Intraoperative observation using a fluorescence imaging instrument during hepatic resection for liver metastasis from colorectal cancer. Hepatogastroenterology 59, 90–92 (2011).
Sahani, D. V. et al. Intraoperative US in patients undergoing surgery for liver neoplasms: comparison with MR imaging. Radiology 232, 810–814 (2004).
Satou, S. et al. Indocyanine green fluorescent imaging for detecting extrahepatic metastasis of hepatocellular carcinoma. J. Gastroenterol. http://dx.doi.org/10.1007/s00535-012-0709-6.
Yokoyama, N. et al. Real-time detection of hepatic micrometastases from pancreatic cancer by intraoperative fluorescence imaging: preliminary results of a prospective study. Cancer 118, 2813–2819 (2012).
Hwang, S. W., Malek, A. M., Schapiro, R. & Wu, J. K. Intraoperative use of indocyanine green fluorescence videography for resection of a spinal cord hemangioblastoma. Neurosurgery 67 (Suppl. 3), ons300–ons303 (2010).
Ferroli, P. et al. Application of intraoperative indocyanine green angiography for CNS tumors: results on the first 100 cases. Acta Neurochir. Suppl. 109, 251–257 (2011).
Hojo, M. et al. Usefulness of tumor blood flow imaging by intraoperative ICG videoangiography in hemangioblastoma surgery. World Neurosurg. http://dx.doi.org/10.1016/j.wneu.2013.02.009.
Keaveny, T. V., Fitzgerald, P. A. & McMullin, J. P. Selective parathyroid and pancreatic staining. Br. J. Surg. 56, 595–597 (1969).
Keaveny, T. V., Tawes, R. & Belzer, F. O. A new method for intra-operative identification of insulinomas. Br. J. Surg. 58, 233–234 (1971).
Roberts, D. W. et al. Coregistered fluorescence-enhanced tumor resection of malignant glioma: relationships between δ-aminolevulinic acid-induced protoporphyrin IX fluorescence, magnetic resonance imaging enhancement, and neuropathological parameters. Clinical article. J. Neurosurg. 114, 595–603 (2011).
Jocham, D., Stepp, H. & Waidelich, R. Photodynamic diagnosis in urology: state-of-the-art. Eur. Urol. 53, 1138–1148 (2008).
Hsiung, P.-L. et al. Detection of colonic dysplasia in vivo using a targeted heptapeptide and confocal microendoscopy. Nat. Med. 14, 454–458 (2008).
Kriegmair, M. et al. Detection of early bladder cancer by 5-aminolevulinic acid induced porphyrin fluorescence. J. Urol. 155, 105–110 (1996).
Jichlinski, P. et al. Clinical evaluation of a method for detecting superficial surgical transitional cell carcinoma of the bladder by light-induced fluorescence of protoporphyrin IX following the topical application of 5-aminolevulinic acid: preliminary results. Lasers Surg. Med. 20, 402–408 (1997).
Jichlinski, P. et al. Hexyl aminolevulinate fluorescence cystoscopy: new diagnostic tool for photodiagnosis of superficial bladder cancer—a multicenter study. J. Urol. 170, 226–229 (2003).
Wallner, C. et al. Causes of fecal and urinary incontinence after total mesorectal excision for rectal cancer based on cadaveric surgery: a study from the Cooperative Clinical Investigators of the Dutch total mesorectal excision trial. J. Clin. Oncol. 26, 4466–4472 (2008).
Miranda-Sousa, A. J., Davila, H. H., Lockhart, J. L., Ordorica, R. C. & Carrion, R. E. Sexual function after surgery for prostate or bladder cancer. Cancer Control 13, 179–187 (2006).
Flum, D. R., Koepsell, T., Heagerty, P., Sinanan, M. & Dellinger, E. P. Common bile duct injury during laparoscopic cholecystectomy and the use of intraoperative cholangiography: adverse outcome or preventable error? Arch. Surg. 136, 1287–1292 (2001).
Selzman, A. A. & Spirnak, J. P. Iatrogenic ureteral injuries: a 20-year experience in treating 165 injuries. J. Urol. 155, 878–881 (1996).
Ishizawa, T. et al. Fluorescent cholangiography illuminating the biliary tree during laparoscopic cholecystectomy. Br. J. Surg. 97, 1369–1377 (2010).
Rasmussen, J. C. et al. Human lymphatic architecture and dynamic transport imaged using near-infrared fluorescence. Transl. Oncol. 3, 362–372 (2010).
Choy, P. Y., Bissett, I. P., Docherty. J. G., Parry, B. R. & Merrie, A. E. Stapled versus handsewn methods for ileocolic anastomoses. Cochrane Database Systematic Reviews, Issue 3. Art. No.: CD004320. http://dx.doi.org/10.1002/14651858.CD004320.pub2.
