Key Points
-
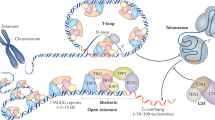
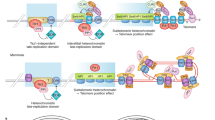
Mammalian telomeres are formed by tandem repeats of the TTAGGG sequence bound by a specialized six-protein complex known as shelterin, which has fundamental roles in the protection of chromosomes and the regulation of telomerase activity at chromosome ends. Excessive telomere shortening and severe telomere uncapping trigger a DNA damage response at chromosome ends, which are then recognized as double-strand breaks. Dysfunctional telomeres can lead to either cancer or ageing pathologies depending on the integrity of the DNA damage response. Studies with mouse models that support a role for these proteins in cancer susceptibility and ageing-related pathologies are discussed in this Review.
-
Telomere dysfunction causes ageing and also constitutes a driving force for cellular transformation by causing genome instability. Molecular mechanisms underlying telomere-induced genomic instability are described.
-
Anti-ageing activity of telomerase has been demonstrated in mice overexpressing TERT genetically engineered to be cancer-resistant by means of enhanced expression of the p53, p16 and ARF tumour suppressors. Telomere-maintenance is the main mechanism underlying the anti-ageing phenotype of TERT-transgenic mice.
-
Telomere-independent functions of TERT have recently been described. Overexpression of TERT is a transcriptional modulator of the Wnt–β-catenin signalling pathway and has RNA-dependent RNA polymerase activity when in a complex with the RNA component of mitochondrial RNA processing endoribonuclease (RMRP).
-
Roles for the shelterin component RAP1 beyond its roles in telomeres have been uncovered. Mammalian RAP1 is involved in subtelomeric gene silencing and transcriptional regulation, and it also acts as a essential modulator of the nuclear factor-κB (NF-κB)-mediated pathway.
-
Telomerase and factors that influence its activity are very attractive targets for the treatment of degenerative diseases and cancer. TPP1 is involved in telomerase recruitment to telomeres. Drugs targeting TPP1 could certainly be a novel strategy for blocking the ultimate goal of telomerase, the lengthening of telomeres.
Abstract
Mammalian telomeres are formed by tandem repeats of the TTAGGG sequence, which are progressively lost with each round of cell division. Telomere protection requires a minimal length of TTAGGG repeats to allow the binding of shelterin, which prevents the activation of a DNA damage response (DDR) at chromosome ends. Telomere elongation is carried out by telomerase. Telomerase can also act as a transcriptional modulator of the Wnt–β-catenin signalling pathway and has RNA-dependent RNA polymerase activity. Dysfunctional telomeres can lead to either cancer or ageing pathologies depending on the integrity of the DDR. This Review discusses the role of telomeric proteins in cancer and ageing through modulating telomere length and protection, as well as regulating gene expression by binding to non-telomeric sites.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
van Steensel, B., Smogorzewska, A. & de Lange, T. TRF2 protects human telomeres from end-to-end fusions. Cell 92, 401–413 (1998).
Celli, G. B. & de Lange, T. DNA processing is not required for ATM-mediated telomere damage response after TRF2 deletion. Nature Cell Biol. 7, 712–718 (2005).
Chin, L. et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 97, 527–538 (1999).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Karlseder, J., Broccoli, D., Dai, Y., Hardy, S. & de Lange, T. p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 283, 1321–1325 (1999).
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003).
Blasco, M. A. The epigenetic regulation of mammalian telomeres. Nature Rev. Genet. 8, 299–309 (2007).
de Lange, T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 19, 2100–2110 (2005).
Palm, W. & de Lange, T. How shelterin protects mammalian telomeres. Annu. Rev. Genet. 42, 301–334 (2008).
Blackburn, E. H. Switching and signaling at the telomere. Cell 106, 661–673 (2001).
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
de Lange, T. How telomeres solve the end-protection problem. Science 326, 948–952 (2009).
Cosme-Blanco, W. & Chang, S. Dual roles of telomere dysfunction in initiation and suppression of tumorigenesis. Exp. Cell Res. 314, 1973–1979 (2008).
Cosme-Blanco, W. et al. Telomere dysfunction suppresses spontaneous tumorigenesis in vivo by initiating p53-dependent cellular senescence. EMBO Rep. 8, 497–503 (2007).
Feldser, D. M. & Greider, C. W. Short telomeres limit tumor progression in vivo by inducing senescence. Cancer Cell 11, 461–469 (2007).
Gonzalez-Suarez, E., Samper, E., Flores, J. M. & Blasco, M. A. Telomerase-deficient mice with short telomeres are resistant to skin tumorigenesis. Nature Genet. 26, 114–117 (2000).
Greenberg, R. A. et al. Short dysfunctional telomeres impair tumorigenesis in the INK4a(delta2/3) cancer-prone mouse. Cell 97, 515–525 (1999).
Herrera, E. et al. Disease states associated with telomerase deficiency appear earlier in mice with short telomeres. EMBO J. 18, 2950–2960 (1999).
Lee, H. W. et al. Essential role of mouse telomerase in highly proliferative organs. Nature 392, 569–574 (1998).
Samper, E. et al. Long-term repopulating ability of telomerase-deficient murine hematopoietic stem cells. Blood 99, 2767–2775 (2002).
Cayuela, M. L., Flores, J. M. & Blasco, M. A. The telomerase RNA component Terc is required for the tumour-promoting effects of Tert overexpression. EMBO Rep. 6, 268–274 (2005).
Flores, I., Cayuela, M. L. & Blasco, M. A. Effects of telomerase and telomere length on epidermal stem cell behavior. Science 309, 1253–1256 (2005).
Gonzalez-Suarez, E. et al. Increased epidermal tumors and increased skin wound healing in transgenic mice overexpressing the catalytic subunit of telomerase, mTERT, in basal keratinocytes. EMBO J. 20, 2619–2630 (2001).
