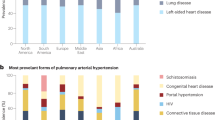
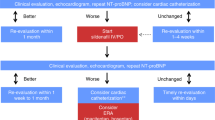
Key Points
-
Pulmonary arterial hypertension (PAH) is a symptom of various pathologies and is frequently associated with progressive deterioration; PAH is an important cause of morbidity and mortality in children
-
Current therapies for PAH target the vascular calcium channel as well as abnormalities in the endothelin, nitric oxide, and prostacyclin signalling pathways
-
Advances in basic and clinical research into PAH have led to improved understanding of disease pathogenesis and identification of novel therapeutic targets
-
Only a minority of drugs for PAH are officially approved in children, because randomized controlled studies are limited by the small number of paediatric patients with PAH
-
Interventional and surgical strategies are increasingly performed in children with severe PAH refractory to medical therapy, to relieve right ventricular pressure overload, improve left ventricular performance, and support systemic circulation
-
Developments in PAH-specific therapy as well as surgical and interventional procedures for severe PAH might delay or even avoid the need for lung transplantation
Abstract
Pulmonary arterial hypertension (PAH) is an important cause of morbidity and mortality in children. Improved understanding of the pathophysiological mechanisms of the underlying diseases has resulted in the development of effective, but not yet curative, therapies. Currently, drugs from three main pharmacological groups targeting specific aberrant pathways (endothelin, nitric oxide, and prostacyclin) and four routes of administration (inhaled, intravenous, oral, and subcutaneous) have been approved for adult patients with PAH. However, only a minority of these drugs has been officially approved for children, mainly because randomized controlled studies are limited by the small number of paediatric patients with PAH worldwide. In children with progressive, severe PAH and an inadequate response to drug therapy, advances in interventional and surgical approaches have provided promising new strategies to avoid right ventricular deterioration. These techniques can delay or even avoid the need for lung transplantation. In this Review, we present an update on developments in drug therapy for patients with PAH and highlight the current status of these treatments in children. We also describe the new surgical, interventional, and hybrid procedures, as well as their practical application in children with severe PAH.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Galiè, N. et al. Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur. Respir. J. 34, 1219–1263 (2009).
Berger, R. M. et al. Clinical features of paediatric pulmonary hypertension: a registry study. Lancet 379, 537–546 (2012).
van Loon, R. L. et al. Pediatric pulmonary hypertension in the Netherlands: epidemiology and characterization during the period 1991 to 2005. Circulation 124, 1755–1764 (2011).
Gaine, S. Pulmonary hypertension. JAMA 284, 3160–3168 (2000).
Zijlstra, W. M. et al. Survival differences in pediatric pulmonary arterial hypertension: clues to a better understanding of outcome and optimal treatment strategies. J. Am. Coll. Cardiol. 63, 2159–2169 (2014).
Barst, R. J. et al. Survival in childhood pulmonary arterial hypertension: insights from the registry to evaluate early and long-term pulmonary arterial hypertension disease management. Circulation 125, 113–122 (2012).
Ivy, D. D. et al. Long-term outcomes in children with pulmonary arterial hypertension treated with bosentan in real-world clinical settings. Am. J. Cardiol. 106, 1332–1338 (2010).
Moledina, S., Hislop, A. A., Foster, H., Schulze-Neick, I. & Haworth, S. G. Childhood idiopathic pulmonary arterial hypertension: a national cohort study. Heart 96, 1401–1406 (2010).
van Loon, R. L. et al. Outcome of pediatric patients with pulmonary arterial hypertension in the era of new medical therapies. Am. J. Cardiol. 106, 117–124 (2010).
Blanc, J., Vouhé, P. & Bonnet, D. Potts shunt in patients with pulmonary hypertension. N. Engl. J. Med. 350, 623 (2004).
Labombarda, F., Maragnes, P., Dupont-Chauvet, P. & Serraf, A. Potts anastomosis for children with idiopathic pulmonary hypertension. Pediatr. Cardiol. 30, 1143–1145 (2009).
Latus, H. et al. Potts shunt and atrial septostomy in pulmonary hypertension caused by left ventricular disease. Ann. Thorac. Surg. 96, 317–319 (2013).
Latus, H. et al. Creation of a functional Potts shunt by stenting the persistent arterial duct in newborns and infants with suprasystemic pulmonary hypertension of various etiologies. J. Heart Lung Transplant. 33, 542–546 (2014).
Baruteau, A. E. et al. Potts shunt in children with idiopathic pulmonary arterial hypertension: long-term results. Ann. Thorac. Surg. 94, 817–824 (2012).
