Abstract
Vascular occlusion remains the leading cause of death in Western countries, despite advances made in balloon angioplasty and conventional surgical intervention. Vascular surgery, such as CABG surgery, arteriovenous shunts, and the treatment of congenital anomalies of the coronary artery and pulmonary tracts, requires biologically responsive vascular substitutes. Autografts, particularly saphenous vein and internal mammary artery, are the gold-standard grafts used to treat vascular occlusions. Prosthetic grafts have been developed as alternatives to autografts, but their low patency owing to short-term and intermediate-term thrombosis still limits their clinical application. Advances in vascular tissue engineering technology—such as self-assembling cell sheets, as well as scaffold-guided and decellularized-matrix approaches—promise to produce responsive, living conduits with properties similar to those of native tissue. Over the past decade, vascular tissue engineering has become one of the fastest-growing areas of research, and is now showing some success in the clinic.
Key Points
-
Coronary artery occlusion accounts for ∼50% of deaths from cardiovascular diseases; given the limited number of autologous vessel substitutes, an urgent need for engineered vascular grafts exists
-
Synthetic polymers were investigated as vascular substitutes because of their availability, structural diversity, and mechanical properties; however, mechanical mismatch and adverse host response remain major impediments to their clinical applicability
-
Natural polymers (collagen, elastin, and fibrin) were originally used as vascular scaffolds; synthetic degradable and nondegradable polymers have subsequently been explored and have improved mechanical properties
-
Self-assembled cell sheets have emerged as alternatives to scaffold-based vascular tissue engineering—vascular cells are cultured on a flat surface, and then the cell sheet is rolled around a mandrel
-
Decellularized, natural matrices from allogenic, heterogenic, or xenogenic sources are an alternative form of scaffold—complete cellular removal is followed by recellularization with cells from a patient
-
None of these approaches has produced ideal small-diameter vascular grafts for reconstructive surgery, but they have greatly advanced our understanding of vascular tissue engineering
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
WHO. Cardiovascular diseases (CVDs) [online], (2013).
Population Reference Bureau. World population data sheet 2012 [online], (2012).
Eisenberg, M. J. et al. Outcomes and cost of coronary artery bypass graft surgery in the United States and Canada. Arch. Intern. Med. 165, 1506–1513 (2005).
Matsuo, Y. et al. Plaque characteristics and arterial remodeling in coronary and peripheral arterial systems. Atherosclerosis 223, 365–371 (2012).
Krueger, K. D., Mitra, A. K., Del Core, M. G., Hunter, W. J. 3rd & Agrawal, D. K. A comparison of stent-induced stenosis in coronary and peripheral arteries. J. Clin. Pathol. 59, 575–579 (2006).
Schwann, T. A., Engoren, M., Bonnell, M., Clancy, C. & Habib, R. H. Comparison of late coronary artery bypass graft survival effects of radial artery versus saphenous vein grafting in male and female patients. Ann. Thorac. Surg. 94, 1485–1491 (2012).
Kannan, R. Y., Salacinski, H. J., Butler, P. E., Hamilton, G. & Seifalian, A. M. Current status of prosthetic bypass grafts: a review. J. Biomed. Mater. Res. B Appl. Biomater. 74, 570–581 (2005).
McBane, J. E. et al. Tissue engineering a small diameter vessel substitute: engineering constructs with select biomaterials and cells. Curr. Vasc. Pharmacol. 10, 347–360 (2012).
Nobel Prize. The Nobel Prize in Physiology or Medicine 1912: Alexis Carrel [online], (2013).
Persijn, G. Alexis Carrel. Transpl. Int. 25, 367–368 (2012).
Chlupác, J., Filová, E. & Bacáková, L. Blood vessel replacement: 50 years of development and tissue engineering paradigms in vascular surgery. Physiol. Res. 58 (Suppl. 2), S119–S139 (2009).
Menzoian, J. O., Koshar, A. L. & Rodrigues, N. Alexis Carrel, Rene Leriche, Jean Kunlin, and the history of bypass surgery. J. Vasc. Surg. 54, 571–574 (2011).