Tomas-Loba, A. et al. Telomerase reverse transcriptase delays aging in cancer-resistant mice. Cell 135, 609–622 (2008). This paper demonstrates for the first time that TERT has an anti-ageing activity and shows that telomerase overexpression in a cancer-resistant genetic background improves the fitness of epithelial barriers and produces a systemic delay in ageing.
Choi, J. et al. TERT promotes epithelial proliferation through transcriptional control of a Myc- and Wnt-related developmental program. PLoS Genet. 4, e10 (2008).
Maida, Y. et al. An RNA-dependent RNA polymerase formed by TERT and the RMRP RNA. Nature 461, 230–235 (2009). This paper demonstrates that the telomerase catalytic subunit TERT exerts an extra-telomeric function by modulating gene transcription.
Park, J. I. et al. Telomerase modulates Wnt signalling by association with target gene chromatin. Nature 460, 66–72 (2009). This papers shows that telomerase modulates Wnt–b-catenin signalling by acting as a cofactor in a b-catenin transcriptional complex. The data presented reinforce the implication of telomerase in stem cell regulation, development and cancer.
Sarin, K. Y. et al. Conditional telomerase induction causes proliferation of hair follicle stem cells. Nature 436, 1048–1052 (2005).
Smogorzewska, A. & de Lange, T. Regulation of telomerase by telomeric proteins. Annu. Rev. Biochem. 73, 177–208 (2004).
Chiang, Y. J., Kim, S. H., Tessarollo, L., Campisi, J. & Hodes, R. J. Telomere-associated protein TIN2 is essential for early embryonic development through a telomerase-independent pathway. Mol. Cell Biol. 24, 6631–6634 (2004).
Hockemeyer, D., Daniels, J. P., Takai, H. & de Lange, T. Recent expansion of the telomeric complex in rodents: Two distinct POT1 proteins protect mouse telomeres. Cell 126, 63–77 (2006).
Karlseder, J. et al. Targeted deletion reveals an essential function for the telomere length regulator Trf1. Mol. Cell Biol. 23, 6533–6541 (2003).
Kibe, T., Osawa, G. A., Keegan, C. E. & de Lange, T. Telomere protection by TPP1 is mediated by POT1a and POT1b. Mol. Cell Biol. 30, 1059–1066 (2010).
Lazzerini Denchi, E., Celli, G. & de Lange, T. Hepatocytes with extensive telomere deprotection and fusion remain viable and regenerate liver mass through endoreduplication. Genes Dev. 20, 2648–2653 (2006).
Wu, L. et al. Pot1 deficiency initiates DNA damage checkpoint activation and aberrant homologous recombination at telomeres. Cell 126, 49–62 (2006).
Martinez, P. & Blasco, M. A. Role of shelterin in cancer and aging. Aging Cell 9, 653–666 (2010).
Martinez, P. et al. Increased telomere fragility and fusions resulting from TRF1 deficiency lead to degenerative pathologies and increased cancer in mice. Genes Dev. 23, 2060–2075 (2009). This paper describes the effect of TRF1 deletion in MEFs and in the stratified epithelia of mice. It demonstrates that TRF1 is required for telomere capping and for proper telomere replication.
Else, T. et al. Genetic p53 deficiency partially rescues the adrenocortical dysplasia phenotype at the expense of increased tumorigenesis. Cancer Cell 15, 465–476 (2009). This paper shows that p53 loss rescues the adrenocortical dysplasia phenotype in mice with a hypomorphic TPP1 mutation but triggers the incidence of carcinomas. It proves the in vivo role of telomere vulnerability in tumorigenesis.
Tejera, A. et al. TPP1 is required for TERT recruitment, telomere elongation during nuclear reprogramming, and normal skin development in mice. Dev. Cell 18, 691–702 (2010). This papers shows that TPP1 is involved in both end protection and telomerase regulation in vivo . It highlights the relevance of telomerase activity in highly proliferative tissues and in the reprogramming of cells to induce pluripotency.
Blasco, M. A. Telomeres and human disease: ageing, cancer and beyond. Nature Rev. Genet. 6, 611–622 (2005).
Munoz, P., Blanco, R., Flores, J. M. & Blasco, M. A. XPF nuclease-dependent telomere loss and increased DNA damage in mice overexpressing TRF2 result in premature aging and cancer. Nature Genet. 37, 1063–1071 (2005). This paper shows that overexpression of TRF2 results in a severe phenotype in the skin in response to light recapitulating the phenotypes observed in xeroderma pigmentosum. These observations provide in vivo links between the NER pathway and telomere maintenance mechanisms.
Poncet, D. et al. Changes in the expression of telomere maintenance genes suggest global telomere dysfunction in B-chronic lymphocytic leukemia. Blood 111, 2388–2391 (2008).
Abreu, E. et al. TIN2-tethered TPP1 recruits human telomerase to telomeres in vivo. Mol. Cell Biol. 30, 2971–2982 (2010). This paper demonstrates that TIN2-anchored TPP1 has a role in telomerase recruitment to telomeres in human cells. It also shows that telomerase recruitment does not depend on POT1 or on interaction of the shelterin complex with the single-stranded region of the telomeres.
Martinez, P. et al. Mammalian Rap1 controls telomere function and gene expression through binding to telomeric and extratelomeric sites. Nature Cell Biol. 12, 768–780 (2010). This paper demonstrates that RAP1 binds to both telomeres and to extratelomeric genomic sites. It also shows that RAP1 is involved in subtelomeric gene silencing and exerts a transcriptional regulatory function.
Teo, H. et al. Telomere-independent Rap1 is an IKK adaptor and regulates NF-kappaB-dependent gene expression. Nature Cell Biol. 12, 758–767 (2010). This paper identifies a cytoplasmatic role of RAP1 and provides a molecular mechanism by which RAP1 exerts a crucial function in the NF-κB signalling pathway, independently of its telomeric function.