Diller, G. P. & Gatzoulis, M. A. Pulmonary vascular disease in adults with congenital heart disease. Circulation 115, 1039–1050 (2007).
Beghetti, M. & Galiè, N. Eisenmenger syndrome a clinical perspective in a new therapeutic era of pulmonary arterial hypertension. J. Am. Coll. Cardiol. 53, 733–740 (2009).
Hopkins, W. E., Ochoa, L. L., Richardson, G. W. & Trulock, E. P. Comparison of the hemodynamics and survival of adults with severe primary pulmonary hypertension or Eisenmenger syndrome. J. Heart Lung Transplant. 15, 100–105 (1996).
Barst, R. J., Ivy, D. D., Foreman, A. J., McGoon, M. D. & Rosenzweig, E. B. Four- and seven-year outcomes of patients with congenital heart disease-associated pulmonary arterial hypertension (from the REVEAL Registry). Am. J. Cardiol. 113, 147–155 (2014).
Ivy, D. D. et al. Pediatric pulmonary hypertension. J. Am. Coll. Cardiol. 62 (Suppl.), D117–D126 (2013).
Beghetti, M. et al. Diagnostic evaluation of paediatric pulmonary hypertension in current clinical practice. Eur. Respir. J. 42, 689–700 (2013).
Yung, D. et al. Outcomes in children with idiopathic pulmonary arterial hypertension. Circulation 110, 660–665 (2004).
Barst, R. J., Maislin, G. & Fishman, A. P. Vasodilator therapy for primary pulmonary hypertension in children. Circulation 99, 1197–1208 (1999).
Barst, R. J., Agnoletti, G., Fraisse, A., Baldassarre, J. & Wessel, D. L. Vasodilator testing with nitric oxide and/or oxygen in pediatric pulmonary hypertension. Pediatr. Cardiol. 31, 598–606 (2010).
Douwes, J. M. et al. Acute pulmonary vasodilator response in paediatric and adult pulmonary arterial hypertension: occurrence and prognostic value when comparing three response criteria. Eur. Heart J. 32, 3137–3146 (2011).
Fraisse, A. et al. Characteristics and prospective 2-year follow-up of children with pulmonary arterial hypertension in France. Arch. Cardiovasc. Dis. 103, 66–74 (2010).
Rich, S., Kaufmann, E. & Levy, P. S. The effect of high doses of calcium-channel blockers on survival in primary pulmonary hypertension. N. Engl. J. Med. 327, 76–81 (1992).
Sitbon, O. et al. Inhaled nitric oxide as a screening agent for safely identifying responders to oral calcium-channel blockers in primary pulmonary hypertension. Eur. Respir. J. 12, 265–270 (1998).
Humbert, M. et al. Pulmonary arterial hypertension in France: results from a national registry. Am. J. Respir. Crit. Care Med. 173, 1023–1030 (2006).
Sitbon, O. et al. Long-term response to calcium channel blockers in idiopathic pulmonary arterial hypertension. Circulation 111, 3105–3111 (2005).
Apitz, C. et al. Assessment of pulmonary endothelial function during invasive testing in children and adolescents with idiopathic pulmonary arterial hypertension. J. Am. Coll. Cardiol. 60, 157–164 (2012).
Galiè, N. et al. Updated treatment algorithm of pulmonary arterial hypertension. J. Am. Coll. Cardiol. 62 (Suppl.), D60–D72 (2013).
Barst, R. J., Ertel, S. I., Beghetti, M. & Ivy, D. D. Pulmonary arterial hypertension: a comparison between children and adults. Eur. Respir. J. 37, 665–677 (2011).
Galié, N., Manes, A. & Branzi, A. The endothelin system in pulmonary arterial hypertension. Cardiovasc. Res. 61, 227–237 (2004).
Rubin, L. J. et al. Bosentan therapy for pulmonary arterial hypertension. N. Engl. J. Med. 346, 896–903 (2002).
Channick, R. N. et al. Effects of the dual endothelin-receptor antagonist bosentan in patients with pulmonary hypertension: a randomised placebo-controlled study. Lancet 358, 1119–1123 (2001).
Beghetti, M. Current treatment options in children with pulmonary arterial hypertension and experiences with oral bosentan. Eur. J. Clin. Invest. 36 (Suppl. 3), 16–24 (2006).
Barst, R. J. et al. Pharmacokinetics, safety, and efficacy of bosentan in pediatric patients with pulmonary arterial hypertension. Clin. Pharmacol. Ther. 73, 372–382 (2003).