Testart, J. Jean Kunlin (1904–1991). Ann. Vasc. Surg. 9 (Suppl.), S1–S6 (1995).
Sabik, J. F. 3rd. Understanding saphenous vein graft patency. Circulation 124, 273–275 (2011).
Gu, C. X., Yang, J. F., Zhang, H. C., Wei, H. & Li, L. K. Off-pump coronary artery bypass grafting using a bilateral internal mammary artery Y graft. J. Geriatr. Cardiol. 9, 247–251 (2012).
Verma, S. et al. Should radial arteries be used routinely for coronary artery bypass grafting? Circulation 110, e40–e46 (2004).
Desai, N. D., Cohen, E. A., Naylor, C. D. & Fremes, S. E. for the Radial Artery Patency Study Investigators. A randomized comparison of radial-artery and saphenous-vein coronary bypass grafts. N. Engl. J. Med. 351, 2302–2309 (2004).
Lee, J. D., Srivastava, M. & Bonatti, J. History and current status of robotic totally endoscopic coronary artery bypass. Circ. J. 76, 2058–2065 (2012).
Levonen, A. L., Vähakangas, E., Koponen, J. K. & Ylä-Herttuala, S. Antioxidant gene therapy for cardiovascular disease: current status and future perspectives. Circulation 117, 2142–2150 (2008).
Akowuah, E. F., Sheridan, P. J., Cooper, G. J. & Newman, C. Preventing saphenous vein graft failure: does gene therapy have a role? Ann. Thorac. Surg. 76, 959–966 (2003).
Owens, C. D., Wake, N., Conte, M. S., Gerhard-Herman, M. & Beckman, J. A. In vivo human lower extremity saphenous vein bypass grafts manifest flow mediated vasodilation. J. Vasc. Surg. 50, 1063–1070 (2009).
Schmitto, J. D., Rajab, T. K. & Cohn, L. H. Prevalence and variability of internal mammary graft use in contemporary multivessel coronary artery bypass graft. Curr. Opin. Cardiol. 25, 609–612 (2010).
Weintraub, W. S., Jones, E. L., Craver, J. M. & Guyton, R. A. Frequency of repeat coronary bypass or coronary angioplasty after coronary artery bypass surgery using saphenous venous grafts. Am. J. Cardiol. 73, 103–112 (1994).
Rashid, S. T., Fuller, B., Hamilton, G. & Seifalian, A. M. Tissue engineering of a hybrid bypass graft for coronary and lower limb bypass surgery. FASEB J. 22, 2084–2089 (2008).
Maisel, W. H. A device for proximal anastomosis of autologous coronary vein grafts: report from the meeting of the Circulatory System Devices Panel of the Food and Drug Administration Center for Devices and Radiologic Health. Circulation 112, 1516–1518 (2005).
Voorhees, A. B. Jr, Jaretzki, A. 3rd & Blakemore, A. H. The use of tubes constructed from vinyon “N” cloth in bridging arterial defects. Ann. Surg. 135, 332–336 (1952).
Abbott, W. M. et al. Evaluation and performance standards for arterial prostheses. J. Vasc. Surg. 21, 746–756 (1993).
Bennion, R. S. et al. Patency of autogenous saphenous vein versus polytetrafluoroethylene grafts in femoropopliteal bypass for advanced ischemia of the extremity. Surg. Gynecol. Obstet. 160, 239–242 (1985).
Desmet, W. et al. Isolated single coronary artery: a review of 50,000 consecutive coronary angiographies. Eur. Heart J. 13, 1637–1640 (1992).
Hoenig, M. R., Campbell, G. R., Rolfe, B. E. & Campbell, J. H. Tissue-engineered blood vessels: alternative to autologous grafts? Arterioscler. Thromb. Vasc. Biol. 25, 1128–1134 (2005).
Baguneid, M. et al. In vivo study of a model tissue-engineered small-diameter vascular bypass graft. Biotechnol. Appl. Biochem. 58, 14–24 (2011).