Hyman, N. N., Manchester, T. L., Osler, T. T., Burns, B. B. & Cataldo, P. A. Anastomotic leaks after intestinal anastomosis: it's later than you think. Ann. Surg. 245, 254–258 (2007).
Kudszus, S., Roesel, C., Schachtrupp, A. & Höer, J. J. Intraoperative laser fluorescence angiography in colorectal surgery: a noninvasive analysis to reduce the rate of anastomotic leakage. Langenbecks Arch. Surg. 395, 1025–1030 (2010).
Pacheco, P. E., Hill, S. M., Henriques, S. M., Paulsen, J. K. & Anderson, R. C. The novel use of intraoperative laser-induced fluorescence of indocyanine green tissue angiography for evaluation of the gastric conduit in esophageal reconstructive surgery. Am. J. Surg. 205, 349–353 (2013).
Jafari, M. D. et al. The use of indocyanine green fluorescence to assess anastomotic perfusion during robotic assisted laparoscopic rectal surgery. Surg. Endosc. http://dx.doi.org/10.1007/s00464-013-2832-8.
Holm, C., Mayr, M., Höfter, E. & Ninkovic, M. Perfusion zones of the DIEP flap revisited: a clinical study. Plast. Reconstr. Surg. 117, 37–43 (2006).
Lee, B. T. et al. The FLARE intraoperative near-infrared fluorescence imaging system: a first-in-human clinical trial in perforator flap breast reconstruction. Plast. Reconstr. Surg. 126, 1472–1481 (2010).
Newman, M. I. & Samson, M. C. The application of laser-assisted indocyanine green fluorescent dye angiography in microsurgical breast reconstruction. J. Reconstr. Microsurg. 25, 21–26 (2009).
Newman, M. I., Samson, M. C., Tamburrino, J. F. & Swartz, K. A. Intraoperative laser-assisted indocyanine green angiography for the evaluation of mastectomy flaps in immediate breast reconstruction. J. Reconstr. Microsurg. 26, 487–492 (2010).
Kumar, A. T. et al. Feasibility of in vivo imaging of fluorescent proteins using lifetime contrast. Opt. Lett. 34, 2066–2068 (2009).
Themelis, G., Yoo, J. S. & Ntziachristos, V. Multispectral imaging using multiple-bandpass filters. Opt. Lett. 33, 1023–1025 (2008).
Marshall, M. V., Draney, D., Sevick-Muraca, E. M. & Olive, D. M. Single-dose intravenous toxicity study of IRDye 800CW in Sprague-Dawley rats. Mol. Imaging Biol. 12, 583–594 (2010).
Oliveira, S. et al. Rapid visualization of human tumor xenografts through optical imaging with a near-infrared fluorescent anti-epidermal growth factor receptor nanobody. Mol. Imaging 11, 33–46 (2012).
Tanaka, E. et al. Real-time intraoperative assessment of the extrahepatic bile ducts in rats and pigs using invisible near-infrared fluorescent light. Surgery 144, 39–48 (2008).
Scheuer, W., van Dam, G. M., Dobosz, M., Schwaiger, M. & Ntziachristos, V. Drug-based optical agents: infiltrating clinics at lower risk. Sci. Transl. Med. 4, 134ps11 (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2012).
Gaertner, F. C., Kessler, H., Wester, H. J., Schwaiger, M. & Beer, A. J. Radiolabelled RGD peptides for imaging and therapy. Eur. J. Nucl. Med. Mol. Imaging 39 (Suppl. 1), 126–138 (2012).
Muyldermans, S., Atarhouch, T., Saldanha, J., Barbosa, J. A. & Hamers, R. Sequence and structure of VH domain from naturally occurring camel heavy chain immunoglobulins lacking light chains. Protein Eng. 7, 1129–1135 (1994).
Bhatia, S., Frangioni, J. V., Hoffman, R. M., Iafrate, A. J. & Polyak, K. The challenges posed by cancer heterogeneity. Nat. Biotechnol. 30, 604–610 (2012).
Gibbs-Strauss, S. L. et al. Nerve-highlighting fluorescent contrast agents for image-guided surgery. Mol. Imaging 10, 91–101 (2011).
Sheth, R. A. et al. Improved detection of ovarian cancer metastases by intraoperative quantitative fluorescence protease imaging in a pre-clinical model. Gynecol. Oncol. 112, 616–622 (2009).
Urano, Y. et al. Selective molecular imaging of viable cancer cells with pH-activatable fluorescence probes. Nat. Med. 15, 104–109 (2009).
Urano, Y. et al. Rapid cancer detection by topically spraying a γ-glutamyltranspeptidase-activated fluorescent probe. Sci. Transl. Med. 3, 110ra119 (2011).
Kosaka, N., Mitsunaga, M., Longmire, M. R., Choyke, P. L. & Kobayashi, H. Near infrared fluorescence-guided real-time endoscopic detection of peritoneal ovarian cancer nodules using intravenously injected indocyanine green. Int. J. Cancer 129, 1671–1677 (2011).