Beghetti, M. et al. Pharmacokinetic and clinical profile of a novel formulation of bosentan in children with pulmonary arterial hypertension: the FUTURE-1 study. Br. J. Clin. Pharmacol. 68, 948–955 (2009).
Beghetti, M. Bosentan in pediatric patients with pulmonary arterial hypertension. Curr. Vasc. Pharmacol. 7, 225–233 (2009).
Hislop, A. A., Moledina, S., Foster, H., Schulze-Neick, I. & Haworth, S. G. Long-term efficacy of bosentan in treatment of pulmonary arterial hypertension in children. Eur. Respir. J. 38, 70–77 (2011).
Rosenzweig, E. B. et al. Effects of long-term bosentan in children with pulmonary arterial hypertension. J. Am. Coll. Cardiol. 46, 697–704 (2005).
Galiè, N. et al. Ambrisentan therapy for pulmonary arterial hypertension. J. Am. Coll. Cardiol. 46, 529–535 (2005).
Galiè, N. et al. Ambrisentan for the treatment of pulmonary arterial hypertension: results of the ambrisentan in pulmonary arterial hypertension, randomized, double-blind, placebo-controlled, multicenter, efficacy (ARIES) study 1 and 2. Circulation 117, 3010–3019 (2008).
McGoon, M. D. et al. Ambrisentan therapy in patients with pulmonary arterial hypertension who discontinued bosentan or sitaxsentan due to liver function test abnormalities. Chest 135, 122–129 (2009).
Takatsuki, S. et al. Clinical safety, pharmacokinetics, and efficacy of ambrisentan therapy in children with pulmonary arterial hypertension. Pediatr. Pulmonol. 48, 27–34 (2013).
Hoeper, M. M., Olsson, K. M., Schneider, A. & Golpon, H. Severe hepatitis associated with sitaxentan and response to glucocorticoid therapy. Eur. Respir. J. 33, 1518–1519 (2009).
Galiè, N., Hoeper, M. M., Gibbs, J. S. & Simonneau, G. Liver toxicity of sitaxentan in pulmonary arterial hypertension. Eur. Respir. J. 37, 475–476 (2011).
Rubin, L. J. et al. Long-term treatment with sildenafil citrate in pulmonary arterial hypertension: the SUPER-2 study. Chest 140, 1274–1283 (2011).
Humpl, T. et al. Sildenafil therapy for neonatal and childhood pulmonary hypertensive vascular disease. Cardiol. Young 21, 187–193 (2011).
Humpl, T., Reyes, J. T., Holtby, H., Stephens, D. & Adatia, I. Beneficial effect of oral sildenafil therapy on childhood pulmonary arterial hypertension: twelve-month clinical trial of a single-drug, open-label, pilot study. Circulation 111, 3274–3280 (2005).
Apitz, C., Reyes, J. T., Holtby, H., Humpl, T. & Redington, A. N. Pharmacokinetic and hemodynamic responses to oral sildenafil during invasive testing in children with pulmonary hypertension. J. Am. Coll. Cardiol. 55, 1456–1462 (2010).
Barst, R. J. et al. A randomized, double-blind, placebo-controlled, dose-ranging study of oral sildenafil citrate in treatment-naive children with pulmonary arterial hypertension. Circulation 125, 324–334 (2012).
Barst, R. J. et al. STARTS-2: long-term survival with oral Sildenafil monotherapy in treatment-naive pediatric pulmonary arterial hypertension. Circulation 129, 1914–1923 (2014).
U.S. Department of Health & Human Services. U.S. FDA. Revatio (sildenafil): Drug Safety Communication—Recommendation Against Use in Children [online], (2012).
Abman, S. H. et al. Implications of the U.S. Food and Drug Administration warning against the use of sildenafil for the treatment of pediatric pulmonary hypertension. Am. J. Respir. Crit. Care Med. 187, 572–575 (2013).
McElhinney, D. B. A new START for sildenafil in pediatric pulmonary hypertension: reframing the dose-survival relationship in the STARTS-2 trial. Circulation 129, 1905–1908 (2014).
Samiee-Zafarghandy, S., Smith, P. B. & van den Anker, J. N. Safety of sildenafil in infants. Pediatr. Crit. Care Med. 15, 362–368 (2014).
Wardle, A. J. & Tulloh, R. M. Paediatric pulmonary hypertension and sildenafil: current practice and controversies. Arch. Dis. Child. Educ. Pract. Ed. 98, 141–147 (2013).
U.S. Department of Health & Human Services. U.S. FDA. FDA Drug Safety Communication: FDA clarifies Warning about Pediatric Use of Revatio (sildenafil) for Pulmonary Arterial Hypertension [online], (2014).