Desai, M., Seifalian, A. M. & Hamilton, G. Role of prosthetic conduits in coronary artery bypass grafting. Eur. J. Cardiothorac. Surg. 40, 394–398 (2011).
Medtech Insight, LLC. European Markets for Prosthetic Vacular Grafts. Ch. 3. (Medtech Insight, LCC, 2006).
Zilla, P., Bezuidenhout, D. & Human, P. Prosthetic vascular grafts: Wrong models, wrong questions and no healing. Biomaterials 28, 5009–5027 (2007).
Annis, D. Polyether-urethane elastomers for small-diameter arterial prostheses. Life Support Syst. 5, 47–52 (1987).
Grasl, C., Bergmeister, H., Stoiber, M., Schima, H. & Weigel, G. Electrospun polyurethane vascular grafts: in vitro mechanical behavior and endothelial adhesion molecule expression. J. Biomed. Mater. Res. A 93, 716–723 (2010).
Brothers, T. E., Stanley, J. C., Burkel, W. E. & Graham, L. M. Small-caliber polyurethane and polytetrafluoroethylene grafts: a comparative study in a canine aortoiliac model. J. Biomed. Mater. Res. 24, 761–771 (1990).
Tiwari, A., Salacinski, H., Seifalian, A. M. & Hamilton, G. New prostheses for use in bypass grafts with special emphasis on polyurethanes. Cardiovasc. Surg. 10, 191–197 (2002).
Rashid, S. T. et al. Cellular engineering of conduits for coronary and lower limb bypass surgery: role of cell attachment peptides and pre-conditioning in optimising smooth muscle cells (SMC) adherence to compliant poly(carbonate-urea)urethane (MyoLink™) scaffolds. Eur. J. Vasc. Endovasc. Surg. 27, 608–616 (2004).
Seifalian, A. M. et al. In vivo biostability of a poly(carbonate-urea)urethane graft. Biomaterials 24, 2549–2557 (2003).
Shin'oka, T., Imai, Y. & Ikada, Y. Transplantation of a tissue-engineered pulmonary artery. N. Engl. J. Med. 344, 532–533 (2001).
L'Heureux, N. et al. Technology insight: the evolution of tissue-engineered vascular grafts—from research to clinical practice. Nat. Clin. Pract. Cardiovasc. Med. 4, 389–395 (2007).
Rabkin, E. & Schoen, F. J. Cardiovascular tissue engineering. Cardiovasc. Pathol. 11, 305–317 (2002).
Platt, J. L. & Nagayasu, T. Current status of xenotransplantation. Clin. Exp. Pharmacol. Physiol. 26, 1026–1032 (1999).
Nerem, R. M. & Seliktar, D. Vascular tissue engineering. Annu. Rev. Biomed. Eng. 3, 225–243 (2001).
Stegemann, J. P., Kaszuba, S. N. & Rowe, S. L. Review: advances in vascular tissue engineering using protein-based biomaterials. Tissue Eng. 13, 2601–2613 (2007).
Poh, M. et al. Blood vessels engineered from human cells. Lancet 365, 2122–2124 (2005).
McKee, J. A. et al. Human arteries engineered in vitro. EMBO Rep. 4, 633–638 (2003).
Kassem, M., Abdallah, B. M., Yu, Z. T., Ditzel, N. & Burns, J. S. The use of hTERT-immortalized cells in tissue engineering. Cytotechnology 45, 39–46 (2004).
Wang, A. J. et al. Derivation of smooth muscle cells with neural crest origin from human induced pluripotent stem cells. Cells Tissues Organs 195, 5–14 (2012).
Sundaram, S. & Niklason, L. E. Smooth muscle and other cell sources for human blood vessel engineering. Cells Tissues Organs 195, 15–25 (2012).
Kane, N. M. et al. Pluripotent stem cell differentiation into vascular cells: a novel technology with promises for vascular re(generation). Pharmacol. Ther. 129, 29–49 (2011).
