Abstract
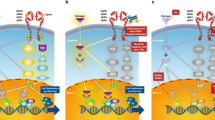
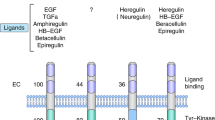
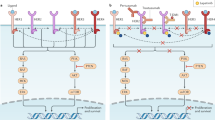
HER2-positive tumors have a worse prognosis than HER2-negative cancers. Although the standard treatment for HER2-positive metastatic breast cancer is chemotherapy and trastuzumab, the combination of aromatase inhibitors with anti-HER2 therapies has become an available strategy in patients with HER2-positive and hormone receptor-positive tumors. However, although this new treatment option is more effective than hormone therapy alone, it has not been compared with the standard chemotherapy and trastuzumab-based regimens. In fact, the activity observed in randomized clinical trials with chemotherapy and anti-HER2 therapies seems to be higher than that observed with aromatase inhibitors and trastuzumab-based or lapatinib-based therapies. In this article, we highlight the importance of considering chemotherapy and anti-HER2 therapy as the standard of care in HER2-positive and hormone receptor-positive tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Piccart-Gebhart, M. J. et al. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N. Engl. J. Med. 353, 1659–1672 (2005).
Romond, E. H. et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N. Engl. J. Med. 353, 1673–1684 (2005).
Untch, M. et al. Estimating the magnitude of trastuzumab effects within patient subgroups in the HERA trial. Ann. Oncol. 19, 1090–1096 (2008).
Perou, C. M. et al. Molecular portraits of human breast tumours. Nature 406, 747–752 (2000).
Pusztai, L. et al. Gene expression profiles obtained from fine-needle aspirations of breast cancer reliably identify routine prognostic markers and reveal large-scale molecular differences between estrogen-negative and estrogen-positive tumors. Clin. Cancer Res. 9, 2406–2415 (2003).
Sørlie, T. et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc. Natl Acad. Sci. USA 100, 8418–8423 (2003).
Sotiriou, C. et al. Breast cancer classification and prognosis based on gene expression profiles from a population-based study. Proc. Natl Acad. Sci. USA 100, 10393–10398 (2003).
Marty, M. et al. Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer administered as first-line treatment: the M77001 study group. J. Clin. Oncol. 23, 4265–4274 (2005).
Brufsky, A., Lembersky, B., Schiffman, K., Lieberman, G. & Paton, V. E. Hormone receptor status does not affect the clinical benefit of trastuzumab therapy for patients with metastatic breast cancer. Clin. Breast Cancer 6, 247–252 (2005).
Kaufman, B. et al. Trastuzumab plus anastrozole versus anastrozole alone for the treatment of postmenopausal women with human epidermal growth factor receptor 2-positive, hormone receptor-positive metastatic breast cancer: results from the randomized phase III TAnDEM study. J. Clin. Oncol. 27, 5529–5537 (2009).
Johnston, S. et al. Lapatinib combined with letrozole versus letrozole and placebo as first-line therapy for postmenopausal hormone receptor-positive metastatic breast cancer. J. Clin. Oncol. 27, 5538–5546 (2009).
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001).
Baselga, J. et al. Phase II study of efficacy, safety, and pharmacokinetics of trastuzumab monotherapy administered on a 3-weekly schedule. J. Clin. Oncol. 23, 2162–2171 (2005).
Vogel, C. L. et al. Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. J. Clin. Oncol. 20, 719–726 (2002).
Gomez, H. L. et al. Efficacy and safety of lapatinib as first-line therapy for ErbB2-amplified locally advanced or metastatic breast cancer. J. Clin. Oncol. 26, 2999–3005 (2008).
Knauer, M. et al. Identification of a low risk subgroup in HER-2-positive breast cancer by the 70-gene prognosis signature. Br. J. Cancer doi:10.1038/sj.bjc.6605916.
Bontenbal, M. et al. Randomized study comparing efficacy/toxicity of monotherapy trastuzumab followed by monotherapy docetaxel at progression, and combination trastuzumab/docetaxel as first line chemotherapy in HER2-neu positive metastatic breast cancer (HERTAX study) [abstract]. J. Clin. Oncol. 26 (Suppl.), a1014 (2008).
Karamouzis, M. V., Ioannidis, G. & Rigatos, G. Quality of life in metastatic breast cancer patients under chemotherapy or supportive care: a single-institution comparative study. Eur. J. Cancer Care (Engl.) 16, 433–438 (2007).
Slamon, D. J. et al. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235, 177–182 (1987).
Arpino, G. et al. HER-2 amplification, HER-1 expression, and tamoxifen response in estrogen receptor-positive metastatic breast cancer. A Southwest Oncology Group Study. Clin. Cancer Res. 10, 5670–5676 (2004).
Sotiriou, C. & Pusztai, L. Gene-expression signatures in breast cancer. N. Engl. J. Med. 360, 790–800 (2009).
Osborne, C. K. & Schiff, R. Estrogen-receptor biology: continuing progress and therapeutic implications. J. Clin. Oncol. 23, 1616–1622 (2005).
Arpino, G. et al. Treatment of human epidermal growth factor receptor 2-overexpressing breast cancer xenografts with multiagent HER-targeted therapy. J. Natl Cancer Inst. 99, 694–705 (2007).
Ellis, M. Overcoming endocrine therapy resistance by signal transduction inhibition. Oncologist 9 (Suppl. 3), 20–26 (2004).
Johnston, S. R., Martin, L. A., Leary, A., Head, J. & Dowsett, M. Clinical strategies for rationale combinations of aromatase inhibitors with novel therapies for breast cancer. J. Steroid Biochem. Mol. Biol. 106, 180–186 (2007).
Cortes, J. & Baselga, J. How to treat hormone receptor-positive, human epidermal growth factor receptor 2-amplified breast cancer. J. Clin. Oncol. 27, 5492–5494 (2009).
Author information
Authors and Affiliations
Contributions
J. Cortés and C. Saura researched data to include in the article. J. Cortés, C. Saura, N. Ramírez-Merino, V. Calvo and M. Vidal wrote the manuscript. All the authors contributed to discussion of content for the article and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Cortés, J., Saura, C., Bellet, M. et al. HER2 and hormone receptor-positive breast cancer—blocking the right target. Nat Rev Clin Oncol 8, 307–311 (2011). https://doi.org/10.1038/nrclinonc.2010.185
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2010.185
This article is cited by
-
Effect of level of hormone-receptor expression on treatment outcomes of “triple-positive” early-stage breast cancer
Breast Cancer Research and Treatment (2021)
-
Monoclonal antibodies for medical oncology: a few critical perspectives
Clinical and Translational Oncology (2011)