Nagendran, J. et al. Phosphodiesterase type 5 is highly expressed in the hypertrophied human right ventricle, and acute inhibition of phosphodiesterase type 5 improves contractility. Circulation 116, 238–248 (2007).
Steinhorn, R. H. et al. Intravenous sildenafil in the treatment of neonates with persistent pulmonary hypertension. J. Pediatr. 155, 841–847.e1 (2009).
Fraisse, A. et al. Intravenous sildenafil for postoperative pulmonary hypertension in children with congenital heart disease. Intensive Care Med. 37, 502–509 (2011).
Oudiz, R. J. et al. Tadalafil for the treatment of pulmonary arterial hypertension: a double-blind 52-week uncontrolled extension study. J. Am. Coll. Cardiol. 60, 768–774 (2012).
Takatsuki, S., Calderbank, M. & Ivy, D. D. Initial experience with tadalafil in pediatric pulmonary arterial hypertension. Pediatr. Cardiol. 33, 683–688 (2012).
Douwes, J. M. et al. Sildenafil add-on therapy in paediatric pulmonary arterial hypertension, experiences of a national referral centre. Heart 100, 224–230 (2014).
Kemp, K. et al. Usefulness of first-line combination therapy with epoprostenol and bosentan in pulmonary arterial hypertension: an observational study. J. Heart Lung Transplant. 31, 150–158 (2012).
Barst, R. J. et al. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension. N. Engl. J. Med. 334, 296–301 (1996).
Rubin, L. J. et al. Treatment of primary pulmonary hypertension with continuous intravenous prostacyclin (epoprostenol): results of a randomized trial. Ann. Intern. Med. 112, 485–491 (1990).
Barst, R. J. et al. Survival in primary pulmonary hypertension with long-term continuous intravenous prostacyclin. Ann. Intern. Med. 121, 409–415 (1994).
Olschewski, H. et al. Inhaled iloprost for severe pulmonary hypertension. N. Engl. J. Med. 347, 322–329 (2002).
Voswinckel, R. et al. Favorable effects of inhaled treprostinil in severe pulmonary hypertension: results from randomized controlled pilot studies. J. Am. Coll. Cardiol. 48, 1672–1681 (2006).
Simonneau, G. et al. Continuous subcutaneous infusion of treprostinil, a prostacyclin analogue, in patients with pulmonary arterial hypertension: a double-blind, randomized, placebo-controlled trial. Am. J. Respir. Crit. Care Med. 165, 800–804 (2002).
Ivy, D. D. et al. Short- and long-term effects of inhaled iloprost therapy in children with pulmonary arterial hypertension. J. Am. Coll. Cardiol. 51, 161–169 (2008).
Krishnan, U. et al. Effectiveness and safety of inhaled treprostinil for the treatment of pulmonary arterial hypertension in children. Am. J. Cardiol. 110, 1704–1709 (2012).
Levy, M. et al. Add-on therapy with subcutaneous treprostinil for refractory pediatric pulmonary hypertension. J. Pediatr. 158, 584–588 (2011).
Siehr, S. L. et al. Children with pulmonary arterial hypertension and prostanoid therapy: long-term hemodynamics. J. Heart Lung Transplant. 32, 546–552 (2013).
Galiè, N. et al. Guidelines for the diagnosis and treatment of pulmonary hypertension: the Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS), endorsed by the International Society of Heart and Lung Transplantation (ISHLT). Eur. Heart J. 30, 2493–2537 (2009).
Nickel, N. et al. The prognostic impact of follow-up assessments in patients with idiopathic pulmonary arterial hypertension. Eur. Respir. J. 39, 589–596 (2012).
Gomberg-Maitland, M. et al. New trial designs and potential therapies for pulmonary artery hypertension. J. Am. Coll. Cardiol. 62 (Suppl.), D82–D91 (2013).
Gatfield, J. et al. Slow receptor dissociation kinetics differentiate macitentan from other endothelin receptor antagonists in pulmonary arterial smooth muscle cells. PLoS ONE 7, e47662 (2012).
Dingemanse, J., Sidharta, P. N., Maddrey, W. C., Rubin, L. J. & Mickail, H. Efficacy, safety and clinical pharmacology of macitentan in comparison to other endothelin receptor antagonists in the treatment of pulmonary arterial hypertension. Expert Opin. Drug Saf. 13, 391–405 (2014).
Pulido, T. et al. Macitentan and morbidity and mortality in pulmonary arterial hypertension. N. Engl. J. Med. 369, 809–818 (2013).