Liu, J. Y., Peng, H. F. & Andreadis, S. T. Contractile smooth muscle cells derived from hair-follicle stem cells. Cardiovasc. Res. 79, 24–33 (2008).
Gong, Z. D. & Niklason, L. E. Small-diameter human vessel wall engineered from bone marrow-derived mesenchymal stem cells (hMSCs). FASEB J. 22, 1635–1648 (2008).
Bajpai, V. K. & Andreadis, S. T. Stem cell sources for vascular tissue engineering and regeneration. Tissue Eng. Part B Rev. 18, 405–425 (2012).
Wagner, W. et al. Comparative characteristics of mesenchymal stem cells from human bone marrow, adipose tissue, and umbilical cord blood. Exp. Hematol. 33, 1402–1416 (2005).
Matsumura, G., Miyagawa-Tomita, S., Shin'oka, T., Ikada, Y. & Kurosawa, H. First evidence that bone marrow cells contribute to the construction of tissue-engineered vascular autografts in vivo. Circulation 108, 1729–1734 (2003).
Cho, S. W. et al. Small-diameter blood vessels engineered with bone marrow-derived cells. Ann. Surg. 241, 506–515 (2005).
L'Heureux, N. et al. Human tissue-engineered blood vessels for adult arterial revascularization. Nat. Med. 12, 361–365 (2006).
L'Heureux, N., Paquet, S., Labbe, R., Germain, L. & Auger, F. A. A completely biological tissue-engineered human blood vessel. FASEB J. 12, 47–56 (1998).
Peck, M., Dusserre, N., McAllister, T. N. & L'Heureux, N. Tissue engineering by self-assembly. Materials Today 14, 218–224 (2011).
Marelli, B. et al. Collagen-reinforced electrospun silk fibroin tubular construct as small calibre vascular graft. Macromol. Biosci. 12, 1566–1574 (2012).
Yao, L., Swartz, D. D., Gugino, S. F., Russell, J. A. & Andreadis, S. T. Fibrin-based tissue-engineered blood vessels: differential effects of biomaterial and culture parameters on mechanical strength and vascular reactivity. Tissue Eng. 11, 991–1003 (2005).
Peng, H. F., Liu, J. Y., Andreadis, S. T. & Swartz, D. D. Hair follicle-derived smooth muscle cells and small intestinal submucosa for engineering mechanically robust and vasoreactive vascular media. Tissue Eng. Part A 17, 981–990 (2011).
Patterson, J. T. et al. Tissue-engineered vascular grafts for use in the treatment of congenital heart disease: from the bench to the clinic and back again. Regen. Med. 7, 409–419 (2012).
Heine, J. et al. Tissue engineering human small-caliber autologous vessels using a xenogenous decellularized connective tissue matrix approach: preclinical comparative biomechanical studies. Artif. Organs 35, 930–940 (2011).
Quint, C., Arief, M., Muto, A., Dardik, A. & Niklason, L. E. Allogeneic human tissue-engineered blood vessel. J. Vasc. Surg. 55, 790–798 (2012).
Birchall, M. & Hamilton, G. Tissue-engineered vascular replacements for children. Lancet 380, 197–198 (2012).
Olausson, M. et al. Transplantation of an allogeneic vein bioengineered with autologous stem cells: a proof-of-concept study. Lancet 380, 230–237 (2012).
Gwyther, T. A., Hu, J. Z., Billiar, K. L. & Rolle, M. W. Directed cellular self-assembly to fabricate cell-derived tissue rings for biomechanical analysis and tissue engineering. J. Vis. Exp. e3366 (2011).
Norotte, C., Marga, F. S., Niklason, L. E. & Forgacs, G. Scaffold-free vascular tissue engineering using bioprinting. Biomaterials 30, 5910–5917 (2009).
Peck, M., Gebhart, D., Dusserre, N., McAllister, T. N. & L'Heureux, N. The evolution of vascular tissue engineering and current state of the art. Cells Tissues Organs 195, 144–158 (2012).
Peck, M. K. et al. New biological solutions for hemodialysis access. J. Vasc. Access 12, 185–192 (2011).