Zaug, A. J., Podell, E. R., Nandakumar, J. & Cech, T. R. Functional interaction between telomere protein TPP1 and telomerase. Genes Dev. 24, 613–622 (2010). This paper shows that POT1–TPP1 binding to telomeres activates telomerase repeat addition processivity and that this activation is dependent on specific interaction with the TEN domain of TERT.
Olovnikov, A. M. A theory of marginotomy. The incomplete copying of template margin in enzymic synthesis of polynucleotides and biological significance of the phenomenon. J. Theor. Biol. 41, 181–190 (1973).
Watson, J. D. Origin of concatemeric T7 DNA. Nat New Biol. 239, 197–201 (1972).
Greider, C. W. & Blackburn, E. H. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 43, 405–413 (1985).
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
Flores, I. et al. The longest telomeres: a general signature of adult stem cell compartments. Genes Dev. 22, 654–667 (2008). This paper shows that the longest telomeres are a general feature of adult stem cell compartments. Telomeres are shown to shorten with age in stem cell compartments concomitantly with a decline in stem cell functionality, suggesting telomere loss as a caue of age-dependent stem cell dysfunction.
Liu, L. et al. Telomere lengthening early in development. Nature Cell Biol. 9, 1436–1441 (2007).
Marion, R. M. et al. Telomeres acquire embryonic stem cell characteristics in induced pluripotent stem cells. Cell Stem Cell 4, 141–154 (2009). This paper demonstrates that telomeres are elongated in a telomerase-dependent manner and acquire the epigenetic markers of ES cells during nuclear reprogramming. That initial telomere length in parental cells is a determinant for the iPS generation efficiency is also shown.
Collado, M., Blasco, M. A. & Serrano, M. Cellular senescence in cancer and aging. Cell 130, 223–233 (2007).
Rudolph, K. L. et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 96, 701–712 (1999).
Armanios, M. Y. et al. Telomerase mutations in families with idiopathic pulmonary fibrosis. N. Engl. J. Med. 356, 1317–1326 (2007).
Mitchell, J. R., Wood, E. & Collins, K. A telomerase component is defective in the human disease dyskeratosis congenita. Nature 402, 551–555 (1999).
Tsakiri, K. D. et al. Adult-onset pulmonary fibrosis caused by mutations in telomerase. Proc. Natl Acad. Sci. USA 104, 7552–7557 (2007).
Vulliamy, T. et al. The RNA component of telomerase is mutated in autosomal dominant dyskeratosis congenita. Nature 413, 432–435 (2001).
Yamaguchi, H. et al. Mutations in TERT, the gene for telomerase reverse transcriptase, in aplastic anemia. N. Engl. J. Med. 352, 1413–1424 (2005).
Schoeftner, S. & Blasco, M. A. A 'higher order' of telomere regulation: telomere heterochromatin and telomeric RNAs. EMBO J. 28, 2323–2336 (2009).
Benetti, R. et al. Suv4–20h deficiency results in telomere elongation and derepression of telomere recombination. J. Cell Biol. 178, 925–936 (2007).
Garcia-Cao, M., O'Sullivan, R., Peters, A. H., Jenuwein, T. & Blasco, M. A. Epigenetic regulation of telomere length in mammalian cells by the Suv39h1 and Suv39h2 histone methyltransferases. Nature Genet. 36, 94–99 (2004).
Gonzalo, S. et al. DNA methyltransferases control telomere length and telomere recombination in mammalian cells. Nature Cell Biol. 8, 416–424 (2006).
Benetti, R. et al. A mammalian microRNA cluster controls DNA methylation and telomere recombination via Rbl2-dependent regulation of DNA methyltransferases. Nature Struct. Mol. Biol. 15, 268–279 (2008).
Azzalin, C. M., Reichenbach, P., Khoriauli, L., Giulotto, E. & Lingner, J. Telomeric repeat containing RNA and RNA surveillance factors at mammalian chromosome ends. Science 318, 798–801 (2007).
Schoeftner, S. & Blasco, M. A. Developmentally regulated transcription of mammalian telomeres by DNA-dependent RNA polymerase II. Nature Cell Biol. 10, 228–236 (2008).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Court, R., Chapman, L., Fairall, L. & Rhodes, D. How the human telomeric proteins TRF1 and TRF2 recognize telomeric DNA: a view from high-resolution crystal structures. EMBO Rep. 6, 39–45 (2005).
Bianchi, A. et al. TRF1 binds a bipartite telomeric site with extreme spatial flexibility. EMBO J. 18, 5735–5744 (1999).
Broccoli, D., Smogorzewska, A., Chong, L. & de Lange, T. Human telomeres contain two distinct Myb-related proteins, TRF1 and TRF2. Nature Genet. 17, 231–235 (1997).
Lei, M., Podell, E. R. & Cech, T. R. Structure of human POT1 bound to telomeric single-stranded DNA provides a model for chromosome end-protection. Nature Struct. Mol. Biol. 11, 1223–1229 (2004).
Baumann, P. & Cech, T. R. Pot1, the putative telomere end-binding protein in fission yeast and humans. Science 292, 1171–1175 (2001).
Loayza, D. & De Lange, T. POT1 as a terminal transducer of TRF1 telomere length control. Nature 423, 1013–1018 (2003).
He, H. et al. POT1b protects telomeres from end-to-end chromosomal fusions and aberrant homologous recombination. EMBO J. 25, 5180–5190 (2006).
Kim, S. H. et al. TIN2 mediates functions of TRF2 at human telomeres. J. Biol. Chem. 279, 43799–43804 (2004).
Ye, J. Z. et al. POT1-interacting protein PIP1: a telomere length regulator that recruits POT1 to the TIN2/TRF1 complex. Genes Dev. 18, 1649–1654 (2004).