Ghofrani, H. A. et al. Riociguat for the treatment of pulmonary arterial hypertension. N. Engl. J. Med. 369, 330–340 (2013).
Hoeper, M. M. et al. Imatinib mesylate as add-on therapy for pulmonary arterial hypertension: results of the randomized IMPRES study. Circulation 127, 1128–1138 (2013).
Simonneau, G. et al. Selexipag: an oral, selective prostacyclin receptor agonist for the treatment of pulmonary arterial hypertension. Eur. Respir. J. 40, 874–880 (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Pullamsetti, S. S. et al. Novel and emerging therapies for pulmonary hypertension. Am. J. Respir. Crit. Care Med. 189, 394–400 (2014).
Wang, X. X. et al. Transplantation of autologous endothelial progenitor cells may be beneficial in patients with idiopathic pulmonary arterial hypertension: a pilot randomized controlled trial. J. Am. Coll. Cardiol. 49, 1566–1571 (2007).
Zhu, J. H. et al. Safety and efficacy of autologous endothelial progenitor cells transplantation in children with idiopathic pulmonary arterial hypertension: open-label pilot study. Pediatr. Transplant. 12, 650–655 (2008).
Fujio, H. et al. Carvedilol inhibits proliferation of cultured pulmonary artery smooth muscle cells of patients with idiopathic pulmonary arterial hypertension. J. Cardiovasc. Pharmacol. 47, 250–255 (2006).
Bogaard, H. J. et al. Adrenergic receptor blockade reverses right heart remodeling and dysfunction in pulmonary hypertensive rats. Am. J. Respir. Crit. Care Med. 182, 652–660 (2010).
Alkon, J. et al. Usefulness of the right ventricular systolic to diastolic duration ratio to predict functional capacity and survival in children with pulmonary arterial hypertension. Am. J. Cardiol. 106, 430–436 (2010).
Thenappan, T., Roy, S. S., Duval, S., Glassner-Kolmin, C. & Gomberg-Maitland, M. Beta-blocker therapy is not associated with adverse outcomes in patients with pulmonary arterial hypertension: a propensity score analysis. Circ. Heart Fail. 7, 903–910 (2014).
Grinnan, D. et al. Treatment of group I pulmonary arterial hypertension with carvedilol is safe. Am. J. Respir. Crit. Care Med. 189, 1562–1564 (2014).
Olsson, K. M. et al. Anticoagulation and survival in pulmonary arterial hypertension: results from the Comparative, Prospective Registry of Newly Initiated Therapies for Pulmonary Hypertension (COMPERA). Circulation 129, 57–65 (2014).
Rich, S. et al. The short-term effects of digoxin in patients with right ventricular dysfunction from pulmonary hypertension. Chest 114, 787–792 (1998).
Grunig, E. et al. Safety and efficacy of exercise training in various forms of pulmonary hypertension. Eur. Respir. J. 40, 84–92 (2012).
Grunig, E. et al. Effect of exercise and respiratory training on clinical progression and survival in patients with severe chronic pulmonary hypertension. Respiration 81, 394–401 (2011).
Mereles, D. et al. Exercise and respiratory training improve exercise capacity and quality of life in patients with severe chronic pulmonary hypertension. Circulation 114, 1482–1489 (2006).
Becker-Grunig, T. et al. Efficacy of exercise training in pulmonary arterial hypertension associated with congenital heart disease. Int. J. Cardiol. 168, 375–381 (2013).
Raymond, R. J. et al. Echocardiographic predictors of adverse outcomes in primary pulmonary hypertension. J. Am. Coll. Cardiol. 39, 1214–1219 (2002).
Benza, R. L. et al. An evaluation of long-term survival from time of diagnosis in pulmonary arterial hypertension from the REVEAL Registry. Chest 142, 448–456 (2012).
Kidd, L. et al. Second natural history study of congenital heart defects: results of treatment of patients with ventricular septal defects. Circulation 87 (Suppl.), I38–I51 (1993).
Saha, A. et al. Prognosis for patients with Eisenmenger syndrome of various aetiology. Int. J. Cardiol. 45, 199–207 (1994).
Diller, G. P. et al. Presentation, survival prospects, and predictors of death in Eisenmenger syndrome: a combined retrospective and case-control study. Eur. Heart J. 27, 1737–1742 (2006).
Hopkins, W. E. & Waggoner, A. D. Severe pulmonary hypertension without right ventricular failure: the unique hearts of patients with Eisenmenger syndrome. Am. J. Cardiol. 89, 34–38 (2002).