Song, L., Wang, L., Shah, P. K., Chaux, A. & Sharifi, B. G. Bioengineered vascular graft grown in the mouse peritoneal cavity. J. Vasc. Surg. 52, 994–1002 (2010).
Campbell, J. H., Efendy, J. L. & Campbell, G. R. Novel vascular graft grown within recipient's own peritoneal cavity. Circ. Res. 85, 1173–1178 (1999).
Patel, A., Fine, B., Sandig, M. & Mequanint, K. Elastin biosynthesis: the missing link in tissue-engineered blood vessels. Cardiovasc. Res. 71, 40–49 (2006).
Tranquillo, R. T. The tissue-engineered small-diameter artery. Ann. N. Y. Acad. Sci. 961, 251–254 (2002).
Kim, B. S. & Mooney, D. J. Development of biocompatible synthetic extracellular matrices for tissue engineering. Trends Biotechnol. 16, 224–230 (1998).
Zhang, W. J., Liu, W., Cui, L. & Cao, Y. Tissue engineering of blood vessel. J. Cell. Mol. Med. 11, 945–957 (2007).
Vats, A., Tolley, N. S., Polak, J. M. & Gough, J. E. Scaffolds and biomaterials for tissue engineering: a review of clinical applications. Clin. Otolaryngol. Allied Sci. 28, 165–172 (2003).
Berglund, J. D. & Galis, Z. S. Designer blood vessels and therapeutic revascularization. Br. J. Pharmacol. 140, 627–636 (2003).
Martin, I., Wendt, D. & Heberer, M. The role of bioreactors in tissue engineering. Trends Biotechnol. 22, 80–86 (2004).
Nilsang, S. et al. Three-dimensional culture for monoclonal antibody production by hybridoma cells immobilized in macroporous gel particles. Biotechnol. Prog. 24, 1122–1131 (2008).
Nilsang, S. et al. Monoclonal antibody production using a new supermacroporous cryogel bioreactor. Biotechnol. Prog. 23, 932–939 (2007).
Isenberg, B. C., Williams, C. & Tranquillo, R. T. Small-diameter artificial arteries engineered in vitro. Circ. Res. 98, 25–35 (2006).
Bilodeau, K. & Mantovani, D. Bioreactors for tissue engineering: focus on mechanical constraints: a comparative review. Tissue Eng. 12, 2367–2383 (2006).
Riha, G. M., Lin, P. H., Lumsden, A. B., Yao, Q. & Chen, C. Roles of hemodynamic forces in vascular cell differentiation. Ann. Biomed. Eng. 33, 772–779 (2005).
Bilodeau, K., Couet, F., Boccafoschi, F. & Mantovani, D. Design of a perfusion bioreactor specific to the regeneration of vascular tissues under mechanical stresses. Artif. Organs 29, 906–912 (2005).
Lee, A. A., Graham, D. A., Dela Cruz, S., Ratcliffe, A. & Karlon, W. J. Fluid shear stress-induced alignment of cultured vascular smooth muscle cells. J. Biomech. Eng. 124, 37–43 (2002).
Zhao, S. et al. Synergistic effects of fluid shear stress and cyclic circumferential stretch on vascular endothelial cell morphology and cytoskeleton. Arterioscler. Thromb. Vasc. Biol. 15, 1781–1786 (1995).
Davies, P. F. Flow-mediated endothelial mechanotransduction. Physiol. Rev. 75, 519–560 (1995).
Weinberg, C. B. & Bell, E. A blood vessel model constructed from collagen and cultured vascular cells. Science 231, 397–400 (1986).
Niklason, L. E. et al. Functional arteries grown in vitro. Science 284, 489–493 (1999).
Niklason, L. E. Medical technology: replacement arteries made to order. Science 286, 1493–1494 (1999).
Kolpakov, V., Rekhter, M. D., Gordon, D., Wang, W. H. & Kulik, T. J. Effect of mechanical forces on growth and matrix protein synthesis in the in vitro pulmonary artery: analysis of the role of individual cell types. Circ. Res. 77, 823–831 (1995).