Chen, Y. et al. A shared docking motif in TRF1 and TRF2 used for differential recruitment of telomeric proteins. Science 319, 1092–1096 (2008).
Liu, D. et al. PTOP interacts with POT1 and regulates its localization to telomeres. Nature Cell Biol. 6, 673–680 (2004).
Chen, L. Y., Liu, D. & Songyang, Z. Telomere maintenance through spatial control of telomeric proteins. Mol. Cell Biol. 27, 5898–5909 (2007).
Xin, H. et al. TPP1 is a homologue of ciliate TEBP-beta and interacts with POT1 to recruit telomerase. Nature 445, 559–562 (2007).
Li, B. & de Lange, T. Rap1 affects the length and heterogeneity of human telomeres. Mol. Biol. Cell 14, 5060–5068 (2003).
Li, B., Oestreich, S. & de Lange, T. Identification of human Rap1: implications for telomere evolution. Cell 101, 471–483 (2000).
Sfeir, A., Kabir, S., van Overbeek, M., Celli, G. B. & de Lange, T. Loss of Rap1 induces telomere recombination in the absence of NHEJ or a DNA damage signal. Science 327, 1657–1661 (2010). This paper describes a crucial role for RAP1 in the inhibition of homology-directed repair at telomeres. RAP1 is shown to be dispensable for TRF2 function, repression of ATM signalling and the NHEJ pathway.
Sfeir, A. et al. Mammalian telomeres resemble fragile sites and require TRF1 for efficient replication. Cell 138, 90–103 (2009). This paper demonstrates that TRF1 is required for proper telomere replication preventing fork stalling. Terf1 deletion activates the ATR signalling pathway and causes a fragile phenotype at telomeres.
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instabilities in human cancers. Nature 396, 643–649 (1998).
Artandi, S. E. & DePinho, R. A. Telomeres and telomerase in cancer. Carcinogenesis 31, 9–18 (2010).
Halazonetis, T. D., Gorgoulis, V. G. & Bartek, J. An oncogene-induced DNA damage model for cancer development. Science 319, 1352–1355 (2008).
Tsantoulis, P. K. et al. Oncogene-induced replication stress preferentially targets common fragile sites in preneoplastic lesions. A genome-wide study. Oncogene 27, 3256–3264 (2008).
Engelhardt, M., Drullinsky, P., Guillem, J. & Moore, M. A. Telomerase and telomere length in the development and progression of premalignant lesions to colorectal cancer. Clin. Cancer Res. 3, 1931–1941 (1997).
Engelhardt, M. et al. Relative contribution of normal and neoplastic cells determines telomerase activity and telomere length in primary cancers of the prostate, colon, and sarcoma. Clin. Cancer Res. 3, 1849–1857 (1997).
Odagiri, E. et al. Reduction of telomeric length and c-erbB-2 gene amplification in human breast cancer, fibroadenoma, and gynecomastia. Relationship to histologic grade and clinical parameters. Cancer 73, 2978–2984 (1994).
Meeker, A. K. et al. Telomere shortening occurs in subsets of normal breast epithelium as well as in situ and invasive carcinoma. Am. J. Pathol. 164, 925–935 (2004).
Meeker, A. K. et al. Telomere length abnormalities occur early in the initiation of epithelial carcinogenesis. Clin. Cancer Res. 10, 3317–3326 (2004).
van Heek, N. T. et al. Telomere shortening is nearly universal in pancreatic intraepithelial neoplasia. Am. J. Pathol. 161, 1541–1547 (2002).
Artandi, S. E. et al. Telomere dysfunction promotes non-reciprocal translocations and epithelial cancers in mice. Nature 406, 641–645 (2000). This paper was the first to demonstrate that critically short telomeres in a Trp53 -deficient genetic background lead to the development of epithelial cancers. It highlights that genomic instability that is induced by telomere dysfunction induces tumorigenesis and establishes a key role for p53 in the cellular response to telomere dysfunction in vivo.
Blanco, R., Munoz, P., Flores, J. M., Klatt, P. & Blasco, M. A. Telomerase abrogation dramatically accelerates TRF2-induced epithelial carcinogenesis. Genes Dev. 21, 206–220 (2007). This paper demonstrates that TRF2 overexpression in combination with telomerase deficiency acts as a very potent oncogene in vivo . The data presented suggest telomerase inhibition might not cease the growth of TRF2-overexpressing tumours.
Denchi, E. L. Give me a break: how telomeres suppress the DNA damage response. DNA Repair (Amst) 8, 1118–1126 (2009).
Rai, R. et al. The function of classical and alternative non-homologous end-joining pathways in the fusion of dysfunctional telomeres. EMBO J. 29, 2598–2610 (2010).
Espejel, S. et al. Mammalian Ku86 mediates chromosomal fusions and apoptosis caused by critically short telomeres. EMBO J. 21, 2207–2219 (2002).
Maser, R. S. et al. DNA-dependent protein kinase catalytic subunit is not required for dysfunctional telomere fusion and checkpoint response in the telomerase-deficient mouse. Mol. Cell Biol. 27, 2253–2265 (2007).
Murnane, J. P. Telomere loss as a mechanism for chromosome instability in human cancer. Cancer Res. 70, 4255–4259 (2010).
Gilson, E. & Geli, V. How telomeres are replicated. Nature Rev. Mol. Cell Biol. 8, 825–838 (2007).
Oganesian, L. & Bryan, T. M. Physiological relevance of telomeric G-quadruplex formation: a potential drug target. Bioessays 29, 155–165 (2007).
Cacchione, S., Cerone, M. A. & Savino, M. In vitro low propensity to form nucleosomes of four telomeric sequences. FEBS Lett. 400, 37–41 (1997).