Kerstein, D. et al. Blade balloon atrial septostomy in patients with severe primary pulmonary hypertension. Circulation 91, 2028–2035 (1995).
Nihill, M. R., O'Laughlin, M. P. & Mullins, C. E. Effects of atrial septostomy in patients with terminal cor pulmonale due to pulmonary vascular disease. Cathet. Cardiovasc. Diagn. 24, 166–172 (1991).
Sandoval, J. et al. Graded balloon dilation atrial septostomy in severe primary pulmonary hypertension: a therapeutic alternative for patients nonresponsive to vasodilator treatment. J. Am. Coll. Cardiol. 32, 297–304 (1998).
Micheletti, A. et al. Role of atrial septostomy in the treatment of children with pulmonary arterial hypertension. Heart 92, 969–972 (2006).
Law, M. A., Grifka, R. G., Mullins, C. E. & Nihill, M. R. Atrial septostomy improves survival in select patients with pulmonary hypertension. Am. Heart J. 153, 779–784 (2007).
Keogh, A. M. et al. Interventional and surgical modalities of treatment in pulmonary hypertension. J. Am. Coll. Cardiol. 54 (Suppl.), S67–S77 (2009).
Lammers, A. E., Haworth, S. G. & Diller, G. P. Atrial septostomy in patients with pulmonary hypertension: should it be recommended? Expert Rev. Respir. Med. 5, 363–376 (2011).
Rich, S. & Lam, W. Atrial septostomy as palliative therapy for refractory primary pulmonary hypertension. Am. J. Cardiol. 51, 1560–1561 (1983).
Ciarka, A. et al. Atrial septostomy decreases sympathetic overactivity in pulmonary arterial hypertension. Chest 131, 1831–1837 (2007).
Velez-Roa, S. et al. Increased sympathetic nerve activity in pulmonary artery hypertension. Circulation 110, 1308–1312 (2004).
Sandoval, J., Rothman, A. & Pulido, T. Atrial septostomy for pulmonary hypertension. Clin. Chest Med. 22, 547–560 (2001).
Diller, G. P. et al. A modelling study of atrial septostomy for pulmonary arterial hypertension, and its effect on the state of tissue oxygenation and systemic blood flow. Cardiol. Young 20, 25–32 (2010).
Espinola-Zavaleta, N. et al. Echocardiographic evaluation of patients with primary pulmonary hypertension before and after atrial septostomy. Echocardiography 16, 625–634 (1999).
O'Byrne, M. L., Rosenzweig, E. S. & Barst, R. J. The effect of atrial septostomy on the concentration of brain-type natriuretic peptide in patients with idiopathic pulmonary arterial hypertension. Cardiol. Young 17, 557–559 (2007).
Klepetko, W. et al. Interventional and surgical modalities of treatment for pulmonary arterial hypertension. J. Am. Coll. Cardiol. 43 (Suppl. S), 73S–80S (2004).
Rich, S., Dodin, E. & McLaughlin, V. V. Usefulness of atrial septostomy as a treatment for primary pulmonary hypertension and guidelines for its application. Am. J. Cardiol. 80, 369–371 (1997).
Potts, W. J., Smith, S. & Gibson, S. Anastomosis of the aorta to a pulmonary artery; certain types in congenital heart disease. J. Am. Med. Assoc. 132, 627–631 (1946).
Esch, J. J. et al. Transcatheter Potts shunt creation in patients with severe pulmonary arterial hypertension: initial clinical experience. J. Heart Lung Transplant. 32, 381–387 (2013).
Guo, K. et al. Anatomical considerations for the development of a new transcatheter aortopulmonary shunt device in patients with severe pulmonary arterial hypertension. Pulm. Circ. 3, 639–646 (2013).
Sayadpour Zanjani, K. Radiofrequency perforation may increase the safety of transcatheter Potts shunt creation. J. Heart Lung Transplant. 32, 938 (2013).
Michel-Behnke, I. et al. Stenting of the ductus arteriosus and banding of the pulmonary arteries: basis for various surgical strategies in newborns with multiple left heart obstructive lesions. Heart 89, 645–650 (2003).
Michel-Behnke, I. et al. Stent implantation in the ductus arteriosus for pulmonary blood supply in congenital heart disease. Catheter. Cardiovasc. Interv. 61, 242–252 (2004).
Schranz, D. et al. Fifteen-year single center experience with the “Giessen hybrid” approach for hypoplastic left heart and variants: current strategies and outcomes. Pediatr. Cardiol. http://dx.doi.org/10.1007/s00246-014-1015-2.