Jackson, Z. S., Gotlieb, A. I. & Langille, B. L. Wall tissue remodeling regulates longitudinal tension in arteries. Circ. Res. 90, 918–925 (2002).
Kanda, K. & Matsuda, T. Behavior of arterial wall cells cultured on periodically stretched substrates. Cell Transplant. 2, 475–484 (1993).
Hirai, J. & Matsuda, T. Self-organized, tubular hybrid vascular tissue composed of vascular cells and collagen for low-pressure-loaded venous system. Cell Transplant. 4, 597–608 (1995).
Hirai, J. & Matsuda, T. Venous reconstruction using hybrid vascular tissue composed of vascular cells and collagen: tissue regeneration process. Cell Transplant. 5, 93–105 (1996).
Bilodeau, K., Couet, F., Boccafoschi, F. & Mantovani, D. Design of a perfusion bioreactor specific to the regeneration of vascular tissues under mechanical stresses. Artif. Organs 29, 906–912 (2005).
Boccafoschi, F., Habermehl, J., Vesentini, S. & Mantovani, D. Biological performances of collagen-based scaffolds tor vascular tissue engineering. Biomaterials 26, 7410–7417 (2005).
Crombez, M. et al. Improving arterial prosthesis neo-endothelialization: application of a proactive VEGF construct onto PTFE surfaces. Biomaterials 26, 7402–7409 (2005).
Rajan, N., Habermehl, J., Cote, M. F., Doillon, C. J. & Mantovani, D. Preparation of ready-to-use, storable and reconstituted type I collagen from rat tail tendon for tissue engineering applications. Nat. Protoc. 1, 2753–2758 (2006).
Boccafoschi, F., Rajan, N., Habermehl, J. & Mantovani, D. Preparation and characterization of a scaffold for vascular tissue engineering by direct-assembling of collagen and cells in a cylindrical geometry. Macromol. Biosci. 7, 719–726 (2007).
Couet, F., Rajan, N. & Mantovani, D. Macromolecular biomaterials for scaffold-based vascular tissue engineering. Macromol. Biosci. 7, 701–718 (2007).
Couet, F., Rajan, N., Vesentini, S. & Mantovani, D. Design of a collagen/silk mechano-compatible composite scaffold for the vascular tissue engineering: focus on compliance. Key Eng. Mater. 334–335, 1169–1172 (2007).
Seliktar, D., Nerem, R. M. & Galis, Z. S. Mechanical strain-stimulated remodeling of tissue-engineered blood vessel constructs. Tissue Eng. 9, 657–666 (2003).
Schutte, S. C., Chen, Z. Z., Brockbank, K. G. M. & Nerem, R. M. Cyclic strain improves strength and function of a collagen-based tissue-engineered vascular media. Tissue Eng. Part A 16, 3149–3157 (2010).
Bulick, A. S. et al. Impact of endothelial cells and mechanical conditioning on smooth muscle cell extracellular matrix production and differentiation. Tissue Eng. Part A 15, 815–825 (2009).
Amadori, L., Rajan, N., Vesentini, S. & Mantovani, D. Atomic force and confocal microscopic studies of collagen-cell-based scaffolds for vascular tissue engineering. Adv. Mater. Res. 15–17, 83–88 (2007).
Couet, F. & Mantovani, D. Experimental validation of a new approach for the development of mechano-compatible composite scaffolds for vascular tissue engineering. J. Mater. Sci. Mater. Med. 19, 2551–2554 (2008).
Couet, F. & Mantovani, D. How to optimise the maturation conditions in a bioreactor? Toward an intelligent bioreactor for vascular tissue engineering [abstract]. Tissue Eng. Part A 14, 858 (2008).
Couet, F. & Mantovani, D. How to optimize maturation in a bioreactor for vascular tissue engineering: focus on a decision algorithm for experimental planning. Ann. Biomed. Eng. 38, 2877–2884 (2010).