Verdun, R. E. & Karlseder, J. The DNA damage machinery and homologous recombination pathway act consecutively to protect human telomeres. Cell 127, 709–720 (2006).
McNees, C. J. et al. ATR suppresses telomere fragility and recombination but is dispensable for elongation of short telomeres by telomerase. J. Cell Biol. 188, 639–652 (2010).
Casper, A. M., Durkin, S. G., Arlt, M. F. & Glover, T. W. Chromosomal instability at common fragile sites in Seckel syndrome. Am. J. Hum. Genet. 75, 654–660 (2004).
Murga, M. et al. A mouse model of ATR-Seckel shows embryonic replicative stress and accelerated aging. Nature Genet. 41, 891–898 (2009).
Okamoto, K., Iwano, T., Tachibana, M. & Shinkai, Y. Distinct roles of TRF1 in the regulation of telomere structure and lengthening. J. Biol. Chem. 283, 23981–23988 (2008).
Else, T. et al. Tpp1/Acd maintains genomic stability through a complex role in telomere protection. Chromosome Res. 15, 1001–1013 (2007).
Keegan, C. E. et al. Urogenital and caudal dysgenesis in adrenocortical dysplasia (acd) mice is caused by a splicing mutation in a novel telomeric regulator. Hum. Mol. Genet. 14, 113–123 (2005).
Savage, S. A. et al. TINF2, a component of the shelterin telomere protection complex, is mutated in dyskeratosis congenita. Am. J. Hum. Genet. 82, 501–509 (2008).
Walne, A. J., Vulliamy, T., Beswick, R., Kirwan, M. & Dokal, I. TINF2 mutations result in very short telomeres: analysis of a large cohort of patients with dyskeratosis congenita and related bone marrow failure syndromes. Blood 112, 3594–3600 (2008).
Carroll, K. A. & Ly, H. Telomere dysfunction in human diseases: the long and short of it! Int. J. Clin. Exp. Pathol. 2, 528–543 (2009).
Henle, E. S. et al. Sequence-specific DNA cleavage by Fe2+-mediated fenton reactions has possible biological implications. J. Biol. Chem. 274, 962–971 (1999).
Oikawa, S., Tada-Oikawa, S. & Kawanishi, S. Site-specific DNA damage at the GGG sequence by UVA involves acceleration of telomere shortening. Biochemistry 40, 4763–4768 (2001).
Ayouaz, A., Raynaud, C., Heride, C., Revaud, D. & Sabatier, L. Telomeres: hallmarks of radiosensitivity. Biochimie 90, 60–72 (2008).
Sitte, N., Saretzki, G. & von Zglinicki, T. Accelerated telomere shortening in fibroblasts after extended periods of confluency. Free Radic Biol. Med. 24, 885–893 (1998).
von Zglinicki, T., Saretzki, G., Docke, W. & Lotze, C. Mild hyperoxia shortens telomeres and inhibits proliferation of fibroblasts: a model for senescence? Exp. Cell Res. 220, 186–193 (1995).
Rochette, P. J. & Brash, D. E. Human telomeres are hypersensitive to UV-induced DNA Damage and refractory to repair. PLoS Genet. 6, e1000926 (2010).
Munoz, P. et al. TRF1 controls telomere length and mitotic fidelity in epithelial homeostasis. Mol. Cell Biol. 29, 1608–1625 (2009).
Zhu, X. D. et al. ERCC1/XPF removes the 3′ overhang from uncapped telomeres and represses formation of telomeric DNA-containing double minute chromosomes. Mol. Cell 12, 1489–1498 (2003).
Munoz, P., Blanco, R. & Blasco, M. A. Role of the TRF2 telomeric protein in cancer and ageing. Cell Cycle 5, 718–721 (2006).
Sands, A. T., Abuin, A., Sanchez, A., Conti, C. J. & Bradley, A. High susceptibility to ultraviolet-induced carcinogenesis in mice lacking XPC. Nature 377, 162–165 (1995).
Nakane, H. et al. High incidence of ultraviolet-B-or chemical-carcinogen-induced skin tumours in mice lacking the xeroderma pigmentosum group A gene. Nature 377, 165–168 (1995).
Rajagopalan, H. & Lengauer, C. Aneuploidy and cancer. Nature 432, 338–341 (2004).
Sen, S. Aneuploidy and cancer. Curr. Opin. Oncol. 12, 82–88 (2000).
Sotillo, R. et al. Mad2 overexpression promotes aneuploidy and tumorigenesis in mice. Cancer Cell 11, 9–23 (2007).
Weaver, B. A., Silk, A. D., Montagna, C., Verdier-Pinard, P. & Cleveland, D. W. Aneuploidy acts both oncogenically and as a tumor suppressor. Cancer Cell 11, 25–36 (2007).
Davoli, T., Denchi, E. L. & de Lange, T. Persistent telomere damage induces bypass of mitosis and tetraploidy. Cell 141, 81–93 (2010). This paper provides a molecular mechanism underlying tetraploidization as a consequence of persistent telomere dysfunction. A prolonged DNA damage signal in a TP53 -deficient background and its consequent activation of ATM and ATR results in the inhibition of CDK1–cyclin B, cells bypass mitosis and enter a second S phase becoming tetraploid. A molecular explanation about the high incidence of aneuploid cells in tumours is given.
Pantic, M. et al. Telomere dysfunction and loss of p53 cooperate in defective mitotic segregation of chromosomes in cancer cells. Oncogene 25, 4413–4420 (2006).
Toledo, L. I., Murga, M., Gutierrez-Martinez, P., Soria, R. & Fernandez-Capetillo, O. ATR signaling can drive cells into senescence in the absence of DNA breaks. Genes Dev. 22, 297–302 (2008).
Denchi, E. L. & de Lange, T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT1. Nature 448, 1068–1071 (2007).