Akinturk, H. et al. Hybrid transcatheter-surgical palliation: basis for univentricular or biventricular repair: the Giessen experience. Pediatr. Cardiol. 28, 79–87 (2007).
Akintuerk, H. et al. Stenting of the arterial duct and banding of the pulmonary arteries: basis for combined Norwood stage I and II repair in hypoplastic left heart. Circulation 105, 1099–1103 (2002).
Schranz, D. et al. Stent implantation of the arterial duct in newborns with a truly duct-dependent pulmonary circulation: a single-center experience with emphasis on aspects of the interventional technique. J. Interv. Cardiol. 23, 581–588 (2010).
Boudjemline, Y. et al. Patent ductus arteriosus stenting (transcatheter Potts shunt) for palliation of suprasystemic pulmonary arterial hypertension: a case series. Circ. Cardiovasc. Interv. 6, e18–e20 (2013).
Petersen, C., Helvind, M., Jensen, T. & Andersen, H. O. Potts shunt in a child with end-stage pulmonary hypertension after late repair of ventricular septal defect. World J. Pediatr. Congenit. Heart Surg. 4, 286–289 (2013).
Bui, M. T. et al. Surgical palliation of primary pulmonary arterial hypertension by a unidirectional valved Potts anastomosis in an animal model. J. Thorac. Cardiovasc. Surg. 142, 1223–1228 (2011).
van Loon, R. L. et al. Pediatric pulmonary hypertension in the Netherlands: epidemiology and characterization during the period 1991 to 2005. Circulation 124, 1755–1764 (2011).
Cavigelli-Brunner, A. et al. Outcome of biventricular repair in infants with multiple left heart obstructive lesions. Pediatr. Cardiol. 33, 506–512 (2012).
Brauner, R. A., Laks, H., Drinkwater, D. C. Jr, Scholl, F. & McCaffery, S. Multiple left heart obstructions (Shone's anomaly) with mitral valve involvement: long-term surgical outcome. Ann. Thorac. Surg. 64, 721–729 (1997).
Malhotra, S. P. et al. Outcomes of reparative and transplantation strategies for multilevel left heart obstructions with mitral stenosis. Ann. Thorac. Surg. 86, 1305–1309 (2008).
Vlahos, A. P., Lock, J. E., McElhinney, D. B. & van der Velde, M. E. Hypoplastic left heart syndrome with intact or highly restrictive atrial septum: outcome after neonatal transcatheter atrial septostomy. Circulation 109, 2326–2330 (2004).
Vida, V. L. et al. Hypoplastic left heart syndrome with intact or highly restrictive atrial septum: surgical experience from a single center. Ann. Thorac. Surg. 84, 581–585 (2007).
Rychik, J., Rome, J. J., Collins, M. H., DeCampli, W. M. & Spray, T. L. The hypoplastic left heart syndrome with intact atrial septum: atrial morphology, pulmonary vascular histopathology and outcome. J. Am. Coll. Cardiol. 34, 554–560 (1999).
Latus, H., Yerebakan, C., Schranz, D. & Akintuerk, H. Right ventricular failure from severe pulmonary hypertension after surgery for shone complex: back to fetal physiology with reducting, atrioseptectomy, and bilateral pulmonary arterial banding. J. Thorac. Cardiovasc. Surg. 148, e226–e228 (2014).
Davlouros, P. A. et al. Right ventricular function in adults with repaired tetralogy of Fallot assessed with cardiovascular magnetic resonance imaging: detrimental role of right ventricular outflow aneurysms or akinesia and adverse right-to-left ventricular interaction. J. Am. Coll. Cardiol. 40, 2044–2052 (2002).
Frigiola, A., Redington, A. N., Cullen, S. & Vogel, M. Pulmonary regurgitation is an important determinant of right ventricular contractile dysfunction in patients with surgically repaired tetralogy of Fallot. Circulation 110 (Suppl. 1), II153–II157 (2004).
Frigiola, A. et al. Biventricular response after pulmonary valve replacement for right ventricular outflow tract dysfunction: is age a predictor of outcome? Circulation 118 (Suppl.), S182–S190 (2008).
Tobler, D. et al. The left heart after pulmonary valve replacement in adults late after tetralogy of Fallot repair. Int. J. Cardiol. 160, 165–170 (2012).
Gan, C. T. et al. Impaired left ventricular filling due to right-to-left ventricular interaction in patients with pulmonary arterial hypertension. Am. J. Physiol. Heart Circ. Physiol. 290, H1528–H1533 (2006).
Friedberg, M. K. & Redington, A. N. Right versus left ventricular failure: differences, similarities, and interactions. Circulation 129, 1033–1044 (2014).