Couet, F. & Mantovani, D. A new bioreactor adapts to materials state and builds a growth model for vascular tissue engineering. Artif. Organs 36, 438–445 (2012).
Couet, F., Meghezi, S. & Mantovani, D. Fetal development, mechanobiology and optimal control processes can improve vascular tissue regeneration in bioreactors: an integrative review. Med. Eng. Phys. 34, 269–278 (2012).
Achilli, M., Lagueux, J. & Mantovani, D. On the effects of UV-C and pH on the mechanical behavior, molecular conformation and cell viability of collagen-based scaffold for vascular tissue engineering. Macromol. Biosci. 10, 307–316 (2010).
Achilli, M., Meghezi, S., Lagueux, J. & Mantovani, D. Mechano-compatible collagen-based scaffolds for vascular tissue engineering: low doses of UV affect the viscoelastic behaviour. Tissue Eng. Part A 14, 878–878 (2008).
Rajan, N. et al. Low doses of ultraviolet radiation stimulate cell activity in collagen-based scaffolds. Biotechnol. Prog. 24, 884–889 (2008).
Haisch, A. et al. Preparation of a pure autologous biodegradable fibrin matrix for tissue engineering. Med. Biol. Eng. Comput. 38, 686–689 (2000).
Swartz, D. D., Russell, J. A. & Andreadis, S. T. Engineering of fibrin-based functional and implantable small-diameter blood vessels. Am. J. Physiol. Heart Circ. Physiol. 288, H1451–H1460 (2005).
Celebi, B., Cloutier, M., Balloni, R., Mantovani, D. & Bandiera, A. Human elastin-based recombinant biopolymers improve mesenchymal stem cell differentiation. Macromol. Biosci. 12, 1546–1554 (2012).
Tiwari, A., Salacinski, H., Seifalian, A. M. & Hamilton, G. New prostheses for use in bypass grafts with special emphasis on polyurethanes. Cardiovasc. Surg. 10, 191–197 (2002).
Kim, B. S., Nikolovski, J., Bonadio, J., Smiley, E. & Mooney, D. J. Engineered smooth muscle tissues: regulating cell phenotype with the scaffold. Exp. Cell Res. 251, 318–328 (1999).
Williams, C. & Wick, T. M. Perfusion bioreactor for small diameter tissue-engineered arteries. Tissue Eng. 10, 930–941 (2004).
Yow, K. H., Ingram, J., Korossis, S. A., Ingham, E. & Homer-Vanniasinkam, S. Tissue engineering of vascular conduits. Br. J. Surg. 93, 652–661 (2006).
Hubbell, J. A. Bioactive biomaterials. Curr. Opin. Biotechnol. 10, 123–129 (1999).
Ito, Y., Kajihara, M. & Imanishi, Y. Materials for enhancing cell adhesion by immobilization of cell-adhesive peptide. J. Biomed. Mater. Res. 25, 1325–1337 (1991).
Ye, Q. et al. Scaffold precoating with human autologous extracellular matrix for improved cell attachment in cardiovascular tissue engineering. ASAIO J. 46, 730–733 (2000).
Higgins, S. P., Solan, A. K. & Niklason, L. E. Effects of polyglycolic acid on porcine smooth muscle cell growth and differentiation. J. Biomed. Mater. Res. A 67, 295–302 (2003).
Kurobe, H., Maxfield, M. W., Breuer, C. K. & Shinoka, T. Concise review: tissue-engineered vascular grafts for cardiac surgery: past, present, and future. Stem Cells Transl. Med. 1, 566–571 (2012).
Vogel, G. Tissue engineering: mending the youngest hearts. Science 333, 1088–1089 (2011).
Dolgin, E. Taking tissue engineering to heart. Nat. Med. 17, 1032–1035 (2011).
Rosellini, E., Vozzi, G., Barbani, N., Giusti, P. & Cristallini, C. Three-dimensional microfabricated scaffolds with cardiac extracellular matrix-like architecture. Int. J. Artif. Organs 33, 885–894 (2010).