Guo, X. et al. Dysfunctional telomeres activate an ATM-ATR-dependent DNA damage response to suppress tumorigenesis. EMBO J. 26, 4709–4719 (2007).
Hockemeyer, D., Palm, W., Wang, R. C., Couto, S. S. & de Lange, T. Engineered telomere degradation models dyskeratosis congenita. Genes Dev. 22, 1773–1785 (2008). This paper shows that mice lacking Pot1b in combination with reduced telomerase activity develop phenotypes that recapitulate those observed in patients with dyskeratosis congenita such as hyperpigmentation and fatal bone marrow failure. It highlights the idea that this disease is caused by telomere dysfunction.
He, H. et al. Pot1b deletion and telomerase haploinsufficiency in mice initiate an ATR-dependent DNA damage response and elicit phenotypes resembling dyskeratosis congenita. Mol. Cell Biol. 29, 229–240 (2009).
Shay, J. W. & Wright, W. E. Telomerase therapeutics for cancer: challenges and new directions. Nature Rev. Drug Discov. 5, 577–584 (2006).
Shore, D. & Nasmyth, K. Purification and cloning of a DNA binding protein from yeast that binds to both silencer and activator elements. Cell 51, 721–732 (1987).
Kyrion, G., Liu, K., Liu, C. & Lustig, A. J. RAP1 and telomere structure regulate telomere position effects in Saccharomyces cerevisiae. Genes Dev. 7, 1146–1159 (1993).
Marcand, S., Gilson, E. & Shore, D. A protein-counting mechanism for telomere length regulation in yeast. Science 275, 986–990 (1997).
Hecht, A., Laroche, T., Strahl-Bolsinger, S., Gasser, S. M. & Grunstein, M. Histone H3 and H4 N-termini interact with SIR3 and SIR4 proteins: a molecular model for the formation of heterochromatin in yeast. Cell 80, 583–592 (1995).
Tanny, J. C., Dowd, G. J., Huang, J., Hilz, H. & Moazed, D. An enzymatic activity in the yeast Sir2 protein that is essential for gene silencing. Cell 99, 735–745 (1999).
Carmen, A. A., Milne, L. & Grunstein, M. Acetylation of the yeast histone H4 N. terminus regulates its binding to heterochromatin protein SIR3. J. Biol. Chem. 277, 4778–4781 (2002).
Imai, S., Armstrong, C. M., Kaeberlein, M. & Guarente, L. Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature 403, 795–800 (2000).
Buchman, A. R., Kimmerly, W. J., Rine, J. & Kornberg, R. D. Two DNA-binding factors recognize specific sequences at silencers, upstream activating sequences, autonomously replicating sequences, and telomeres in Saccharomyces cerevisiae. Mol. Cell Biol. 8, 210–225 (1988).
Capieaux, E., Vignais, M. L., Sentenac, A. & Goffeau, A. The yeast H+-ATPase gene is controlled by the promoter binding factor TUF. J. Biol. Chem. 264, 7437–7446 (1989).
Yang, X., Figueiredo, L. M., Espinal, A., Okubo, E. & Li, B. RAP1 is essential for silencing telomeric variant surface glycoprotein genes in Trypanosoma brucei. Cell 137, 99–109 (2009).
Hanaoka, S. et al. NMR structure of the hRap1 Myb motif reveals a canonical three-helix bundle lacking the positive surface charge typical of Myb DNA-binding domains. J. Mol. Biol. 312, 167–175 (2001).
Ghosh, S. & Karin, M. Missing pieces in the NF-κB puzzle. Cell 109, S81–S96 (2002).
Weigelt, B., Peterse, J. L. & van 't Veer, L. J. Breast cancer metastasis: markers and models. Nature Rev. Cancer 5, 591–602 (2005).
Basseres, D. S. & Baldwin, A. S. Nuclear factor-kappaB and inhibitor of kappaB kinase pathways in oncogenic initiation and progression. Oncogene 25, 6817–6830 (2006).
Akiyama, M. et al. Nuclear factor-κB p65 mediates tumor necrosis factor alpha-induced nuclear translocation of telomerase reverse transcriptase protein. Cancer Res. 63, 18–21 (2003).
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998).
Artandi, S. E. et al. Constitutive telomerase expression promotes mammary carcinomas in aging mice. Proc. Natl Acad. Sci. USA 99, 8191–8196 (2002).
Gonzalez-Suarez, E., Flores, J. M. & Blasco, M. A. Cooperation between p53 mutation and high telomerase transgenic expression in spontaneous cancer development. Mol. Cell Biol. 22, 7291–7301 (2002).
Canela, A., Martin-Caballero, J., Flores, J. M. & Blasco, M. A. Constitutive expression of tert in thymocytes leads to increased incidence and dissemination of T-cell lymphoma in Lck-Tert mice. Mol. Cell Biol. 24, 4275–4293 (2004).
Shete, S. et al. Genome-wide association study identifies five susceptibility loci for glioma. Nature Genet. 41, 899–904 (2009).
Petersen, G. M. et al. A genome-wide association study identifies pancreatic cancer susceptibility loci on chromosomes 13q22.1, 1q32.1 and 5p15.33. Nature Genet. 42, 224–228 (2010).
McKay, J. D. et al. Lung cancer susceptibility locus at 5p15.33. Nature Genet. 40, 1404–1406 (2008).
Rafnar, T. et al. Sequence variants at the TERT-CLPTM1L locus associate with many cancer types. Nature Genet. 41, 221–227 (2009).
Wang, Y. et al. Common 5p15.33 and 6p21.33 variants influence lung cancer risk. Nature Genet. 40, 1407–1409 (2008).
Zhang, A. et al. Genetic alterations in cervical carcinomas: frequent low-level amplifications of oncogenes are associated with human papillomavirus infection. Int. J. Cancer 101, 427–433 (2002).