Dengel, L. T. et al. Intraoperative imaging guidance for sentinel node biopsy in melanoma using a mobile gamma camera. Ann. Surg. 253, 774–778 (2011).
Brouwer, O. R. et al. Image navigation as a means to expand the boundaries of fluorescence-guided surgery. Phys. Med. Biol. 57, 3123–3136 (2012).
Frangioni, J. V. Translating in vivo diagnostics into clinical reality. Nat. Biotechnol. 24, 909–913 (2006).
Yuasa, Y. et al. Sentinel lymph node biopsy using intraoperative indocyanine green fluorescence imaging navigated with preoperative CT lymphography for superficial esophageal cancer. Ann. Surg. Oncol. 19, 486–493 (2011).
Miyashiro, I. et al. Intraoperative diagnosis using sentinel node biopsy with indocyanine green dye in gastric cancer surgery: an institutional trial by experienced surgeons. Ann. Surg. Oncol. 20, 542–546 (2012).
Miyashiro, I. et al. Laparoscopic detection of sentinel node in gastric cancer surgery by indocyanine green fluorescence imaging. Surg. Endosc. 25, 1672–1676 (2010).
Kusano, M. et al. Sentinel node mapping guided by indocyanine green fluorescence imaging: a new method for sentinel node navigation surgery in gastrointestinal cancer. Dig. Surg. 25, 103–108 (2008).
Jeschke, S. et al. Visualisation of the lymph node pathway in real time by laparoscopic radioisotope- and fluorescence-guided sentinel lymph node dissection in prostate cancer staging. Urology 80, 1080–1086 (2012).
Ishizawa, T. et al. Indocyanine green-fluorescent imaging of hepatocellular carcinoma during laparoscopic hepatectomy: an initial experience. Asian J. Endosc. Surg. 3, 42–45 (2010).
Mitsuhashi, N. et al. Usefulness of intraoperative fluorescence imaging to evaluate local anatomy in hepatobiliary surgery. J. Hepatobiliary Pancreat. Surg. 15, 508–514 (2008).
Hutteman, M. et al. Near-infrared fluorescence imaging in patients undergoing pancreaticoduodenectomy. Eur. Surg. Res. 47, 90–97 (2011).
Tagaya, N. et al. Intraoperative exploration of biliary anatomy using fluorescence imaging of indocyanine green in experimental and clinical cholecystectomies. J. Hepatobiliary Pancreat. Surg. 17, 595–600 (2010).
Aoki, T. et al. Intraoperative fluorescent imaging using indocyanine green for liver mapping and cholangiography. J. Hepatobiliary Pancreat. Surg. 17, 590–594 (2009).
Acknowledgements
The authors are supported by the NIH (award numbers R01-CA-115296, R01-EB-011523, R01-EB-010022 and R21-CA-130297), The Dutch Cancer Society (grant UL2010-4732) and The Center for Translational Molecular Medicine (DeCoDe project, grant 03O-101). The content is solely the responsibility of the authors and does not necessarily represent the official views of these agencies.
Author information
Authors and Affiliations
Contributions
A. L. Vahrmeijer and M. Hutteman researched the data for the Review. A. L. Vahrmeijer, M. Hutteman and J. V. Frangioni wrote the manuscript. All authors contributed to the discussion of the manuscript content and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J. V. Frangioni is a founding member of the following companies: Curadel, Curadel ResVet Imaging, Curadel Surgical Innovations, and the founder and chairman of the FLARE Foundation, which licenses the FLARE technology from Beth Israel Deaconess Medical Center, Harvard Medical School. J. V. Frangioni has elected to surrender postmarket royalties for FLARE, which he would otherwise be entitled to as inventor, and has elected to donate premarket proceeds to the FLARE Foundation.
Rights and permissions
About this article
Cite this article
Vahrmeijer, A., Hutteman, M., van der Vorst, J. et al. Image-guided cancer surgery using near-infrared fluorescence. Nat Rev Clin Oncol 10, 507–518 (2013). https://doi.org/10.1038/nrclinonc.2013.123
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2013.123
This article is cited by
-
Visual extended reality tools in image-guided surgery in urology: a systematic review
European Journal of Nuclear Medicine and Molecular Imaging (2024)
-
High-precision detection and navigation surgery of colorectal cancer micrometastases
Journal of Nanobiotechnology (2023)
-
Dual tracer navigation for lymph node dissection in laparoscopic radical gastrectomy (DANCE trial): a protocol for a prospective, randomized clinical trial
Trials (2023)
-
Safety and efficacy of precision hepatectomy in the treatment of primary liver cancer
BMC Surgery (2023)
-
Fluorescence image-guided tumour surgery
Nature Reviews Bioengineering (2023)