Quint, C. et al. Decellularized tissue-engineered blood vessel as an arterial conduit. Proc. Natl Acad. Sci. USA 108, 9214–9219 (2011).
Keane, T. J., Londono, R. Turner, N. J. & Badylak, S. F. Consequences of ineffective decellularization of biologic scaffolds on the host response. Biomaterials 33, 1771–1781 (2012).
Mitchell, S., Koh, J., Probhakar, V. & Niklason, L. Decellularized tissue engineered constructs and tissues. US Patent 6,962,814 (2001).
Wolfinbarger, L. Jr, Lange, P., Linhurst Jones, A., Moore, E. & Nolf, B. Process for decellularizing soft-tissue engineered medical implants, and decellularized soft-tissue medical implants produced. US Patent 6,734,018 (2000).
Wolfinbarger, L. Jr, Lange, P., Linhurst Jones, A., Moore, E. & Nolf, B. Process for decellularizing soft-tissue engineered medical implants, and decellularized soft-tissue medical implants produced. US Patent 7,338,757 (2003).
Leyh, R. G. et al. Acellularized porcine heart valve scaffolds for heart valve tissue engineering and the risk of cross-species transmission of porcine endogenous retrovirus. J. Thorac. Cardiovasc. Surg. 126, 1000–1004 (2003).
Pasterkamp, G. et al. Atherosclerotic arterial remodeling and the localization of macrophages and matrix metalloproteases 1, 2 and 9 in the human coronary artery. Atherosclerosis 150, 245–253 (2000).
Villalona, G. A. et al. Cell-seeding techniques in vascular tissue engineering. Tissue Eng. Part B Rev. 16, 341–350 (2010).
Cleary, M. A. et al. Vascular tissue engineering: the next generation. Trends Mol. Med. 18, 394–404 (2012).
Vilain, K. R. et al. Costs and cost-effectiveness of carotid stenting versus endarterectomy for patients at standard surgical risk: results from the Carotid Revascularization Endarterectomy versus Stenting Trial (CREST). Stroke 43, 2408–2416 (2012).
Shinoka, T. et al. Substrate for culture of cardiovascular tissue. US Patent 8,372,433 (2008).
McAllister, T. N., Garrido, S. & L'Heureux, N. Guided percutaneous bypass. US Patent 2012/0150092 (2010).
Smith, M. J. et al. Suture-reinforced electrospun polydioxanone-elastin small-diameter tubes for use in vascular tissue engineering: a feasibility study. Acta Biomater. 4, 58–66 (2008).
Author information
Authors and Affiliations
Contributions
All the authors researched data for the article, contributed substantially to discussion of its content, wrote the manuscript, and reviewed/edited the article before submission.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Seifu, D., Purnama, A., Mequanint, K. et al. Small-diameter vascular tissue engineering. Nat Rev Cardiol 10, 410–421 (2013). https://doi.org/10.1038/nrcardio.2013.77
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2013.77
This article is cited by
-
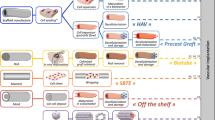
Clopidogrel-loaded vascular grafts prepared using digital light processing 3D printing
Drug Delivery and Translational Research (2023)
-
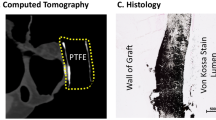
A novel small diameter nanotextile arterial graft is associated with surgical feasibility and safety and increased transmural endothelial ingrowth in pig
Journal of Nanobiotechnology (2022)
-
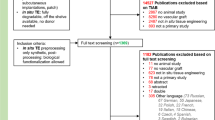
Animal studies for the evaluation of in situ tissue-engineered vascular grafts — a systematic review, evidence map, and meta-analysis
npj Regenerative Medicine (2022)
-
Biological small-calibre tissue engineered blood vessels developed by electrospinning and in-body tissue architecture
Journal of Materials Science: Materials in Medicine (2022)
-
Protection of rat artery grafts from tissue damage by voltage-applied supercooling
Medical Molecular Morphology (2022)