Kang, J. U., Koo, S. H., Kwon, K. C., Park, J. W. & Kim, J. M. Gain at chromosomal region 5p15.33, containing TERT, is the most frequent genetic event in early stages of non-small cell lung cancer. Cancer Genet. Cytogenet. 182, 1–11 (2008).
Hemann, M. T., Strong, M. A., Hao, L. Y. & Greider, C. W. The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell 107, 67–77 (2001).
Armanios, M. et al. Short telomeres are sufficient to cause the degenerative defects associated with aging. Am. J. Hum. Genet. 85, 823–832 (2009).
Hao, L. Y. et al. Short telomeres, even in the presence of telomerase, limit tissue renewal capacity. Cell 123, 1121–1131 (2005).
Reya, T. & Clevers, H. Wnt signalling in stem cells and cancer. Nature 434, 843–850 (2005).
Lo Celso, C., Prowse, D. M. & Watt, F. M. Transient activation of beta-catenin signalling in adult mouse epidermis is sufficient to induce new hair follicles but continuous activation is required to maintain hair follicle tumours. Development 131, 1787–1799 (2004).
Van Mater., D., Kolligs, F. T., Dlugosz, A. A. & Fearon, E. R. Transient activation of beta -catenin signaling in cutaneous keratinocytes is sufficient to trigger the active growth phase of the hair cycle in mice. Genes Dev. 17, 1219–1224 (2003).
Cong, Y. & Shay, J. W. Actions of human telomerase beyond telomeres. Cell Res. 18, 725–732 (2008).
Ridanpaa, M. et al. Mutations in the RNA component of RNase MRP cause a pleiotropic human disease, cartilage-hair hypoplasia. Cell 104, 195–203 (2001).
Santos, J. H., Meyer, J. N., Skorvaga, M., Annab, L. A. & Van Houten, B. Mitochondrial hTERT exacerbates free-radical-mediated mtDNA damage. Aging Cell 3, 399–411 (2004).
Santos, J. H., Meyer, J. N. & Van Houten, B. Mitochondrial localization of telomerase as a determinant for hydrogen peroxide-induced mitochondrial DNA damage and apoptosis. Hum. Mol. Genet. 15, 1757–1768 (2006).
Haendeler, J. et al. Antioxidants inhibit nuclear export of telomerase reverse transcriptase and delay replicative senescence of endothelial cells. Circ. Res. 94, 768–775 (2004).
Zhang, P., Chan, S. L., Fu, W., Mendoza, M. & Mattson, M. P. TERT suppresses apoptotis at a premitochondrial step by a mechanism requiring reverse transcriptase activity and 14-3-3 protein-binding ability. FASEB J. 17, 767–769 (2003).
Massard, C. et al. hTERT: a novel endogenous inhibitor of the mitochondrial cell death pathway. Oncogene 25, 4505–4514 (2006).
Lee, J. et al. TERT promotes cellular and organismal survival independently of telomerase activity. Oncogene 27, 3754–3760 (2008).
Vidal-Cardenas, S. L. & Greider, C. W. Comparing effects of mTR and mTERT deletion on gene expression and DNA damage response: a critical examination of telomere length maintenance-independent roles of telomerase. Nucleic Acids Res. 38, 60–71 (2010).
Erdmann, N. & Harrington, L. A. No attenuation of the ATM-dependent DNA damage response in murine telomerase-deficient cells. DNA Repair (Amst) 8, 347–353 (2009).
Zhang, J. Z. Overexpression analysis of plant transcription factors. Curr. Opin. Plant Biol. 6, 30–440 (2003).
Barolo, S. Transgenic Wnt/TCF pathway reporters: all you need is Lef? Oncogene 25, 7505–7511 (2006).
Cortez-Gonzalez, X. & Zanetti, M. Telomerase immunity from bench to bedside: round one. J. Transl. Med. 5, 12 (2007).
Harley, C. B. Telomerase and cancer therapeutics. Nature Rev. Cancer 8, 167–179 (2008).
Wang, F. et al. The POT1-TPP1 telomere complex is a telomerase processivity factor. Nature 445, 506–510 (2007).
Latrick, C. M. & Cech, T. R. POT1-TPP1 enhances telomerase processivity by slowing primer dissociation and aiding translocation. EMBO J. 29, 924–933 (2010).
Marion, R. M. et al. A p53-mediated DNA damage response limits reprogramming to ensure iPS cell genomic integrity. Nature 460, 1149–1153 (2009). This paper demonstrates the role of p53 as a barrier for reprogramming. p53 initiates apoptotic pathways in response to critically short telomeres, as well as other types of DNA damage, and thereby inhibits the generation of pluripotent stem cells from suboptimal parental cells, including those with short telomeres.
Walne, A. J. et al. Genetic heterogeneity in autosomal recessive dyskeratosis congenita with one subtype due to mutations in the telomerase-associated protein NOP10. Hum. Mol. Genet. 16, 1619–1629 (2007).
Vulliamy, T. et al. Mutations in the telomerase component NHP2 cause the premature ageing syndrome dyskeratosis congenita. Proc. Natl Acad. Sci. USA 105, 8073–8078 (2008).
Savage, S. A. et al. Genetic variation in telomeric repeat binding factors 1 and 2 in aplastic anemia. Exp. Hematol. 34, 664–671 (2006).
Blasco, M. A. & Hahn, W. C. Evolving views of telomerase and cancer. Trends Cell Biol. 13, 289–294 (2003).
Artandi, S. E. & DePinho, R. A. A critical role for telomeres in suppressing and facilitating carcinogenesis. Curr. Opin. Genet. Dev. 10, 39–46 (2000).
Flores, I. & Blasco, M. A. A p53-dependent response limits epidermal stem cell functionality and organismal size in mice with short telomeres. PLoS One 4, e4934 (2009).