Damiano, R. J. Jr, La Follette, P. Jr, Cox, J. L., Lowe, J. E. & Santamore, W. P. Significant left ventricular contribution to right ventricular systolic function. Am. J. Physiol. 261, H1514–H1524 (1991).
Apitz, C. et al. Biventricular structural and functional responses to aortic constriction in a rabbit model of chronic right ventricular pressure overload. J. Thorac. Cardiovasc. Surg. 144, 1494–1501 (2012).
Schranz, D. et al. Pulmonary artery banding in infants and young children with left ventricular dilated cardiomyopathy: a novel therapeutic strategy before heart transplantation. J. Heart Lung Transplant. 32, 475–481 (2013).
Burns, K. M. et al. New mechanistic and therapeutic targets for pediatric heart failure: report from a National Heart, Lung, and Blood Institute working group. Circulation 130, 79–86 (2014).
Calkins, H. et al. 2012 HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design: a report of the Heart Rhythm Society (HRS) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. Developed in partnership with the European Heart Rhythm Association (EHRA), a registered branch of the European Society of Cardiology (ESC) and the European Cardiac Arrhythmia Society (ECAS); and in collaboration with the American College of Cardiology (ACC), American Heart Association (AHA), the Asia Pacific Heart Rhythm Society (APHRS), and the Society of Thoracic Surgeons (STS). Endorsed by the governing bodies of the American College of Cardiology Foundation, the American Heart Association, the European Cardiac Arrhythmia Society, the European Heart Rhythm Association, the Society of Thoracic Surgeons, the Asia Pacific Heart Rhythm Society, and the Heart Rhythm Society. Heart Rhythm 9, 632–696.e21 (2012).
Schlaich, M. P. et al. International expert consensus statement: percutaneous transluminal renal denervation for the treatment of resistant hypertension. J. Am. Coll. Cardiol. 62, 2031–2045 (2013).
Chen, S. L. et al. Pulmonary artery denervation to treat pulmonary arterial hypertension: the single-center, prospective, first-in-man PADN-1 study (first-in-man pulmonary artery denervation for treatment of pulmonary artery hypertension). J. Am. Coll. Cardiol. 62, 1092–1100 (2013).
Benden, C. et al. The registry of the International Society for Heart and Lung Transplantation: sixteenth official pediatric lung and heart–lung transplantation report—2013; focus theme: age. J. Heart Lung Transplant. 32, 989–997 (2013).
Trulock, E. P. et al. Registry of the International Society for Heart and Lung Transplantation: twenty-third official adult lung and heart–lung transplantation report—2006. J. Heart Lung Transplant. 25, 880–892 (2006).
Valapour, M. et al. OPTN/SRTR 2011 annual data report: lung. Am. J. Transplant. 13 (Suppl. 1), 149–177 (2013).
Goldstein, B. S., Sweet, S. C., Mao, J., Huddleston, C. B. & Grady, R. M. Lung transplantation in children with idiopathic pulmonary arterial hypertension: an 18-year experience. J. Heart Lung Transplant. 30, 1148–1152 (2011).
Adatia, I. et al. Clinical trials in neonates and children: report of the pulmonary hypertension academic research consortium pediatric advisory committee. Pulm. Circ. 3, 252–266 (2013).
Author information
Authors and Affiliations
Contributions
H.L. and C.A. researched data for the article, and all the authors discussed its content. H.L. and C.A. wrote the manuscript. T.D. and D.S. reviewed and edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Latus, H., Delhaas, T., Schranz, D. et al. Treatment of pulmonary arterial hypertension in children. Nat Rev Cardiol 12, 244–254 (2015). https://doi.org/10.1038/nrcardio.2015.6
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2015.6
This article is cited by
-
In vivo assessment of a single adenine mutation in 5′UTR of Endothelin-1 gene in paediatric cases with severe pulmonary hypertension: an observational study
BMC Research Notes (2021)
-
Therapeutic effects of the selective farnesoid X receptor agonist obeticholic acid in a monocrotaline-induced pulmonary hypertension rat model
Journal of Endocrinological Investigation (2019)
-
Impact of the Right Ventricular Sokolow–Lyon Index in Children with Idiopathic Pulmonary Arterial Hypertension
Pediatric Cardiology (2018)
-
Home Exercise Training in Children and Adolescents with Pulmonary Arterial Hypertension: A Pilot Study
Pediatric Cardiology (2017)
-
Hemodynamic Evaluation of Children with Persistent or Recurrent Pulmonary Arterial Hypertension Following Complete Repair of Congenital Heart Disease
Pediatric Cardiology (2017)