Begus-Nahrmann, Y. et al. p53 deletion impairs clearance of chromosomal-instable stem cells in aging telomere-dysfunctional mice. Nature Genet. 41, 1138–1143 (2009).
Khoo, C. M., Carrasco, D. R., Bosenberg, M. W., Paik, J. H. & Depinho, R. A. Ink4a/Arf tumor suppressor does not modulate the degenerative conditions or tumor spectrum of the telomerase-deficient mouse. Proc. Natl Acad. Sci. USA 104, 3931–3936 (2007).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Stadtfeld, M., Maherali, N., Breault, D. T. & Hochedlinger, K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell 2, 230–240 (2008).
Tsangaris, E. et al. Ataxia and pancytopenia caused by a mutation in TINF2. Hum. Genet. 124, 507–513 (2008).
Ye, J. Z. et al. TIN2 binds TRF1 and TRF2 simultaneously and stabilizes the TRF2 complex on telomeres. J. Biol. Chem. 279, 47264–47271 (2004).
Acknowledgements
P.M. is a 'Ramón y Cajal' senior scientist. M.A.B.'s laboratory is funded by the Spanish Ministry of Innovation and Science, the European Union (géenica, FP7), the European Reseach Council (ERC Advance Grants), the Spanish Association Against Cancer (AECC) and the Körber European Science Award. The authors apologize to all authors whose work has not been cited owing to space limitations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Telomere uncapping
-
Loss of proper telomere structure owing to either loss of telomeric repeat sequences or alteration in telomere proteins that leads to the activation of the DNA damage response.
- Mismatch repair pathway
-
DNA repair mechanism that corrects mispaired nucleotides that originate during DNA replication and recombination.
- Oral leukoplakia
-
The most common premalignant or potentially malignant disorder of the oral mucosa characterized by presenting several degrees of epithelia dysplasia. Defined as a white patch or plaque of the oral mucosa that cannot be characterized clinically or pathologically as any other disease.
- Chromatid dicentrics
-
Aberrant dicentric chromosome that results when a chromosome lacking a telomere or with a dysfunctional telomere replicates and its sister chromatids fuse at their ends. At anaphase, the fused sister chromatids break owing to the presence of two centromeres.
- Dicentric chromosome
-
Chromosome with two centromeres that results from the fusion of two monocentric chromosome pieces.
- Multicentric chromosome
-
Chromosome with multiple centromeres that results from the fusion of several chromosome pieces.
- Ring
-
Aberrant monocentric chromosome the arms of which have fused together to form a ring.
- Double-minute chromosome
-
Acentric chromatin circle of variable size that consists of multiple copies of a short rearranged DNA segment that has undergone amplification.
- G-quadruplex
-
Four-stranded structure formed by nucleic acids rich in guanine that consists of a square arrangement of guanines stabilized by Hoogsteen hydrogen bonds. Can form within or between G-rich strands of telomeric DNA.
- Fragile site
-
Genomic region prone to replication fork stalling in which gaps and breaks frequently occur and is thus a hotspot for recombination events.
- Multiple telomeric signal (MTS)
-
An aberrant structure at telomeres that is enhanced by conditions known to cause replication fork collapse, such as aphidicolin treatment. By immunofluorescence technique, an MTS is seen as several fluorescent dots instead of just one.
- Hypomorphic mutation
-
A type of mutation that results in an altered gene product with reduced enzymatic activity or a lower expression level than the wild-type allele.
- Chromosome concatenation
-
Aberrant chromosomal structure in which several chromosomes are fused.
- Cyclobutane pyrimidine dimer (CPD)
-
Interstrand DNA lesion formed when two adjacent pyrimidines are joined across their 5–6 double bonds owing to the UV radiation excitation of one of the pyrimidines.
- Nucleotide excision repair (NER)
-
DNA repair mechanism that removes UV radiation-induced helix-distorting lesions such as pyrimidine dimers and 6,4 photoproducts. Two subpathways exist: global genomic NER and transcription-coupled NER.
- Xeroderma pigmentosum
-
Autosomal recessive genetic disorder linked to mutations in components within the NER pathway. Patients are deficient in repairing UV radiation-induced DNA damage and are prone to develop skin cancers and several skin malignancies such as keratoses, hyperpigmentation and blistering.
- Spindle assembly checkpoint (SAC)
-
Monitors proper chromosome attachment to spindle microtubules. SCA prevents anaphase until all chromosomes are properly attached to the spindle. To achieve proper segregation, the two kinetochores on the sister chromatids must be attached to opposite spindle poles. It is composed of mitotic checkpoint proteins MAD and BUB. These reside on kinetochores and show changes in phosphorylation and localization as cells proceed through mitosis. Failure of SAC can result in aneuploidy.
- Gene set enrichment analysis (GSEA)
-
A computational method that determines whether an a priori defined set of genes shows significant differences between two biological states.
Rights and permissions
About this article
Cite this article
Martínez, P., Blasco, M. Telomeric and extra-telomeric roles for telomerase and the telomere-binding proteins. Nat Rev Cancer 11, 161–176 (2011). https://doi.org/10.1038/nrc3025
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3025
This article is cited by
-
Genomic alterations in thyroid cancer: biological and clinical insights
Nature Reviews Endocrinology (2024)
-
Telomere-based treatment strategy of cardiovascular diseases: imagination comes to reality
Genome Instability & Disease (2024)
-
Endothelial progenitor cells systemic administration alleviates multi-organ senescence by down-regulating USP7/p300 pathway in chronic obstructive pulmonary disease
Journal of Translational Medicine (2023)
-
Aging and the impact of global DNA methylation, telomere shortening, and total oxidative status on sarcopenia and frailty syndrome
Immunity & Ageing (2023)
-
Telomerase RNA-based aptamers restore defective myelopoiesis in congenital neutropenic syndromes
Nature Communications (2023)